Vaccine Residual Process Reagents Market
Vaccine Residual Process Reagents Market Analysis - Size, Share, and Forecast Outlook 2025 to 2035
Historical Data Covered: 2015 to 2023 | Base Year: 2024 | Estimated Year: 2025 | Forecast Period: 2026 to 2035
Vaccine Residual Process Reagents Market Forecast and Outlook 2025 to 2035
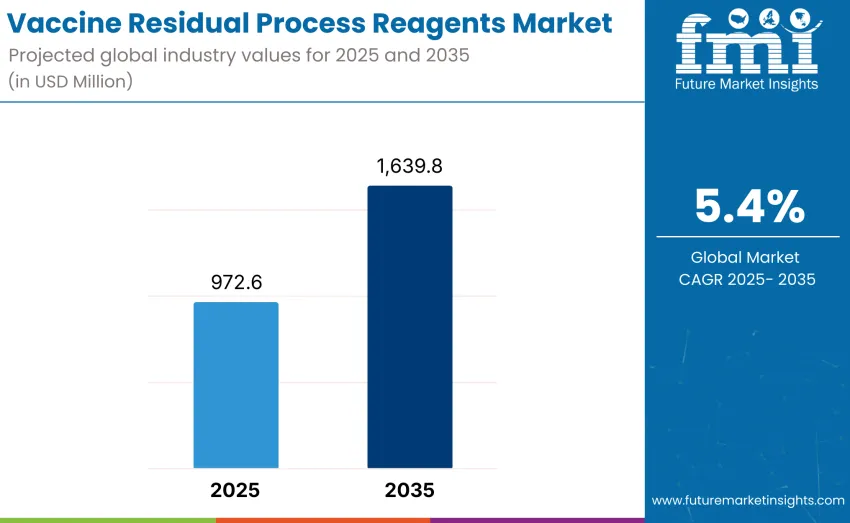
The global vaccine residual process reagents market is projected to reach USD 1,639.8 million by 2035, recording an absolute increase of USD 667.2 million over the forecast period. This market is valued at USD 972.6 million in 2025 and is set to rise at a CAGR of 5.4% during the assessment period. Overall size is expected to grow by nearly 1.5 times over the same period, supported by increasing demand for high-quality vaccines worldwide, driving adoption of advanced quality control solutions and growing investments in biopharmaceutical manufacturing infrastructure and process optimization initiatives globally.
Increasing complexity of vaccine platforms and rising global immunization targets create demand for high-precision reagents capable of detecting host cell proteins, nucleic acids, and endotoxins at trace levels. Technological advancements in assay design, reagent formulation, and automated detection platforms are reshaping the vaccine residual process reagents landscape. Modern systems integrate with ELISA, HPLC, PCR, and chromatography tools, enabling precise detection of residual contaminants and supporting streamlined quality control workflows. Innovations in reagent sensitivity, stability, and reproducibility allow manufacturers to maintain regulatory compliance while reducing batch release times.
Quick Stats for Vaccine Residual Process Reagents Market
- Vaccine Residual Process Reagents Market Value (2025): USD 972.6 million
- Vaccine Residual Process Reagents Market Value (2035): USD 1,639.8 million
- Vaccine Residual Process Reagents Market CAGR: 5.4%
- Leading Reagent Class in Vaccine Residual Process Reagents Market: Nucleases for Residual DNA Removal with 23.7% share
- Key Growth Regions in Vaccine Residual Process Reagents Market: Asia Pacific, Europe, and North America
- Top Players in Vaccine Residual Process Reagents Market: Merck MilliporeSigma (incl. Benzonase), Thermo Fisher Scientific, Cytiva, Sartorius, Croda Pharma (incl. Avanti), BASF (Kolliphor/solvents), Seppic (Air Liquide Group), NOF Corporation (PEG & lipids), ArcticZymes (SAN nucleases), Eastman (BPL/formulation solvents), Promega (nucleases & enzymes)
Government vaccination programs, global immunization campaigns, and public health initiatives accelerate growth. Biopharmaceutical companies and research institutions worldwide are investing in laboratory infrastructure and process optimization to ensure vaccine safety and efficacy. Industry adoption of large-scale automated production platforms drives sustained demand for high-performance reagents that support rapid vaccine development, efficient batch testing, and global distribution. Training programs for laboratory personnel and quality control teams enhance adoption of sophisticated reagents, enabling manufacturers to maintain consistent product quality across diverse vaccine pipelines.
Vaccine Residual Process Reagents Market Year-over-Year Forecast (2025 to 2035)
Between 2025 and 2030, the vaccine residual process reagents market is projected to expand from USD 972.6 million to USD 1,262.9 million, resulting in a value increase of USD 290.3 million, which represents 43.5% of total forecast growth for the decade. This growth phase will be driven by rising global demand for vaccines, increased production capacities, innovation in residual process reagents, and adoption of advanced purification and formulation technologies. Companies are strengthening positions through investment in high-quality reagents, enzyme technologies, process optimization, and strategic expansion in emerging and established markets.
From 2030 to 2035, growth is forecast from USD 1,262.9 million to USD 1,639.8 million, adding USD 376.9 million, constituting 56.5% of overall ten-year expansion. This period is expected to see increased adoption of specialized reagents for next-generation vaccines, collaborations between reagent suppliers and vaccine manufacturers, and focus on scalable, efficient, and sustainable manufacturing processes. Rising global vaccination programs, demand for higher purity reagents, and regulatory compliance will continue to drive growth, along with innovation in enzyme and solvent formulations tailored for advanced vaccine production processes.
Vaccine Residual Process Reagents Market Key Takeaways
| Metric | Value |
|---|---|
| Market Value (2025) | USD 972.6 million |
| Market Forecast Value (2035) | USD 1,639.8 million |
| Forecast CAGR (2025 to 2035) | 5.4% |
Why Is the Vaccine Residual Process Reagents Market Growing Worldwide?
The vaccine residual process reagents market grows by enabling biopharmaceutical manufacturers to achieve superior vaccine purity and safety while maintaining efficient production workflows for regulatory compliance. Vaccine producers face mounting pressure to deliver high-quality products within stringent regulatory timelines, with advanced residual process reagents providing significant improvement in contaminant detection and removal efficiency over conventional methods, making these technologies essential for modern vaccine manufacturing operations. Growing global vaccination programs create demand for professional-grade reagents that can deliver consistent quality across mRNA, viral vector, and protein subunit vaccine production requiring precise impurity control.
Government initiatives promoting vaccine development and manufacturing infrastructure drive adoption in biopharmaceutical facilities, contract manufacturing organizations, and research institutions, where high-quality reagents have direct impact on product safety and regulatory approval success. The global shift toward automated production platforms and quality control systems accelerates demand for reagents that enable rapid batch testing and release processes while maintaining compliance with international standards. Premium pricing of specialized reagents and complex integration requirements with sophisticated purification systems may limit adoption among smaller manufacturers.
How Is the Vaccine Residual Process Reagents Market Segmented by Reagent Class, Application, and Region?
The vaccine residual process reagents market is segmented by reagent class, application, and region. By reagent class, division includes nucleases for residual DNA removal, detergents & solubilizers, inactivation agents, precipitation polymers & flocculants, extraction solvents & phase-transfer aids, buffer additives & scavengers, and LNP/wash & auxiliary process reagents. Based on application, categorization covers mRNA & LNP vaccines, viral vector vaccines, protein subunit & inactivated vaccines, live-attenuated & oral vaccines, and combination/platform-agnostic programs. Regionally, segmentation spans Asia Pacific, Europe, North America, Latin America, and Middle East & Africa.
By Reagent Class, Nucleases for Residual DNA Removal Lead
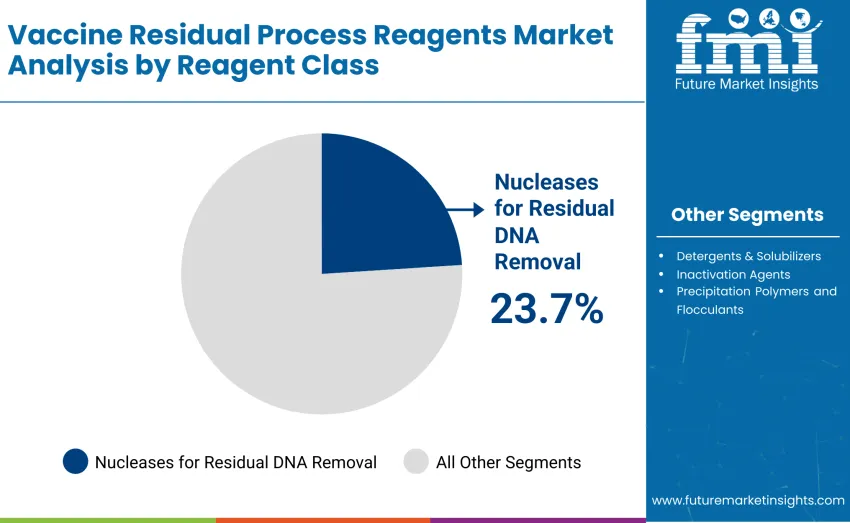
The nucleases for residual DNA removal segment represents the leading force, capturing approximately 23.7% of total revenue in 2025. This segment includes high-purity enzymatic reagents used to degrade residual host-cell DNA during vaccine production, ensuring regulatory compliance and product safety. Leadership stems from essential role in maintaining vaccine quality, reducing contaminants, and supporting high-volume manufacturing workflows where process consistency and regulatory adherence are critical for successful commercialization.
The detergents and solubilizers segment holds 19.6% of share, supporting protein extraction, solubilization, and formulation processes. Inactivation agents account for 16.4%, aiding viral inactivation and stabilization, while precipitation polymers and flocculants contribute 14.8%, helping remove impurities and clarify production streams. Extraction solvents and phase-transfer aids represent 9.7%, buffer additives and scavengers 8.9%, and LNP/wash & auxiliary process reagents 6.9%, providing support for lipid nanoparticle formulation and auxiliary purification steps across diverse vaccine manufacturing platforms.
Key advantages driving the nucleases segment include:
- High specificity in degrading residual DNA to meet regulatory thresholds
- Compatibility with multiple vaccine platforms, including mRNA, viral vector, and protein subunit vaccines
- Integration into automated purification workflows to improve consistency and throughput
By Application, Protein Subunit & Inactivated Vaccines Lead
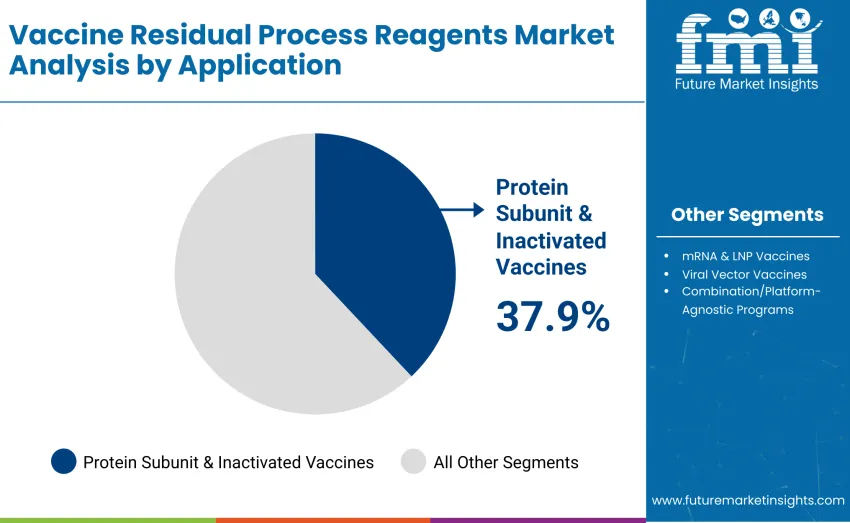
Protein subunit and inactivated vaccines dominate with 37.9% revenue share in 2025, reflecting their widespread adoption and stringent purification requirements these platforms demand. Viral vector vaccines account for 24.6%, and mRNA & LNP vaccines represent 22.8%. Live-attenuated and oral vaccines contribute 9.8%, while combination/platform-agnostic programs account for 4.9%, serving diverse therapeutic applications and research initiatives requiring specialized reagent solutions.
Key market dynamics supporting application preferences include:
- High global demand for protein subunit and inactivated vaccines, particularly in routine immunization programs
- Increasing adoption of viral vector and mRNA platforms requiring specialized reagents for residual DNA removal and process optimization
- Regulatory emphasis on residual impurity control, driving consistent use of high-quality reagents
What Are the Major Drivers, Restraints, and Emerging Trends Influencing the Vaccine Residual Process Reagents Market?
The vaccine residual process reagents market is driven by several key factors tied to growth of global vaccine production and increasing focus on product safety and regulatory compliance. Expansion of vaccination programs worldwide, including COVID-19, influenza, and other emerging infectious diseases, creates higher demand for high-quality reagents that ensure vaccine purity and efficacy. Scaling up of vaccine manufacturing capacities, including mRNA, viral vector, and protein subunit platforms, requires reliable residual process reagents to support automated workflows, high-throughput purification, and formulation processes across diverse production environments.
What Are the Major Restraints Affecting the Adoption of Vaccine Residual Process Reagents?
Stringent regulatory requirements from agencies such as the FDA, EMA, and WHO necessitate precise control over residual DNA, proteins, and other impurities, reinforcing the need for advanced enzymatic and chemical reagents in vaccine production. Restraints include high costs associated with premium reagents, which can pose adoption barriers for small-scale manufacturers or emerging regions. Technical complexity in integrating reagents into sophisticated purification and formulation workflows requires skilled personnel and strict adherence to protocols, which may limit efficiency if not properly managed.
What Emerging Trends Are Shaping the Vaccine Residual Process Reagents Market?
Key emerging trends in the reagent market include the increasing adoption of next-generation reagents designed for mRNA and lipid nanoparticle (LNP) vaccine platforms, which are crucial for the development of novel vaccines. There is also a focus on the creation of multi-functional reagent solutions that streamline the DNA removal, inactivation, and purification processes, enhancing overall efficiency. Additionally, there is growing investment in sustainable and scalable reagent manufacturing processes to meet the rising global demand, driven by the expansion of vaccine production and biotechnology research in response to global health challenges.
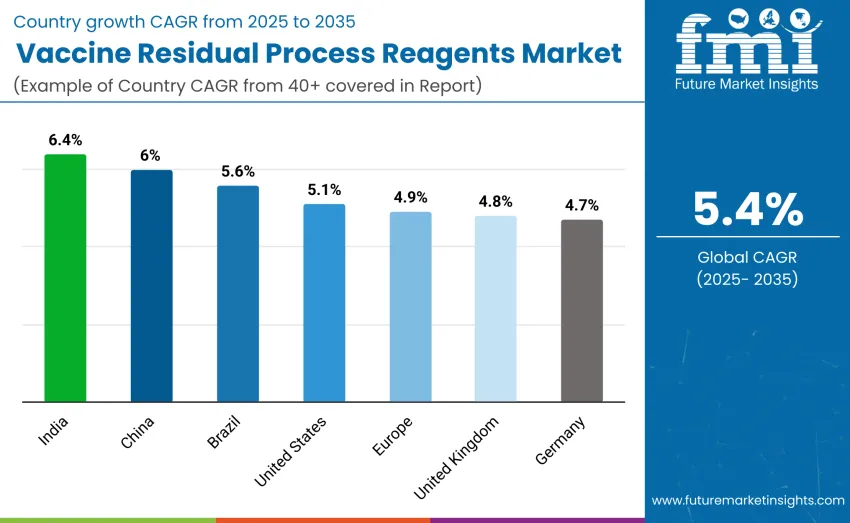
How Are Leading Countries Driving Growth in the Vaccine Residual Process Reagents Market?
| Country/Region | CAGR 2025 to 2035 (%) |
|---|---|
| United States | 5.1 |
| Brazil | 5.6 |
| China | 6.0 |
| India | 6.4 |
| Europe | 4.9 |
| Germany | 4.7 |
| United Kingdom | 4.8 |
The vaccine residual process reagents market is growing globally, with India leading at a 6.4% CAGR due to government programs promoting local vaccine production and adoption of advanced reagents. China follows with a 6.0% CAGR, driven by expanded vaccine manufacturing and government support for mRNA production. Germany grows at 4.7%, supported by strong manufacturing infrastructure and regulatory standards. Brazil shows a 5.6% CAGR, fueled by expanding vaccine production. The USA maintains a 5.1% growth rate, while the UK shows steady progress at 4.8%, driven by research and manufacturing advancements in vaccine production.
The report covers an in-depth analysis of 40+ countries top-performing countries are highlighted below.
How Are Government Policies and Manufacturing Modernization Accelerating Growth in China?
The vaccine residual process reagents market in China is projected to grow at a CAGR of 6.0% from 2025 to 2035. Growth is primarily driven by national initiatives to modernize vaccine manufacturing facilities, including large-scale adoption of nucleases, inactivation agents, and detergents to optimize DNA removal and virus inactivation processes. Industrial adoption has been facilitated by government-funded programs aimed at strengthening domestic biopharmaceutical infrastructure, with significant investment in mRNA and viral vector vaccine production hubs located in Beijing, Shanghai, Shenzhen, and Hangzhou.
Key market factors:
- Large-scale vaccine manufacturing modernization supported by government policies and industrial adoption of nucleases, inactivation agents, and detergents.
- Integration of automated purification systems and high-quality reagents in mRNA and viral vector vaccine production hubs.
- Policy incentives for domestic reagent production and technology transfer to strengthen biopharma infrastructure.
How Are Biopharma Clusters and Workforce Development Driving Adoption in India?
The vaccine residual process reagents market in India is forecasted to grow at a CAGR of 6.4% through 2035. Expansion is supported by enhanced local manufacturing capacity for mRNA, viral vector, and protein subunit vaccines, underpinned by government programs promoting pharmaceutical infrastructure development and public-private partnerships. Adoption of nucleases, precipitation polymers, and buffer additives has been accelerated by establishment of regional biopharma clusters in Bangalore, Hyderabad, Mumbai, and Pune.
- Expansion of regional biopharma clusters in Bangalore, Hyderabad, Mumbai, and Pune boosting adoption of nucleases, polymers, and buffer additives.
- Workforce skill development through training programs in residual DNA removal, purification, and inactivation processes.
- Government initiatives like Make in India and National Health Mission supporting infrastructure, quality certification, and accessibility to reagents.
How Are Engineering Excellence and Regulatory Standards Supporting Growth in Germany?
Germany's vaccine residual process reagents market is expected to grow at a CAGR of 4.7% through 2035. Growth is driven by engineering excellence in vaccine manufacturing facilities and rigorous regulatory standards under the European Medicines Agency. Adoption of detergents, solubilizers, and LNP/wash auxiliary reagents has been prioritized in production sites located in Berlin, Munich, Hamburg, and Frankfurt, enabling consistent viral vector and protein subunit vaccine quality. Sustainability initiatives, including solvent recycling and energy-efficient purification systems, have been integrated into manufacturing workflows.
Key development areas:
- Advanced R&D-driven innovation optimizing nucleases, inactivation agents, and extraction solvents for improved yield and safety.
- Strong regulatory oversight by the FDA ensuring product reliability, consistency, and compliance.
- Adoption of automated purification systems in commercial and contract development vaccine production hubs.
How Are Manufacturing Modernization and Public Health Initiatives Driving Growth in Brazil?
The vaccine residual process reagents market in Brazil is forecasted to expand at a CAGR of 5.6% from 2025 to 2035. Growth is fueled by modernization of local vaccine production facilities and increasing demand for domestic immunization programs. Adoption of precipitation polymers, inactivation agents, and buffer additives has been supported by public health initiatives and partnerships with international reagent suppliers to ensure high-quality materials are available for mRNA, viral vector, and protein subunit vaccines.
Leading market segments:
- Engineering excellence in vaccine manufacturing facilities ensuring precision and reproducibility in reagent application.
- Adoption of detergents, solubilizers, and LNP/wash auxiliary reagents for viral vector and protein subunit vaccines.
- Integration of sustainable practices including solvent recycling and energy-efficient purification systems.
How Are Advanced R&D and Regulatory Oversight Driving Adoption in the United States?
The USA vaccine residual process reagents market is projected to register a CAGR of 5.1% through 2035. Growth is supported by advanced adoption of automated purification systems and high-performance reagents in both commercial and contract development organizations. Extensive research investment has facilitated innovation in nucleases, inactivation agents, and extraction solvents, optimizing yield, safety, and regulatory compliance. Adoption is concentrated in key vaccine manufacturing hubs in California, Washington, Texas, and New York.
Key market characteristics:
- Expansion of vaccine manufacturing capacity and modernization of local production facilities.
- Cost-efficiency initiatives in process reagent adoption improving operational performance.
- Workforce training programs for biopharma personnel enhancing process knowledge and efficiency.
How Are Advanced Production Facilities and Public Sector Support Enhancing Growth in the United Kingdom?
The vaccine residual process reagents market in the UK is projected to grow at a CAGR of 4.8% through 2035. Expansion is supported by advanced vaccine production infrastructure concentrated in London, Manchester, Bristol, and Edinburgh. High-quality nucleases, detergents, and extraction solvents have been integrated into purification and inactivation workflows to enhance yield and consistency in mRNA and viral vector vaccine production. Public sector support and government-funded research initiatives have accelerated adoption of process reagents.
Market development factors:
- Advanced vaccine production infrastructure supporting integration of high-purity nucleases, detergents, and solvents.
- Government funding and research initiatives accelerating adoption of residual process reagents.
- Collaboration with biotech startups and reagent suppliers promoting technology transfer and innovation.
How Are Creative Economy Investments Advancing Expansion Across Europe?
The vaccine residual process reagents market in Europe is projected to grow from USD 254.8 million in 2025 to USD 408.3 million by 2035, registering a CAGR of 4.9% over the forecast period. Germany is expected to maintain leadership with a 28.5% share in 2025, slightly declining to 27.8% by 2035, supported by advanced vaccine manufacturing infrastructure, robust regulatory compliance under the EMA, and major production hubs in Berlin, Munich, and Hamburg.
France follows with a 22.3% share in 2025, increasing to 22.6% by 2035, driven by strong biotechnology clusters and high-quality reagent adoption in Paris and Lyon. The United Kingdom holds a 19.5% share in 2025, reaching 19.8% by 2035 through expansion of modern vaccine production facilities and public-private collaborations. Italy, Spain, the Netherlands, and the Rest of Europe contribute steadily through localized infrastructure upgrades, workforce training programs, and adoption of advanced process reagents in regional biopharma clusters.
How Competitive Is the Global Vaccine Residual Process Reagents Market and What Defines Its Structure?
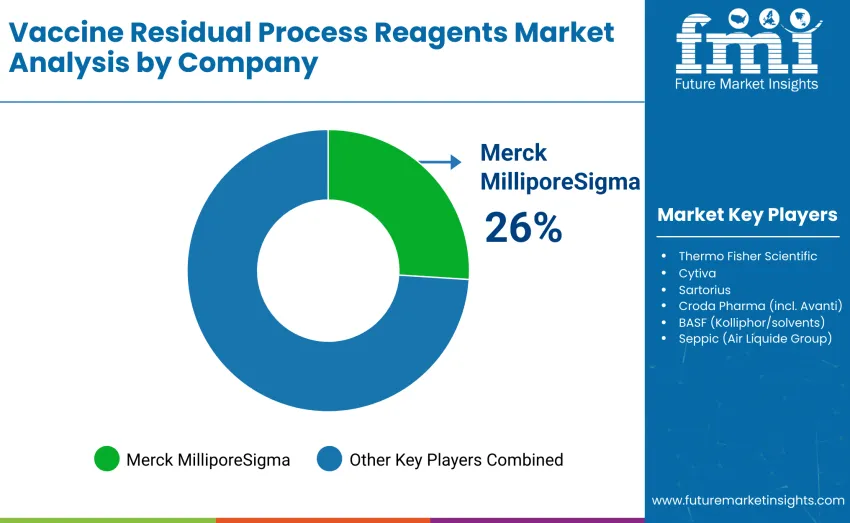
The global vaccine residual process reagents market is moderately concentrated, with 12–15 key players driving most commercial activity. The top three companies, Merck MilliporeSigma (including Benzonase), Thermo Fisher Scientific, and Cytiva, collectively hold roughly 55–60% of the global market share, with Merck MilliporeSigma holding a 26% share. These leaders benefit from established brand reputations, comprehensive product portfolios, and strong regulatory compliance records. Competition is primarily shaped by reagent purity, process compatibility, and scalability for mRNA, viral vector, and protein subunit vaccine production, rather than pricing alone.
Leaders maintain competitive advantages through extensive research investments, high-purity nucleases, inactivation agents, and solvents, as well as integration with automated purification and inactivation workflows. Mid-tier competitors like Sartorius, Croda Pharma (including Avanti), and BASF focus on specialized reagents such as surfactants, lipids, and formulation solvents, with regional penetration strategies. Emerging players such as ArcticZymes, Eastman, NOF Corporation, and Promega drive competition with niche offerings like SAN nucleases, PEG/lipids, and high-quality enzymes.
Competition is particularly fierce in cost-sensitive regions across Asia-Pacific, Latin America, and emerging economies, where pricing pressure drives adoption of alternative suppliers. Innovation in specialized reagents, sustainable manufacturing processes, and workflow integration continues to define competitive positioning.
Key Players in the Vaccine Residual Process Reagents Market
- Merck MilliporeSigma (incl. Benzonase)
- Thermo Fisher Scientific
- Cytiva
- Sartorius
- Croda Pharma (incl. Avanti)
- BASF (Kolliphor/solvents)
- Seppic (Air Liquide Group)
- NOF Corporation (PEG & lipids)
- ArcticZymes (SAN nucleases)
- Eastman (BPL/formulation solvents)
- Promega (nucleases & enzymes)
Scope of the Report
| Items | Values |
|---|---|
| Quantitative Units | USD 972.6 million |
| Reagent Class | Nucleases for Residual DNA Removal, Detergents & Solubilizers, Inactivation Agents, Precipitation Polymers & Flocculants, Extraction Solvents & Phase-Transfer Aids, Buffer Additives & Scavengers, LNP/Wash & Auxiliary Process Reagents |
| Application | mRNA & LNP Vaccines, Viral Vector Vaccines, Protein Subunit & Inactivated Vaccines, Live-Attenuated & Oral Vaccines, Combination/Platform-Agnostic Programs |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Country Covered | USA, Brazil, China, India, Europe, Germany, France, UK |
| Key Companies Profiled | Merck MilliporeSigma (incl. Benzonase ), Thermo Fisher Scientific, Cytiva , Sartorius, Croda Pharma (incl. Avanti), BASF ( Kolliphor /solvents), Seppic (Air Liquide Group), NOF Corporation (PEG & lipids), ArcticZymes (SAN nucleases), Eastman (BPL/formulation solvents), Promega (nucleases & enzymes), Others |
| Additional Attributes | Dollar sales by reagent type and application, regional trends across Asia Pacific, Europe, and North America, competitive landscape of key manufacturers, technical specifications, workflow integration, innovations in reagent chemistry and detection methods, and development of specialized configurations with enhanced sensitivity, specificity, and regulatory compliance |
Vaccine Residual Process Reagents Market by Segments
By Reagent Class:
- Nucleases for Residual DNA Removal
- Detergents & Solubilizers
- Inactivation Agents
- Precipitation Polymers & Flocculants
- Extraction Solvents & Phase-Transfer Aids
- Buffer Additives & Scavengers
- LNP/Wash & Auxiliary Process Reagents
By Application:
- mRNA & LNP Vaccines
- Viral Vector Vaccines
- Protein Subunit & Inactivated Vaccines
- Live-Attenuated & Oral Vaccines
- Combination / Platform-Agnostic Programs
By Region:
- Asia Pacific
- China
- India
- Japan
- South Korea
- Vietnam
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Netherlands
- Nordic
- BENELUX
- Rest of Europe
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Argentina
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Other African Union
- Rest of Middle East & Africa
Frequently Asked Questions
How big is the vaccine residual process reagents market in 2025?
The global vaccine residual process reagents market is valued at USD 972.6 million in 2025.
What will be the size of the vaccine residual process reagents market in 2035?
The market is projected to reach USD 1,639.8 million by 2035.
How fast will the vaccine residual process reagents market grow between 2025 and 2035?
The market will grow at a CAGR of 5.4% from 2025 to 2035.
Which reagent class leads the vaccine residual process reagents market in 2025?
Nucleases for residual DNA removal lead the market with a 23.7% share in 2025.
Who are the major players in the vaccine residual process reagents market?
Key players include Merck MilliporeSigma, Thermo Fisher Scientific, Cytiva, Sartorius, Croda Pharma, BASF, Seppic, NOF Corporation, ArcticZymes, Eastman, and Promega.
Table of Content
- Executive Summary
- Regional Level Market Outlook
- Demand-side Trends
- Supply-side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Assesment
- Profit Margin Analysis
- Value Chain Analysis
- Raw Material Suppliers
- Reagent Manufacturers
- Mid-Level Participants
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- By Key Regions
- By Key Countries
- Regional Parent Market Outlook
- Technological Innovations
- White-Space Opportunity Assessment
- Market Dynamics
- Regional Level Market Analysis 2020 to 2024 and Forecast, 2025 to 2035
- Historical Market Size USD Million Analysis, 2020 to 2024
- Current and Future Market Size USD Million Projections, 2025 to 2035
- Y-o-Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Regional Level Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Reagent Class
- Introduction / Key Findings
- Historical Market Size USD Million Analysis By Reagent Class, 2020 to 2024
- Current and Future Market Size USD Million Analysis and Forecast By Reagent Class, 2025 to 2035
- Nucleases for Residual DNA Removal
- Detergents & Solubilizers (e.g., polysorbates, zwitterionics)
- Inactivation Agents (BPL, Formaldehyde)
- Precipitation Polymers & Flocculants (PEG, PAEs)
- Extraction Solvents & Phase-Transfer Aids
- Buffer Additives & Scavengers (antioxidants, quenchers)
- LNP/Wash & Auxiliary Process Reagents
- Y-o-Y Growth Trend Analysis By Reagent Class, 2020 to 2024
- Absolute $ Opportunity Analysis By Reagent Class, 2025 to 2035
- Regional Level Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Application
- Introduction / Key Findings
- Historical Market Size USD Million Analysis By Application, 2020 to 2024
- Current and Future Market Size USD Million Analysis and Forecast By Application, 2025 to 2035
- mRNA & LNP Vaccines
- Viral Vector Vaccines
- Protein Subunit & Inactivated Vaccines
- Live-Attenuated & Oral Vaccines
- Combination / Platform-Agnostic Programs
- Y-o-Y Growth Trend Analysis By Application, 2020 to 2024
- Absolute $ Opportunity Analysis By Application, 2025 to 2035
- Regional Level Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By End User
- Introduction / Key Findings
- Historical Market Size USD Million Analysis By End User, 2020 to 2024
- Current and Future Market Size USD Million Analysis and Forecast By End User, 2025 to 2035
- Vaccine Manufacturers (Innovators & Generics)
- CDMOs & Fill-Finish Providers
- Public Sector Institutes
- Academic & NGO Labs
- Y-o-Y Growth Trend Analysis By End User, 2020 to 2024
- Absolute $ Opportunity Analysis By End User, 2025 to 2035
- Regional Level Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Region
- Introduction
- Historical Market Size USD Million Analysis By Region, 2020 to 2024
- Current Market Size USD Million Analysis and Forecast By Region, 2025 to 2035
- North America
- Latin America
- East Asia
- South Asia
- Western Europe
- Eastern Europe
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size USD Million Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size USD Million Forecast By Market Taxonomy, 2025 to 2035
- By Country
- USA
- Canada
- Mexico
- By Reagent Class
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Reagent Class
- By Application
- By End User
- Key Takeaways
- Latin America Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size USD Million Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size USD Million Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Brazil
- Argentina
- Rest of Latin America
- By Reagent Class
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Reagent Class
- By Application
- By End User
- Key Takeaways
- East Asia Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size USD Million Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size USD Million Forecast By Market Taxonomy, 2025 to 2035
- By Country
- China
- Japan
- South Korea
- By Reagent Class
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Reagent Class
- By Application
- By End User
- Key Takeaways
- South Asia Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size USD Million Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size USD Million Forecast By Market Taxonomy, 2025 to 2035
- By Country
- India
- ASEAN Countries
- Australia & New Zealand
- Rest of South Asia
- By Reagent Class
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Reagent Class
- By Application
- By End User
- Key Takeaways
- Western Europe Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size USD Million Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size USD Million Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Germany
- UK
- France
- Italy
- Spain
- BENELUX
- Nordic Countries
- Rest of Western Europe
- By Reagent Class
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Reagent Class
- By Application
- By End User
- Key Takeaways
- Eastern Europe Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size USD Million Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size USD Million Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Russia
- Hungary
- Poland
- Rest of Eastern Europe
- By Reagent Class
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Reagent Class
- By Application
- By End User
- Key Takeaways
- Middle East & Africa Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size USD Million Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size USD Million Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Saudi Arabia
- Other GCC Countries
- Türkiye
- South Africa
- Rest of MEA
- By Reagent Class
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Reagent Class
- By Application
- By End User
- Key Takeaways
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Reagent Class
- By Application
- By End User
- Competition Analysis
- Competition Deep Dive
- Merck MilliporeSigma (incl. Benzonase)
- Overview
- Product Portfolio
- Key Financials
- Key Developments
- Global/Regional Footprint
- Strategy Overview
- Marketing Strategy
- SWOT
- Analyst Comment
- Thermo Fisher Scientific
- Overview
- Product Portfolio
- Key Financials
- Key Developments
- Global/Regional Footprint
- Strategy Overview
- Marketing Strategy
- SWOT
- Analyst Comment
- Cytiva
- Overview
- Product Portfolio
- Key Financials
- Key Developments
- Global/Regional Footprint
- Strategy Overview
- Marketing Strategy
- SWOT
- Analyst Comment
- Sartorius
- Overview
- Product Portfolio
- Key Financials
- Key Developments
- Global/Regional Footprint
- Strategy Overview
- Marketing Strategy
- SWOT
- Analyst Comment
- Croda Pharma (incl. Avanti)
- Overview
- Product Portfolio
- Key Financials
- Key Developments
- Global/Regional Footprint
- Strategy Overview
- Marketing Strategy
- SWOT
- Analyst Comment
- BASF (Kolliphor/solvents)
- Overview
- Product Portfolio
- Key Financials
- Key Developments
- Global/Regional Footprint
- Strategy Overview
- Marketing Strategy
- SWOT
- Analyst Comment
- Seppic (Air Liquide Group)
- Overview
- Product Portfolio
- Key Financials
- Key Developments
- Global/Regional Footprint
- Strategy Overview
- Marketing Strategy
- SWOT
- Analyst Comment
- NOF Corporation (PEG & lipids)
- Overview
- Product Portfolio
- Key Financials
- Key Developments
- Global/Regional Footprint
- Strategy Overview
- Marketing Strategy
- SWOT
- Analyst Comment
- ArcticZymes (SAN nucleases)
- Overview
- Product Portfolio
- Key Financials
- Key Developments
- Global/Regional Footprint
- Strategy Overview
- Marketing Strategy
- SWOT
- Analyst Comment
- Eastman (BPL/formulation solvents)
- Overview
- Product Portfolio
- Key Financials
- Key Developments
- Global/Regional Footprint
- Strategy Overview
- Marketing Strategy
- SWOT
- Analyst Comment
- Promega (nucleases & enzymes)
- Overview
- Product Portfolio
- Key Financials
- Key Developments
- Global/Regional Footprint
- Strategy Overview
- Marketing Strategy
- SWOT
- Analyst Comment
- Merck MilliporeSigma (incl. Benzonase)
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: Global Market Size (USD Million), by Reagent Class, 2020-2035
- Table 2: Global Market Size (USD Million), by Nucleases for Residual DNA Removal, 2020-2035
- Table 3: Global Market Size (USD Million), by Detergents & Solubilizers (e.g., polysorbates, zwitterionics), 2020-2035
- Table 4: Global Market Size (USD Million), by Inactivation Agents (BPL, Formaldehyde), 2020-2035
- Table 5: Global Market Size (USD Million), by Precipitation Polymers & Flocculants (PEG, PAEs), 2020-2035
- Table 6: Global Market Size (USD Million), by Extraction Solvents & Phase-Transfer Aids, 2020-2035
- Table 7: Global Market Size (USD Million), by Buffer Additives & Scavengers (antioxidants, quenchers), 2020-2035
- Table 8: Global Market Size (USD Million), by LNP/Wash & Auxiliary Process Reagents, 2020-2035
- Table 9: Global Market Size (USD Million), By Application, 2020-2035
- Table 10: Global Market Size (USD Million), by mRNA & LNP Vaccines, 2020-2035
- Table 11: Global Market Size (USD Million), by Viral Vector Vaccines, 2020-2035
- Table 12: Global Market Size (USD Million), by Protein Subunit & Inactivated Vaccines, 2020-2035
- Table 13: Global Market Size (USD Million), by Live-Attenuated & Oral Vaccines, 2020-2035
- Table 14: Global Market Size (USD Million), by Combination / Platform-Agnostic Programs, 2020-2035
- Table 15: Global Market Size (USD Million), by End User, 2020-2035
- Table 16: Global Market Size (USD Million), by Vaccine Manufacturers (Innovators & Generics), 2020-2035
- Table 17: Global Market Size (USD Million), by CDMOs & Fill-Finish Providers, 2020-2035
- Table 18: Global Market Size (USD Million), by Public Sector Institutes, 2020-2035
- Table 19: Global Market Size (USD Million), by Academic & NGO Labs, 2020-2035
- Table 20: North America Market Size (USD Million), by Reagent Class, 2020-2035
- Table 21: North America Market Size (USD Million), by Nucleases for Residual DNA Removal, 2020-2035
- Table 22: North America Market Size (USD Million), by Detergents & Solubilizers (e.g., polysorbates, zwitterionics), 2020-2035
- Table 23: North America Market Size (USD Million), by Inactivation Agents (BPL, Formaldehyde), 2020-2035
- Table 24: North America Market Size (USD Million), by Precipitation Polymers & Flocculants (PEG, PAEs), 2020-2035
- Table 25: North America Market Size (USD Million), by Extraction Solvents & Phase-Transfer Aids, 2020-2035
- Table 26: North America Market Size (USD Million), by Buffer Additives & Scavengers (antioxidants, quenchers), 2020-2035
- Table 27: North America Market Size (USD Million), by LNP/Wash & Auxiliary Process Reagents, 2020-2035
- Table 28: North America Market Size (USD Million), By Application, 2020-2035
- Table 29: North America Market Size (USD Million), by mRNA & LNP Vaccines, 2020-2035
- Table 30: North America Market Size (USD Million), by Viral Vector Vaccines, 2020-2035
- Table 31: North America Market Size (USD Million), by Protein Subunit & Inactivated Vaccines, 2020-2035
- Table 32: North America Market Size (USD Million), by Live-Attenuated & Oral Vaccines, 2020-2035
- Table 33: North America Market Size (USD Million), by Combination / Platform-Agnostic Programs, 2020-2035
- Table 34: North America Market Size (USD Million), by End User, 2020-2035
- Table 35: North America Market Size (USD Million), by Vaccine Manufacturers (Innovators & Generics), 2020-2035
- Table 36: North America Market Size (USD Million), by CDMOs & Fill-Finish Providers, 2020-2035
- Table 37: North America Market Size (USD Million), by Public Sector Institutes, 2020-2035
- Table 38: North America Market Size (USD Million), by Academic & NGO Labs, 2020-2035
- Table 39: USA Market Size (USD Million), by Reagent Class, 2020-2035
- Table 40: USA Market Size (USD Million), By Application, 2020-2035
- Table 41: USA Market Size (USD Million), by End User, 2020-2035
- Table 42: Canada Market Size (USD Million), by Reagent Class, 2020-2035
- Table 43: Canada Market Size (USD Million), By Application, 2020-2035
- Table 44: Canada Market Size (USD Million), by End User, 2020-2035
- Table 45: Mexico Market Size (USD Million), by Reagent Class, 2020-2035
- Table 46: Mexico Market Size (USD Million), By Application, 2020-2035
- Table 47: Mexico Market Size (USD Million), by End User, 2020-2035
- Table 48: Latin America Market Size (USD Million), by Reagent Class, 2020-2035
- Table 49: Latin America Market Size (USD Million), by Nucleases for Residual DNA Removal, 2020-2035
- Table 50: Latin America Market Size (USD Million), by Detergents & Solubilizers (e.g., polysorbates, zwitterionics), 2020-2035
- Table 51: Latin America Market Size (USD Million), by Inactivation Agents (BPL, Formaldehyde), 2020-2035
- Table 52: Latin America Market Size (USD Million), by Precipitation Polymers & Flocculants (PEG, PAEs), 2020-2035
- Table 53: Latin America Market Size (USD Million), by Extraction Solvents & Phase-Transfer Aids, 2020-2035
- Table 54: Latin America Market Size (USD Million), by Buffer Additives & Scavengers (antioxidants, quenchers), 2020-2035
- Table 55: Latin America Market Size (USD Million), by LNP/Wash & Auxiliary Process Reagents, 2020-2035
- Table 56: Latin America Market Size (USD Million), By Application, 2020-2035
- Table 57: Latin America Market Size (USD Million), by mRNA & LNP Vaccines, 2020-2035
- Table 58: Latin America Market Size (USD Million), by Viral Vector Vaccines, 2020-2035
- Table 59: Latin America Market Size (USD Million), by Protein Subunit & Inactivated Vaccines, 2020-2035
- Table 60: Latin America Market Size (USD Million), by Live-Attenuated & Oral Vaccines, 2020-2035
- Table 61: Latin America Market Size (USD Million), by Combination / Platform-Agnostic Programs, 2020-2035
- Table 62: Latin America Market Size (USD Million), by End User, 2020-2035
- Table 63: Latin America Market Size (USD Million), by Vaccine Manufacturers (Innovators & Generics), 2020-2035
- Table 64: Latin America Market Size (USD Million), by CDMOs & Fill-Finish Providers, 2020-2035
- Table 65: Latin America Market Size (USD Million), by Public Sector Institutes, 2020-2035
- Table 66: Latin America Market Size (USD Million), by Academic & NGO Labs, 2020-2035
- Table 67: Brazil Market Size (USD Million), by Reagent Class, 2020-2035
- Table 68: Brazil Market Size (USD Million), By Application, 2020-2035
- Table 69: Brazil Market Size (USD Million), by End User, 2020-2035
- Table 70: Argentina Market Size (USD Million), by Reagent Class, 2020-2035
- Table 71: Argentina Market Size (USD Million), By Application, 2020-2035
- Table 72: Argentina Market Size (USD Million), by End User, 2020-2035
- Table 73: Rest of Latin America Market Size (USD Million), by Reagent Class, 2020-2035
- Table 74: Rest of Latin America Market Size (USD Million), By Application, 2020-2035
- Table 75: Rest of Latin America Market Size (USD Million), by End User, 2020-2035
- Table 76: East Asia Market Size (USD Million), by Reagent Class, 2020-2035
- Table 77: East Asia Market Size (USD Million), by Nucleases for Residual DNA Removal, 2020-2035
- Table 78: East Asia Market Size (USD Million), by Detergents & Solubilizers (e.g., polysorbates, zwitterionics), 2020-2035
- Table 79: East Asia Market Size (USD Million), by Inactivation Agents (BPL, Formaldehyde), 2020-2035
- Table 80: East Asia Market Size (USD Million), by Precipitation Polymers & Flocculants (PEG, PAEs), 2020-2035
- Table 81: East Asia Market Size (USD Million), by Extraction Solvents & Phase-Transfer Aids, 2020-2035
- Table 82: East Asia Market Size (USD Million), by Buffer Additives & Scavengers (antioxidants, quenchers), 2020-2035
- Table 83: East Asia Market Size (USD Million), by LNP/Wash & Auxiliary Process Reagents, 2020-2035
- Table 84: East Asia Market Size (USD Million), By Application, 2020-2035
- Table 85: East Asia Market Size (USD Million), by mRNA & LNP Vaccines, 2020-2035
- Table 86: East Asia Market Size (USD Million), by Viral Vector Vaccines, 2020-2035
- Table 87: East Asia Market Size (USD Million), by Protein Subunit & Inactivated Vaccines, 2020-2035
- Table 88: East Asia Market Size (USD Million), by Live-Attenuated & Oral Vaccines, 2020-2035
- Table 89: East Asia Market Size (USD Million), by Combination / Platform-Agnostic Programs, 2020-2035
- Table 90: East Asia Market Size (USD Million), by End User, 2020-2035
- Table 91: East Asia Market Size (USD Million), by Vaccine Manufacturers (Innovators & Generics), 2020-2035
- Table 92: East Asia Market Size (USD Million), by CDMOs & Fill-Finish Providers, 2020-2035
- Table 93: East Asia Market Size (USD Million), by Public Sector Institutes, 2020-2035
- Table 94: East Asia Market Size (USD Million), by Academic & NGO Labs, 2020-2035
- Table 95: China Market Size (USD Million), by Reagent Class, 2020-2035
- Table 96: China Market Size (USD Million), By Application, 2020-2035
- Table 97: China Market Size (USD Million), by End User, 2020-2035
- Table 98: Japan Market Size (USD Million), by Reagent Class, 2020-2035
- Table 99: Japan Market Size (USD Million), By Application, 2020-2035
- Table 100: Japan Market Size (USD Million), by End User, 2020-2035
- Table 101: South Korea Market Size (USD Million), by Reagent Class, 2020-2035
- Table 102: South Korea Market Size (USD Million), By Application, 2020-2035
- Table 103: South Korea Market Size (USD Million), by End User, 2020-2035
- Table 104: South Asia Market Size (USD Million), by Reagent Class, 2020-2035
- Table 105: South Asia Market Size (USD Million), by Nucleases for Residual DNA Removal, 2020-2035
- Table 106: South Asia Market Size (USD Million), by Detergents & Solubilizers (e.g., polysorbates, zwitterionics), 2020-2035
- Table 107: South Asia Market Size (USD Million), by Inactivation Agents (BPL, Formaldehyde), 2020-2035
- Table 108: South Asia Market Size (USD Million), by Precipitation Polymers & Flocculants (PEG, PAEs), 2020-2035
- Table 109: South Asia Market Size (USD Million), by Extraction Solvents & Phase-Transfer Aids, 2020-2035
- Table 110: South Asia Market Size (USD Million), by Buffer Additives & Scavengers (antioxidants, quenchers), 2020-2035
- Table 111: South Asia Market Size (USD Million), by LNP/Wash & Auxiliary Process Reagents, 2020-2035
- Table 112: South Asia Market Size (USD Million), By Application, 2020-2035
- Table 113: South Asia Market Size (USD Million), by mRNA & LNP Vaccines, 2020-2035
- Table 114: South Asia Market Size (USD Million), by Viral Vector Vaccines, 2020-2035
- Table 115: South Asia Market Size (USD Million), by Protein Subunit & Inactivated Vaccines, 2020-2035
- Table 116: South Asia Market Size (USD Million), by Live-Attenuated & Oral Vaccines, 2020-2035
- Table 117: South Asia Market Size (USD Million), by Combination / Platform-Agnostic Programs, 2020-2035
- Table 118: South Asia Market Size (USD Million), by End User, 2020-2035
- Table 119: South Asia Market Size (USD Million), by Vaccine Manufacturers (Innovators & Generics), 2020-2035
- Table 120: South Asia Market Size (USD Million), by CDMOs & Fill-Finish Providers, 2020-2035
- Table 121: South Asia Market Size (USD Million), by Public Sector Institutes, 2020-2035
- Table 122: South Asia Market Size (USD Million), by Academic & NGO Labs, 2020-2035
- Table 123: India Market Size (USD Million), by Reagent Class, 2020-2035
- Table 124: India Market Size (USD Million), By Application, 2020-2035
- Table 125: India Market Size (USD Million), by End User, 2020-2035
- Table 126: ASEAN Countries Market Size (USD Million), by Reagent Class, 2020-2035
- Table 127: ASEAN Countries Market Size (USD Million), By Application, 2020-2035
- Table 128: ASEAN Countries Market Size (USD Million), by End User, 2020-2035
- Table 129: Australia & New Zealand Market Size (USD Million), by Reagent Class, 2020-2035
- Table 130: Australia & New Zealand Market Size (USD Million), By Application, 2020-2035
- Table 131: Australia & New Zealand Market Size (USD Million), by End User, 2020-2035
- Table 132: Rest of South Asia Market Size (USD Million), by Reagent Class, 2020-2035
- Table 133: Rest of South Asia Market Size (USD Million), By Application, 2020-2035
- Table 134: Rest of South Asia Market Size (USD Million), by End User, 2020-2035
- Table 135: Western Europe Market Size (USD Million), by Reagent Class, 2020-2035
- Table 136: Western Europe Market Size (USD Million), by Nucleases for Residual DNA Removal, 2020-2035
- Table 137: Western Europe Market Size (USD Million), by Detergents & Solubilizers (e.g., polysorbates, zwitterionics), 2020-2035
- Table 138: Western Europe Market Size (USD Million), by Inactivation Agents (BPL, Formaldehyde), 2020-2035
- Table 139: Western Europe Market Size (USD Million), by Precipitation Polymers & Flocculants (PEG, PAEs), 2020-2035
- Table 140: Western Europe Market Size (USD Million), by Extraction Solvents & Phase-Transfer Aids, 2020-2035
- Table 141: Western Europe Market Size (USD Million), by Buffer Additives & Scavengers (antioxidants, quenchers), 2020-2035
- Table 142: Western Europe Market Size (USD Million), by LNP/Wash & Auxiliary Process Reagents, 2020-2035
- Table 143: Western Europe Market Size (USD Million), By Application, 2020-2035
- Table 144: Western Europe Market Size (USD Million), by mRNA & LNP Vaccines, 2020-2035
- Table 145: Western Europe Market Size (USD Million), by Viral Vector Vaccines, 2020-2035
- Table 146: Western Europe Market Size (USD Million), by Protein Subunit & Inactivated Vaccines, 2020-2035
- Table 147: Western Europe Market Size (USD Million), by Live-Attenuated & Oral Vaccines, 2020-2035
- Table 148: Western Europe Market Size (USD Million), by Combination / Platform-Agnostic Programs, 2020-2035
- Table 149: Western Europe Market Size (USD Million), by End User, 2020-2035
- Table 150: Western Europe Market Size (USD Million), by Vaccine Manufacturers (Innovators & Generics), 2020-2035
- Table 151: Western Europe Market Size (USD Million), by CDMOs & Fill-Finish Providers, 2020-2035
- Table 152: Western Europe Market Size (USD Million), by Public Sector Institutes, 2020-2035
- Table 153: Western Europe Market Size (USD Million), by Academic & NGO Labs, 2020-2035
- Table 154: Germany Market Size (USD Million), by Reagent Class, 2020-2035
- Table 155: Germany Market Size (USD Million), By Application, 2020-2035
- Table 156: Germany Market Size (USD Million), by End User, 2020-2035
- Table 157: UK Market Size (USD Million), by Reagent Class, 2020-2035
- Table 158: UK Market Size (USD Million), By Application, 2020-2035
- Table 159: UK Market Size (USD Million), by End User, 2020-2035
- Table 160: France Market Size (USD Million), by Reagent Class, 2020-2035
- Table 161: France Market Size (USD Million), By Application, 2020-2035
- Table 162: France Market Size (USD Million), by End User, 2020-2035
- Table 163: Italy Market Size (USD Million), by Reagent Class, 2020-2035
- Table 164: Italy Market Size (USD Million), By Application, 2020-2035
- Table 165: Italy Market Size (USD Million), by End User, 2020-2035
- Table 166: Spain Market Size (USD Million), by Reagent Class, 2020-2035
- Table 167: Spain Market Size (USD Million), By Application, 2020-2035
- Table 168: Spain Market Size (USD Million), by End User, 2020-2035
- Table 169: BENELUX Market Size (USD Million), by Reagent Class, 2020-2035
- Table 170: BENELUX Market Size (USD Million), By Application, 2020-2035
- Table 171: BENELUX Market Size (USD Million), by End User, 2020-2035
- Table 172: Nordic Countries Market Size (USD Million), by Reagent Class, 2020-2035
- Table 173: Nordic Countries Market Size (USD Million), By Application, 2020-2035
- Table 174: Nordic Countries Market Size (USD Million), by End User, 2020-2035
- Table 175: Rest of Western Europe Market Size (USD Million), by Reagent Class, 2020-2035
- Table 176: Rest of Western Europe Market Size (USD Million), By Application, 2020-2035
- Table 177: Rest of Western Europe Market Size (USD Million), by End User, 2020-2035
- Table 178: Eastern Europe Market Size (USD Million), by Reagent Class, 2020-2035
- Table 179: Eastern Europe Market Size (USD Million), by Nucleases for Residual DNA Removal, 2020-2035
- Table 180: Eastern Europe Market Size (USD Million), by Detergents & Solubilizers (e.g., polysorbates, zwitterionics), 2020-2035
- Table 181: Eastern Europe Market Size (USD Million), by Inactivation Agents (BPL, Formaldehyde), 2020-2035
- Table 182: Eastern Europe Market Size (USD Million), by Precipitation Polymers & Flocculants (PEG, PAEs), 2020-2035
- Table 183: Eastern Europe Market Size (USD Million), by Extraction Solvents & Phase-Transfer Aids, 2020-2035
- Table 184: Eastern Europe Market Size (USD Million), by Buffer Additives & Scavengers (antioxidants, quenchers), 2020-2035
- Table 185: Eastern Europe Market Size (USD Million), by LNP/Wash & Auxiliary Process Reagents, 2020-2035
- Table 186: Eastern Europe Market Size (USD Million), By Application, 2020-2035
- Table 187: Eastern Europe Market Size (USD Million), by mRNA & LNP Vaccines, 2020-2035
- Table 188: Eastern Europe Market Size (USD Million), by Viral Vector Vaccines, 2020-2035
- Table 189: Eastern Europe Market Size (USD Million), by Protein Subunit & Inactivated Vaccines, 2020-2035
- Table 190: Eastern Europe Market Size (USD Million), by Live-Attenuated & Oral Vaccines, 2020-2035
- Table 191: Eastern Europe Market Size (USD Million), by Combination / Platform-Agnostic Programs, 2020-2035
- Table 192: Eastern Europe Market Size (USD Million), by End User, 2020-2035
- Table 193: Eastern Europe Market Size (USD Million), by Vaccine Manufacturers (Innovators & Generics), 2020-2035
- Table 194: Eastern Europe Market Size (USD Million), by CDMOs & Fill-Finish Providers, 2020-2035
- Table 195: Eastern Europe Market Size (USD Million), by Public Sector Institutes, 2020-2035
- Table 196: Eastern Europe Market Size (USD Million), by Academic & NGO Labs, 2020-2035
- Table 197: Russia Market Size (USD Million), by Reagent Class, 2020-2035
- Table 198: Russia Market Size (USD Million), By Application, 2020-2035
- Table 199: Russia Market Size (USD Million), by End User, 2020-2035
- Table 200: Hungary Market Size (USD Million), by Reagent Class, 2020-2035
- Table 201: Hungary Market Size (USD Million), By Application, 2020-2035
- Table 202: Hungary Market Size (USD Million), by End User, 2020-2035
- Table 203: Poland Market Size (USD Million), by Reagent Class, 2020-2035
- Table 204: Poland Market Size (USD Million), By Application, 2020-2035
- Table 205: Poland Market Size (USD Million), by End User, 2020-2035
- Table 206: Rest of Eastern Europe Market Size (USD Million), by Reagent Class, 2020-2035
- Table 207: Rest of Eastern Europe Market Size (USD Million), By Application, 2020-2035
- Table 208: Rest of Eastern Europe Market Size (USD Million), by End User, 2020-2035
- Table 209: Middle East & Africa Market Size (USD Million), by Reagent Class, 2020-2035
- Table 210: Middle East & Africa Market Size (USD Million), by Nucleases for Residual DNA Removal, 2020-2035
- Table 211: Middle East & Africa Market Size (USD Million), by Detergents & Solubilizers (e.g., polysorbates, zwitterionics), 2020-2035
- Table 212: Middle East & Africa Market Size (USD Million), by Inactivation Agents (BPL, Formaldehyde), 2020-2035
- Table 213: Middle East & Africa Market Size (USD Million), by Precipitation Polymers & Flocculants (PEG, PAEs), 2020-2035
- Table 214: Middle East & Africa Market Size (USD Million), by Extraction Solvents & Phase-Transfer Aids, 2020-2035
- Table 215: Middle East & Africa Market Size (USD Million), by Buffer Additives & Scavengers (antioxidants, quenchers), 2020-2035
- Table 216: Middle East & Africa Market Size (USD Million), by LNP/Wash & Auxiliary Process Reagents, 2020-2035
- Table 217: Middle East & Africa Market Size (USD Million), By Application, 2020-2035
- Table 218: Middle East & Africa Market Size (USD Million), by mRNA & LNP Vaccines, 2020-2035
- Table 219: Middle East & Africa Market Size (USD Million), by Viral Vector Vaccines, 2020-2035
- Table 220: Middle East & Africa Market Size (USD Million), by Protein Subunit & Inactivated Vaccines, 2020-2035
- Table 221: Middle East & Africa Market Size (USD Million), by Live-Attenuated & Oral Vaccines, 2020-2035
- Table 222: Middle East & Africa Market Size (USD Million), by Combination / Platform-Agnostic Programs, 2020-2035
- Table 223: Middle East & Africa Market Size (USD Million), by End User, 2020-2035
- Table 224: Middle East & Africa Market Size (USD Million), by Vaccine Manufacturers (Innovators & Generics), 2020-2035
- Table 225: Middle East & Africa Market Size (USD Million), by CDMOs & Fill-Finish Providers, 2020-2035
- Table 226: Middle East & Africa Market Size (USD Million), by Public Sector Institutes, 2020-2035
- Table 227: Middle East & Africa Market Size (USD Million), by Academic & NGO Labs, 2020-2035
- Table 228: Saudi Arabia Market Size (USD Million), by Reagent Class, 2020-2035
- Table 229: Saudi Arabia Market Size (USD Million), By Application, 2020-2035
- Table 230: Saudi Arabia Market Size (USD Million), by End User, 2020-2035
- Table 231: Other GCC Countries Market Size (USD Million), by Reagent Class, 2020-2035
- Table 232: Other GCC Countries Market Size (USD Million), By Application, 2020-2035
- Table 233: Other GCC Countries Market Size (USD Million), by End User, 2020-2035
- Table 234: Türkiye Market Size (USD Million), by Reagent Class, 2020-2035
- Table 235: Türkiye Market Size (USD Million), By Application, 2020-2035
- Table 236: Türkiye Market Size (USD Million), by End User, 2020-2035
- Table 237: South Africa Market Size (USD Million), by Reagent Class, 2020-2035
- Table 238: South Africa Market Size (USD Million), By Application, 2020-2035
- Table 239: South Africa Market Size (USD Million), by End User, 2020-2035
- Table 240: Rest of MEA Market Size (USD Million), by Reagent Class, 2020-2035
- Table 241: Rest of MEA Market Size (USD Million), By Application, 2020-2035
- Table 242: Rest of MEA Market Size (USD Million), by End User, 2020-2035
List of Figures
- Figure 1: Global Market (USD Million) Forecast, by Reagent Class, 2020-2035
- Figure 2: Global Market (USD Million) Forecast, by Nucleases for Residual DNA Removal, 2020-2035
- Figure 3: Global Market (USD Million) Forecast, by Detergents & Solubilizers (e.g., polysorbates, zwitterionics), 2020-2035
- Figure 4: Global Market (USD Million) Forecast, by Inactivation Agents (BPL, Formaldehyde), 2020-2035
- Figure 5: Global Market (USD Million) Forecast, by Precipitation Polymers & Flocculants (PEG, PAEs), 2020-2035
- Figure 6: Global Market (USD Million) Forecast, by Extraction Solvents & Phase-Transfer Aids, 2020-2035
- Figure 7: Global Market (USD Million) Forecast, by Buffer Additives & Scavengers (antioxidants, quenchers), 2020-2035
- Figure 8: Global Market (USD Million) Forecast, by LNP/Wash & Auxiliary Process Reagents, 2020-2035
- Figure 9: Global Market (USD Million) Forecast, By Application, 2020-2035
- Figure 10: Global Market (USD Million) Forecast, by mRNA & LNP Vaccines, 2020-2035
- Figure 11: Global Market (USD Million) Forecast, by Viral Vector Vaccines, 2020-2035
- Figure 12: Global Market (USD Million) Forecast, by Protein Subunit & Inactivated Vaccines, 2020-2035
- Figure 13: Global Market (USD Million) Forecast, by Live-Attenuated & Oral Vaccines, 2020-2035
- Figure 14: Global Market (USD Million) Forecast, by Combination / Platform-Agnostic Programs, 2020-2035
- Figure 15: Global Market (USD Million) Forecast, by End User, 2020-2035
- Figure 16: Global Market (USD Million) Forecast, by Vaccine Manufacturers (Innovators & Generics), 2020-2035
- Figure 17: Global Market (USD Million) Forecast, by CDMOs & Fill-Finish Providers, 2020-2035
- Figure 18: Global Market (USD Million) Forecast, by Public Sector Institutes, 2020-2035
- Figure 19: Global Market (USD Million) Forecast, by Academic & NGO Labs, 2020-2035
- Figure 20: North America Market (USD Million) Forecast, by Reagent Class, 2020-2035
- Figure 21: North America Market (USD Million) Forecast, by Nucleases for Residual DNA Removal, 2020-2035
- Figure 22: North America Market (USD Million) Forecast, by Detergents & Solubilizers (e.g., polysorbates, zwitterionics), 2020-2035
- Figure 23: North America Market (USD Million) Forecast, by Inactivation Agents (BPL, Formaldehyde), 2020-2035
- Figure 24: North America Market (USD Million) Forecast, by Precipitation Polymers & Flocculants (PEG, PAEs), 2020-2035
- Figure 25: North America Market (USD Million) Forecast, by Extraction Solvents & Phase-Transfer Aids, 2020-2035
- Figure 26: North America Market (USD Million) Forecast, by Buffer Additives & Scavengers (antioxidants, quenchers), 2020-2035
- Figure 27: North America Market (USD Million) Forecast, by LNP/Wash & Auxiliary Process Reagents, 2020-2035
- Figure 28: North America Market (USD Million) Forecast, By Application, 2020-2035
- Figure 29: North America Market (USD Million) Forecast, by mRNA & LNP Vaccines, 2020-2035
- Figure 30: North America Market (USD Million) Forecast, by Viral Vector Vaccines, 2020-2035
- Figure 31: North America Market (USD Million) Forecast, by Protein Subunit & Inactivated Vaccines, 2020-2035
- Figure 32: North America Market (USD Million) Forecast, by Live-Attenuated & Oral Vaccines, 2020-2035
- Figure 33: North America Market (USD Million) Forecast, by Combination / Platform-Agnostic Programs, 2020-2035
- Figure 34: North America Market (USD Million) Forecast, by End User, 2020-2035
- Figure 35: North America Market (USD Million) Forecast, by Vaccine Manufacturers (Innovators & Generics), 2020-2035
- Figure 36: North America Market (USD Million) Forecast, by CDMOs & Fill-Finish Providers, 2020-2035
- Figure 37: North America Market (USD Million) Forecast, by Public Sector Institutes, 2020-2035
- Figure 38: North America Market (USD Million) Forecast, by Academic & NGO Labs, 2020-2035
- Figure 39: USA Market (USD Million) Forecast, by Reagent Class, 2020-2035
- Figure 40: USA Market (USD Million) Forecast, by Nucleases for Residual DNA Removal, 2020-2035
- Figure 41: USA Market (USD Million) Forecast, By Application, 2020-2035
- Figure 42: USA Market (USD Million) Forecast, by mRNA & LNP Vaccines, 2020-2035
- Figure 43: USA Market (USD Million) Forecast, by End User, 2020-2035
- Figure 44: USA Market (USD Million) Forecast, by Vaccine Manufacturers (Innovators & Generics), 2020-2035
- Figure 45: Canada Market (USD Million) Forecast, by Reagent Class, 2020-2035
- Figure 46: Canada Market (USD Million) Forecast, by Nucleases for Residual DNA Removal, 2020-2035
- Figure 47: Canada Market (USD Million) Forecast, By Application, 2020-2035
- Figure 48: Canada Market (USD Million) Forecast, by mRNA & LNP Vaccines, 2020-2035
- Figure 49: Canada Market (USD Million) Forecast, by End User, 2020-2035
- Figure 50: Canada Market (USD Million) Forecast, by Vaccine Manufacturers (Innovators & Generics), 2020-2035
- Figure 51: Mexico Market (USD Million) Forecast, by Reagent Class, 2020-2035
- Figure 52: Mexico Market (USD Million) Forecast, by Nucleases for Residual DNA Removal, 2020-2035
- Figure 53: Mexico Market (USD Million) Forecast, By Application, 2020-2035
- Figure 54: Mexico Market (USD Million) Forecast, by mRNA & LNP Vaccines, 2020-2035
- Figure 55: Mexico Market (USD Million) Forecast, by End User, 2020-2035
- Figure 56: Mexico Market (USD Million) Forecast, by Vaccine Manufacturers (Innovators & Generics), 2020-2035
- Figure 57: Latin America Market (USD Million) Forecast, by Reagent Class, 2020-2035
- Figure 58: Latin America Market (USD Million) Forecast, by Nucleases for Residual DNA Removal, 2020-2035
- Figure 59: Latin America Market (USD Million) Forecast, by Detergents & Solubilizers (e.g., polysorbates, zwitterionics), 2020-2035
- Figure 60: Latin America Market (USD Million) Forecast, by Inactivation Agents (BPL, Formaldehyde), 2020-2035
- Figure 61: Latin America Market (USD Million) Forecast, by Precipitation Polymers & Flocculants (PEG, PAEs), 2020-2035
- Figure 62: Latin America Market (USD Million) Forecast, by Extraction Solvents & Phase-Transfer Aids, 2020-2035
- Figure 63: Latin America Market (USD Million) Forecast, by Buffer Additives & Scavengers (antioxidants, quenchers), 2020-2035
- Figure 64: Latin America Market (USD Million) Forecast, by LNP/Wash & Auxiliary Process Reagents, 2020-2035
- Figure 65: Latin America Market (USD Million) Forecast, By Application, 2020-2035
- Figure 66: Latin America Market (USD Million) Forecast, by mRNA & LNP Vaccines, 2020-2035
- Figure 67: Latin America Market (USD Million) Forecast, by Viral Vector Vaccines, 2020-2035
- Figure 68: Latin America Market (USD Million) Forecast, by Protein Subunit & Inactivated Vaccines, 2020-2035
- Figure 69: Latin America Market (USD Million) Forecast, by Live-Attenuated & Oral Vaccines, 2020-2035
- Figure 70: Latin America Market (USD Million) Forecast, by Combination / Platform-Agnostic Programs, 2020-2035
- Figure 71: Latin America Market (USD Million) Forecast, by End User, 2020-2035
- Figure 72: Latin America Market (USD Million) Forecast, by Vaccine Manufacturers (Innovators & Generics), 2020-2035
- Figure 73: Latin America Market (USD Million) Forecast, by CDMOs & Fill-Finish Providers, 2020-2035
- Figure 74: Latin America Market (USD Million) Forecast, by Public Sector Institutes, 2020-2035
- Figure 75: Latin America Market (USD Million) Forecast, by Academic & NGO Labs, 2020-2035
- Figure 76: Brazil Market (USD Million) Forecast, by Reagent Class, 2020-2035
- Figure 77: Brazil Market (USD Million) Forecast, by Nucleases for Residual DNA Removal, 2020-2035
- Figure 78: Brazil Market (USD Million) Forecast, By Application, 2020-2035
- Figure 79: Brazil Market (USD Million) Forecast, by mRNA & LNP Vaccines, 2020-2035
- Figure 80: Brazil Market (USD Million) Forecast, by End User, 2020-2035
- Figure 81: Brazil Market (USD Million) Forecast, by Vaccine Manufacturers (Innovators & Generics), 2020-2035
- Figure 82: Argentina Market (USD Million) Forecast, by Reagent Class, 2020-2035
- Figure 83: Argentina Market (USD Million) Forecast, by Nucleases for Residual DNA Removal, 2020-2035
- Figure 84: Argentina Market (USD Million) Forecast, By Application, 2020-2035
- Figure 85: Argentina Market (USD Million) Forecast, by mRNA & LNP Vaccines, 2020-2035
- Figure 86: Argentina Market (USD Million) Forecast, by End User, 2020-2035
- Figure 87: Argentina Market (USD Million) Forecast, by Vaccine Manufacturers (Innovators & Generics), 2020-2035
- Figure 88: Rest of Latin America Market (USD Million) Forecast, by Reagent Class, 2020-2035
- Figure 89: Rest of Latin America Market (USD Million) Forecast, by Nucleases for Residual DNA Removal, 2020-2035
- Figure 90: Rest of Latin America Market (USD Million) Forecast, By Application, 2020-2035
- Figure 91: Rest of Latin America Market (USD Million) Forecast, by mRNA & LNP Vaccines, 2020-2035
- Figure 92: Rest of Latin America Market (USD Million) Forecast, by End User, 2020-2035
- Figure 93: Rest of Latin America Market (USD Million) Forecast, by Vaccine Manufacturers (Innovators & Generics), 2020-2035
- Figure 94: East Asia Market (USD Million) Forecast, by Reagent Class, 2020-2035
- Figure 95: East Asia Market (USD Million) Forecast, by Nucleases for Residual DNA Removal, 2020-2035
- Figure 96: East Asia Market (USD Million) Forecast, by Detergents & Solubilizers (e.g., polysorbates, zwitterionics), 2020-2035
- Figure 97: East Asia Market (USD Million) Forecast, by Inactivation Agents (BPL, Formaldehyde), 2020-2035
- Figure 98: East Asia Market (USD Million) Forecast, by Precipitation Polymers & Flocculants (PEG, PAEs), 2020-2035
- Figure 99: East Asia Market (USD Million) Forecast, by Extraction Solvents & Phase-Transfer Aids, 2020-2035
- Figure 100: East Asia Market (USD Million) Forecast, by Buffer Additives & Scavengers (antioxidants, quenchers), 2020-2035
- Figure 101: East Asia Market (USD Million) Forecast, by LNP/Wash & Auxiliary Process Reagents, 2020-2035
- Figure 102: East Asia Market (USD Million) Forecast, By Application, 2020-2035
- Figure 103: East Asia Market (USD Million) Forecast, by mRNA & LNP Vaccines, 2020-2035
- Figure 104: East Asia Market (USD Million) Forecast, by Viral Vector Vaccines, 2020-2035
- Figure 105: East Asia Market (USD Million) Forecast, by Protein Subunit & Inactivated Vaccines, 2020-2035
- Figure 106: East Asia Market (USD Million) Forecast, by Live-Attenuated & Oral Vaccines, 2020-2035
- Figure 107: East Asia Market (USD Million) Forecast, by Combination / Platform-Agnostic Programs, 2020-2035
- Figure 108: East Asia Market (USD Million) Forecast, by End User, 2020-2035
- Figure 109: East Asia Market (USD Million) Forecast, by Vaccine Manufacturers (Innovators & Generics), 2020-2035
- Figure 110: East Asia Market (USD Million) Forecast, by CDMOs & Fill-Finish Providers, 2020-2035
- Figure 111: East Asia Market (USD Million) Forecast, by Public Sector Institutes, 2020-2035
- Figure 112: East Asia Market (USD Million) Forecast, by Academic & NGO Labs, 2020-2035
- Figure 113: China Market (USD Million) Forecast, by Reagent Class, 2020-2035
- Figure 114: China Market (USD Million) Forecast, by Nucleases for Residual DNA Removal, 2020-2035
- Figure 115: China Market (USD Million) Forecast, By Application, 2020-2035
- Figure 116: China Market (USD Million) Forecast, by mRNA & LNP Vaccines, 2020-2035
- Figure 117: China Market (USD Million) Forecast, by End User, 2020-2035
- Figure 118: China Market (USD Million) Forecast, by Vaccine Manufacturers (Innovators & Generics), 2020-2035
- Figure 119: Japan Market (USD Million) Forecast, by Reagent Class, 2020-2035
- Figure 120: Japan Market (USD Million) Forecast, by Nucleases for Residual DNA Removal, 2020-2035
- Figure 121: Japan Market (USD Million) Forecast, By Application, 2020-2035
- Figure 122: Japan Market (USD Million) Forecast, by mRNA & LNP Vaccines, 2020-2035
- Figure 123: Japan Market (USD Million) Forecast, by End User, 2020-2035
- Figure 124: Japan Market (USD Million) Forecast, by Vaccine Manufacturers (Innovators & Generics), 2020-2035
- Figure 125: South Korea Market (USD Million) Forecast, by Reagent Class, 2020-2035
- Figure 126: South Korea Market (USD Million) Forecast, by Nucleases for Residual DNA Removal, 2020-2035
- Figure 127: South Korea Market (USD Million) Forecast, By Application, 2020-2035
- Figure 128: South Korea Market (USD Million) Forecast, by mRNA & LNP Vaccines, 2020-2035
- Figure 129: South Korea Market (USD Million) Forecast, by End User, 2020-2035
- Figure 130: South Korea Market (USD Million) Forecast, by Vaccine Manufacturers (Innovators & Generics), 2020-2035
- Figure 131: South Asia Market (USD Million) Forecast, by Reagent Class, 2020-2035
- Figure 132: South Asia Market (USD Million) Forecast, by Nucleases for Residual DNA Removal, 2020-2035
- Figure 133: South Asia Market (USD Million) Forecast, by Detergents & Solubilizers (e.g., polysorbates, zwitterionics), 2020-2035
- Figure 134: South Asia Market (USD Million) Forecast, by Inactivation Agents (BPL, Formaldehyde), 2020-2035
- Figure 135: South Asia Market (USD Million) Forecast, by Precipitation Polymers & Flocculants (PEG, PAEs), 2020-2035
- Figure 136: South Asia Market (USD Million) Forecast, by Extraction Solvents & Phase-Transfer Aids, 2020-2035
- Figure 137: South Asia Market (USD Million) Forecast, by Buffer Additives & Scavengers (antioxidants, quenchers), 2020-2035
- Figure 138: South Asia Market (USD Million) Forecast, by LNP/Wash & Auxiliary Process Reagents, 2020-2035
- Figure 139: South Asia Market (USD Million) Forecast, By Application, 2020-2035
- Figure 140: South Asia Market (USD Million) Forecast, by mRNA & LNP Vaccines, 2020-2035
- Figure 141: South Asia Market (USD Million) Forecast, by Viral Vector Vaccines, 2020-2035
- Figure 142: South Asia Market (USD Million) Forecast, by Protein Subunit & Inactivated Vaccines, 2020-2035
- Figure 143: South Asia Market (USD Million) Forecast, by Live-Attenuated & Oral Vaccines, 2020-2035
- Figure 144: South Asia Market (USD Million) Forecast, by Combination / Platform-Agnostic Programs, 2020-2035
- Figure 145: South Asia Market (USD Million) Forecast, by End User, 2020-2035
- Figure 146: South Asia Market (USD Million) Forecast, by Vaccine Manufacturers (Innovators & Generics), 2020-2035
- Figure 147: South Asia Market (USD Million) Forecast, by CDMOs & Fill-Finish Providers, 2020-2035
- Figure 148: South Asia Market (USD Million) Forecast, by Public Sector Institutes, 2020-2035
- Figure 149: South Asia Market (USD Million) Forecast, by Academic & NGO Labs, 2020-2035
- Figure 150: India Market (USD Million) Forecast, by Reagent Class, 2020-2035
- Figure 151: India Market (USD Million) Forecast, by Nucleases for Residual DNA Removal, 2020-2035
- Figure 152: India Market (USD Million) Forecast, By Application, 2020-2035
- Figure 153: India Market (USD Million) Forecast, by mRNA & LNP Vaccines, 2020-2035
- Figure 154: India Market (USD Million) Forecast, by End User, 2020-2035
- Figure 155: India Market (USD Million) Forecast, by Vaccine Manufacturers (Innovators & Generics), 2020-2035
- Figure 156: ASEAN Countries Market (USD Million) Forecast, by Reagent Class, 2020-2035
- Figure 157: ASEAN Countries Market (USD Million) Forecast, by Nucleases for Residual DNA Removal, 2020-2035
- Figure 158: ASEAN Countries Market (USD Million) Forecast, By Application, 2020-2035
- Figure 159: ASEAN Countries Market (USD Million) Forecast, by mRNA & LNP Vaccines, 2020-2035
- Figure 160: ASEAN Countries Market (USD Million) Forecast, by End User, 2020-2035
- Figure 161: ASEAN Countries Market (USD Million) Forecast, by Vaccine Manufacturers (Innovators & Generics), 2020-2035
- Figure 162: Australia & New Zealand Market (USD Million) Forecast, by Reagent Class, 2020-2035
- Figure 163: Australia & New Zealand Market (USD Million) Forecast, by Nucleases for Residual DNA Removal, 2020-2035
- Figure 164: Australia & New Zealand Market (USD Million) Forecast, By Application, 2020-2035
- Figure 165: Australia & New Zealand Market (USD Million) Forecast, by mRNA & LNP Vaccines, 2020-2035
- Figure 166: Australia & New Zealand Market (USD Million) Forecast, by End User, 2020-2035
- Figure 167: Australia & New Zealand Market (USD Million) Forecast, by Vaccine Manufacturers (Innovators & Generics), 2020-2035
- Figure 168: Rest of South Asia Market (USD Million) Forecast, by Reagent Class, 2020-2035
- Figure 169: Rest of South Asia Market (USD Million) Forecast, by Nucleases for Residual DNA Removal, 2020-2035
- Figure 170: Rest of South Asia Market (USD Million) Forecast, By Application, 2020-2035
- Figure 171: Rest of South Asia Market (USD Million) Forecast, by mRNA & LNP Vaccines, 2020-2035
- Figure 172: Rest of South Asia Market (USD Million) Forecast, by End User, 2020-2035
- Figure 173: Rest of South Asia Market (USD Million) Forecast, by Vaccine Manufacturers (Innovators & Generics), 2020-2035
- Figure 174: Western Europe Market (USD Million) Forecast, by Reagent Class, 2020-2035
- Figure 175: Western Europe Market (USD Million) Forecast, by Nucleases for Residual DNA Removal, 2020-2035
- Figure 176: Western Europe Market (USD Million) Forecast, by Detergents & Solubilizers (e.g., polysorbates, zwitterionics), 2020-2035
- Figure 177: Western Europe Market (USD Million) Forecast, by Inactivation Agents (BPL, Formaldehyde), 2020-2035
- Figure 178: Western Europe Market (USD Million) Forecast, by Precipitation Polymers & Flocculants (PEG, PAEs), 2020-2035
- Figure 179: Western Europe Market (USD Million) Forecast, by Extraction Solvents & Phase-Transfer Aids, 2020-2035
- Figure 180: Western Europe Market (USD Million) Forecast, by Buffer Additives & Scavengers (antioxidants, quenchers), 2020-2035
- Figure 181: Western Europe Market (USD Million) Forecast, by LNP/Wash & Auxiliary Process Reagents, 2020-2035
- Figure 182: Western Europe Market (USD Million) Forecast, By Application, 2020-2035
- Figure 183: Western Europe Market (USD Million) Forecast, by mRNA & LNP Vaccines, 2020-2035
- Figure 184: Western Europe Market (USD Million) Forecast, by Viral Vector Vaccines, 2020-2035
- Figure 185: Western Europe Market (USD Million) Forecast, by Protein Subunit & Inactivated Vaccines, 2020-2035
- Figure 186: Western Europe Market (USD Million) Forecast, by Live-Attenuated & Oral Vaccines, 2020-2035
- Figure 187: Western Europe Market (USD Million) Forecast, by Combination / Platform-Agnostic Programs, 2020-2035
- Figure 188: Western Europe Market (USD Million) Forecast, by End User, 2020-2035
- Figure 189: Western Europe Market (USD Million) Forecast, by Vaccine Manufacturers (Innovators & Generics), 2020-2035
- Figure 190: Western Europe Market (USD Million) Forecast, by CDMOs & Fill-Finish Providers, 2020-2035
- Figure 191: Western Europe Market (USD Million) Forecast, by Public Sector Institutes, 2020-2035
- Figure 192: Western Europe Market (USD Million) Forecast, by Academic & NGO Labs, 2020-2035
- Figure 193: Germany Market (USD Million) Forecast, by Reagent Class, 2020-2035
- Figure 194: Germany Market (USD Million) Forecast, by Nucleases for Residual DNA Removal, 2020-2035
- Figure 195: Germany Market (USD Million) Forecast, By Application, 2020-2035
- Figure 196: Germany Market (USD Million) Forecast, by mRNA & LNP Vaccines, 2020-2035
- Figure 197: Germany Market (USD Million) Forecast, by End User, 2020-2035
- Figure 198: Germany Market (USD Million) Forecast, by Vaccine Manufacturers (Innovators & Generics), 2020-2035
- Figure 199: UK Market (USD Million) Forecast, by Reagent Class, 2020-2035
- Figure 200: UK Market (USD Million) Forecast, by Nucleases for Residual DNA Removal, 2020-2035
- Figure 201: UK Market (USD Million) Forecast, By Application, 2020-2035
- Figure 202: UK Market (USD Million) Forecast, by mRNA & LNP Vaccines, 2020-2035
- Figure 203: UK Market (USD Million) Forecast, by End User, 2020-2035
- Figure 204: UK Market (USD Million) Forecast, by Vaccine Manufacturers (Innovators & Generics), 2020-2035
- Figure 205: France Market (USD Million) Forecast, by Reagent Class, 2020-2035
- Figure 206: France Market (USD Million) Forecast, by Nucleases for Residual DNA Removal, 2020-2035
- Figure 207: France Market (USD Million) Forecast, By Application, 2020-2035
- Figure 208: France Market (USD Million) Forecast, by mRNA & LNP Vaccines, 2020-2035
- Figure 209: France Market (USD Million) Forecast, by End User, 2020-2035
- Figure 210: France Market (USD Million) Forecast, by Vaccine Manufacturers (Innovators & Generics), 2020-2035
- Figure 211: Italy Market (USD Million) Forecast, by Reagent Class, 2020-2035
- Figure 212: Italy Market (USD Million) Forecast, by Nucleases for Residual DNA Removal, 2020-2035
- Figure 213: Italy Market (USD Million) Forecast, By Application, 2020-2035
- Figure 214: Italy Market (USD Million) Forecast, by mRNA & LNP Vaccines, 2020-2035
- Figure 215: Italy Market (USD Million) Forecast, by End User, 2020-2035
- Figure 216: Italy Market (USD Million) Forecast, by Vaccine Manufacturers (Innovators & Generics), 2020-2035
- Figure 217: Spain Market (USD Million) Forecast, by Reagent Class, 2020-2035
- Figure 218: Spain Market (USD Million) Forecast, by Nucleases for Residual DNA Removal, 2020-2035
- Figure 219: Spain Market (USD Million) Forecast, By Application, 2020-2035
- Figure 220: Spain Market (USD Million) Forecast, by mRNA & LNP Vaccines, 2020-2035
- Figure 221: Spain Market (USD Million) Forecast, by End User, 2020-2035
- Figure 222: Spain Market (USD Million) Forecast, by Vaccine Manufacturers (Innovators & Generics), 2020-2035
- Figure 223: BENELUX Market (USD Million) Forecast, by Reagent Class, 2020-2035
- Figure 224: BENELUX Market (USD Million) Forecast, by Nucleases for Residual DNA Removal, 2020-2035
- Figure 225: BENELUX Market (USD Million) Forecast, By Application, 2020-2035
- Figure 226: BENELUX Market (USD Million) Forecast, by mRNA & LNP Vaccines, 2020-2035
- Figure 227: BENELUX Market (USD Million) Forecast, by End User, 2020-2035
- Figure 228: BENELUX Market (USD Million) Forecast, by Vaccine Manufacturers (Innovators & Generics), 2020-2035
- Figure 229: Nordic Countries Market (USD Million) Forecast, by Reagent Class, 2020-2035
- Figure 230: Nordic Countries Market (USD Million) Forecast, by Nucleases for Residual DNA Removal, 2020-2035
- Figure 231: Nordic Countries Market (USD Million) Forecast, By Application, 2020-2035
- Figure 232: Nordic Countries Market (USD Million) Forecast, by mRNA & LNP Vaccines, 2020-2035
- Figure 233: Nordic Countries Market (USD Million) Forecast, by End User, 2020-2035
- Figure 234: Nordic Countries Market (USD Million) Forecast, by Vaccine Manufacturers (Innovators & Generics), 2020-2035
- Figure 235: Rest of Western Europe Market (USD Million) Forecast, by Reagent Class, 2020-2035
- Figure 236: Rest of Western Europe Market (USD Million) Forecast, by Nucleases for Residual DNA Removal, 2020-2035
- Figure 237: Rest of Western Europe Market (USD Million) Forecast, By Application, 2020-2035
- Figure 238: Rest of Western Europe Market (USD Million) Forecast, by mRNA & LNP Vaccines, 2020-2035
- Figure 239: Rest of Western Europe Market (USD Million) Forecast, by End User, 2020-2035
- Figure 240: Rest of Western Europe Market (USD Million) Forecast, by Vaccine Manufacturers (Innovators & Generics), 2020-2035
- Figure 241: Eastern Europe Market (USD Million) Forecast, by Reagent Class, 2020-2035
- Figure 242: Eastern Europe Market (USD Million) Forecast, by Nucleases for Residual DNA Removal, 2020-2035
- Figure 243: Eastern Europe Market (USD Million) Forecast, by Detergents & Solubilizers (e.g., polysorbates, zwitterionics), 2020-2035
- Figure 244: Eastern Europe Market (USD Million) Forecast, by Inactivation Agents (BPL, Formaldehyde), 2020-2035
- Figure 245: Eastern Europe Market (USD Million) Forecast, by Precipitation Polymers & Flocculants (PEG, PAEs), 2020-2035
- Figure 246: Eastern Europe Market (USD Million) Forecast, by Extraction Solvents & Phase-Transfer Aids, 2020-2035
- Figure 247: Eastern Europe Market (USD Million) Forecast, by Buffer Additives & Scavengers (antioxidants, quenchers), 2020-2035
- Figure 248: Eastern Europe Market (USD Million) Forecast, by LNP/Wash & Auxiliary Process Reagents, 2020-2035
- Figure 249: Eastern Europe Market (USD Million) Forecast, By Application, 2020-2035
- Figure 250: Eastern Europe Market (USD Million) Forecast, by mRNA & LNP Vaccines, 2020-2035
- Figure 251: Eastern Europe Market (USD Million) Forecast, by Viral Vector Vaccines, 2020-2035
- Figure 252: Eastern Europe Market (USD Million) Forecast, by Protein Subunit & Inactivated Vaccines, 2020-2035
- Figure 253: Eastern Europe Market (USD Million) Forecast, by Live-Attenuated & Oral Vaccines, 2020-2035
- Figure 254: Eastern Europe Market (USD Million) Forecast, by Combination / Platform-Agnostic Programs, 2020-2035
- Figure 255: Eastern Europe Market (USD Million) Forecast, by End User, 2020-2035
- Figure 256: Eastern Europe Market (USD Million) Forecast, by Vaccine Manufacturers (Innovators & Generics), 2020-2035
- Figure 257: Eastern Europe Market (USD Million) Forecast, by CDMOs & Fill-Finish Providers, 2020-2035
- Figure 258: Eastern Europe Market (USD Million) Forecast, by Public Sector Institutes, 2020-2035
- Figure 259: Eastern Europe Market (USD Million) Forecast, by Academic & NGO Labs, 2020-2035
- Figure 260: Russia Market (USD Million) Forecast, by Reagent Class, 2020-2035
- Figure 261: Russia Market (USD Million) Forecast, by Nucleases for Residual DNA Removal, 2020-2035
- Figure 262: Russia Market (USD Million) Forecast, By Application, 2020-2035
- Figure 263: Russia Market (USD Million) Forecast, by mRNA & LNP Vaccines, 2020-2035
- Figure 264: Russia Market (USD Million) Forecast, by End User, 2020-2035
- Figure 265: Russia Market (USD Million) Forecast, by Vaccine Manufacturers (Innovators & Generics), 2020-2035
- Figure 266: Hungary Market (USD Million) Forecast, by Reagent Class, 2020-2035
- Figure 267: Hungary Market (USD Million) Forecast, by Nucleases for Residual DNA Removal, 2020-2035
- Figure 268: Hungary Market (USD Million) Forecast, By Application, 2020-2035
- Figure 269: Hungary Market (USD Million) Forecast, by mRNA & LNP Vaccines, 2020-2035
- Figure 270: Hungary Market (USD Million) Forecast, by End User, 2020-2035
- Figure 271: Hungary Market (USD Million) Forecast, by Vaccine Manufacturers (Innovators & Generics), 2020-2035
- Figure 272: Poland Market (USD Million) Forecast, by Reagent Class, 2020-2035
- Figure 273: Poland Market (USD Million) Forecast, by Nucleases for Residual DNA Removal, 2020-2035
- Figure 274: Poland Market (USD Million) Forecast, By Application, 2020-2035
- Figure 275: Poland Market (USD Million) Forecast, by mRNA & LNP Vaccines, 2020-2035
- Figure 276: Poland Market (USD Million) Forecast, by End User, 2020-2035
- Figure 277: Poland Market (USD Million) Forecast, by Vaccine Manufacturers (Innovators & Generics), 2020-2035
- Figure 278: Rest of Eastern Europe Market (USD Million) Forecast, by Reagent Class, 2020-2035
- Figure 279: Rest of Eastern Europe Market (USD Million) Forecast, by Nucleases for Residual DNA Removal, 2020-2035
- Figure 280: Rest of Eastern Europe Market (USD Million) Forecast, By Application, 2020-2035
- Figure 281: Rest of Eastern Europe Market (USD Million) Forecast, by mRNA & LNP Vaccines, 2020-2035
- Figure 282: Rest of Eastern Europe Market (USD Million) Forecast, by End User, 2020-2035
- Figure 283: Rest of Eastern Europe Market (USD Million) Forecast, by Vaccine Manufacturers (Innovators & Generics), 2020-2035
- Figure 284: Middle East & Africa Market (USD Million) Forecast, by Reagent Class, 2020-2035
- Figure 285: Middle East & Africa Market (USD Million) Forecast, by Nucleases for Residual DNA Removal, 2020-2035
- Figure 286: Middle East & Africa Market (USD Million) Forecast, by Detergents & Solubilizers (e.g., polysorbates, zwitterionics), 2020-2035
- Figure 287: Middle East & Africa Market (USD Million) Forecast, by Inactivation Agents (BPL, Formaldehyde), 2020-2035
- Figure 288: Middle East & Africa Market (USD Million) Forecast, by Precipitation Polymers & Flocculants (PEG, PAEs), 2020-2035
- Figure 289: Middle East & Africa Market (USD Million) Forecast, by Extraction Solvents & Phase-Transfer Aids, 2020-2035
- Figure 290: Middle East & Africa Market (USD Million) Forecast, by Buffer Additives & Scavengers (antioxidants, quenchers), 2020-2035
- Figure 291: Middle East & Africa Market (USD Million) Forecast, by LNP/Wash & Auxiliary Process Reagents, 2020-2035
- Figure 292: Middle East & Africa Market (USD Million) Forecast, By Application, 2020-2035
- Figure 293: Middle East & Africa Market (USD Million) Forecast, by mRNA & LNP Vaccines, 2020-2035
- Figure 294: Middle East & Africa Market (USD Million) Forecast, by Viral Vector Vaccines, 2020-2035
- Figure 295: Middle East & Africa Market (USD Million) Forecast, by Protein Subunit & Inactivated Vaccines, 2020-2035
- Figure 296: Middle East & Africa Market (USD Million) Forecast, by Live-Attenuated & Oral Vaccines, 2020-2035
- Figure 297: Middle East & Africa Market (USD Million) Forecast, by Combination / Platform-Agnostic Programs, 2020-2035
- Figure 298: Middle East & Africa Market (USD Million) Forecast, by End User, 2020-2035
- Figure 299: Middle East & Africa Market (USD Million) Forecast, by Vaccine Manufacturers (Innovators & Generics), 2020-2035
- Figure 300: Middle East & Africa Market (USD Million) Forecast, by CDMOs & Fill-Finish Providers, 2020-2035
- Figure 301: Middle East & Africa Market (USD Million) Forecast, by Public Sector Institutes, 2020-2035
- Figure 302: Middle East & Africa Market (USD Million) Forecast, by Academic & NGO Labs, 2020-2035
- Figure 303: Saudi Arabia Market (USD Million) Forecast, by Reagent Class, 2020-2035
- Figure 304: Saudi Arabia Market (USD Million) Forecast, by Nucleases for Residual DNA Removal, 2020-2035
- Figure 305: Saudi Arabia Market (USD Million) Forecast, By Application, 2020-2035
- Figure 306: Saudi Arabia Market (USD Million) Forecast, by mRNA & LNP Vaccines, 2020-2035
- Figure 307: Saudi Arabia Market (USD Million) Forecast, by End User, 2020-2035
- Figure 308: Saudi Arabia Market (USD Million) Forecast, by Vaccine Manufacturers (Innovators & Generics), 2020-2035
- Figure 309: Other GCC Countries Market (USD Million) Forecast, by Reagent Class, 2020-2035
- Figure 310: Other GCC Countries Market (USD Million) Forecast, by Nucleases for Residual DNA Removal, 2020-2035
- Figure 311: Other GCC Countries Market (USD Million) Forecast, By Application, 2020-2035
- Figure 312: Other GCC Countries Market (USD Million) Forecast, by mRNA & LNP Vaccines, 2020-2035
- Figure 313: Other GCC Countries Market (USD Million) Forecast, by End User, 2020-2035
- Figure 314: Other GCC Countries Market (USD Million) Forecast, by Vaccine Manufacturers (Innovators & Generics), 2020-2035
- Figure 315: Türkiye Market (USD Million) Forecast, by Reagent Class, 2020-2035
- Figure 316: Türkiye Market (USD Million) Forecast, by Nucleases for Residual DNA Removal, 2020-2035
- Figure 317: Türkiye Market (USD Million) Forecast, By Application, 2020-2035
- Figure 318: Türkiye Market (USD Million) Forecast, by mRNA & LNP Vaccines, 2020-2035
- Figure 319: Türkiye Market (USD Million) Forecast, by End User, 2020-2035
- Figure 320: Türkiye Market (USD Million) Forecast, by Vaccine Manufacturers (Innovators & Generics), 2020-2035
- Figure 321: South Africa Market (USD Million) Forecast, by Reagent Class, 2020-2035
- Figure 322: South Africa Market (USD Million) Forecast, by Nucleases for Residual DNA Removal, 2020-2035
- Figure 323: South Africa Market (USD Million) Forecast, By Application, 2020-2035
- Figure 324: South Africa Market (USD Million) Forecast, by mRNA & LNP Vaccines, 2020-2035
- Figure 325: South Africa Market (USD Million) Forecast, by End User, 2020-2035
- Figure 326: South Africa Market (USD Million) Forecast, by Vaccine Manufacturers (Innovators & Generics), 2020-2035
- Figure 327: Rest of MEA Market (USD Million) Forecast, by Reagent Class, 2020-2035
- Figure 328: Rest of MEA Market (USD Million) Forecast, by Nucleases for Residual DNA Removal, 2020-2035
- Figure 329: Rest of MEA Market (USD Million) Forecast, By Application, 2020-2035
- Figure 330: Rest of MEA Market (USD Million) Forecast, by mRNA & LNP Vaccines, 2020-2035
- Figure 331: Rest of MEA Market (USD Million) Forecast, by End User, 2020-2035
- Figure 332: Rest of MEA Market (USD Million) Forecast, by Vaccine Manufacturers (Innovators & Generics), 2020-2035
- Figure 333: Merck MilliporeSigma (incl. Benzonase) - Company Snapshot
- Figure 334: Merck MilliporeSigma (incl. Benzonase) - SWOT Analysis
- Figure 335: Thermo Fisher Scientific - Company Snapshot
- Figure 336: Thermo Fisher Scientific - SWOT Analysis
- Figure 337: Cytiva - Company Snapshot
- Figure 338: Cytiva - SWOT Analysis
- Figure 339: Sartorius - Company Snapshot
- Figure 340: Sartorius - SWOT Analysis
- Figure 341: Croda Pharma (incl. Avanti) - Company Snapshot
- Figure 342: Croda Pharma (incl. Avanti) - SWOT Analysis
- Figure 343: BASF (Kolliphor/solvents) - Company Snapshot
- Figure 344: BASF (Kolliphor/solvents) - SWOT Analysis
- Figure 345: Seppic (Air Liquide Group) - Company Snapshot
- Figure 346: Seppic (Air Liquide Group) - SWOT Analysis
- Figure 347: NOF Corporation (PEG & lipids) - Company Snapshot
- Figure 348: NOF Corporation (PEG & lipids) - SWOT Analysis
- Figure 349: ArcticZymes (SAN nucleases) - Company Snapshot
- Figure 350: ArcticZymes (SAN nucleases) - SWOT Analysis
- Figure 351: Eastman (BPL/formulation solvents) - Company Snapshot
- Figure 352: Eastman (BPL/formulation solvents) - SWOT Analysis
- Figure 353: Promega (nucleases & enzymes) - Company Snapshot
- Figure 354: Promega (nucleases & enzymes) - SWOT Analysis
- Figure 355: Others - Company Snapshot
- Figure 356: Others - SWOT Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

