Biological Indicator Vial Market
Biological Indicator Vial Market Analysis Size and Share Forecast Outlook 2026 to 2036
Biological Indicator Vial Market Forecast and Outlook 2026 to 2036
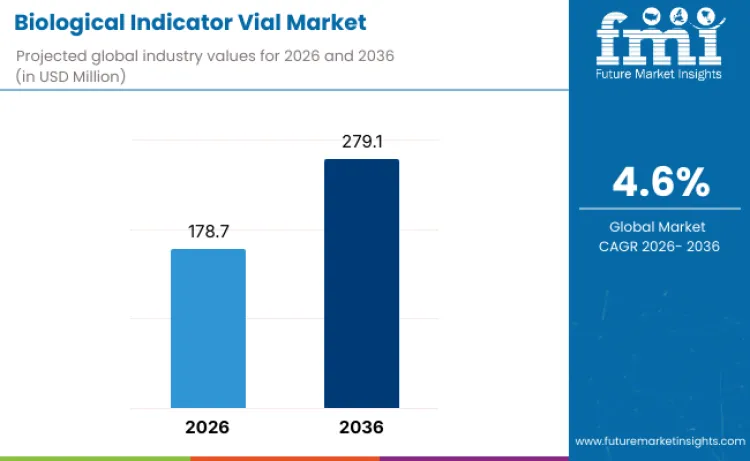
The biological indicator vial market is valued at USD 178.7 million in 2026 and is projected to reach USD 279.1 million by 2036, reflecting a CAGR of 4.6% during the forecast period. Market expansion is underpinned by the rising emphasis on sterilization validation across hospitals, pharmaceutical manufacturing, dental practices, and laboratory environments. North America and Europe remain key consumption centers due to stringent regulatory frameworks and established healthcare infrastructure, while Asia Pacific is expected to grow rapidly as healthcare capacity expands and pharmaceutical production scales across emerging economies.
Demand growth is closely tied to the critical role biological indicators play in validating steam, ethylene oxide, and hydrogen peroxide sterilization processes. Increasing procedural volumes, tighter infection control protocols, and audits in medical device and drug production facilities continue to reinforce recurring demand. The shift toward reusable instruments in surgical settings and the expansion of contract sterilization services further contribute to steady market adoption.
Quick Stats for Biological Indicator Vial Market
- Market Value 2026: USD 178.7 million
- Market Forecast Value 2036: USD 279.1 million
- Forecast CAGR: 4.6%
- Leading Sterilization Type: Steam sterilization
- Key Growth Regions: East Asia, South Asia Pacific, Western Europe
- Key Players: VWR Corporation, STERIS plc., Mesa Labs, 3M Company, Siltex Australia
Despite favorable growth prospects, the biological indicator vial market faces operational and regulatory challenges. Compliance requirements, time-intensive incubation cycles, and the need for precise handling raise costs and complexity for end users. Manufacturers are focusing on faster readout technologies, improved packaging reliability, and integrated monitoring systems that reduce error risk while aligning with evolving quality standards.
Biological Indicator Vial Market Key Takeaways
| Metric | Value |
|---|---|
| Market Value (2026) | USD 178.7 million |
| Market Forecast Value (2036) | USD 279.1 million |
| Forecast CAGR (2026 to 2036) | 4.6% |
How Are the Segments Classified in the Biological Indicator Vial Market?
The biological indicator vial market is segmented by sterilization type, incubation time, end use, and region. By sterilization type, the industry includes steam sterilization and ethylene oxide sterilization, reflecting the evolution from traditional thermal sterilization validation toward advanced chemical sterilization monitoring. By incubation time, segments include up to 24 hours, 24 to 32 hours, 32 to 48 hours, and above 48 hours, supporting different validation turnaround expectations and sterilization assurance strategies.
End-use industries span food and beverages, pharmaceuticals, and cosmetics, while regional coverage includes North America, Latin America, Western Europe, Eastern Europe, East Asia, South Asia Pacific, and the Middle East & Africa. Segmentation trends highlight a gradual shift from long-incubation indicators toward faster readout systems designed to improve operational efficiency, reduce production delays, and support rigorous process validation standards across regulated environments.
Which Sterilization Type Accounts for the Largest Share of Global Demand?
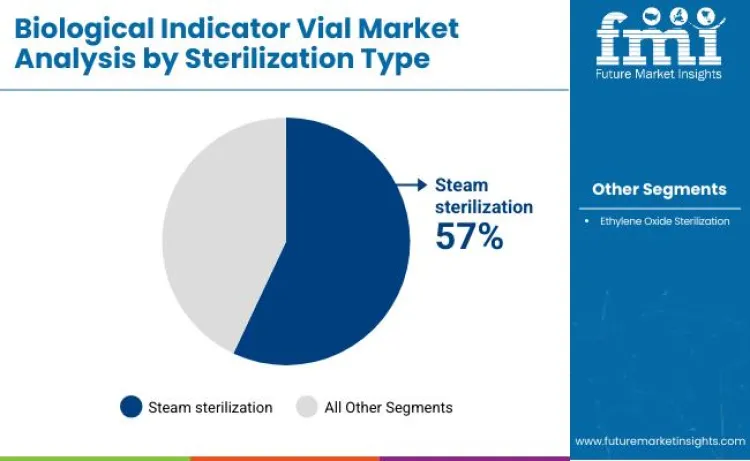
Steam sterilization indicators hold approximately 57% market share, making them the dominant sterilization type worldwide. These indicators support validation of autoclave cycles, offering proven spore resistance, robust thermal stability, and strong regulatory acceptance. Their compatibility with standard sterilization equipment enables effective verification across hospitals, pharmaceutical facilities, food processors, and contract sterilization providers.
Ethylene oxide biological indicators maintain a 43% market contribution, primarily serving applications where low-temperature sterilization is required for heat-sensitive medical devices and specialized manufacturing environments.
Key Points:
- Steam indicators dominate due to reliability and widespread acceptance
- Well suited for autoclave validation and routine cycle monitoring
- Strong role in regulated pharmaceutical and food processing environments
- EtO indicators support specialized, low-temperature device sterilization needs
Which Incubation Time Segment Represents the Largest Share of Market Demand?
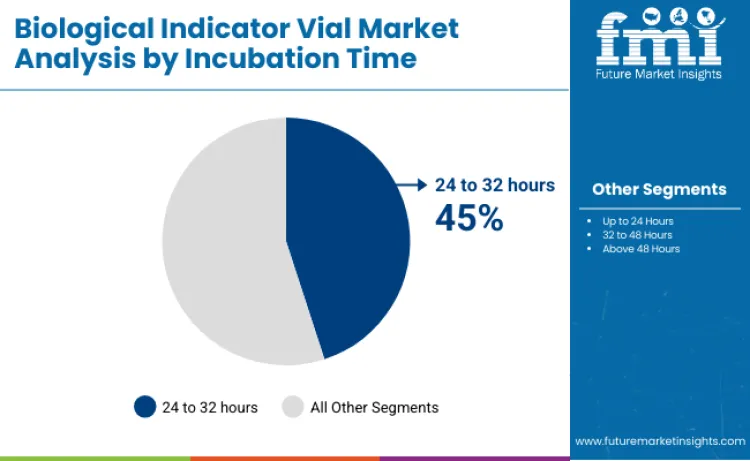
The 24 to 32 hours incubation segment accounts for 45% of global demand, providing a balance between validation reliability and turnaround efficiency. Quality assurance teams favor this segment because it supports compliant documentation and timely batch release decisions without significant production delays.
Up to 24 hours indicators hold 19% share, reflecting rapid-readout adoption in time-sensitive environments. Extended incubation segments, including 32 to 48 hours and above 48 hours at 18% each, continue to serve legacy validation practices, specialized sterility assurance protocols, and applications requiring enhanced detection sensitivity.
Key Points:
- 24-32 hours remains the industry standard for routine validation
- Rapid-readout indicators gain traction in manufacturing environments
- Extended incubation supports specialized and conservative validation protocols
Which End Use Segment Represents the Largest Share of Global Demand?
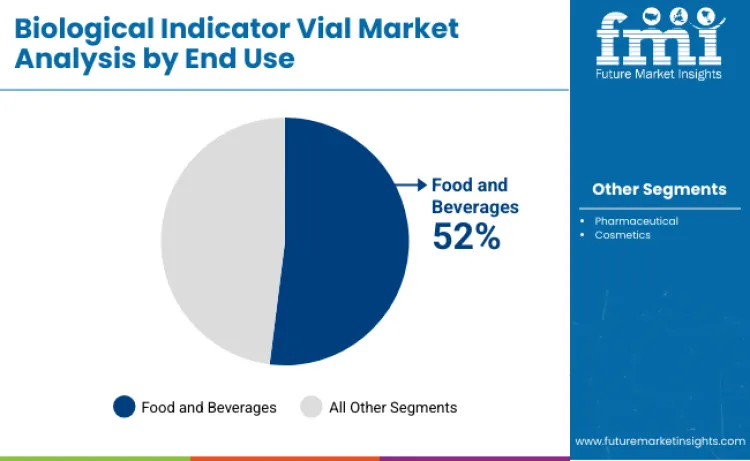
Food and beverages account for 52% of total demand, making the sector the leading end-use category. Biological indicators play a critical role in validating retort processes, aseptic packaging systems, and canning operations to ensure food safety compliance. Pharmaceutical applications represent 28% share, driven by sterile manufacturing expansion, injectable production, and strict regulatory oversight. Cosmetics account for 20%, emphasizing quality control, contamination prevention, and product safety assurance.
Key Points:
- Food processing dominates due to strict safety regulations
- Pharmaceuticals rely on indicators for aseptic and terminal sterilization validation
- Cosmetics require microbial control and batch verification processes
What Are the Key Dynamics in the Biological Indicator Vial Market?
Market growth is supported by rising global emphasis on sterilization validation, infection control, and documented proof of sterility assurance. Expanding pharmaceutical manufacturing capacity, stricter food safety regulations, and increased focus on contamination prevention drive recurring demand. Technology innovation is shifting the industry toward rapid-readout indicators and self-contained vial systems that minimize handling risk, shorten incubation timelines, and improve workflow efficiency.
Manufacturers focus on advancing spore technology, strengthening regulatory compliance documentation, and developing faster result systems that align with production scheduling needs and evolving quality expectations.
How Are Sterility Requirements and Manufacturing Workflows Influencing Adoption?
Tighter regulatory expectations across pharmaceuticals, food processing, and healthcare environments reinforce biological indicator use as a core validation tool. Facilities depend on indicators to verify sterilization lethality, document compliance, and reduce contamination risks that threaten patient safety and product integrity.
Production workflows increasingly prioritize faster batch release timelines, encouraging adoption of shorter-incubation technologies. Food processors rely on indicators to meet HACCP and thermal validation requirements, while pharmaceutical operators integrate biological indicators into routine cycle qualification, equipment commissioning, and aseptic process verification.
Across industries, adoption reflects the need for reliability, traceability, and documented proof of sterility assurance.
How Do Regulatory Constraints and Operational Limitations Affect Scalability?
Regulatory mandates from bodies including FDA, ISO organizations, and pharmacopeias shape development, validation, labeling, and documentation requirements, raising product design complexity and extending approval timelines. Manufacturers must maintain high-cost quality systems and validation records across multiple jurisdictions, limiting rapid scale-up.
Operational challenges include lengthy incubation cycles, which delay production release, and cost pressures among smaller facilities, which may limit indicator frequency. Transitioning to rapid-readout platforms requires workflow changes and new capital investments, creating adoption barriers in budget-sensitive markets.
Long-term scalability depends on accelerating readout capability, improving accuracy, and reducing validation burden while maintaining strict regulatory alignment.
What Are the Drivers, Restraints, and Key Trends in the Biological Indicator Vial Market?
The biological indicator vial market is driven by intensifying regulatory compliance requirements across pharmaceuticals, medical devices, and food processing, where agencies such as the FDA and global ISO bodies mandate documented sterilization validation as part of quality assurance. Expanding sterile manufacturing capacity, particularly in biologics, injectables, and aseptic processing environments, further increases validation frequency and recurring indicator use. The industry faces restraints linked to long incubation times that delay batch release and create production bottlenecks, along with cost sensitivity among smaller manufacturers and emerging markets that limits testing intensity and slows technology upgrades. Against this backdrop, key trends are reshaping the competitive landscape, including rapid-readout indicators that enable same-day verification and self-contained systems that integrate growth media, reduce contamination risk, simplify workflows, and improve traceability. Together, these developments reflect a market moving toward faster validation, stronger documentation, and greater operational efficiency while balancing regulatory expectations and cost pressures.
How Is Demand for the Biological Indicator Vial Market Shifting Across Major Countries?
Demand for biological indicator vials is rising as global industries strengthen sterilization validation requirements, modernize production systems, and respond to stricter regulatory oversight. Growth is led by regions expanding pharmaceutical capacity and advancing food safety initiatives. China grows at 6.2% driven by pharmaceutical expansion programs and food processing modernization that require validated sterilization cycles. India records 5.8% growth as contract manufacturing and export certification increase validation frequency.
Germany grows at 5.3%, reflecting EU GMP compliance and mature quality systems. The United States posts 4.4% growth through strong sterility assurance culture and adoption of rapid-readout technologies. The United Kingdom records 3.9%, supported by MHRA oversight and pharmaceutical production needs. Japan maintains 3.5% growth driven by disciplined validation practices and preference for premium biological indicators. Together, these markets reflect rising emphasis on traceability, faster validation turnaround, and documented proof of sterilization across regulated manufacturing environments.
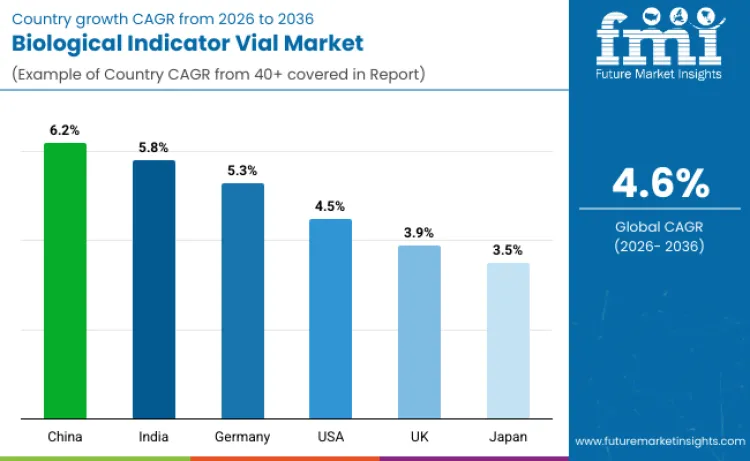
| Country | CAGR |
|---|---|
| China | 6.2% |
| India | 5.8% |
| Germany | 5.3% |
| United States | 4.5% |
| United Kingdom | 3.9% |
| Japan | 3.5% |
Why Is China Emerging as the Fastest-Growing Biological Indicator Vial Market?
China is emerging as the fastest-growing biological indicator vial market, advancing at a 6.2% CAGR as national pharmaceutical expansion programs and sweeping food safety reforms intensify sterilization validation requirements. Government policies promoting domestic drug production mandate validated autoclave cycles, aseptic process qualification, and documented sterility assurance across sterile injectable facilities. Modernization in canning and aseptic food processing drives greater reliance on biological indicators to verify thermal lethality and compliance. Indicators are now systematically integrated into qualification protocols across major hubs such as Shanghai, Jiangsu, and Beijing, supporting export readiness and regulatory audit performance. Local manufacturers leverage competitive pricing, localization advantages, and strong distribution networks, improving accessibility while aligning with evolving GMP standards. As enforcement strengthens and production scales, recurring demand for reliable, traceable validation tools positions biological indicator vials as essential components of China’s expanding pharmaceutical and food processing ecosystems.
What Is Fueling India’s High-Growth Adoption of Biological Indicator Vials?
India’s biological indicator vial market expands at 5.8% as pharmaceutical exports, contract manufacturing activity, and food safety modernization programs intensify validation needs. Biological indicators are increasingly adopted across major pharmaceutical clusters such as Hyderabad, Mumbai, and Ahmedabad, where facilities require documented sterilization proof to meet global agency expectations. Contract manufacturing organizations rely on indicators to support qualification, batch release, and inspection readiness, while food processors deploy validation tools for canning and aseptic packaging operations. Cost-effective products, combined with supplier training and technical guidance, help organizations transition from minimal testing toward structured validation planning. Growing international audits, evolving domestic regulations, and customer expectations for traceability further reinforce recurring use. As pharmaceutical investments expand and export ambitions increase, biological indicator vials continue to play a central role in helping Indian manufacturers demonstrate process integrity and meet stringent global market requirements.
How Does Germany Maintain Leadership in Sterilization Validation Markets?
Germany maintains leadership in the biological indicator vial market with 5.3% growth, supported by advanced pharmaceutical manufacturing, stringent EU GMP frameworks, and long-established validation culture. Biological indicators are deeply integrated into qualification protocols for autoclaves, aseptic processing lines, and medical device sterilization systems. Quality assurance teams rely on standardized procedures, strong documentation practices, and regulatory expertise to ensure consistent sterility assurance. Laboratory suppliers and validation service providers strengthen adoption by offering technical support, regulatory interpretation, and training resources that simplify implementation. Ongoing investment in rapid-readout technologies enables faster decision-making while maintaining compliance with conservative risk controls. The country’s mature healthcare infrastructure, combined with export-oriented pharmaceutical operations, ensures stable, recurring demand. Germany’s emphasis on precision, safety, and lifecycle validation positions biological indicators as essential tools supporting reliability, regulatory readiness, and continuous process improvement across highly regulated production environments.
What Factors Are Driving the United States Biological Indicator Market?
The United States biological indicator vial market grows at 4.5%, shaped by rigorous FDA oversight, sophisticated pharmaceutical infrastructure, and increasing interest in faster validation turnaround. Biological indicators are central to autoclave qualification, aseptic processing verification, and ongoing process monitoring across sterile injectable and biologics manufacturing facilities. Manufacturers prioritize indicators that support rapid-readout capabilities, traceable results, and alignment with evolving regulatory expectations. Extensive laboratory distribution networks, contract manufacturing partnerships, and validation consulting services reinforce adoption by providing expertise and supply reliability. Frequent inspections, heightened quality expectations, and continuous process verification requirements sustain recurring demand. As manufacturers seek to balance safety assurance with production efficiency, biological indicators remain indispensable tools for documenting sterility, supporting risk mitigation, and maintaining regulatory confidence across complex pharmaceutical production systems.
Why Does the United Kingdom Show Steady but Mature Market Growth?
The United Kingdom records steady 3.9% growth in the biological indicator vial market, reflecting a mature pharmaceutical base, stringent MHRA oversight, and strong quality assurance culture. Biological indicators are widely used across parenteral manufacturing, biotech facilities, and contract organizations where documented sterilization validation remains essential. Manufacturers value proven indicator systems with extensive regulatory precedent and reliable performance records that support inspection readiness and ongoing compliance. Contract development and manufacturing activity reinforces recurring demand as facilities validate processes for multiple clients under strict documentation requirements. Market dynamics emphasize service quality, technical expertise, and consistent validation methodology rather than rapid expansion. While growth rates are modest compared with emerging markets, reliance on validated, traceable sterilization verification ensures biological indicators remain integral to the United Kingdom’s regulated pharmaceutical production environment.
How Does Japan’s Quality-Driven Approach Shape Market Expansion?
Japan’s biological indicator vial market expands at 3.5% due to its strong culture of quality discipline and stringent regulatory documentation expectations. Pharmaceutical and medical device manufacturers integrate biological indicators into routine sterilization validation to support compliance with PMDA guidelines and international export standards. Facilities emphasize premium indicator systems that deliver reliable performance, traceable results, and compatibility with comprehensive validation protocols. Adoption progresses steadily rather than rapidly, reflecting conservative decision-making processes and careful evaluation of new technologies. Growth is supported by rising biologics production, continued modernization of healthcare sterilization practices, and demand for proven sterilization assurance tools. As manufacturers prioritize risk reduction and long-term reliability, biological indicators remain fundamental to ensuring process integrity, patient safety, and regulatory alignment across Japan’s highly disciplined manufacturing environment.
What Is the Competitive Landscape of the Biological Indicator Vial Market Globally?
The biological indicator vial market is moderately consolidated, characterized by a concentrated group of manufacturers with strong regulatory capability and technical depth. Leading players benefit from integrated spore production, validated vial manufacturing processes, and established relationships across pharmaceutical, food processing, and healthcare sterilization environments. Competitive positioning is shaped by product reliability, regulatory documentation strength, speed of innovation in rapid-readout technologies, and the breadth of validation support services offered to end users. Companies expand biological indicator portfolios to support autoclave, ethylene oxide, and hydrogen peroxide sterilization while advancing digital documentation platforms that align with evolving inspection requirements. Demand growth supports players that combine proven sterility assurance performance with innovation in turnaround time, automation, and lifecycle validation support.
Within the competitive structure, infection-prevention companies with comprehensive sterilization portfolios hold strong positions due to regulatory credibility and global distribution reach. Specialized biological indicator manufacturers differentiate through spore technology expertise, custom validation programs, and rapid-readout system development. Laboratory supply distributors provide broad market coverage and bundled quality assurance offerings, while emerging technology innovators introduce automated incubators, fluorescence-based detection, and digital reporting systems that enhance efficiency. Leadership across the industry is sustained through documented performance claims, reliable supply chains, technical training resources, and established channel partnerships serving pharmaceutical and healthcare customers.
Commoditization pressures are visible in traditional steam indicators with conventional incubation times, basic spore strips, and legacy 7-day biological indicators. Margin expansion increasingly shifts toward rapid-readout solutions, self-contained vials with integrated media, specialized ethylene oxide indicators, automated readers, and consulting services that support validation planning and electronic batch record integration. Competitive dynamics therefore balance price pressure in standard segments with premium innovation opportunities tied to workflow optimization and regulatory readiness. Companies capable of combining cost-effective manufacturing with advanced validation technologies are positioned to capture share as users prioritize traceability, faster decision making, and audit-ready documentation.
Competitive rivalry further reflects the roles of different stakeholders across the supply chain. Infection-prevention leaders control global sterilization portfolios and hospital relationships, supported by strong compliance credibility but often slower innovation cycle times. Specialized biological indicator manufacturers control spore production expertise and deep validation knowledge, although they may lack broad distribution scale. Laboratory distributors control customer access and purchasing convenience but rely on external product innovation. Rapid-technology innovators control fast-detection platforms and automation capabilities but face slower regulatory acceptance. Regional producers control local manufacturing and pricing advantages yet have limited reach into premium segments. Market competition intensifies around rapid-readout adoption, regulatory documentation excellence, and self-contained system development as facilities seek validated solutions that reduce risk and accelerate batch release across regulated industries.
Key Players in the Biological Indicator Vial Market
- VWR Corporation, Avantor
- STERIS plc
- Mesa Laboratories Inc.
- 3M Company
- Siltex Australia Pty Ltd.
- Getinge AB
- Terragene S.A.
- Crosstex International Inc.
- STERIS
- Raven Biological Laboratories
- ProSpore
- Hardy Diagnostics
- Excelsior Scientific
- Benchmark Indicator Technologies
- Hu-Friedy Mfg. Co. LLC
Scope of Report
| Item | Value |
|---|---|
| Quantitative Units | USD million |
| Sterilization Type | Steam Sterilization, Ethylene Oxide Sterilization |
| Incubation Time | 24 to 32 Hours, Up to 24 Hours, 32 to 48 Hours, Above 48 Hours |
| End Use | Food & Beverages, Pharmaceutical, Cosmetics |
| Regions Covered | North America, Latin America, Western Europe, Eastern Europe, East Asia, South Asia Pacific, Middle East & Africa |
| Countries Covered | China, India, Germany, United States, United Kingdom, Japan, South Korea, France, Italy, Australia, plus 25+ additional countries |
| Key Companies Profiled | VWR Corporation Avantor, STERIS plc, Mesa Laboratories Inc., 3M Company, Siltex Australia Pty Ltd., Getinge AB, Terragene S.A., Crosstex International Inc., STERIS |
| Additional Attributes | Dollar sales by sterilization type and end-use categories; regional adoption trends across East Asia, North America, and Western Europe; competitive landscape with sterilization equipment manufacturers and quality assurance suppliers; quality professional preferences for validation reliability and regulatory compliance; integration with sterilization equipment and quality management systems; innovations in rapid-readout technology and self-contained designs; development of enhanced-performance biological indicators optimized for validation efficiency |
Biological Indicator Vial Market by Segments
By Sterilization Type:
- Steam Sterilization
- Ethylene Oxide Sterilization
By Incubation Time:
- 24 to 32 Hours
- Up to 24 Hours
- 32 to 48 Hours
- Above 48 Hours
By End Use:
- Food & Beverages
- Pharmaceutical
- Cosmetics
By Region:
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Argentina
- Rest of Latin America
- Western Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Nordic
- BENELUX
- Rest of Western Europe
- Eastern Europe
- Russia
- Poland
- Rest of Eastern Europe
- East Asia
- China
- Japan
- South Korea
- South Asia Pacific
- India
- Australia
- ASEAN
- New Zealand
- Rest of South Asia Pacific
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Other African Union
- Rest of Middle East & Africa
Bibliography
- STERIS plc. (2023). Biological indicators and sterility assurance solutions for healthcare and pharmaceutical applications. STERIS plc.
- Mesa Laboratories Inc. (2023). Advances in biological indicator vial technology for sterilization validation. Mesa Laboratories Inc.
- 3M Company. (2023). Biological monitoring systems for steam and ethylene oxide sterilization processes. 3M Company.
- Getinge AB. (2023). Sterilization validation tools and biological indicators for regulated environments. Getinge AB.
- Terragene S.A. (2023). Rapid readout biological indicators and automated incubation systems. Terragene S.A.
Frequently Asked Questions
How big is the biological indicator vial market in 2026?
The global biological indicator vial market is valued at USD 178.7 million in 2026.
What will be the size of the biological indicator vial market in 2036?
The biological indicator vial market is projected to reach USD 279.1 million by 2036.
How much will the biological indicator vial market grow between 2026 and 2036?
The biological indicator vial market is expected to grow at a CAGR of 4.6% between 2026 and 2036.
What are the key sterilization types in the biological indicator vial market?
The primary sterilization categories include steam sterilization and ethylene oxide sterilization indicators.
Which incubation time segment contributes the largest share to the industry?
The 24 to 32 hours incubation segment represents the largest share, accounting for about 45% of the biological indicator vial market.
Table of Content
- Executive Summary
- Global Market Outlook
- Key Market Metrics
- Segment Highlights
- Regional Insights
- Key Recommendations
- Market Overview
- Market Definition and Scope
- Market Taxonomy
- Assumptions and Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunities
- Trends
- Scenario Forecast
- Optimistic Scenario
- Likely Scenario
- Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Five Forces
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Biological Indicator Vial Market Analysis 2026 to 2036
- Market Value Analysis, 2026 to 2036
- Market Growth Outlook and CAGR Analysis
- Year-on-Year Growth Trend Analysis
- Absolute Dollar Opportunity Analysis
- Pricing Analysis 2026 to 2036
- Market Analysis by Sterilization Type
- Steam Sterilization
- Ethylene Oxide Sterilization
- Market Value and Forecast by Sterilization Type
- Growth Trend and Opportunity Assessment
- Market Analysis by Incubation Time
- Up to 24 Hours
- 24 to 32 Hours
- 32 to 48 Hours
- Above 48 Hours
- Market Value and Forecast by Incubation Time
- Opportunity Assessment
- Market Analysis by End Use
- Food and Beverages
- Pharmaceutical
- Cosmetics
- Market Value and Forecast by End Use
- Opportunity Assessment
- Regional Market Analysis
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia Pacific
- Middle East and Africa
- Market Structure Analysis
- Competition Dashboard
- Market Share Analysis
- Competition Benchmarking
- Competitive Landscape
- Company Profiles
- Overview
- Product Portfolio
- Strategy Overview
- Regional Footprint
- Financial and Operational Highlights
- Company Profiles
- Research Methodology
- Acronyms and Abbreviations
- Bibliography
List of Tables
- Table 1: Global Market Value Forecast, 2026 to 2036
- Table 2: Market Value by Sterilization Type, 2026 to 2036
- Table 3: Market Value by Incubation Time, 2026 to 2036
- Table 4: Market Value by End Use, 2026 to 2036
- Table 5: Regional Market Value Forecast, 2026 to 2036
- Table 6: Country-Level Market Growth Rates
- Table 7: Pricing Analysis by Region
- Table 8: Opportunity Analysis by Segment
- Table 9: Competitive Presence by Region
- Table 10: Company Market Share Analysis
List of Figures
- Figure 1: Global Market Value and Growth Trend, 2026 to 2036
- Figure 2: Market Share by Sterilization Type
- Figure 3: Market Share by Incubation Time
- Figure 4: Market Share by End Use
- Figure 5: Regional Market Contribution
- Figure 6: Demand Outlook by Key Countries
- Figure 7: Drivers and Restraints Impact Analysis
- Figure 8: Value Chain Overview
- Figure 9: Competitive Landscape Mapping
- Figure 10: Technology Roadmap for Biological Indicators
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

