Vaccine and Biologic Product Distribution Optimization Services Market
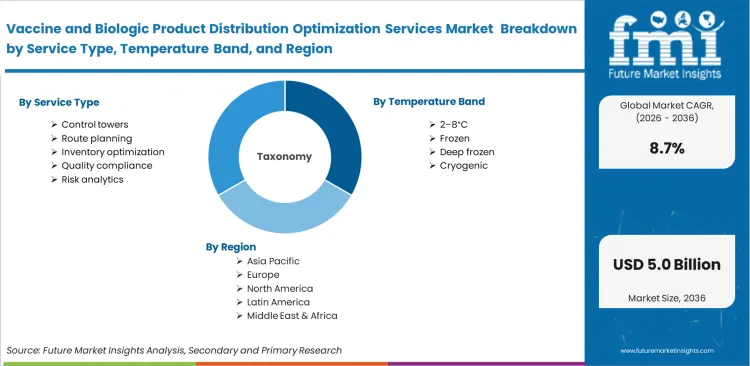
The Vaccine and Biologic Product Distribution Optimization Services Market is segmented by Service Type (Control towers, Route planning, Inventory optimization, Quality compliance, Risk analytics), Temperature Band (2–8°C, Frozen, Deep frozen, Cryogenic), Transport Mode (Air freight, Road freight, Multimodal, Ocean freight), Product Focus (Vaccines, Monoclonal antibodies, Plasma biologics, Cell therapies, Gene therapies), Buyer Type (Manufacturers, Government agencies, Specialty distributors, Hospitals, CDMOs), and Region. Forecast for 2026 to 2036.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Vaccine and Biologic Product Distribution Optimization Services Market Size, Market Forecast and Outlook By FMI
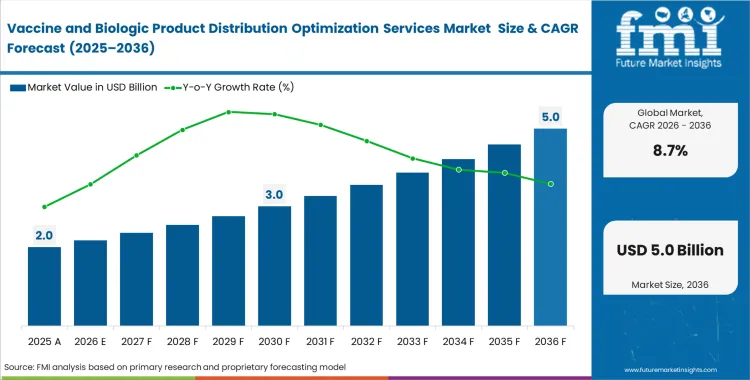
The Vaccine and Biologic Product Distribution Optimization Services Market crossed a valuation of USD 1.9 billion in 2025. Demand is poised to surpass USD 2.0 billion in 2026 at a CAGR of 8.7% during the forecast period. Revenue expansion lifts total valuation to USD 4.6 billion through 2036 as pharmaceutical procurement directors mandate strict zero-excursion qualification standards before assigning high-value cellular therapy loads.
Live-tracking gaps during multimodal transfers are creating a major risk point for temperature-sensitive biologics distribution. Manufacturers are under pressure to reduce product loss, since weak handoff standardization can disrupt shipment integrity and lead to batch write-offs. Procurement teams now place strong emphasis on cold chain exception management for vaccines because visibility failures can trigger compliance issues and financial losses. Buyers often assume freezer capacity is the main growth constraint, yet routing intelligence and response speed often have a greater effect on throughput across complex networks. Vendors without predictive alerting capabilities are finding it harder to compete, as biologics distribution optimization services are increasingly valued for the data layer they add to compliance control and operational stability.
Summary of Vaccine and Biologic Product Distribution Optimization Services Market
- Market Snapshot
- The Vaccine and Biologic Product Distribution Optimization Services Market is valued at USD 1.86 billion in 2025 and is projected to reach USD 4.61 billion by 2036.
- The market is forecast to expand at 8.6% CAGR from 2026 to 2036, creating an incremental opportunity of USD 2.59 billion over the period.
- This industry sits inside temperature-sensitive healthcare logistics and covers planning, orchestration, monitoring, and compliance services used to move vaccines and biologics without potency loss.
- The market remains compliance-led because vaccines and biologics move through multi-country chains that demand validated storage, qualified lanes, and GDP-aligned handling.
- Demand and Growth Drivers
- Demand is increasing as large-scale vaccine distribution programs continue to require tighter control over routing, inventory, and exception handling across multiple countries.
- Growth is being shaped by a wider mix of innovative therapies, which is making biologics distribution more complex and increasing the need for stronger optimization support.
- Demand for these services is also supported by persistent operational delays in customs, warehousing, and last-mile movement, where better service-layer coordination can help reduce inefficiencies.
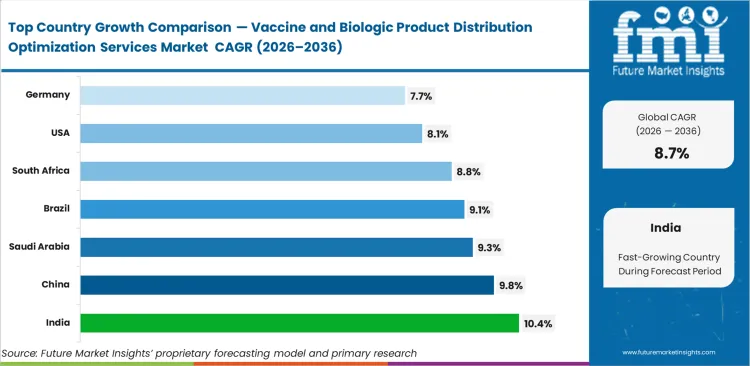
- Among key countries, India leads at 10.4% CAGR, followed by China at 9.8%, Saudi Arabia at 9.3%, Brazil at 9.1%, South Africa at 8.8%, the United States at 8.1%, and Germany at 7.7%.
- Growth is moderated by public-sector procurement cycles, fragmented last-mile infrastructure in lower-income markets, and the cost of meeting GDP, validation, and monitoring requirements across multiple temperature bands.
- Product and Segment View
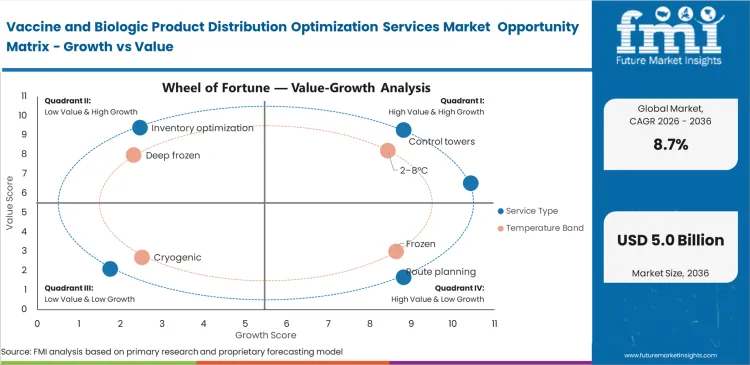
- The market covers distribution control towers, route planning, inventory optimization, quality-compliance support, and risk analytics used for vaccines, monoclonal antibodies, plasma biologics, cell therapies, and gene therapies.
- These services are applied across 2-8°C, frozen, deep-frozen, and cryogenic distribution flows and are most critical when air freight, cross-border warehousing, and high-value last-mile delivery intersect.
- Control towers lead the Service Type segment with 29.0% share, because centralized orchestration is the clearest way to manage temperature excursions, customs events, and inventory priorities in real time.
- 2-8°C leads the Temperature Band segment with 41.0% share, reflecting the continuing scale of routine vaccines and a broad installed base of refrigerated distribution assets.
- Air freight leads the Transport Mode segment with 46.0% share, supported by IATA’s CEIV Pharma network and the need for speed, consistency, and validated handling of time-sensitive biologics.
- Vaccines lead the Product Focus segment with 36.0% share, supported by global immunization campaigns, public procurement systems, and repeated outbreak-response deployments.
- Manufacturers lead the Buyer Type segment with 44.0% share, since biologic producers are under the greatest pressure to maintain product integrity, launch continuity, and compliant global distribution.
- The scope includes optimization services tied to warehousing, transport planning, monitoring, and GDP execution, while excluding basic parcel delivery, generic pharma wholesaling, non-temperature-sensitive drug logistics, and standalone packaging sales.
- Geography and Competitive Outlook
- India, China, Brazil, Saudi Arabia, and South Africa are the fastest-growing geographies, while the United States and Germany remain stable high-value markets with denser validated infrastructure and more sophisticated biologics portfolios.
- Competition is being shaped by global network density, certified cold-chain stations, digital visibility, and the ability to combine transport execution with control-tower planning and compliance support.
- Key participants include UPS Healthcare, DHL Supply Chain, Kuehne+Nagel, World Courier, FedEx, Cryoport Systems, and McKesson, with UPS Healthcare estimated to be the leading single player in this services niche.
- The market is moderately fragmented because the largest players have broad global networks, while specialized biologics operators still retain strong positions in high-touch and high-risk lanes.
Pharmaceutical sponsors are accelerating requirements for real-time visibility across third-party logistics networks, which is raising the standard for service providers. Companies that meet this validation threshold are in a stronger position to secure priority contracts from biological distributors. Regional carriers are also being pushed to operate as connected nodes within broader global supply chains rather than as isolated transport partners. Vaccine logistics control tower services support this shift by linking fragmented movements into a more coordinated monitoring structure. Logistics directors depend on centralized dashboards to identify disruptions early and protect shipment value.
Demand for biologics distribution optimization services in India is anticipated to rise at a CAGR of 10.4% through 2036. China is expected to expand at 9.8% CAGR during the forecast period as biotechnology activity lifts the need for precise material handling. Saudi Arabia is set to grow at 9.3% CAGR through 2036, supported by medical infrastructure modernization. Brazil is anticipated to record 9.1% CAGR, driven by stricter transport compliance needs. South Africa is likely to see 8.8% CAGR by 2036 as donor-backed healthcare distribution programs improve coordination needs. The United States is expected to advance at 8.1% CAGR through 2036 with continued commercialization of advanced therapies. Germany is forecast to post 7.7% CAGR over the study period, supported by established bio-manufacturing capacity.
Segmental Analysis
Vaccine and Biologic Product Distribution Optimization Services Market Analysis by Service Type
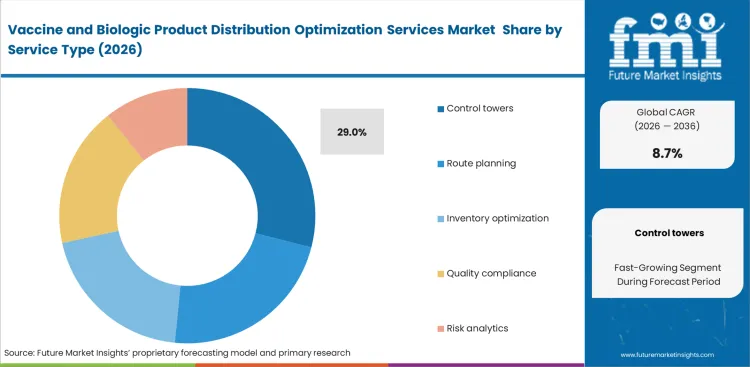
The control towers segment is estimated to account for 29.0% share in 2026, as logistics directors increasingly rely on centralized platforms to manage complex international handoffs. FMI estimates that vendors without integrated tracking infrastructure face weaker positioning when competing for tier-one biological distribution contracts. Standalone route algorithms often fall short when airport delays or cross-border disruptions interrupt planned movements. A less visible benefit of control tower adoption is its link with lower insurance exposure for high-value shipments, since stronger oversight improves response during transit exceptions. Operators without cold chain logistics synchronization remain more vulnerable to financial losses when disruptions occur. Vaccine logistics control tower services help convert scattered transport data into timely operational decisions.
- Tender qualification: Major pharmaceutical sponsors require centralized visibility platforms during initial vendor vetting. Logistics directors secure lucrative long-term contracts solely by demonstrating this capability.
- Data synchronization: Handoffs between air carriers and ground fleets expose cargo to extreme risk. IT directors implement seamless integration to prevent regulatory violations.
- Insurance reduction: Insurers lower rates for shipments tracked via advanced telemetry. Procurement heads capture immediate margin improvements through this risk mitigation.
Vaccine and Biologic Product Distribution Optimization Services Market Analysis by Temperature Band
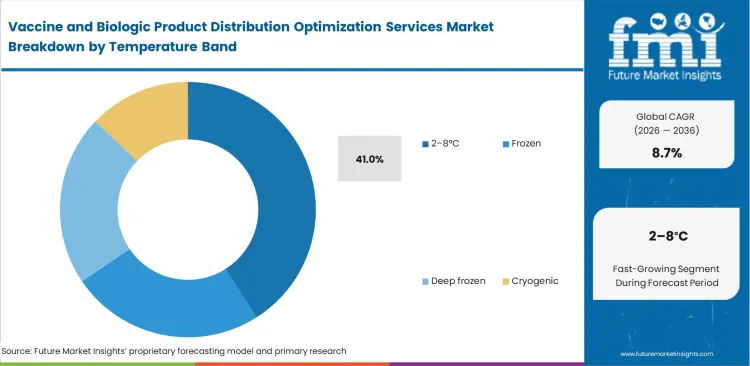
Conventional protein formulations still shape bulk shipment conditions across global biologics supply chains. The 2–8°C segment is estimated to account for 41.0% share in 2026 because most facilities are already built around this established temperature range. Existing storage and transport systems are easier to manage within these parameters, which keeps this range central to day-to-day operations. FMI notes that deep frozen alternatives require more specialized handling and tighter process control. Any shift away from standard refrigeration can bring added validation work, which makes execution more demanding for logistics teams. Ambient temperature controlled pharma packaging solutions can sometimes hide weak temperature control during tarmac transfers, where exposure risks rise quickly. Third-party providers still using older refrigeration systems face a higher chance of product loss when handling sensitive biologics. Biologics distribution optimization services help keep these thermal boundaries under closer control across the network.
- Infrastructure compatibility: Existing hospital refrigeration networks universally support standard cold ranges. Facility managers avoid adopting therapies requiring expensive cryogenic storage retrofits.
- Bulk economics: Shipping standard refrigerated loads costs significantly less than deep frozen cargo. Supply chain vice presidents maximize budgets by prioritizing stable formulations.
- Validation continuity: Changing established temperature parameters voids previous stability data. QA directors maintain existing bands to prevent costly regulatory delays.
Vaccine and Biologic Product Distribution Optimization Services Market Analysis by Transport Mode
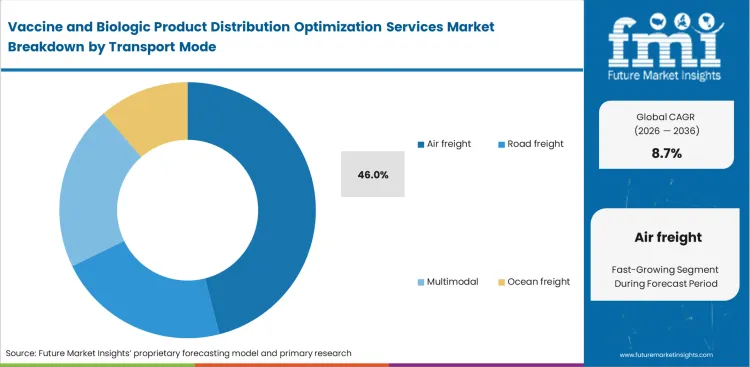
Short half-life therapies require fast transit across international markets. That urgency is expected to place air freight at 46.0% share in 2026. Logistics teams choose speed over freight savings when moving these high-value therapies. FMI analysis suggests ocean shipping cannot deliver within the narrow treatment windows tied to personalized cell therapies. Ground transport creates added handling stress and raises vibration-related risk during long-distance movement. Air freight also remains central because major airport hubs streamline customs clearance for temperature-sensitive biologics. Service providers that fail to secure lift capacity in peak periods often weaken their position with pharmaceutical shippers.
- Time criticality: Autologous cell therapies demand immediate return to patients. Operations managers select rapid aviation routes to preserve cellular viability.
- Customs bypass: Pre-cleared aviation channels bypass congested surface borders. Logistics planners utilize these expedited lanes for sensitive imports.
- Capacity bidding: Limited active container slots on passenger flights create fierce competition. Procurement directors must pre-purchase capacity to guarantee delivery windows.
Vaccine and Biologic Product Distribution Optimization Services Market Analysis by Product Focus
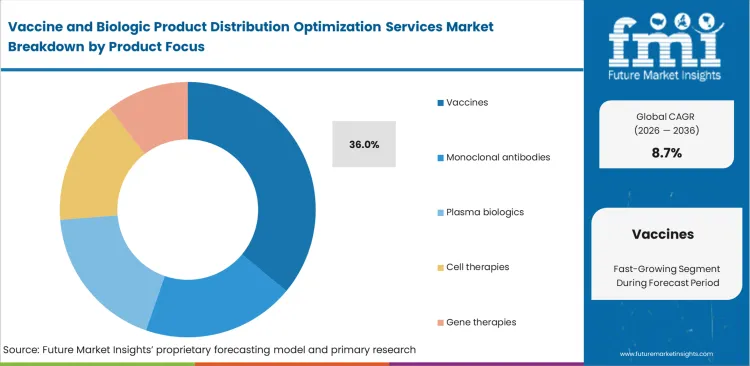
Mass immunization programs require steady replenishment across domestic and cross-border distribution networks. That scale is expected to place vaccines at 36.0% share in 2026. Public health agencies manage repeated high-volume movements to support routine immunization schedules, campaign rollouts, and emergency stock deployment. FMI analysis suggests specialized therapies serve smaller patient groups and move through more selective delivery channels. Vaccine logistics depend on broad cold chain coverage, coordinated procurement, and reliable distribution across regional healthcare systems. Long acting monoclonal antibodies move through separate channels and are not typically handled through the same networks used for large adult vaccine programs. Distributors without proven high-volume capabilities often struggle to qualify for major government tenders. Vaccine distribution optimization services help manage these large and recurring public health shipments.
- Tender volume: Government agencies purchase immunizations in massive annual blocks. Public health directors require distributors capable of handling sudden volume surges.
- Last-mile friction: Delivering vials to remote rural clinics tests network durability. Operations directors must establish specialized local partnerships to succeed.
- Formulation complexity: Novel mRNA constructs require stricter handling than traditional adjuvanted types. QA officers update transit protocols constantly to prevent degradation.
Vaccine and Biologic Product Distribution Optimization Services Market Drivers, Restraints, and Opportunities
Clinical logistics teams increasingly require predictive risk routing for monoclonal antibodies and advanced therapies to reduce that exposure. Vendors without live telemetry are becoming less viable in high-value pharmaceutical supply chains. Regulatory oversight also requires continuous temperature records from point of origin to final delivery. Freight providers are under growing pressure to move beyond basic tracking and build broader control tower capabilities. Pharmaceutical cold chain optimization services support this shift by improving shipment visibility, route control, and lane monitoring. Route optimization helps teams respond earlier to weather disruptions. Manufacturers also expect lane qualification before approving high-value transfers.
Legacy IT environments continue to limit data exchange across regional carrier networks. Transport teams often struggle to connect local tracking systems with global cold chain platforms, especially during multi-leg international movements. These disconnects reduce visibility at key airport handoff points where timing and temperature control matter most. Smaller carriers face a further disadvantage because network upgrades require capital many of them cannot easily commit. Industry groups are working toward open data standards, yet competing proprietary systems continue to slow alignment. These integration barriers raise the operating complexity and cost of outsourced vaccine distribution planning.
Opportunities in the Vaccine and Biologic Product Distribution Optimization Services Market
- AI routing: Integrating machine learning algorithms prevents weather-related transit delays. Operations managers ensure timely deliveries for veterinary vaccines.
- Return tracking: Monitoring return logistics for reusable thermal shippers improves asset utilization. Procurement heads lower overall transit expenses significantly.
- Direct care: Managing complex home infusions requires specialized micro-logistics. Pharmacy directors capture new revenue streams through personalized delivery models.
Regional Analysis
Based on regional analysis, vaccine and biologic product distribution optimization services market is segmented into Asia Pacific, Middle East and Africa, Latin America, North America, and Europe.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| India | 10.4% |
| China | 9.8% |
| Saudi Arabia | 9.3% |
| Brazil | 9.1% |
| South Africa | 8.8% |
| United States | 8.1% |
| Germany | 7.7% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Asia Pacific Vaccine and Biologic Product Distribution Optimization Services Market Analysis
Massive domestic manufacturing expansion forces rapid supply chain modernization across eastern hubs. Local pharmaceutical originators increasingly demand global-standard logistics to support aggressive export strategies. As per FMI's projection, regional carriers must upgrade tracking capabilities to secure outbound contracts. Fragmented provincial transport networks complicate unified visibility. Advanced tracking solutions bridge these infrastructural gaps. Upgrading local fleets with specialized telemetry ensures compliance with strict international quality standards. Biologics logistics service providers expand aggressively across this geography.
- India: Demand for high-value healthcare asset tracking and recovery platforms in India is anticipated to rise at a CAGR of 10.4% through 2036, supported by large biosimilars production volumes and stricter outbound shipment control requirements. Export compliance teams are placing greater emphasis on verified temperature records for Europe-bound consignments, where documentation quality directly affects shipment acceptance. Vendors that cannot offer strong digital integration are losing access to higher-value international forwarding programs, which is sharpening the market gap between basic tracking providers and integrated platform players.
- China: The market for high-value healthcare asset tracking and recovery platforms in China is expected to grow at a CAGR of 9.8% during the forecast period, supported by state-backed biotechnology parks and rising complexity in inbound materials logistics. Facility managers are demanding tighter delivery coordination for fragile cellular and biologic inputs, especially where timing and handling conditions influence production continuity. Regional forwarding companies that combine predictive routing with stronger platform connectivity are improving service reliability and securing a stronger competitive position.
Middle East and Africa Vaccine and Biologic Product Distribution Optimization Services Market Analysis
Centralized medical infrastructure investments attract specialized logistics providers. Government procurement agencies demand robust cold chain integrity for imported therapeutics. Supply chain directors establish unified distribution hubs to serve broader regional populations. FMI's analysis indicates that extreme ambient temperatures force rigorous lane validation protocols. Operators deploy advanced control towers to monitor sensitive cargo continuously. Vendors demonstrating flawless track records secure multi-year government contracts.
- Saudi Arabia: Sales of high-value healthcare asset tracking and recovery platforms in Saudi Arabia are set to record a CAGR of 9.3% during the assessment period, supported by Ministry of Health initiatives that are strengthening specialized medical logistics networks. Operations directors are adopting real-time monitoring systems to reduce the risk of heat-related product excursions across temperature-sensitive healthcare shipments. Companies that build resilient local distribution channels are likely to improve service continuity and secure stronger long-term commercial potential in the country.
- South Africa: The high-value healthcare asset tracking and recovery platforms market in South Africa is projected to witness a CAGR of 8.8% through 2036, driven by international donor-backed distribution programs and the need for better regional healthcare logistics coordination. Public health authorities are placing greater emphasis on routing efficiency for deliveries moving into remote clinics, where service gaps can disrupt medicine access and cold-chain reliability. Providers that reduce last-mile delivery friction are in a stronger position to capture meaningful public sector demand.
Latin America Vaccine and Biologic Product Distribution Optimization Services Market Analysis
Strict local transport regulations compel regional forwarders to adopt digital visibility platforms. Regulatory bodies mandate detailed temperature logs for all imported biologicals. Compliance officers audit inbound shipments rigorously. FMI observes that fragmented geography necessitates sophisticated multimodal planning. Upgrading legacy tracking systems becomes mandatory for carriers handling specialty pharma. Logistics directors partner with global control tower providers to ensure compliance.
- Brazil: Demand for high-value healthcare asset tracking and recovery platforms in Brazil is expected to increase at a CAGR of 9.1% through 2036, as complex domestic geography raises the need for tighter multimodal coordination for sensitive healthcare cargo. Transport directors are using predictive routing tools to work around infrastructure bottlenecks that can disrupt shipment timing and handling stability. Companies that build stronger domestic route optimization capabilities are better placed to win business from private hospital networks that value reliability across longer and more fragmented delivery corridors.
North America Vaccine and Biologic Product Distribution Optimization Services Market Analysis
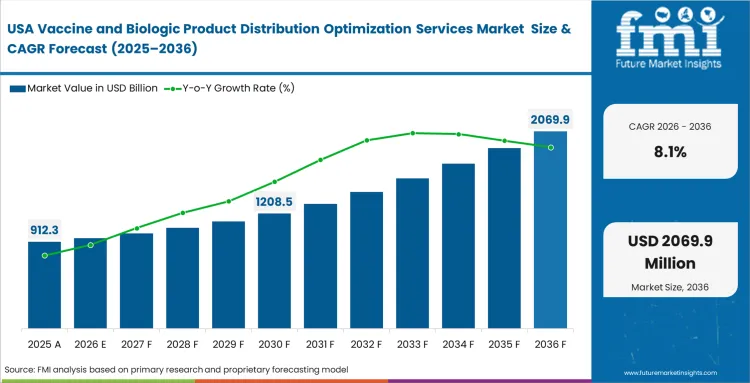
Advanced therapy commercialization dictates ultra-precise logistics across this territory. Clinical trial directors require zero-excursion guarantees for individualized patient treatments. High insurance liabilities compel operators to utilize predictive risk software. Cross-border coordination remains essential for complex manufacturing supply chains. Utilizing specialized optimization tools mitigates severe financial exposure during transit delays. Vendors demonstrating flawless track records command premium pricing. Pharmaceutical cold chain optimization services define operational baselines here.
- United States: Adoption of high-value healthcare asset tracking and recovery platforms in the United States is likely to advance at a CAGR of 8.1% by 2036, supported by the expansion of cellular therapy logistics and the need for flawless multimodal shipment handoffs. Clinical operations teams are relying on advanced control tower systems to monitor high-value patient-linked consignments where timing, condition, and chain of custody all matter. Providers that manage return logistics well for specialized packaging formats are likely to stand out more clearly in this market.
Europe Vaccine and Biologic Product Distribution Optimization Services Market Analysis
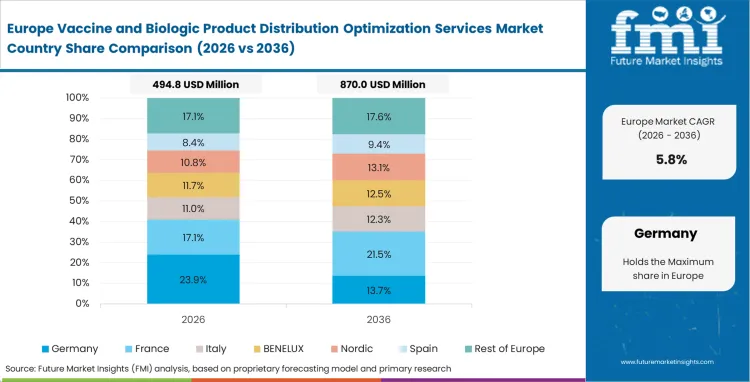
Strict good distribution practice enforcement shapes vendor selection across continental borders. Quality assurance officers reject carriers lacking continuous telemetry capabilities. Unified regulatory frameworks simplify cross-border software integration. Originators mandate complete visibility during complex pan-European transit routes. Sophisticated analytical platforms prevent costly compliance violations. Forwarders invest heavily in predictive software to maintain tier-one status. GDP-compliant vaccine logistics vendors dominate procurement discussions.
- Germany: The high-value healthcare asset tracking and recovery platforms sector in Germany is expected to register a CAGR of 7.7% through 2036, supported by strong bio-manufacturing hubs that ship large volumes of sensitive therapeutics into global markets. Logistics procurement managers are showing greater preference for vendors using AI-driven route optimization to improve shipment predictability and reduce handling risk. Deep integration with originator ERP systems is also raising switching costs, which strengthens the position of providers already embedded in established pharmaceutical logistics workflows.
Competitive Aligners for Market Players
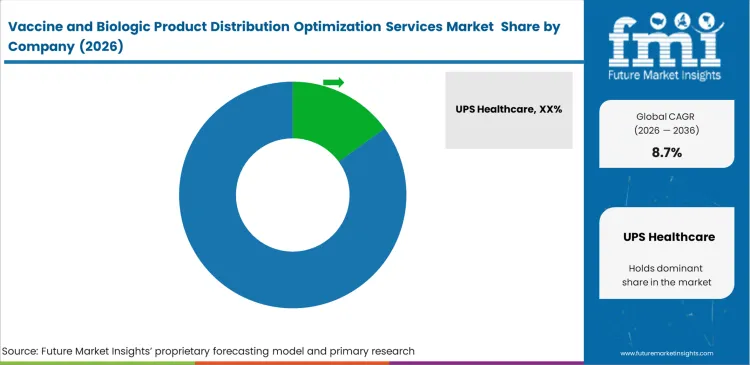
Specialized certification requirements create a clear barrier for general freight operators trying to enter medical logistics. Large players such as UPS Healthcare and DHL Supply Chain hold an advantage because they operate extensive networks of GDP-compliant and validated facilities across major trade lanes. Supply chain leaders in pharmaceutical companies do not select partners on price alone. Past excursion performance, lane reliability, and the ability to integrate operational data into wider planning systems carry far more weight in contract decisions. Smaller regional carriers rarely secure core originator programs unless they work alongside larger certified networks. This is becoming more visible as vaccine distribution planning shifts toward integrated providers that can manage compliance, monitoring, and execution at scale.
Established incumbents also benefit from deeper software integration with pharmaceutical originator ERP systems. Kuehne+Nagel and World Courier, for example, are better positioned where customers expect continuous digital visibility across shipment flows. Newer challengers cannot compete with basic tracking alone. They need stronger API connectivity between specialized fleets and centralized control towers, along with predictive risk tools that help operations teams act before a shipment issue turns into a product loss event. Temperature data by itself no longer meets the standard for high-value biological logistics. Buyers are also assessing companies such as FedEx and Cryoport Systems on their ability to validate lanes, manage exceptions, and support demanding biologic transport requirements.
Large pharmaceutical manufacturers are also taking steps to avoid overdependence on any one logistics partner. Many are dividing shipment volumes across multiple certified carriers while using neutral control tower platforms to compare performance across providers in real time. This reduces vendor lock-in and gives originators more leverage during procurement and network planning. Over time, wider adoption of interoperable data standards is likely to make basic tracking more interchangeable across the market. That shift will put more pressure on logistics providers to stand out through stronger exception response, better predictive routing, and tighter execution in complex healthcare distribution programs.
Key Players in Vaccine and Biologic Product Distribution Optimization Services Market
- UPS Healthcare
- DHL Supply Chain
- Kuehne+Nagel
- World Courier
- FedEx
- Cryoport Systems
- McKesson
Scope of the Report
| Metric | Value |
|---|---|
| Quantitative Units | USD 2.0 billion to USD 4.6 billion, at a CAGR of 8.7% |
| Market Definition | Digital tracking and predictive routing services govern complex biological supply chains. Such solutions prevent temperature excursions during high-value medical transit. |
| Segmentation | Service Type, Temperature Band, Transport Mode, Product Focus, Buyer Type, Region |
| Regions Covered | North America, Latin America, Europe, Asia Pacific, Middle East and Africa |
| Countries Covered | India, China, Saudi Arabia, Brazil, South Africa, United States, Germany |
| Key Companies Profiled | UPS Healthcare, DHL Supply Chain, Kuehne+Nagel, World Courier, FedEx, Cryoport Systems, McKesson |
| Forecast Period | 2026 to 2036 |
| Approach | Valuation based on specialized contract values and vendor earnings cross-referenced against global biological approval rates |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Vaccine and Biologic Product Distribution Optimization Services Market Analysis by Segments
Service Type
- Control towers
- Route planning
- Inventory optimization
- Quality compliance
- Risk analytics
Temperature Band
- 2-8°C
- Frozen
- Deep frozen
- Cryogenic
Transport Mode
- Air freight
- Road freight
- Multimodal
- Ocean freight
Product Focus
- Vaccines
- Monoclonal antibodies
- Plasma biologics
- Cell therapies
- Gene therapies
Buyer Type
- Manufacturers
- Government agencies
- Specialty distributors
- Hospitals
- CDMOs
Region
- North America
- Latin America
- Europe
- Asia Pacific
- Middle East and Africa
Bibliography
- Centers for Disease Control and Prevention. (2024). Vaccine storage and handling. U.S. Department of Health and Human Services.
- Food and Drug Administration. (2025). Vaccine and related biological product guidances. U.S. Department of Health and Human Services.
- IQVIA Institute for Human Data Science. (2025). The global use of medicines outlook through 2029.
- Nkwain, J., Ndzi, E., Ndzi, J., Ndzi, C., Ndzi, M., Ndzi, P., & collaborators. (2025). Deployment of vaccine cold chain equipment in resource-limited settings: Lessons from the Gavi Cold Chain Equipment Optimization Platform in Cameroon. International Health, 17(1), 33-41.
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary source documentation.
This Report Addresses
- Software integration latency between regional transport nodes.
- Autologous cell therapies demanding immediate return to patients.
- Deep frozen alternatives demanding specialized handling procedures.
- Originating entities controlling outbound logistics decisions.
- State-funded biotechnology parks demanding inbound materials logistics.
- Established bio-manufacturing hubs exporting sensitive therapeutics.
- Major incumbents possessing deep software integration.
- Interoperable data standards commoditizing basic tracking systems.
Frequently Asked Questions
What is the value of this sector in 2026?
Revenue reaches USD 2.0 billion in 2026. This figure reflects massive investments by major pharmaceutical originators securing global distribution capacity.
What is the projected value by 2036?
Sales climb to USD 4.6 billion by 2036. Strict regulatory audits require continuous telemetry systems across all biological shipments.
What is the expected CAGR?
Demand expands at 8.7% between 2026 and 2036. Cell therapy commercialization forces rapid upgrades to predictive risk routing tools.
Why do control towers lead the service segment?
Centralized platforms resolve fragmented visibility across multimodal transport networks. Logistics directors depend on these systems to manage complex international handoffs.
Why does 2-8°C hold the largest temperature share?
Conventional protein formulations dictate bulk shipment parameters globally. Facility managers optimize existing infrastructure around this established stability standard.
Why does air freight dominate transport modes?
Short half-life therapies dictate rapid global transit regardless of expense. Operations managers prioritize speed over unit cost for autologous cell treatments.
Why do vaccines lead the product focus?
Mass immunization campaigns demand continuous global replenishment cycles. Public health directors coordinate massive regional distributions annually to maintain coverage.
Why do manufacturers control the buyer segment?
Originating entities dictate which third-party carriers touch active pharmaceutical ingredients. Supply chain vice presidents instantly disqualify carriers failing GDP adherence.
What drives growth in India?
Massive biosimilars production volumes require strict outbound tracking. Export compliance officers mandate verified temperature logs for European shipments.
What drives growth in China?
State-funded biotechnology parks demand sophisticated inbound materials logistics. Facility managers require precise delivery windows for fragile cellular inputs.
What drives growth in Saudi Arabia?
Ministry of Health initiatives prioritize specialized medical logistics networks. Operations directors deploy real-time monitoring to prevent heat-related excursions.
What drives growth in Brazil?
Complex geography requires precise multimodal orchestration for sensitive cargo. Transport directors utilize predictive algorithms to navigate infrastructure bottlenecks.
What drives growth in South Africa?
International donor coordination programs rely on optimized regional distribution. Public health officials require sophisticated routing to reach remote clinics safely.
What drives growth in the United States?
Cellular therapy scale-up necessitates flawless multimodal handoffs. Clinical operations directors deploy advanced control towers to monitor critical patient shipments.
What drives growth in Germany?
Established bio-manufacturing hubs export immense volumes of sensitive therapeutics globally. Logistics procurement managers prioritize vendors utilizing AI-driven route optimization.
How does software integration affect operations?
Handoffs between air carriers and ground fleets expose cargo to extreme risk. Seamless digital connections prevent costly regulatory violations during transfers.
How do custom boundaries shape strategy?
Pre-cleared aviation channels bypass congested surface borders. Logistics planners utilize these expedited lanes for sensitive medical imports.
How do capacity constraints impact planning?
Limited active container slots on passenger flights create fierce competition. Procurement directors must pre-purchase space to guarantee delivery windows.
How do manufacturers manage vendor risk?
Handing off cargo to certified forwarders transfers financial risk. Procurement directors prioritize vendors offering complete insurance coverage.
How do data silos restrict efficiency?
Siloed legacy IT systems prevent seamless data sharing across multiple regional carriers. Data fragmentation causes severe visibility gaps during airport handoffs.
How do AI routing algorithms help?
Integrating machine learning algorithms prevents weather-related transit delays. Operations managers ensure timely deliveries by predicting disruptive events proactively.
How does return tracking reduce costs?
Monitoring return logistics for reusable cold chain shippers improves asset utilization. Procurement heads lower overall transit expenses significantly through tracking.
How do direct care models work?
Managing complex home infusions requires specialized micro-logistics. Pharmacy directors capture new revenue streams through personalized care models.
How do originators prevent lock-in?
Large pharmaceutical manufacturers mitigate vendor lock-in by splitting volumes across multiple certified carriers. Originators utilize agnostic control tower software to monitor competing providers simultaneously.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Service Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Service Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Service Type , 2026 to 2036
- Control towers
- Route planning
- Inventory optimization
- Quality compliance
- Risk analytics
- Control towers
- Y to o to Y Growth Trend Analysis By Service Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Service Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Temperature Band
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Temperature Band, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Temperature Band, 2026 to 2036
- 2–8°C
- Frozen
- Deep frozen
- Cryogenic
- 2–8°C
- Y to o to Y Growth Trend Analysis By Temperature Band, 2021 to 2025
- Absolute $ Opportunity Analysis By Temperature Band, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Transport Mode
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Transport Mode, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Transport Mode, 2026 to 2036
- Air freight
- Road freight
- Multimodal
- Ocean freight
- Air freight
- Y to o to Y Growth Trend Analysis By Transport Mode, 2021 to 2025
- Absolute $ Opportunity Analysis By Transport Mode, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product Focus
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product Focus, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product Focus, 2026 to 2036
- Vaccines
- Monoclonal antibodies
- Plasma biologics
- Cell therapies
- Gene therapies
- Vaccines
- Y to o to Y Growth Trend Analysis By Product Focus, 2021 to 2025
- Absolute $ Opportunity Analysis By Product Focus, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Service Type
- By Temperature Band
- By Transport Mode
- By Product Focus
- By Country
- Market Attractiveness Analysis
- By Country
- By Service Type
- By Temperature Band
- By Transport Mode
- By Product Focus
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Service Type
- By Temperature Band
- By Transport Mode
- By Product Focus
- By Country
- Market Attractiveness Analysis
- By Country
- By Service Type
- By Temperature Band
- By Transport Mode
- By Product Focus
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Service Type
- By Temperature Band
- By Transport Mode
- By Product Focus
- By Country
- Market Attractiveness Analysis
- By Country
- By Service Type
- By Temperature Band
- By Transport Mode
- By Product Focus
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Service Type
- By Temperature Band
- By Transport Mode
- By Product Focus
- By Country
- Market Attractiveness Analysis
- By Country
- By Service Type
- By Temperature Band
- By Transport Mode
- By Product Focus
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Service Type
- By Temperature Band
- By Transport Mode
- By Product Focus
- By Country
- Market Attractiveness Analysis
- By Country
- By Service Type
- By Temperature Band
- By Transport Mode
- By Product Focus
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Service Type
- By Temperature Band
- By Transport Mode
- By Product Focus
- By Country
- Market Attractiveness Analysis
- By Country
- By Service Type
- By Temperature Band
- By Transport Mode
- By Product Focus
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Service Type
- By Temperature Band
- By Transport Mode
- By Product Focus
- By Country
- Market Attractiveness Analysis
- By Country
- By Service Type
- By Temperature Band
- By Transport Mode
- By Product Focus
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Temperature Band
- By Transport Mode
- By Product Focus
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Temperature Band
- By Transport Mode
- By Product Focus
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Temperature Band
- By Transport Mode
- By Product Focus
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Temperature Band
- By Transport Mode
- By Product Focus
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Temperature Band
- By Transport Mode
- By Product Focus
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Temperature Band
- By Transport Mode
- By Product Focus
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Temperature Band
- By Transport Mode
- By Product Focus
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Temperature Band
- By Transport Mode
- By Product Focus
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Temperature Band
- By Transport Mode
- By Product Focus
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Temperature Band
- By Transport Mode
- By Product Focus
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Temperature Band
- By Transport Mode
- By Product Focus
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Temperature Band
- By Transport Mode
- By Product Focus
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Temperature Band
- By Transport Mode
- By Product Focus
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Temperature Band
- By Transport Mode
- By Product Focus
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Temperature Band
- By Transport Mode
- By Product Focus
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Temperature Band
- By Transport Mode
- By Product Focus
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Temperature Band
- By Transport Mode
- By Product Focus
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Temperature Band
- By Transport Mode
- By Product Focus
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Temperature Band
- By Transport Mode
- By Product Focus
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Temperature Band
- By Transport Mode
- By Product Focus
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Temperature Band
- By Transport Mode
- By Product Focus
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Temperature Band
- By Transport Mode
- By Product Focus
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Service Type
- By Temperature Band
- By Transport Mode
- By Product Focus
- Competition Analysis
- Competition Deep Dive
- UPS Healthcare
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- DHL Supply Chain
- Kuehne+Nagel
- World Courier
- FedEx
- Cryoport Systems
- McKesson
- UPS Healthcare
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Service Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Temperature Band, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Transport Mode, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by Product Focus, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Service Type , 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Temperature Band, 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Transport Mode, 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by Product Focus, 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by Service Type , 2021 to 2036
- Table 13: Latin America Market Value (USD Million) Forecast by Temperature Band, 2021 to 2036
- Table 14: Latin America Market Value (USD Million) Forecast by Transport Mode, 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Product Focus, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: Western Europe Market Value (USD Million) Forecast by Service Type , 2021 to 2036
- Table 18: Western Europe Market Value (USD Million) Forecast by Temperature Band, 2021 to 2036
- Table 19: Western Europe Market Value (USD Million) Forecast by Transport Mode, 2021 to 2036
- Table 20: Western Europe Market Value (USD Million) Forecast by Product Focus, 2021 to 2036
- Table 21: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: Eastern Europe Market Value (USD Million) Forecast by Service Type , 2021 to 2036
- Table 23: Eastern Europe Market Value (USD Million) Forecast by Temperature Band, 2021 to 2036
- Table 24: Eastern Europe Market Value (USD Million) Forecast by Transport Mode, 2021 to 2036
- Table 25: Eastern Europe Market Value (USD Million) Forecast by Product Focus, 2021 to 2036
- Table 26: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 27: East Asia Market Value (USD Million) Forecast by Service Type , 2021 to 2036
- Table 28: East Asia Market Value (USD Million) Forecast by Temperature Band, 2021 to 2036
- Table 29: East Asia Market Value (USD Million) Forecast by Transport Mode, 2021 to 2036
- Table 30: East Asia Market Value (USD Million) Forecast by Product Focus, 2021 to 2036
- Table 31: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 32: South Asia and Pacific Market Value (USD Million) Forecast by Service Type , 2021 to 2036
- Table 33: South Asia and Pacific Market Value (USD Million) Forecast by Temperature Band, 2021 to 2036
- Table 34: South Asia and Pacific Market Value (USD Million) Forecast by Transport Mode, 2021 to 2036
- Table 35: South Asia and Pacific Market Value (USD Million) Forecast by Product Focus, 2021 to 2036
- Table 36: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 37: Middle East & Africa Market Value (USD Million) Forecast by Service Type , 2021 to 2036
- Table 38: Middle East & Africa Market Value (USD Million) Forecast by Temperature Band, 2021 to 2036
- Table 39: Middle East & Africa Market Value (USD Million) Forecast by Transport Mode, 2021 to 2036
- Table 40: Middle East & Africa Market Value (USD Million) Forecast by Product Focus, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Service Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Service Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Service Type
- Figure 6: Global Market Value Share and BPS Analysis by Temperature Band, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Temperature Band, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Temperature Band
- Figure 9: Global Market Value Share and BPS Analysis by Transport Mode, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Transport Mode, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Transport Mode
- Figure 12: Global Market Value Share and BPS Analysis by Product Focus, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Product Focus, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Product Focus
- Figure 15: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 17: Global Market Attractiveness Analysis by Region
- Figure 18: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 20: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 21: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 23: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 24: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 25: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 26: North America Market Value Share and BPS Analysis by Service Type , 2026 and 2036
- Figure 27: North America Market Y-o-Y Growth Comparison by Service Type , 2026-2036
- Figure 28: North America Market Attractiveness Analysis by Service Type
- Figure 29: North America Market Value Share and BPS Analysis by Temperature Band, 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Temperature Band, 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Temperature Band
- Figure 32: North America Market Value Share and BPS Analysis by Transport Mode, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Transport Mode, 2026-2036
- Figure 34: North America Market Attractiveness Analysis by Transport Mode
- Figure 35: North America Market Value Share and BPS Analysis by Product Focus, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by Product Focus, 2026-2036
- Figure 37: North America Market Attractiveness Analysis by Product Focus
- Figure 38: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 39: Latin America Market Value Share and BPS Analysis by Service Type , 2026 and 2036
- Figure 40: Latin America Market Y-o-Y Growth Comparison by Service Type , 2026-2036
- Figure 41: Latin America Market Attractiveness Analysis by Service Type
- Figure 42: Latin America Market Value Share and BPS Analysis by Temperature Band, 2026 and 2036
- Figure 43: Latin America Market Y-o-Y Growth Comparison by Temperature Band, 2026-2036
- Figure 44: Latin America Market Attractiveness Analysis by Temperature Band
- Figure 45: Latin America Market Value Share and BPS Analysis by Transport Mode, 2026 and 2036
- Figure 46: Latin America Market Y-o-Y Growth Comparison by Transport Mode, 2026-2036
- Figure 47: Latin America Market Attractiveness Analysis by Transport Mode
- Figure 48: Latin America Market Value Share and BPS Analysis by Product Focus, 2026 and 2036
- Figure 49: Latin America Market Y-o-Y Growth Comparison by Product Focus, 2026-2036
- Figure 50: Latin America Market Attractiveness Analysis by Product Focus
- Figure 51: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 52: Western Europe Market Value Share and BPS Analysis by Service Type , 2026 and 2036
- Figure 53: Western Europe Market Y-o-Y Growth Comparison by Service Type , 2026-2036
- Figure 54: Western Europe Market Attractiveness Analysis by Service Type
- Figure 55: Western Europe Market Value Share and BPS Analysis by Temperature Band, 2026 and 2036
- Figure 56: Western Europe Market Y-o-Y Growth Comparison by Temperature Band, 2026-2036
- Figure 57: Western Europe Market Attractiveness Analysis by Temperature Band
- Figure 58: Western Europe Market Value Share and BPS Analysis by Transport Mode, 2026 and 2036
- Figure 59: Western Europe Market Y-o-Y Growth Comparison by Transport Mode, 2026-2036
- Figure 60: Western Europe Market Attractiveness Analysis by Transport Mode
- Figure 61: Western Europe Market Value Share and BPS Analysis by Product Focus, 2026 and 2036
- Figure 62: Western Europe Market Y-o-Y Growth Comparison by Product Focus, 2026-2036
- Figure 63: Western Europe Market Attractiveness Analysis by Product Focus
- Figure 64: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 65: Eastern Europe Market Value Share and BPS Analysis by Service Type , 2026 and 2036
- Figure 66: Eastern Europe Market Y-o-Y Growth Comparison by Service Type , 2026-2036
- Figure 67: Eastern Europe Market Attractiveness Analysis by Service Type
- Figure 68: Eastern Europe Market Value Share and BPS Analysis by Temperature Band, 2026 and 2036
- Figure 69: Eastern Europe Market Y-o-Y Growth Comparison by Temperature Band, 2026-2036
- Figure 70: Eastern Europe Market Attractiveness Analysis by Temperature Band
- Figure 71: Eastern Europe Market Value Share and BPS Analysis by Transport Mode, 2026 and 2036
- Figure 72: Eastern Europe Market Y-o-Y Growth Comparison by Transport Mode, 2026-2036
- Figure 73: Eastern Europe Market Attractiveness Analysis by Transport Mode
- Figure 74: Eastern Europe Market Value Share and BPS Analysis by Product Focus, 2026 and 2036
- Figure 75: Eastern Europe Market Y-o-Y Growth Comparison by Product Focus, 2026-2036
- Figure 76: Eastern Europe Market Attractiveness Analysis by Product Focus
- Figure 77: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 78: East Asia Market Value Share and BPS Analysis by Service Type , 2026 and 2036
- Figure 79: East Asia Market Y-o-Y Growth Comparison by Service Type , 2026-2036
- Figure 80: East Asia Market Attractiveness Analysis by Service Type
- Figure 81: East Asia Market Value Share and BPS Analysis by Temperature Band, 2026 and 2036
- Figure 82: East Asia Market Y-o-Y Growth Comparison by Temperature Band, 2026-2036
- Figure 83: East Asia Market Attractiveness Analysis by Temperature Band
- Figure 84: East Asia Market Value Share and BPS Analysis by Transport Mode, 2026 and 2036
- Figure 85: East Asia Market Y-o-Y Growth Comparison by Transport Mode, 2026-2036
- Figure 86: East Asia Market Attractiveness Analysis by Transport Mode
- Figure 87: East Asia Market Value Share and BPS Analysis by Product Focus, 2026 and 2036
- Figure 88: East Asia Market Y-o-Y Growth Comparison by Product Focus, 2026-2036
- Figure 89: East Asia Market Attractiveness Analysis by Product Focus
- Figure 90: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 91: South Asia and Pacific Market Value Share and BPS Analysis by Service Type , 2026 and 2036
- Figure 92: South Asia and Pacific Market Y-o-Y Growth Comparison by Service Type , 2026-2036
- Figure 93: South Asia and Pacific Market Attractiveness Analysis by Service Type
- Figure 94: South Asia and Pacific Market Value Share and BPS Analysis by Temperature Band, 2026 and 2036
- Figure 95: South Asia and Pacific Market Y-o-Y Growth Comparison by Temperature Band, 2026-2036
- Figure 96: South Asia and Pacific Market Attractiveness Analysis by Temperature Band
- Figure 97: South Asia and Pacific Market Value Share and BPS Analysis by Transport Mode, 2026 and 2036
- Figure 98: South Asia and Pacific Market Y-o-Y Growth Comparison by Transport Mode, 2026-2036
- Figure 99: South Asia and Pacific Market Attractiveness Analysis by Transport Mode
- Figure 100: South Asia and Pacific Market Value Share and BPS Analysis by Product Focus, 2026 and 2036
- Figure 101: South Asia and Pacific Market Y-o-Y Growth Comparison by Product Focus, 2026-2036
- Figure 102: South Asia and Pacific Market Attractiveness Analysis by Product Focus
- Figure 103: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 104: Middle East & Africa Market Value Share and BPS Analysis by Service Type , 2026 and 2036
- Figure 105: Middle East & Africa Market Y-o-Y Growth Comparison by Service Type , 2026-2036
- Figure 106: Middle East & Africa Market Attractiveness Analysis by Service Type
- Figure 107: Middle East & Africa Market Value Share and BPS Analysis by Temperature Band, 2026 and 2036
- Figure 108: Middle East & Africa Market Y-o-Y Growth Comparison by Temperature Band, 2026-2036
- Figure 109: Middle East & Africa Market Attractiveness Analysis by Temperature Band
- Figure 110: Middle East & Africa Market Value Share and BPS Analysis by Transport Mode, 2026 and 2036
- Figure 111: Middle East & Africa Market Y-o-Y Growth Comparison by Transport Mode, 2026-2036
- Figure 112: Middle East & Africa Market Attractiveness Analysis by Transport Mode
- Figure 113: Middle East & Africa Market Value Share and BPS Analysis by Product Focus, 2026 and 2036
- Figure 114: Middle East & Africa Market Y-o-Y Growth Comparison by Product Focus, 2026-2036
- Figure 115: Middle East & Africa Market Attractiveness Analysis by Product Focus
- Figure 116: Global Market - Tier Structure Analysis
- Figure 117: Global Market - Company Share Analysis

