Lipid Nanoparticle Reagents Kits Market : Global Industry Analysis 2015 - 2024 and Opportunity Assessment 2025 - 2035
Lipid Nanoparticle Reagents Kits Market Analysis - Size, Share, and Forecast Outlook 2025 to 2035
Historical Data Covered: 2015 - 2024 | Base Year: 2024 | Estimated Year: 2025 | Forecast Period: 2025 - 2035
Lipid Nanoparticle Reagents Kits Market Forecast and Outlook 2025 to 2035
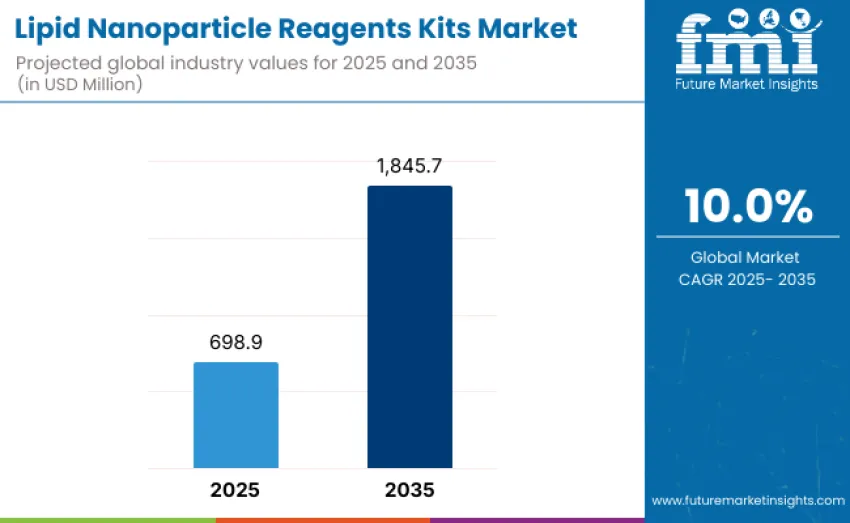
The global lipid nanoparticle reagents kits market is projected to reach USD 1,845.7 million by 2035, recording an absolute increase of USD 1,146.8 million over the forecast period. This market is valued at USD 698.9 million in 2025 and is set to rise at a CAGR of 10% during the assessment period. Overall size is anticipated to expand during this timeframe, supported by rising investments in mRNA-based vaccine development, gene therapy platforms, and next-generation drug delivery research across biotechnology and pharmaceutical sectors globally.
Technological advancements in lipid composition optimization, microfluidic mixing systems, and formulation stability are reshaping the lipid nanoparticle reagents kits landscape. Modern kits integrate proprietary cationic and ionizable lipid formulations designed for improved endosomal escape and payload release, while modular reagent systems support flexible screening of lipid ratios to achieve desired physicochemical properties. Compatibility with leading automated synthesis and analytical platforms including microfluidic mixers, nanoparticle tracking analyzers, and dynamic light scattering systems facilitates seamless integration into preclinical and process development workflows.
Quick Stats for Lipid Nanoparticle Reagents Kits Market
- Lipid Nanoparticle Reagents Kits Market Value (2025): USD 698.9 million
- Lipid Nanoparticle Reagents Kits Market Forecast Value (2035): USD 1,845.7 million
- Lipid Nanoparticle Reagents Kits Market Forecast CAGR: 10%
- Leading Product Type in Lipid Nanoparticle Reagents Kits Market: Lipid Formulation Kits with 38.5% share
- Key Growth Regions in Lipid Nanoparticle Reagents Kits Market: Asia Pacific, Europe, and North America
- Top Players in Lipid Nanoparticle Reagents Kits Market: Precision NanoSystems (Danaher), Evonik Industries AG, Polymun Scientific, Acuitas Therapeutics, Merck KGaA, Avanti Polar Lipids (Croda), CordenPharma International, BioNTech Manufacturing Services, Nitto Denko/Reprocell
Government funding for RNA-based therapeutic development, along with public-private partnerships supporting pandemic preparedness and personalized medicine research, are accelerating growth. Academic and translational research institutions increasingly adopt LNP reagent kits to support RNA delivery studies, oncology research, and vaccine platform development, while biotechnology companies leverage them to streamline early-stage formulation screening. The continued expansion of mRNA vaccine pipelines, gene therapy clinical trials, and nanomedicine innovation programs across North America, Europe, and Asia-Pacific sustains long-term demand for high-quality reagent kits.
Lipid Nanoparticle Reagents Kits Market Year-over-Year Forecast (2025 to 2035)
Between 2025 and 2030, the Lipid Nanoparticle Reagents Kits Market is projected to expand from USD 698.9 million to USD 1,182 million, resulting in a value increase of USD 483.1 million, which represents 42.1% of total forecast growth for the decade. This phase will be shaped by rising demand for mRNA-based vaccine research, increasing adoption of lipid nanoparticle delivery systems in gene therapy and RNA interference studies, and expanding use of LNP formulation kits in preclinical and translational research laboratories.
From 2030 to 2035, growth is forecast from USD 1,182 million to USD 1,845.7 million, adding USD 663.7 million, which constitutes 57.9% of overall ten-year expansion. This period is expected to be characterized by commercial maturation of RNA-based therapeutics, large-scale adoption of LNP technologies in gene therapy manufacturing, and integration of automated formulation and analytical platforms into drug development workflows. Strategic collaborations between biotechnology companies, academic research centers, and reagent manufacturers will drive innovation in lipid chemistry, process scalability, and reproducibility.
Lipid Nanoparticle Reagents Kits Market Key Takeaways
| Metric | Value |
|---|---|
| Market Value (2025) | USD 698.9 million |
| Market Forecast Value (2035) | USD 1,845.7 million |
| Forecast CAGR (2025 to 2035) | 10% |
Why Is the Lipid Nanoparticle Reagents Kits Market Growing Worldwide?
The Lipid Nanoparticle Reagents Kits Market is growing worldwide by enabling biotechnology and pharmaceutical researchers to achieve superior precision and formulation efficiency in developing RNA-based therapeutics and advanced drug delivery systems. Scientists and process engineers face increasing pressure to produce stable, high-performance lipid nanoparticle formulations that ensure efficient nucleic acid delivery, consistent encapsulation, and reproducible particle size distribution across large-scale research and production environments. LNP reagent kits provide optimized lipid mixtures and standardized formulation protocols that improve workflow efficiency by 30-50% compared to conventional formulation methods.
Government initiatives promoting biopharmaceutical research and national vaccine self-reliance programs are driving adoption across academic institutions, contract research organizations, and clinical-stage biotech firms. Public-private collaborations supporting pandemic preparedness, oncology research, and personalized medicine development stimulate the use of LNP reagent kits in translational and applied research projects. The global shift toward advanced RNA therapeutics, accelerated by continuous progress in microfluidic formulation systems and automated process technologies, reinforces long-term demand for these reagents.
How Is the Lipid Nanoparticle Reagents Kits Market Segmented by Product Type, Application, and Region?
The lipid nanoparticle reagents kits market is segmented by product type, application, and region. By product type, division includes lipid formulation kits, nucleic acid encapsulation reagents, stabilization & cryoprotectant kits, surface functionalization reagents, and purification & characterization kits. Based on application, categorization covers mRNA vaccines, gene therapy & siRNA delivery, drug delivery research, cell therapy manufacturing, and protein replacement therapy. Regionally, segmentation spans Asia Pacific, Europe, North America, Latin America, and Middle East & Africa.
By Product Type, Lipid Formulation Kits Account for Largest Share
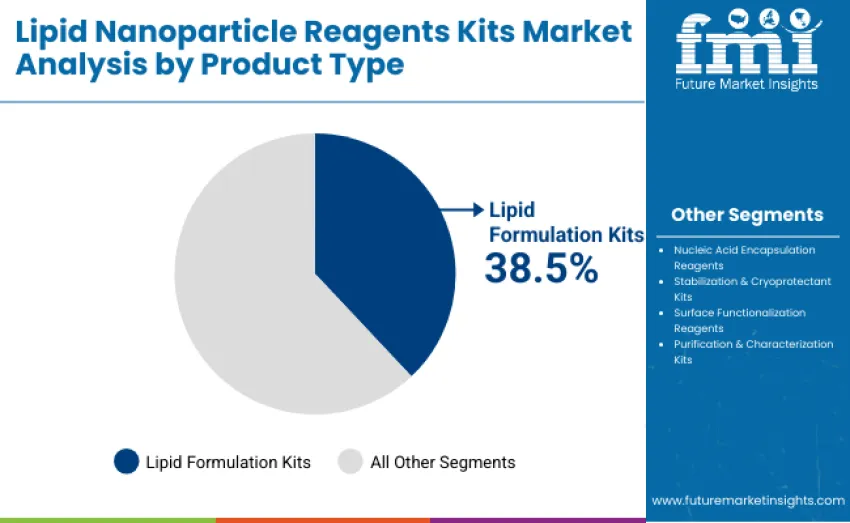
The lipid formulation kits segment represents the dominant force, capturing approximately 38.5% of total share in 2025. This advanced category encompasses essential lipid component mixtures designed to optimize nanoparticle formation, control particle uniformity, and enhance nucleic acid encapsulation efficiency, delivering consistent performance across research, clinical, and large-scale biomanufacturing applications. Leadership stems from its critical role in enabling reproducible formulation of mRNA vaccines, gene therapies, and RNA-based therapeutics where stability, scalability, and delivery efficiency determine project success.
The nucleic acid encapsulation reagents segment maintains a substantial 26.0% share, serving researchers and biopharmaceutical manufacturers developing RNA and DNA-based delivery systems that require precise encapsulation ratios and enhanced payload protection. Stabilization and cryoprotectant kits account for 13.0%, supporting long-term LNP preservation, cold-chain stability, and bioactivity retention during storage and transportation. Surface functionalization reagents contribute 12.5%, while purification & characterization kits represent 10.0% of revenue.
Key advantages driving the lipid formulation kits segment include:
- High formulation precision supporting consistent particle size and lipid composition critical for reproducible therapeutic performance
- Compatibility with diverse lipid chemistries enabling flexible formulation design for mRNA, siRNA, and gene delivery platforms
- Scalability across R&D to GMP manufacturing workflows ensuring seamless process translation
By Application, mRNA Vaccines Account for Largest Share
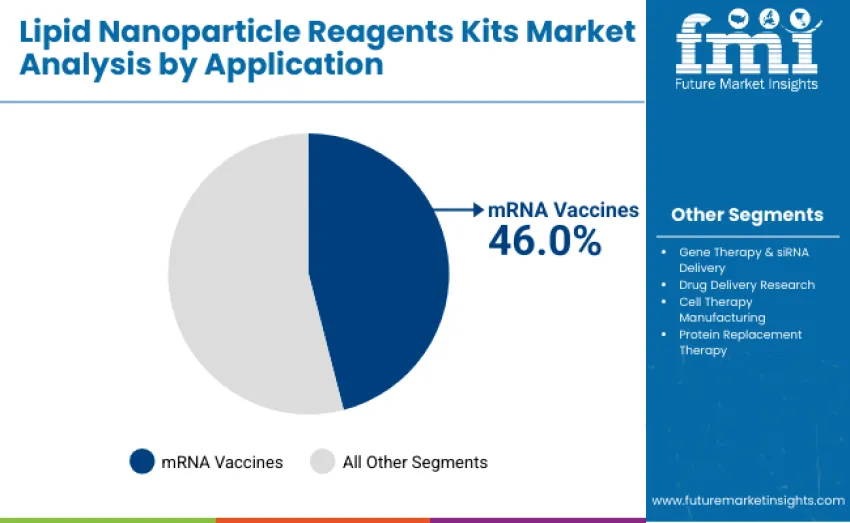
The mRNA vaccines segment dominates, capturing approximately 46.0% of total share in 2025. This leadership is attributed to the expanding adoption of mRNA vaccine platforms across infectious diseases, oncology, and pandemic preparedness programs. Pharmaceutical and biotech companies continue to invest heavily in lipid formulation optimization and scalable reagent systems that ensure high encapsulation efficiency and stability for mRNA payloads. Strong performance is reinforced by ongoing global vaccination initiatives and next-generation mRNA pipelines targeting influenza, RSV, cytomegalovirus, and personalized cancer vaccines.
The gene therapy and siRNA delivery segment follows with 26.5%, supported by increasing utilization of lipid nanoparticles in delivering genetic payloads for rare diseases, immune disorders, and gene-editing applications. Drug delivery research accounts for 15.0%, cell therapy manufacturing contributes 7.5%, and protein replacement therapy represents 5.0% of revenue.
Key market dynamics supporting application preferences include:
- Strong pipeline growth in mRNA-based vaccines targeting influenza, RSV, Zika, and cancer neoantigens, reinforcing large-scale adoption of lipid nanoparticle reagent kits
- Expansion of gene therapy research utilizing lipid-based vectors for safe and efficient genetic material delivery
- Increased investment by CDMOs and biopharma companies in scalable lipid nanoparticle manufacturing capabilities for clinical and commercial supply
What Are the Major Drivers, Restraints, and Emerging Trends Influencing the Lipid Nanoparticle Reagents Kits Market?
The lipid nanoparticle reagents kits market is driven by three concrete demand factors tied to the rapid evolution of biopharmaceutical research and therapeutic development workflows. Expansion of mRNA-based vaccine and therapeutics pipelines creates sustained requirements for standardized lipid nanoparticle formulation tools, with global RNA-based drug development activities growing by 10-15% annually and demanding proven reagent technologies that ensure encapsulation efficiency, reproducibility, and scalability. Acceleration of gene therapy and RNA interference research drives adoption of LNP reagent kits across both academic and industrial research programs, as these products enable streamlined formulation optimization.
What Are the Major Restraints Limiting Adoption in the LNP Reagents Kits Market?
Restraints in the market stem primarily from the high costs linked to specialized lipid components and formulation-grade reagents, which pose financial challenges for smaller laboratories, academic groups, and early-stage biotechnology companies operating with limited budgets. These expenses can restrict experimental scale, slow project timelines, and hinder broader adoption of advanced formulation technologies. Additionally, the need for precise handling conditions, strict environmental controls, and highly trained technical personnel further limits accessibility. Many low-resource research environments also lack the advanced instrumentation required, creating operational barriers and reducing opportunities for wider technology implementation.
What Emerging Trends Are Shaping the Lipid Nanoparticle Reagent Kits Market?
Key trends indicate accelerated adoption of lipid nanoparticle reagent kits in North America, Europe, and Asia-Pacific, particularly in countries investing heavily in RNA-based drug development and translational medicine infrastructure. Increasing integration of automation, microfluidic mixing, and sophisticated formulation design tools transforms the LNP preparation landscape by improving precision, reducing waste, and shortening optimization cycles. Collaborative research models between pharmaceutical companies, reagent manufacturers, and academic institutions advance next-generation lipid chemistries with enhanced endosomal escape and targeted tissue delivery capabilities.
How Are Leading Countries Driving Growth in the Lipid Nanoparticle Reagents Kits Market?
| Country | CAGR (2025 to 2035)% |
|---|---|
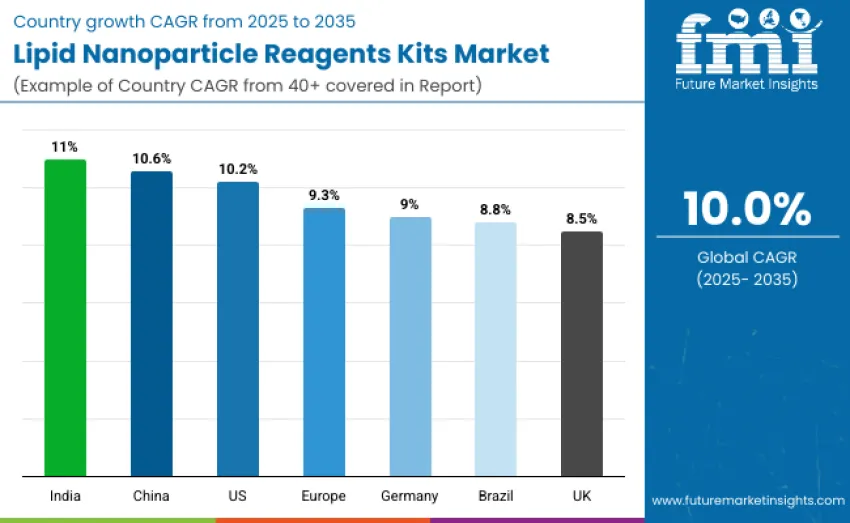
| United States | 10.2 |
| Brazil | 8.8 |
| China | 10.6 |
| India | 11.0 |
| Europe | 9.3 |
| Germany | 9.0 |
| United Kingdom | 8.5 |
The lipid nanoparticle reagent kits market is expanding globally, with India leading at an 11.0% CAGR due to its growing biotech ecosystem, vaccine self-reliance programs, and strengthened LNP research capabilities. China follows at 10.6%, driven by government-backed RNA therapeutic initiatives and rising reagent manufacturing capacity. The U.S. grows at 10.2% through strong mRNA vaccine, gene therapy, and biomanufacturing investments. Europe records 9.3% growth, with Germany at 9.0% supported by pharmaceutical research strength and translational medicine programs. Brazil advances at 8.8%, while the U.K. grows at 8.5% through academic–industry partnerships and oncology-focused LNP adoption.
The report covers an in-depth analysis of 40+ countries top-performing countries are highlighted below.
How Is Biotechnology and Therapeutic Research Infrastructure Accelerating Growth in China?
China demonstrates the strongest growth potential with a CAGR of 10.6% through 2035. Leadership stems from large-scale investment in mRNA vaccine and gene therapy research, expanding biopharmaceutical industrial parks, and government programs promoting biotechnology innovation that drive adoption of standardized LNP reagent kits. Growth is concentrated in major research and industrial hubs, including Beijing, Shanghai, Shenzhen, and Suzhou, where academic institutions, contract research organizations, and pharmaceutical companies are equipping research teams with LNP reagent kits for formulation optimization.
Distribution channels through specialized life sciences suppliers, e-commerce platforms, and direct manufacturer partnerships expand deployment across commercial labs, academic research centers, and translational medicine facilities. The country's "Made in China 2025" initiative provides policy support for high-value pharmaceutical research, including subsidies for lab equipment procurement, research funding, and biotechnology startup incubation programs. Large-scale biotechnology and RNA therapeutics research in eastern provinces includes researchers engaged in vaccine, gene therapy, and RNAi formulation studies.
Why Are Creative Skill Development Programs Enhancing Adoption in India?
India's lipid nanoparticle reagents kits market demonstrates strong growth momentum with a CAGR of 11% through 2035, linked to comprehensive pharmaceutical sector diversification into RNA therapeutics and increasing focus on advanced drug delivery capability development. Indian researchers and biopharma companies are implementing standardized LNP formulation workflows and reagent systems to serve global therapeutic development pipelines while meeting regulatory and quality standards required for clinical applications. Leading biotechnology and research clusters, including Bangalore, Hyderabad, Mumbai, and Pune, drive LNP reagent kit adoption.
Government incentive programs enable funding support for laboratory infrastructure, reagent procurement, and skill development through national and state biotechnology promotion schemes. Technology collaboration agreements accelerate adoption with international reagent manufacturers establishing distribution partnerships and local technical support infrastructure. Policy support through DBT Translational Research programs, Biotechnology Industry Research Assistance Council initiatives, and skill development training programs provide equipment access, technical education, and hands-on formulation training. National biotechnology development programs and vaccine innovation initiatives create sustained demand.
How Does Biotechnology and Pharmaceutical Research Excellence Sustain Leadership in Germany?
Germany's advanced biotechnology and pharmaceutical research ecosystem demonstrates sophisticated implementation of lipid nanoparticle reagent kits, with documented case studies showing improvement in formulation workflow efficiency through standardized kit adoption across academic laboratories, contract research organizations, and industrial research facilities. Research infrastructure in major biotech hubs, including Berlin, Munich, Hamburg, and Frankfurt regions, showcases integration of LNP reagent kits with existing laboratory workflows, leveraging expertise in precision chemistry, process development, and quality assurance traditions.
German researchers and companies emphasize reagent quality, reproducibility, and regulatory compliance, creating demand for premium LNP kits that support rigorous experimental protocols and meet strict GMP and clinical research standards. Growth maintains steady momentum through focus on professional research institutions and premium product segments, with a CAGR of 9.0% through 2035. Academic and industrial laboratories implement standardized LNP formulation workflows with comprehensive training programs. Strategic collaborations between European distributors and international manufacturers ensure reliable product availability.
Why Is Expanding Biotechnology and Research Community Fueling Demand in Brazil?
The Brazilian lipid nanoparticle reagents kits market leads Latin American adoption based on expansion of biotechnology research infrastructure and the growing community of academic and industrial researchers supporting domestic RNA therapeutic development and international collaboration projects. Growth shows solid potential with a CAGR of 8.8% through 2035, driven by investment in translational medicine, vaccine research, and growing demand for standardized LNP formulation reagents across São Paulo, Rio de Janeiro, Brasília, and Porto Alegre metropolitan areas.
Brazilian scientists and biopharma teams are adopting lipid nanoparticle reagent kits for mRNA vaccine development, gene therapy studies, and RNAi applications, supporting both domestic research programs and global therapeutic pipelines. Academic laboratories and contract research organizations implement standardized LNP formulation workflows for clinical and preclinical projects. Technology partnerships with international reagent manufacturers enable Brazilian access through localized pricing strategies and flexible procurement options addressing budget constraints. Strategic collaborations between biotechnology associations, research networks, and equipment suppliers enable group purchasing programs.
How Do Established Biotechnology and Pharmaceutical Industries Strengthen Performance in the United States?
The USA Lipid Nanoparticle Reagents Kits Market demonstrates mature implementation focused on established biopharmaceutical companies, academic research institutions, and contract research organization adoption, with documented integration across major mRNA vaccine developers, gene therapy companies, and RNA therapeutics research centers operating throughout California, Massachusetts, New York, and Texas biotech clusters. Growth maintains steady momentum with a CAGR of 10.2% through 2035, driven by continued expansion of RNA-based therapeutic pipelines, robust preclinical and clinical research activity, and a mature biotechnology ecosystem.
Professional adoption across biopharmaceutical research teams includes standardized LNP reagent kit protocols ensuring reproducible formulation performance and regulatory compliance. Established distribution infrastructure through specialized life sciences suppliers and online marketplaces provides competitive pricing, rapid delivery, and technical support services. Collaboration between industry associations, academic consortia, and reagent manufacturers enables product testing, protocol optimization, and feedback integration into next-generation kit development. Emphasis on workflow efficiency, scalability, and reagent reliability drives professional preference for premium kits.
How Is Biotechnology and Therapeutic Research Expansion Supporting Growth in the United Kingdom?
In London, Cambridge, Oxford, and Edinburgh biotechnology and pharmaceutical research clusters, academic institutions, contract research organizations, and pharmaceutical companies are implementing lipid nanoparticle reagent kit workflows to address research and therapeutic development requirements in mRNA vaccines, gene therapy, and RNAi applications. Growth shows solid potential with a CAGR of 8.5% through 2035, linked to the UK biotechnology sector's economic contribution and increasing emphasis on translational research, nanomedicine innovation, and RNA therapeutic development programs.
British researchers and biopharma companies are adopting standardized LNP reagent workflows and formulation techniques to maintain competitive positioning in European and global therapeutic development while advancing domestic research capabilities. Laboratory adoption across pharmaceutical and academic research centers implements standardized LNP formulation workflows with comprehensive team training and protocol standardization programs. Educational partnerships between universities, graduate programs, and reagent manufacturers provide student and early-career researcher access through subsidized or academic pricing schemes.
How Are Regional R&D Investments Driving Expansion Across Europe?
The Lipid Nanoparticle Reagents Kits Market in Europe is projected to grow from USD 206.1 million in 2025 to USD 498.3 million by 2035, registering a CAGR of 9.3% over the forecast period. Germany leads the regional landscape with a CAGR of 9.0%, backed by a robust biotechnology base and extensive investment in RNA and gene delivery innovation. The country's strong academic-industry collaborations, particularly across Berlin, Munich, and Heidelberg, foster advancements in lipid formulation, nucleic acid encapsulation, and nanocarrier optimization.
France follows closely with a CAGR of 8.8%, supported by increasing government-backed research programs in nanomedicine and expanding biotech clusters around Paris and Lyon. Growing participation of domestic firms in global mRNA and siRNA development projects enhances outlook. The United Kingdom demonstrates a CAGR of 8.5%, driven by its mature life sciences sector and expanding research investments in advanced therapeutics manufacturing. Innovation hubs across Oxford, Cambridge, and London play a central role in integrating lipid nanoparticle technologies into clinical-stage drug delivery and vaccine development pipelines.
How Competitive Is the Global Lipid Nanoparticle Reagents Kits Market and What Defines Its Structure?
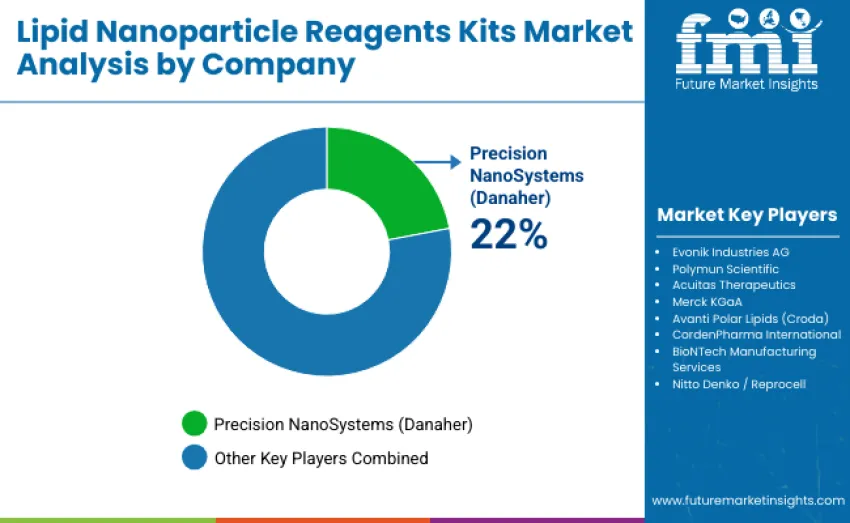
The Lipid Nanoparticle Reagent Kits Market is moderately concentrated, with 12–15 participants shaping global competition. The top three companies collectively command around 55–60% of global share, supported by strong technological expertise, validated formulation platforms, and long-term partnerships with pharmaceutical and academic institutions. Precision NanoSystems (Danaher) leads with a 22% share, leveraging advanced lipid chemistry capabilities, proprietary nanoparticle technologies, and robust cGMP-compliant production capacities. Competition is driven by formulation precision, reagent consistency, clinical scalability, and comprehensive technical support rather than pricing alone.
Precision NanoSystems, Evonik Industries AG, and Polymun Scientific dominate the landscape through extensive lipid formulation portfolios, scalable reagent systems, and deep integration with global mRNA and gene therapy manufacturing workflows. Their innovations in microfluidic assembly systems, ionizable lipid optimization, and encapsulation efficiency ensure high reproducibility across preclinical and clinical environments.
Challengers such as Acuitas Therapeutics, Merck KGaA, and Avanti Polar Lipids compete through specialized lipid chemistry expertise, proprietary lipid libraries, and flexible formulation capabilities supporting both academic and commercial therapeutic pipelines. Additional players including CordenPharma International, BioNTech Manufacturing Services, and Nitto Denko/Reprocell strengthen market depth through custom lipid synthesis, encapsulation process optimization, and contract manufacturing solutions, enhancing global development and production capacity.
Key Players in the Lipid Nanoparticle Reagents Kits Market
- Precision NanoSystems (Danaher)
- Evonik Industries AG
- Polymun Scientific
- Acuitas Therapeutics
- Merck KGaA
- Avanti Polar Lipids (Croda)
- CordenPharma International
- BioNTech Manufacturing Services
- Nitto Denko / Reprocell
Scope of the Report
| Items | Values |
|---|---|
| Quantitative Units | USD 698.9 million |
| Product Type | Lipid Formulation Kits, Nucleic Acid Encapsulation Reagents, Stabilization & Cryoprotectant Kits, Surface Functionalization Reagents, Purification & Characterization Kits |
| Application | mRNA Vaccines, Gene Therapy & siRNA Delivery, Drug Delivery Research, Cell Therapy Manufacturing, Protein Replacement Therapy |
| Regions Covered | North America, Europe, East Asia, South Asia & Pacific, Latin America, Middle East & Africa |
| Country Covered | China, India, Germany, Brazil, USA, UK, Japan, and 40+ countries |
| Key Companies Profiled | Precision NanoSystems (Danaher), Evonik Industries AG, Polymun Scientific, Acuitas Therapeutics, Merck KGaA, Avanti Polar Lipids (Croda), CordenPharma International, BioNTech Manufacturing Services, Nitto Denko/ Reprocell |
| Additional Attributes | Dollar sales by product type and application, regional trends across Asia Pacific, Europe, and North America, competitive landscape of LNP reagent kit manufacturers, technical specifications, integration with microfluidic and mRNA workflows, innovations in ionizable lipids and stabilization, and development of scalable, high-efficiency LNP kits |
Lipid Nanoparticle Reagents Kits Market by Segments
By Product Type:
- Lipid Formulation Kits
- Nucleic Acid Encapsulation Reagents
- Stabilization & Cryoprotectant Kits
- Surface Functionalization Reagents
- Purification & Characterization Kits
By Application:
- mRNA Vaccines
- Gene Therapy & siRNA Delivery
- Drug Delivery Research
- Cell Therapy Manufacturing
- Protein Replacement Therapy
By Region:
- Asia Pacific
- China
- India
- Japan
- South Korea
- Vietnam
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Netherlands
- Nordic
- BENELUX
- Rest of Europe
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Argentina
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Other African Union
- Rest of Middle East & Africa
Frequently Asked Questions
How big is the lipid nanoparticle reagents kits market in 2025?
The global lipid nanoparticle reagents kits market is valued at USD 698.9 million in 2025.
What will be the size of the lipid nanoparticle reagents kits market in 2035?
The market is projected to reach USD 1,845.7 million by 2035.
How fast will the lipid nanoparticle reagents kits market grow between 2025 and 2035?
The market will grow at a CAGR of 10% from 2025 to 2035.
Which product type leads the lipid nanoparticle reagents kits market in 2025?
Lipid formulation kits lead the market with a 38.5% share in 2025.
Who are the major players in the lipid nanoparticle reagents kits market?
Key players include Precision NanoSystems, Evonik Industries, Polymun Scientific, Acuitas Therapeutics, Merck KGaA, Avanti Polar Lipids, CordenPharma, BioNTech Manufacturing Services, and Nitto Denko/Reprocell.
Table of Content
- Executive Summary
- Regional Level Market Outlook
- Demand-side Trends
- Supply-side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Assessment
- Products Market Assessment
- Value Chain Analysis
- Participants and their Roles
- Raw Material Suppliers
- Manufacturers
- Mid-Level Participants
- Wholesalers and Distributors
- Profit Margin Analysis
- Operating Margins By Region
- Notes on Value Added at Each Node in the Value Chain
- Participants and their Roles
- Strategic Divestments/ Investments
- Market Trade Assessment
- Import-Export Analysis
- Supply Demand Gap Assessment
- White –Space Opportunity Assessment and Mapping
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- By Key Regions
- By Key Countries
- Regional Parent Market Outlook
- Product Launches & Development
- Technology Innovation/Advancement
- Joint Ventures, M&A, Licensing & Partnerships
- Market Dynamics
- Regional Level Market Analysis 2020 to 2024 and Forecast, 2025 to 2035
- Historical Market Size USD Million Analysis, 2020 to 2024
- Current and Future Market Size USD Million Projections, 2025 to 2035
- Y-o-Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Regional Level Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Product Type
- Introduction / Key Findings
- Historical Market Size USD Million Analysis By Product Type, 2020 to 2024
- Current and Future Market Size USD Million Analysis and Forecast By Product Type, 2025 to 2035
- Lipid Formulation Kits
- Nucleic Acid Encapsulation Reagents
- Stabilization & Cryoprotectant Kits
- Surface Functionalization Reagents
- Purification & Characterization Kits
- Y-o-Y Growth Trend Analysis By Product Type, 2020 to 2024
- Absolute $ Opportunity Analysis By Product Type, 2025 to 2035
- Regional Level Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Application
- Introduction / Key Findings
- Historical Market Size USD Million Analysis By Application, 2020 to 2024
- Current and Future Market Size USD Million Analysis and Forecast By Application, 2025 to 2035
- mRNA Vaccines
- Gene Therapy & siRNA Delivery
- Drug Delivery Research
- Cell Therapy Manufacturing
- Protein Replacement Therapy
- Y-o-Y Growth Trend Analysis By Application, 2020 to 2024
- Absolute $ Opportunity Analysis By Application, 2025 to 2035
- Regional Level Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Technology
- Introduction / Key Findings
- Historical Market Size USD Million Analysis By Technology, 2020 to 2024
- Current and Future Market Size USD Million Analysis and Forecast By Technology, 2025 to 2035
- Microfluidic Mixing Systems
- Ethanol Injection / Solvent Exchange
- pH Gradient-Based Assembly
- TFF & Size-Exclusion Purification Kits
- Hybrid AI/ML Formulation Platforms
- Y-o-Y Growth Trend Analysis By Technology, 2020 to 2024
- Absolute $ Opportunity Analysis By Technology, 2025 to 2035
- Regional Level Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By End User
- Introduction / Key Findings
- Historical Market Size USD Million Analysis By End User, 2020 to 2024
- Current and Future Market Size USD Million Analysis and Forecast By End User, 2025 to 2035
- Pharma & Biotech Companies
- Academic & Research Institutes
- CDMOs / CROs
- Reagent Manufacturers & Distributors
- Y-o-Y Growth Trend Analysis By End User, 2020 to 2024
- Absolute $ Opportunity Analysis By End User, 2025 to 2035
- Regional Level Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Region
- Introduction
- Historical Market Size USD Million Analysis By Region, 2020 to 2024
- Current Market Size USD Million Analysis and Forecast By Region, 2025 to 2035
- North America
- Latin America
- East Asia
- South Asia
- Western Europe
- Eastern Europe
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size USD Million Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size USD Million Forecast By Market Taxonomy, 2025 to 2035
- By Country
- USA
- Canada
- Mexico
- By Product Type
- By Application
- By Technology
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By Technology
- By End User
- Key Takeaways
- Latin America Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size USD Million Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size USD Million Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Brazil
- Argentina
- Rest of Latin America
- By Product Type
- By Application
- By Technology
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By Technology
- By End User
- Key Takeaways
- East Asia Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size USD Million Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size USD Million Forecast By Market Taxonomy, 2025 to 2035
- By Country
- China
- Japan
- South Korea
- By Product Type
- By Application
- By Technology
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By Technology
- By End User
- Key Takeaways
- South Asia Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size USD Million Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size USD Million Forecast By Market Taxonomy, 2025 to 2035
- By Country
- India
- ASEAN Countries
- Australia & New Zealand
- Rest of South Asia
- By Product Type
- By Application
- By Technology
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By Technology
- By End User
- Key Takeaways
- Western Europe Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size USD Million Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size USD Million Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Germany
- UK
- France
- Italy
- Spain
- BENELUX
- Nordic Countries
- Rest of Western Europe
- By Product Type
- By Application
- By Technology
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By Technology
- By End User
- Key Takeaways
- Eastern Europe Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size USD Million Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size USD Million Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Russia
- Hungary
- Poland
- Rest of Eastern Europe
- By Product Type
- By Application
- By Technology
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By Technology
- By End User
- Key Takeaways
- Middle East & Africa Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size USD Million Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size USD Million Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Saudi Arabia
- Other GCC Countries
- Türkiye
- South Africa
- Rest of MEA
- By Product Type
- By Application
- By Technology
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By Technology
- By End User
- Key Takeaways
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product Type
- By Application
- By Technology
- By End User
- Competition Analysis
- Competition Deep Dive
- Precision NanoSystems (Danaher)
- Overview
- Product Portfolio
- Key Financials
- Key Developments
- Global/Regional Footprint
- Strategy Overview
- Marketing Strategy
- SWOT
- Analyst Comment
- Evonik Industries AG
- Overview
- Product Portfolio
- Key Financials
- Key Developments
- Global/Regional Footprint
- Strategy Overview
- Marketing Strategy
- SWOT
- Analyst Comment
- Polymun Scientific
- Overview
- Product Portfolio
- Key Financials
- Key Developments
- Global/Regional Footprint
- Strategy Overview
- Marketing Strategy
- SWOT
- Analyst Comment
- Acuitas Therapeutics
- Overview
- Product Portfolio
- Key Financials
- Key Developments
- Global/Regional Footprint
- Strategy Overview
- Marketing Strategy
- SWOT
- Analyst Comment
- Merck KGaA
- Overview
- Product Portfolio
- Key Financials
- Key Developments
- Global/Regional Footprint
- Strategy Overview
- Marketing Strategy
- SWOT
- Analyst Comment
- Avanti Polar Lipids (Croda)
- Overview
- Product Portfolio
- Key Financials
- Key Developments
- Global/Regional Footprint
- Strategy Overview
- Marketing Strategy
- SWOT
- Analyst Comment
- CordenPharma International
- Overview
- Product Portfolio
- Key Financials
- Key Developments
- Global/Regional Footprint
- Strategy Overview
- Marketing Strategy
- SWOT
- Analyst Comment
- BioNTech Manufacturing Services
- Overview
- Product Portfolio
- Key Financials
- Key Developments
- Global/Regional Footprint
- Strategy Overview
- Marketing Strategy
- SWOT
- Analyst Comment
- Nitto Denko / Reprocell
- Overview
- Product Portfolio
- Key Financials
- Key Developments
- Global/Regional Footprint
- Strategy Overview
- Marketing Strategy
- SWOT
- Analyst Comment
- Precision NanoSystems (Danaher)
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: Global Market Size (USD Million), by Product Type, 2020-2035
- Table 2: Global Market Size (USD Million), by Lipid Formulation Kits, 2020-2035
- Table 3: Global Market Size (USD Million), by Nucleic Acid Encapsulation Reagents, 2020-2035
- Table 4: Global Market Size (USD Million), by Stabilization & Cryoprotectant Kits, 2020-2035
- Table 5: Global Market Size (USD Million), by Surface Functionalization Reagents, 2020-2035
- Table 6: Global Market Size (USD Million), by Purification & Characterization Kits, 2020-2035
- Table 7: Global Market Size (USD Million), by Application, 2020-2035
- Table 8: Global Market Size (USD Million), by mRNA Vaccines, 2020-2035
- Table 9: Global Market Size (USD Million), by Gene Therapy & siRNA Delivery, 2020-2035
- Table 10: Global Market Size (USD Million), by Drug Delivery Research, 2020-2035
- Table 11: Global Market Size (USD Million), by Cell Therapy Manufacturing, 2020-2035
- Table 12: Global Market Size (USD Million), by Protein Replacement Therapy, 2020-2035
- Table 13: Global Market Size (USD Million), by Technology, 2020-2035
- Table 14: Global Market Size (USD Million), by Microfluidic Mixing Systems, 2020-2035
- Table 15: Global Market Size (USD Million), by Ethanol Injection / Solvent Exchange, 2020-2035
- Table 16: Global Market Size (USD Million), by pH Gradient-Based Assembly, 2020-2035
- Table 17: Global Market Size (USD Million), by TFF & Size-Exclusion Purification Kits, 2020-2035
- Table 18: Global Market Size (USD Million), by Hybrid AI/ML Formulation Platforms, 2020-2035
- Table 19: Global Market Size (USD Million), by End User, 2020-2035
- Table 20: Global Market Size (USD Million), by Pharma & Biotech Companies, 2020-2035
- Table 21: Global Market Size (USD Million), by Academic & Research Institutes, 2020-2035
- Table 22: Global Market Size (USD Million), by CDMOs / CROs, 2020-2035
- Table 23: Global Market Size (USD Million), by Reagent Manufacturers & Distributors, 2020-2035
- Table 24: North America Market Size (USD Million), by Product Type, 2020-2035
- Table 25: North America Market Size (USD Million), by Lipid Formulation Kits, 2020-2035
- Table 26: North America Market Size (USD Million), by Nucleic Acid Encapsulation Reagents, 2020-2035
- Table 27: North America Market Size (USD Million), by Stabilization & Cryoprotectant Kits, 2020-2035
- Table 28: North America Market Size (USD Million), by Surface Functionalization Reagents, 2020-2035
- Table 29: North America Market Size (USD Million), by Purification & Characterization Kits, 2020-2035
- Table 30: North America Market Size (USD Million), by Application, 2020-2035
- Table 31: North America Market Size (USD Million), by mRNA Vaccines, 2020-2035
- Table 32: North America Market Size (USD Million), by Gene Therapy & siRNA Delivery, 2020-2035
- Table 33: North America Market Size (USD Million), by Drug Delivery Research, 2020-2035
- Table 34: North America Market Size (USD Million), by Cell Therapy Manufacturing, 2020-2035
- Table 35: North America Market Size (USD Million), by Protein Replacement Therapy, 2020-2035
- Table 36: North America Market Size (USD Million), by Technology, 2020-2035
- Table 37: North America Market Size (USD Million), by Microfluidic Mixing Systems, 2020-2035
- Table 38: North America Market Size (USD Million), by Ethanol Injection / Solvent Exchange, 2020-2035
- Table 39: North America Market Size (USD Million), by pH Gradient-Based Assembly, 2020-2035
- Table 40: North America Market Size (USD Million), by TFF & Size-Exclusion Purification Kits, 2020-2035
- Table 41: North America Market Size (USD Million), by Hybrid AI/ML Formulation Platforms, 2020-2035
- Table 42: North America Market Size (USD Million), by End User, 2020-2035
- Table 43: North America Market Size (USD Million), by Pharma & Biotech Companies, 2020-2035
- Table 44: North America Market Size (USD Million), by Academic & Research Institutes, 2020-2035
- Table 45: North America Market Size (USD Million), by CDMOs / CROs, 2020-2035
- Table 46: North America Market Size (USD Million), by Reagent Manufacturers & Distributors, 2020-2035
- Table 47: USA Market Size (USD Million), by Product Type, 2020-2035
- Table 48: USA Market Size (USD Million), by Application, 2020-2035
- Table 49: USA Market Size (USD Million), by Technology, 2020-2035
- Table 50: USA Market Size (USD Million), by End User, 2020-2035
- Table 51: Canada Market Size (USD Million), by Product Type, 2020-2035
- Table 52: Canada Market Size (USD Million), by Application, 2020-2035
- Table 53: Canada Market Size (USD Million), by Technology, 2020-2035
- Table 54: Canada Market Size (USD Million), by End User, 2020-2035
- Table 55: Mexico Market Size (USD Million), by Product Type, 2020-2035
- Table 56: Mexico Market Size (USD Million), by Application, 2020-2035
- Table 57: Mexico Market Size (USD Million), by Technology, 2020-2035
- Table 58: Mexico Market Size (USD Million), by End User, 2020-2035
- Table 59: Latin America Market Size (USD Million), by Product Type, 2020-2035
- Table 60: Latin America Market Size (USD Million), by Lipid Formulation Kits, 2020-2035
- Table 61: Latin America Market Size (USD Million), by Nucleic Acid Encapsulation Reagents, 2020-2035
- Table 62: Latin America Market Size (USD Million), by Stabilization & Cryoprotectant Kits, 2020-2035
- Table 63: Latin America Market Size (USD Million), by Surface Functionalization Reagents, 2020-2035
- Table 64: Latin America Market Size (USD Million), by Purification & Characterization Kits, 2020-2035
- Table 65: Latin America Market Size (USD Million), by Application, 2020-2035
- Table 66: Latin America Market Size (USD Million), by mRNA Vaccines, 2020-2035
- Table 67: Latin America Market Size (USD Million), by Gene Therapy & siRNA Delivery, 2020-2035
- Table 68: Latin America Market Size (USD Million), by Drug Delivery Research, 2020-2035
- Table 69: Latin America Market Size (USD Million), by Cell Therapy Manufacturing, 2020-2035
- Table 70: Latin America Market Size (USD Million), by Protein Replacement Therapy, 2020-2035
- Table 71: Latin America Market Size (USD Million), by Technology, 2020-2035
- Table 72: Latin America Market Size (USD Million), by Microfluidic Mixing Systems, 2020-2035
- Table 73: Latin America Market Size (USD Million), by Ethanol Injection / Solvent Exchange, 2020-2035
- Table 74: Latin America Market Size (USD Million), by pH Gradient-Based Assembly, 2020-2035
- Table 75: Latin America Market Size (USD Million), by TFF & Size-Exclusion Purification Kits, 2020-2035
- Table 76: Latin America Market Size (USD Million), by Hybrid AI/ML Formulation Platforms, 2020-2035
- Table 77: Latin America Market Size (USD Million), by End User, 2020-2035
- Table 78: Latin America Market Size (USD Million), by Pharma & Biotech Companies, 2020-2035
- Table 79: Latin America Market Size (USD Million), by Academic & Research Institutes, 2020-2035
- Table 80: Latin America Market Size (USD Million), by CDMOs / CROs, 2020-2035
- Table 81: Latin America Market Size (USD Million), by Reagent Manufacturers & Distributors, 2020-2035
- Table 82: Brazil Market Size (USD Million), by Product Type, 2020-2035
- Table 83: Brazil Market Size (USD Million), by Application, 2020-2035
- Table 84: Brazil Market Size (USD Million), by Technology, 2020-2035
- Table 85: Brazil Market Size (USD Million), by End User, 2020-2035
- Table 86: Argentina Market Size (USD Million), by Product Type, 2020-2035
- Table 87: Argentina Market Size (USD Million), by Application, 2020-2035
- Table 88: Argentina Market Size (USD Million), by Technology, 2020-2035
- Table 89: Argentina Market Size (USD Million), by End User, 2020-2035
- Table 90: Rest of Latin America Market Size (USD Million), by Product Type, 2020-2035
- Table 91: Rest of Latin America Market Size (USD Million), by Application, 2020-2035
- Table 92: Rest of Latin America Market Size (USD Million), by Technology, 2020-2035
- Table 93: Rest of Latin America Market Size (USD Million), by End User, 2020-2035
- Table 94: East Asia Market Size (USD Million), by Product Type, 2020-2035
- Table 95: East Asia Market Size (USD Million), by Lipid Formulation Kits, 2020-2035
- Table 96: East Asia Market Size (USD Million), by Nucleic Acid Encapsulation Reagents, 2020-2035
- Table 97: East Asia Market Size (USD Million), by Stabilization & Cryoprotectant Kits, 2020-2035
- Table 98: East Asia Market Size (USD Million), by Surface Functionalization Reagents, 2020-2035
- Table 99: East Asia Market Size (USD Million), by Purification & Characterization Kits, 2020-2035
- Table 100: East Asia Market Size (USD Million), by Application, 2020-2035
- Table 101: East Asia Market Size (USD Million), by mRNA Vaccines, 2020-2035
- Table 102: East Asia Market Size (USD Million), by Gene Therapy & siRNA Delivery, 2020-2035
- Table 103: East Asia Market Size (USD Million), by Drug Delivery Research, 2020-2035
- Table 104: East Asia Market Size (USD Million), by Cell Therapy Manufacturing, 2020-2035
- Table 105: East Asia Market Size (USD Million), by Protein Replacement Therapy, 2020-2035
- Table 106: East Asia Market Size (USD Million), by Technology, 2020-2035
- Table 107: East Asia Market Size (USD Million), by Microfluidic Mixing Systems, 2020-2035
- Table 108: East Asia Market Size (USD Million), by Ethanol Injection / Solvent Exchange, 2020-2035
- Table 109: East Asia Market Size (USD Million), by pH Gradient-Based Assembly, 2020-2035
- Table 110: East Asia Market Size (USD Million), by TFF & Size-Exclusion Purification Kits, 2020-2035
- Table 111: East Asia Market Size (USD Million), by Hybrid AI/ML Formulation Platforms, 2020-2035
- Table 112: East Asia Market Size (USD Million), by End User, 2020-2035
- Table 113: East Asia Market Size (USD Million), by Pharma & Biotech Companies, 2020-2035
- Table 114: East Asia Market Size (USD Million), by Academic & Research Institutes, 2020-2035
- Table 115: East Asia Market Size (USD Million), by CDMOs / CROs, 2020-2035
- Table 116: East Asia Market Size (USD Million), by Reagent Manufacturers & Distributors, 2020-2035
- Table 117: China Market Size (USD Million), by Product Type, 2020-2035
- Table 118: China Market Size (USD Million), by Application, 2020-2035
- Table 119: China Market Size (USD Million), by Technology, 2020-2035
- Table 120: China Market Size (USD Million), by End User, 2020-2035
- Table 121: Japan Market Size (USD Million), by Product Type, 2020-2035
- Table 122: Japan Market Size (USD Million), by Application, 2020-2035
- Table 123: Japan Market Size (USD Million), by Technology, 2020-2035
- Table 124: Japan Market Size (USD Million), by End User, 2020-2035
- Table 125: South Korea Market Size (USD Million), by Product Type, 2020-2035
- Table 126: South Korea Market Size (USD Million), by Application, 2020-2035
- Table 127: South Korea Market Size (USD Million), by Technology, 2020-2035
- Table 128: South Korea Market Size (USD Million), by End User, 2020-2035
- Table 129: South Asia Market Size (USD Million), by Product Type, 2020-2035
- Table 130: South Asia Market Size (USD Million), by Lipid Formulation Kits, 2020-2035
- Table 131: South Asia Market Size (USD Million), by Nucleic Acid Encapsulation Reagents, 2020-2035
- Table 132: South Asia Market Size (USD Million), by Stabilization & Cryoprotectant Kits, 2020-2035
- Table 133: South Asia Market Size (USD Million), by Surface Functionalization Reagents, 2020-2035
- Table 134: South Asia Market Size (USD Million), by Purification & Characterization Kits, 2020-2035
- Table 135: South Asia Market Size (USD Million), by Application, 2020-2035
- Table 136: South Asia Market Size (USD Million), by mRNA Vaccines, 2020-2035
- Table 137: South Asia Market Size (USD Million), by Gene Therapy & siRNA Delivery, 2020-2035
- Table 138: South Asia Market Size (USD Million), by Drug Delivery Research, 2020-2035
- Table 139: South Asia Market Size (USD Million), by Cell Therapy Manufacturing, 2020-2035
- Table 140: South Asia Market Size (USD Million), by Protein Replacement Therapy, 2020-2035
- Table 141: South Asia Market Size (USD Million), by Technology, 2020-2035
- Table 142: South Asia Market Size (USD Million), by Microfluidic Mixing Systems, 2020-2035
- Table 143: South Asia Market Size (USD Million), by Ethanol Injection / Solvent Exchange, 2020-2035
- Table 144: South Asia Market Size (USD Million), by pH Gradient-Based Assembly, 2020-2035
- Table 145: South Asia Market Size (USD Million), by TFF & Size-Exclusion Purification Kits, 2020-2035
- Table 146: South Asia Market Size (USD Million), by Hybrid AI/ML Formulation Platforms, 2020-2035
- Table 147: South Asia Market Size (USD Million), by End User, 2020-2035
- Table 148: South Asia Market Size (USD Million), by Pharma & Biotech Companies, 2020-2035
- Table 149: South Asia Market Size (USD Million), by Academic & Research Institutes, 2020-2035
- Table 150: South Asia Market Size (USD Million), by CDMOs / CROs, 2020-2035
- Table 151: South Asia Market Size (USD Million), by Reagent Manufacturers & Distributors, 2020-2035
- Table 152: India Market Size (USD Million), by Product Type, 2020-2035
- Table 153: India Market Size (USD Million), by Application, 2020-2035
- Table 154: India Market Size (USD Million), by Technology, 2020-2035
- Table 155: India Market Size (USD Million), by End User, 2020-2035
- Table 156: ASEAN Countries Market Size (USD Million), by Product Type, 2020-2035
- Table 157: ASEAN Countries Market Size (USD Million), by Application, 2020-2035
- Table 158: ASEAN Countries Market Size (USD Million), by Technology, 2020-2035
- Table 159: ASEAN Countries Market Size (USD Million), by End User, 2020-2035
- Table 160: Australia & New Zealand Market Size (USD Million), by Product Type, 2020-2035
- Table 161: Australia & New Zealand Market Size (USD Million), by Application, 2020-2035
- Table 162: Australia & New Zealand Market Size (USD Million), by Technology, 2020-2035
- Table 163: Australia & New Zealand Market Size (USD Million), by End User, 2020-2035
- Table 164: Rest of South Asia Market Size (USD Million), by Product Type, 2020-2035
- Table 165: Rest of South Asia Market Size (USD Million), by Application, 2020-2035
- Table 166: Rest of South Asia Market Size (USD Million), by Technology, 2020-2035
- Table 167: Rest of South Asia Market Size (USD Million), by End User, 2020-2035
- Table 168: Western Europe Market Size (USD Million), by Product Type, 2020-2035
- Table 169: Western Europe Market Size (USD Million), by Lipid Formulation Kits, 2020-2035
- Table 170: Western Europe Market Size (USD Million), by Nucleic Acid Encapsulation Reagents, 2020-2035
- Table 171: Western Europe Market Size (USD Million), by Stabilization & Cryoprotectant Kits, 2020-2035
- Table 172: Western Europe Market Size (USD Million), by Surface Functionalization Reagents, 2020-2035
- Table 173: Western Europe Market Size (USD Million), by Purification & Characterization Kits, 2020-2035
- Table 174: Western Europe Market Size (USD Million), by Application, 2020-2035
- Table 175: Western Europe Market Size (USD Million), by mRNA Vaccines, 2020-2035
- Table 176: Western Europe Market Size (USD Million), by Gene Therapy & siRNA Delivery, 2020-2035
- Table 177: Western Europe Market Size (USD Million), by Drug Delivery Research, 2020-2035
- Table 178: Western Europe Market Size (USD Million), by Cell Therapy Manufacturing, 2020-2035
- Table 179: Western Europe Market Size (USD Million), by Protein Replacement Therapy, 2020-2035
- Table 180: Western Europe Market Size (USD Million), by Technology, 2020-2035
- Table 181: Western Europe Market Size (USD Million), by Microfluidic Mixing Systems, 2020-2035
- Table 182: Western Europe Market Size (USD Million), by Ethanol Injection / Solvent Exchange, 2020-2035
- Table 183: Western Europe Market Size (USD Million), by pH Gradient-Based Assembly, 2020-2035
- Table 184: Western Europe Market Size (USD Million), by TFF & Size-Exclusion Purification Kits, 2020-2035
- Table 185: Western Europe Market Size (USD Million), by Hybrid AI/ML Formulation Platforms, 2020-2035
- Table 186: Western Europe Market Size (USD Million), by End User, 2020-2035
- Table 187: Western Europe Market Size (USD Million), by Pharma & Biotech Companies, 2020-2035
- Table 188: Western Europe Market Size (USD Million), by Academic & Research Institutes, 2020-2035
- Table 189: Western Europe Market Size (USD Million), by CDMOs / CROs, 2020-2035
- Table 190: Western Europe Market Size (USD Million), by Reagent Manufacturers & Distributors, 2020-2035
- Table 191: Germany Market Size (USD Million), by Product Type, 2020-2035
- Table 192: Germany Market Size (USD Million), by Application, 2020-2035
- Table 193: Germany Market Size (USD Million), by Technology, 2020-2035
- Table 194: Germany Market Size (USD Million), by End User, 2020-2035
- Table 195: UK Market Size (USD Million), by Product Type, 2020-2035
- Table 196: UK Market Size (USD Million), by Application, 2020-2035
- Table 197: UK Market Size (USD Million), by Technology, 2020-2035
- Table 198: UK Market Size (USD Million), by End User, 2020-2035
- Table 199: France Market Size (USD Million), by Product Type, 2020-2035
- Table 200: France Market Size (USD Million), by Application, 2020-2035
- Table 201: France Market Size (USD Million), by Technology, 2020-2035
- Table 202: France Market Size (USD Million), by End User, 2020-2035
- Table 203: Italy Market Size (USD Million), by Product Type, 2020-2035
- Table 204: Italy Market Size (USD Million), by Application, 2020-2035
- Table 205: Italy Market Size (USD Million), by Technology, 2020-2035
- Table 206: Italy Market Size (USD Million), by End User, 2020-2035
- Table 207: Spain Market Size (USD Million), by Product Type, 2020-2035
- Table 208: Spain Market Size (USD Million), by Application, 2020-2035
- Table 209: Spain Market Size (USD Million), by Technology, 2020-2035
- Table 210: Spain Market Size (USD Million), by End User, 2020-2035
- Table 211: BENELUX Market Size (USD Million), by Product Type, 2020-2035
- Table 212: BENELUX Market Size (USD Million), by Application, 2020-2035
- Table 213: BENELUX Market Size (USD Million), by Technology, 2020-2035
- Table 214: BENELUX Market Size (USD Million), by End User, 2020-2035
- Table 215: Nordic Countries Market Size (USD Million), by Product Type, 2020-2035
- Table 216: Nordic Countries Market Size (USD Million), by Application, 2020-2035
- Table 217: Nordic Countries Market Size (USD Million), by Technology, 2020-2035
- Table 218: Nordic Countries Market Size (USD Million), by End User, 2020-2035
- Table 219: Rest of Western Europe Market Size (USD Million), by Product Type, 2020-2035
- Table 220: Rest of Western Europe Market Size (USD Million), by Application, 2020-2035
- Table 221: Rest of Western Europe Market Size (USD Million), by Technology, 2020-2035
- Table 222: Rest of Western Europe Market Size (USD Million), by End User, 2020-2035
- Table 223: Eastern Europe Market Size (USD Million), by Product Type, 2020-2035
- Table 224: Eastern Europe Market Size (USD Million), by Lipid Formulation Kits, 2020-2035
- Table 225: Eastern Europe Market Size (USD Million), by Nucleic Acid Encapsulation Reagents, 2020-2035
- Table 226: Eastern Europe Market Size (USD Million), by Stabilization & Cryoprotectant Kits, 2020-2035
- Table 227: Eastern Europe Market Size (USD Million), by Surface Functionalization Reagents, 2020-2035
- Table 228: Eastern Europe Market Size (USD Million), by Purification & Characterization Kits, 2020-2035
- Table 229: Eastern Europe Market Size (USD Million), by Application, 2020-2035
- Table 230: Eastern Europe Market Size (USD Million), by mRNA Vaccines, 2020-2035
- Table 231: Eastern Europe Market Size (USD Million), by Gene Therapy & siRNA Delivery, 2020-2035
- Table 232: Eastern Europe Market Size (USD Million), by Drug Delivery Research, 2020-2035
- Table 233: Eastern Europe Market Size (USD Million), by Cell Therapy Manufacturing, 2020-2035
- Table 234: Eastern Europe Market Size (USD Million), by Protein Replacement Therapy, 2020-2035
- Table 235: Eastern Europe Market Size (USD Million), by Technology, 2020-2035
- Table 236: Eastern Europe Market Size (USD Million), by Microfluidic Mixing Systems, 2020-2035
- Table 237: Eastern Europe Market Size (USD Million), by Ethanol Injection / Solvent Exchange, 2020-2035
- Table 238: Eastern Europe Market Size (USD Million), by pH Gradient-Based Assembly, 2020-2035
- Table 239: Eastern Europe Market Size (USD Million), by TFF & Size-Exclusion Purification Kits, 2020-2035
- Table 240: Eastern Europe Market Size (USD Million), by Hybrid AI/ML Formulation Platforms, 2020-2035
- Table 241: Eastern Europe Market Size (USD Million), by End User, 2020-2035
- Table 242: Eastern Europe Market Size (USD Million), by Pharma & Biotech Companies, 2020-2035
- Table 243: Eastern Europe Market Size (USD Million), by Academic & Research Institutes, 2020-2035
- Table 244: Eastern Europe Market Size (USD Million), by CDMOs / CROs, 2020-2035
- Table 245: Eastern Europe Market Size (USD Million), by Reagent Manufacturers & Distributors, 2020-2035
- Table 246: Russia Market Size (USD Million), by Product Type, 2020-2035
- Table 247: Russia Market Size (USD Million), by Application, 2020-2035
- Table 248: Russia Market Size (USD Million), by Technology, 2020-2035
- Table 249: Russia Market Size (USD Million), by End User, 2020-2035
- Table 250: Hungary Market Size (USD Million), by Product Type, 2020-2035
- Table 251: Hungary Market Size (USD Million), by Application, 2020-2035
- Table 252: Hungary Market Size (USD Million), by Technology, 2020-2035
- Table 253: Hungary Market Size (USD Million), by End User, 2020-2035
- Table 254: Poland Market Size (USD Million), by Product Type, 2020-2035
- Table 255: Poland Market Size (USD Million), by Application, 2020-2035
- Table 256: Poland Market Size (USD Million), by Technology, 2020-2035
- Table 257: Poland Market Size (USD Million), by End User, 2020-2035
- Table 258: Rest of Eastern Europe Market Size (USD Million), by Product Type, 2020-2035
- Table 259: Rest of Eastern Europe Market Size (USD Million), by Application, 2020-2035
- Table 260: Rest of Eastern Europe Market Size (USD Million), by Technology, 2020-2035
- Table 261: Rest of Eastern Europe Market Size (USD Million), by End User, 2020-2035
- Table 262: Middle East & Africa Market Size (USD Million), by Product Type, 2020-2035
- Table 263: Middle East & Africa Market Size (USD Million), by Lipid Formulation Kits, 2020-2035
- Table 264: Middle East & Africa Market Size (USD Million), by Nucleic Acid Encapsulation Reagents, 2020-2035
- Table 265: Middle East & Africa Market Size (USD Million), by Stabilization & Cryoprotectant Kits, 2020-2035
- Table 266: Middle East & Africa Market Size (USD Million), by Surface Functionalization Reagents, 2020-2035
- Table 267: Middle East & Africa Market Size (USD Million), by Purification & Characterization Kits, 2020-2035
- Table 268: Middle East & Africa Market Size (USD Million), by Application, 2020-2035
- Table 269: Middle East & Africa Market Size (USD Million), by mRNA Vaccines, 2020-2035
- Table 270: Middle East & Africa Market Size (USD Million), by Gene Therapy & siRNA Delivery, 2020-2035
- Table 271: Middle East & Africa Market Size (USD Million), by Drug Delivery Research, 2020-2035
- Table 272: Middle East & Africa Market Size (USD Million), by Cell Therapy Manufacturing, 2020-2035
- Table 273: Middle East & Africa Market Size (USD Million), by Protein Replacement Therapy, 2020-2035
- Table 274: Middle East & Africa Market Size (USD Million), by Technology, 2020-2035
- Table 275: Middle East & Africa Market Size (USD Million), by Microfluidic Mixing Systems, 2020-2035
- Table 276: Middle East & Africa Market Size (USD Million), by Ethanol Injection / Solvent Exchange, 2020-2035
- Table 277: Middle East & Africa Market Size (USD Million), by pH Gradient-Based Assembly, 2020-2035
- Table 278: Middle East & Africa Market Size (USD Million), by TFF & Size-Exclusion Purification Kits, 2020-2035
- Table 279: Middle East & Africa Market Size (USD Million), by Hybrid AI/ML Formulation Platforms, 2020-2035
- Table 280: Middle East & Africa Market Size (USD Million), by End User, 2020-2035
- Table 281: Middle East & Africa Market Size (USD Million), by Pharma & Biotech Companies, 2020-2035
- Table 282: Middle East & Africa Market Size (USD Million), by Academic & Research Institutes, 2020-2035
- Table 283: Middle East & Africa Market Size (USD Million), by CDMOs / CROs, 2020-2035
- Table 284: Middle East & Africa Market Size (USD Million), by Reagent Manufacturers & Distributors, 2020-2035
- Table 285: Saudi Arabia Market Size (USD Million), by Product Type, 2020-2035
- Table 286: Saudi Arabia Market Size (USD Million), by Application, 2020-2035
- Table 287: Saudi Arabia Market Size (USD Million), by Technology, 2020-2035
- Table 288: Saudi Arabia Market Size (USD Million), by End User, 2020-2035
- Table 289: Other GCC Countries Market Size (USD Million), by Product Type, 2020-2035
- Table 290: Other GCC Countries Market Size (USD Million), by Application, 2020-2035
- Table 291: Other GCC Countries Market Size (USD Million), by Technology, 2020-2035
- Table 292: Other GCC Countries Market Size (USD Million), by End User, 2020-2035
- Table 293: Türkiye Market Size (USD Million), by Product Type, 2020-2035
- Table 294: Türkiye Market Size (USD Million), by Application, 2020-2035
- Table 295: Türkiye Market Size (USD Million), by Technology, 2020-2035
- Table 296: Türkiye Market Size (USD Million), by End User, 2020-2035
- Table 297: South Africa Market Size (USD Million), by Product Type, 2020-2035
- Table 298: South Africa Market Size (USD Million), by Application, 2020-2035
- Table 299: South Africa Market Size (USD Million), by Technology, 2020-2035
- Table 300: South Africa Market Size (USD Million), by End User, 2020-2035
- Table 301: Rest of MEA Market Size (USD Million), by Product Type, 2020-2035
- Table 302: Rest of MEA Market Size (USD Million), by Application, 2020-2035
- Table 303: Rest of MEA Market Size (USD Million), by Technology, 2020-2035
- Table 304: Rest of MEA Market Size (USD Million), by End User, 2020-2035
List of Figures
- Figure 1: Global Market (USD Million) Forecast, by Product Type, 2020-2035
- Figure 2: Global Market (USD Million) Forecast, by Lipid Formulation Kits, 2020-2035
- Figure 3: Global Market (USD Million) Forecast, by Nucleic Acid Encapsulation Reagents, 2020-2035
- Figure 4: Global Market (USD Million) Forecast, by Stabilization & Cryoprotectant Kits, 2020-2035
- Figure 5: Global Market (USD Million) Forecast, by Surface Functionalization Reagents, 2020-2035
- Figure 6: Global Market (USD Million) Forecast, by Purification & Characterization Kits, 2020-2035
- Figure 7: Global Market (USD Million) Forecast, by Application, 2020-2035
- Figure 8: Global Market (USD Million) Forecast, by mRNA Vaccines, 2020-2035
- Figure 9: Global Market (USD Million) Forecast, by Gene Therapy & siRNA Delivery, 2020-2035
- Figure 10: Global Market (USD Million) Forecast, by Drug Delivery Research, 2020-2035
- Figure 11: Global Market (USD Million) Forecast, by Cell Therapy Manufacturing, 2020-2035
- Figure 12: Global Market (USD Million) Forecast, by Protein Replacement Therapy, 2020-2035
- Figure 13: Global Market (USD Million) Forecast, by Technology, 2020-2035
- Figure 14: Global Market (USD Million) Forecast, by Microfluidic Mixing Systems, 2020-2035
- Figure 15: Global Market (USD Million) Forecast, by Ethanol Injection / Solvent Exchange, 2020-2035
- Figure 16: Global Market (USD Million) Forecast, by pH Gradient-Based Assembly, 2020-2035
- Figure 17: Global Market (USD Million) Forecast, by TFF & Size-Exclusion Purification Kits, 2020-2035
- Figure 18: Global Market (USD Million) Forecast, by Hybrid AI/ML Formulation Platforms, 2020-2035
- Figure 19: Global Market (USD Million) Forecast, by End User, 2020-2035
- Figure 20: Global Market (USD Million) Forecast, by Pharma & Biotech Companies, 2020-2035
- Figure 21: Global Market (USD Million) Forecast, by Academic & Research Institutes, 2020-2035
- Figure 22: Global Market (USD Million) Forecast, by CDMOs / CROs, 2020-2035
- Figure 23: Global Market (USD Million) Forecast, by Reagent Manufacturers & Distributors, 2020-2035
- Figure 24: North America Market (USD Million) Forecast, by Product Type, 2020-2035
- Figure 25: North America Market (USD Million) Forecast, by Lipid Formulation Kits, 2020-2035
- Figure 26: North America Market (USD Million) Forecast, by Nucleic Acid Encapsulation Reagents, 2020-2035
- Figure 27: North America Market (USD Million) Forecast, by Stabilization & Cryoprotectant Kits, 2020-2035
- Figure 28: North America Market (USD Million) Forecast, by Surface Functionalization Reagents, 2020-2035
- Figure 29: North America Market (USD Million) Forecast, by Purification & Characterization Kits, 2020-2035
- Figure 30: North America Market (USD Million) Forecast, by Application, 2020-2035
- Figure 31: North America Market (USD Million) Forecast, by mRNA Vaccines, 2020-2035
- Figure 32: North America Market (USD Million) Forecast, by Gene Therapy & siRNA Delivery, 2020-2035
- Figure 33: North America Market (USD Million) Forecast, by Drug Delivery Research, 2020-2035
- Figure 34: North America Market (USD Million) Forecast, by Cell Therapy Manufacturing, 2020-2035
- Figure 35: North America Market (USD Million) Forecast, by Protein Replacement Therapy, 2020-2035
- Figure 36: North America Market (USD Million) Forecast, by Technology, 2020-2035
- Figure 37: North America Market (USD Million) Forecast, by Microfluidic Mixing Systems, 2020-2035
- Figure 38: North America Market (USD Million) Forecast, by Ethanol Injection / Solvent Exchange, 2020-2035
- Figure 39: North America Market (USD Million) Forecast, by pH Gradient-Based Assembly, 2020-2035
- Figure 40: North America Market (USD Million) Forecast, by TFF & Size-Exclusion Purification Kits, 2020-2035
- Figure 41: North America Market (USD Million) Forecast, by Hybrid AI/ML Formulation Platforms, 2020-2035
- Figure 42: North America Market (USD Million) Forecast, by End User, 2020-2035
- Figure 43: North America Market (USD Million) Forecast, by Pharma & Biotech Companies, 2020-2035
- Figure 44: North America Market (USD Million) Forecast, by Academic & Research Institutes, 2020-2035
- Figure 45: North America Market (USD Million) Forecast, by CDMOs / CROs, 2020-2035
- Figure 46: North America Market (USD Million) Forecast, by Reagent Manufacturers & Distributors, 2020-2035
- Figure 47: USA Market (USD Million) Forecast, by Product Type, 2020-2035
- Figure 48: USA Market (USD Million) Forecast, by Lipid Formulation Kits, 2020-2035
- Figure 49: USA Market (USD Million) Forecast, by Application, 2020-2035
- Figure 50: USA Market (USD Million) Forecast, by mRNA Vaccines, 2020-2035
- Figure 51: USA Market (USD Million) Forecast, by Technology, 2020-2035
- Figure 52: USA Market (USD Million) Forecast, by Microfluidic Mixing Systems, 2020-2035
- Figure 53: USA Market (USD Million) Forecast, by End User, 2020-2035
- Figure 54: USA Market (USD Million) Forecast, by Pharma & Biotech Companies, 2020-2035
- Figure 55: Canada Market (USD Million) Forecast, by Product Type, 2020-2035
- Figure 56: Canada Market (USD Million) Forecast, by Lipid Formulation Kits, 2020-2035
- Figure 57: Canada Market (USD Million) Forecast, by Application, 2020-2035
- Figure 58: Canada Market (USD Million) Forecast, by mRNA Vaccines, 2020-2035
- Figure 59: Canada Market (USD Million) Forecast, by Technology, 2020-2035
- Figure 60: Canada Market (USD Million) Forecast, by Microfluidic Mixing Systems, 2020-2035
- Figure 61: Canada Market (USD Million) Forecast, by End User, 2020-2035
- Figure 62: Canada Market (USD Million) Forecast, by Pharma & Biotech Companies, 2020-2035
- Figure 63: Mexico Market (USD Million) Forecast, by Product Type, 2020-2035
- Figure 64: Mexico Market (USD Million) Forecast, by Lipid Formulation Kits, 2020-2035
- Figure 65: Mexico Market (USD Million) Forecast, by Application, 2020-2035
- Figure 66: Mexico Market (USD Million) Forecast, by mRNA Vaccines, 2020-2035
- Figure 67: Mexico Market (USD Million) Forecast, by Technology, 2020-2035
- Figure 68: Mexico Market (USD Million) Forecast, by Microfluidic Mixing Systems, 2020-2035
- Figure 69: Mexico Market (USD Million) Forecast, by End User, 2020-2035
- Figure 70: Mexico Market (USD Million) Forecast, by Pharma & Biotech Companies, 2020-2035
- Figure 71: Latin America Market (USD Million) Forecast, by Product Type, 2020-2035
- Figure 72: Latin America Market (USD Million) Forecast, by Lipid Formulation Kits, 2020-2035
- Figure 73: Latin America Market (USD Million) Forecast, by Nucleic Acid Encapsulation Reagents, 2020-2035
- Figure 74: Latin America Market (USD Million) Forecast, by Stabilization & Cryoprotectant Kits, 2020-2035
- Figure 75: Latin America Market (USD Million) Forecast, by Surface Functionalization Reagents, 2020-2035
- Figure 76: Latin America Market (USD Million) Forecast, by Purification & Characterization Kits, 2020-2035
- Figure 77: Latin America Market (USD Million) Forecast, by Application, 2020-2035
- Figure 78: Latin America Market (USD Million) Forecast, by mRNA Vaccines, 2020-2035
- Figure 79: Latin America Market (USD Million) Forecast, by Gene Therapy & siRNA Delivery, 2020-2035
- Figure 80: Latin America Market (USD Million) Forecast, by Drug Delivery Research, 2020-2035
- Figure 81: Latin America Market (USD Million) Forecast, by Cell Therapy Manufacturing, 2020-2035
- Figure 82: Latin America Market (USD Million) Forecast, by Protein Replacement Therapy, 2020-2035
- Figure 83: Latin America Market (USD Million) Forecast, by Technology, 2020-2035
- Figure 84: Latin America Market (USD Million) Forecast, by Microfluidic Mixing Systems, 2020-2035
- Figure 85: Latin America Market (USD Million) Forecast, by Ethanol Injection / Solvent Exchange, 2020-2035
- Figure 86: Latin America Market (USD Million) Forecast, by pH Gradient-Based Assembly, 2020-2035
- Figure 87: Latin America Market (USD Million) Forecast, by TFF & Size-Exclusion Purification Kits, 2020-2035
- Figure 88: Latin America Market (USD Million) Forecast, by Hybrid AI/ML Formulation Platforms, 2020-2035
- Figure 89: Latin America Market (USD Million) Forecast, by End User, 2020-2035
- Figure 90: Latin America Market (USD Million) Forecast, by Pharma & Biotech Companies, 2020-2035
- Figure 91: Latin America Market (USD Million) Forecast, by Academic & Research Institutes, 2020-2035
- Figure 92: Latin America Market (USD Million) Forecast, by CDMOs / CROs, 2020-2035
- Figure 93: Latin America Market (USD Million) Forecast, by Reagent Manufacturers & Distributors, 2020-2035
- Figure 94: Brazil Market (USD Million) Forecast, by Product Type, 2020-2035
- Figure 95: Brazil Market (USD Million) Forecast, by Lipid Formulation Kits, 2020-2035
- Figure 96: Brazil Market (USD Million) Forecast, by Application, 2020-2035
- Figure 97: Brazil Market (USD Million) Forecast, by mRNA Vaccines, 2020-2035
- Figure 98: Brazil Market (USD Million) Forecast, by Technology, 2020-2035
- Figure 99: Brazil Market (USD Million) Forecast, by Microfluidic Mixing Systems, 2020-2035
- Figure 100: Brazil Market (USD Million) Forecast, by End User, 2020-2035
- Figure 101: Brazil Market (USD Million) Forecast, by Pharma & Biotech Companies, 2020-2035
- Figure 102: Argentina Market (USD Million) Forecast, by Product Type, 2020-2035
- Figure 103: Argentina Market (USD Million) Forecast, by Lipid Formulation Kits, 2020-2035
- Figure 104: Argentina Market (USD Million) Forecast, by Application, 2020-2035
- Figure 105: Argentina Market (USD Million) Forecast, by mRNA Vaccines, 2020-2035
- Figure 106: Argentina Market (USD Million) Forecast, by Technology, 2020-2035
- Figure 107: Argentina Market (USD Million) Forecast, by Microfluidic Mixing Systems, 2020-2035
- Figure 108: Argentina Market (USD Million) Forecast, by End User, 2020-2035
- Figure 109: Argentina Market (USD Million) Forecast, by Pharma & Biotech Companies, 2020-2035
- Figure 110: Rest of Latin America Market (USD Million) Forecast, by Product Type, 2020-2035
- Figure 111: Rest of Latin America Market (USD Million) Forecast, by Lipid Formulation Kits, 2020-2035
- Figure 112: Rest of Latin America Market (USD Million) Forecast, by Application, 2020-2035
- Figure 113: Rest of Latin America Market (USD Million) Forecast, by mRNA Vaccines, 2020-2035
- Figure 114: Rest of Latin America Market (USD Million) Forecast, by Technology, 2020-2035
- Figure 115: Rest of Latin America Market (USD Million) Forecast, by Microfluidic Mixing Systems, 2020-2035
- Figure 116: Rest of Latin America Market (USD Million) Forecast, by End User, 2020-2035
- Figure 117: Rest of Latin America Market (USD Million) Forecast, by Pharma & Biotech Companies, 2020-2035
- Figure 118: East Asia Market (USD Million) Forecast, by Product Type, 2020-2035
- Figure 119: East Asia Market (USD Million) Forecast, by Lipid Formulation Kits, 2020-2035
- Figure 120: East Asia Market (USD Million) Forecast, by Nucleic Acid Encapsulation Reagents, 2020-2035
- Figure 121: East Asia Market (USD Million) Forecast, by Stabilization & Cryoprotectant Kits, 2020-2035
- Figure 122: East Asia Market (USD Million) Forecast, by Surface Functionalization Reagents, 2020-2035
- Figure 123: East Asia Market (USD Million) Forecast, by Purification & Characterization Kits, 2020-2035
- Figure 124: East Asia Market (USD Million) Forecast, by Application, 2020-2035
- Figure 125: East Asia Market (USD Million) Forecast, by mRNA Vaccines, 2020-2035
- Figure 126: East Asia Market (USD Million) Forecast, by Gene Therapy & siRNA Delivery, 2020-2035
- Figure 127: East Asia Market (USD Million) Forecast, by Drug Delivery Research, 2020-2035
- Figure 128: East Asia Market (USD Million) Forecast, by Cell Therapy Manufacturing, 2020-2035
- Figure 129: East Asia Market (USD Million) Forecast, by Protein Replacement Therapy, 2020-2035
- Figure 130: East Asia Market (USD Million) Forecast, by Technology, 2020-2035
- Figure 131: East Asia Market (USD Million) Forecast, by Microfluidic Mixing Systems, 2020-2035
- Figure 132: East Asia Market (USD Million) Forecast, by Ethanol Injection / Solvent Exchange, 2020-2035
- Figure 133: East Asia Market (USD Million) Forecast, by pH Gradient-Based Assembly, 2020-2035
- Figure 134: East Asia Market (USD Million) Forecast, by TFF & Size-Exclusion Purification Kits, 2020-2035
- Figure 135: East Asia Market (USD Million) Forecast, by Hybrid AI/ML Formulation Platforms, 2020-2035
- Figure 136: East Asia Market (USD Million) Forecast, by End User, 2020-2035
- Figure 137: East Asia Market (USD Million) Forecast, by Pharma & Biotech Companies, 2020-2035
- Figure 138: East Asia Market (USD Million) Forecast, by Academic & Research Institutes, 2020-2035
- Figure 139: East Asia Market (USD Million) Forecast, by CDMOs / CROs, 2020-2035
- Figure 140: East Asia Market (USD Million) Forecast, by Reagent Manufacturers & Distributors, 2020-2035
- Figure 141: China Market (USD Million) Forecast, by Product Type, 2020-2035
- Figure 142: China Market (USD Million) Forecast, by Lipid Formulation Kits, 2020-2035
- Figure 143: China Market (USD Million) Forecast, by Application, 2020-2035
- Figure 144: China Market (USD Million) Forecast, by mRNA Vaccines, 2020-2035
- Figure 145: China Market (USD Million) Forecast, by Technology, 2020-2035
- Figure 146: China Market (USD Million) Forecast, by Microfluidic Mixing Systems, 2020-2035
- Figure 147: China Market (USD Million) Forecast, by End User, 2020-2035
- Figure 148: China Market (USD Million) Forecast, by Pharma & Biotech Companies, 2020-2035
- Figure 149: Japan Market (USD Million) Forecast, by Product Type, 2020-2035
- Figure 150: Japan Market (USD Million) Forecast, by Lipid Formulation Kits, 2020-2035
- Figure 151: Japan Market (USD Million) Forecast, by Application, 2020-2035
- Figure 152: Japan Market (USD Million) Forecast, by mRNA Vaccines, 2020-2035
- Figure 153: Japan Market (USD Million) Forecast, by Technology, 2020-2035
- Figure 154: Japan Market (USD Million) Forecast, by Microfluidic Mixing Systems, 2020-2035
- Figure 155: Japan Market (USD Million) Forecast, by End User, 2020-2035
- Figure 156: Japan Market (USD Million) Forecast, by Pharma & Biotech Companies, 2020-2035
- Figure 157: South Korea Market (USD Million) Forecast, by Product Type, 2020-2035
- Figure 158: South Korea Market (USD Million) Forecast, by Lipid Formulation Kits, 2020-2035
- Figure 159: South Korea Market (USD Million) Forecast, by Application, 2020-2035
- Figure 160: South Korea Market (USD Million) Forecast, by mRNA Vaccines, 2020-2035
- Figure 161: South Korea Market (USD Million) Forecast, by Technology, 2020-2035
- Figure 162: South Korea Market (USD Million) Forecast, by Microfluidic Mixing Systems, 2020-2035
- Figure 163: South Korea Market (USD Million) Forecast, by End User, 2020-2035
- Figure 164: South Korea Market (USD Million) Forecast, by Pharma & Biotech Companies, 2020-2035
- Figure 165: South Asia Market (USD Million) Forecast, by Product Type, 2020-2035
- Figure 166: South Asia Market (USD Million) Forecast, by Lipid Formulation Kits, 2020-2035
- Figure 167: South Asia Market (USD Million) Forecast, by Nucleic Acid Encapsulation Reagents, 2020-2035
- Figure 168: South Asia Market (USD Million) Forecast, by Stabilization & Cryoprotectant Kits, 2020-2035
- Figure 169: South Asia Market (USD Million) Forecast, by Surface Functionalization Reagents, 2020-2035
- Figure 170: South Asia Market (USD Million) Forecast, by Purification & Characterization Kits, 2020-2035
- Figure 171: South Asia Market (USD Million) Forecast, by Application, 2020-2035
- Figure 172: South Asia Market (USD Million) Forecast, by mRNA Vaccines, 2020-2035
- Figure 173: South Asia Market (USD Million) Forecast, by Gene Therapy & siRNA Delivery, 2020-2035
- Figure 174: South Asia Market (USD Million) Forecast, by Drug Delivery Research, 2020-2035
- Figure 175: South Asia Market (USD Million) Forecast, by Cell Therapy Manufacturing, 2020-2035
- Figure 176: South Asia Market (USD Million) Forecast, by Protein Replacement Therapy, 2020-2035
- Figure 177: South Asia Market (USD Million) Forecast, by Technology, 2020-2035
- Figure 178: South Asia Market (USD Million) Forecast, by Microfluidic Mixing Systems, 2020-2035
- Figure 179: South Asia Market (USD Million) Forecast, by Ethanol Injection / Solvent Exchange, 2020-2035
- Figure 180: South Asia Market (USD Million) Forecast, by pH Gradient-Based Assembly, 2020-2035
- Figure 181: South Asia Market (USD Million) Forecast, by TFF & Size-Exclusion Purification Kits, 2020-2035
- Figure 182: South Asia Market (USD Million) Forecast, by Hybrid AI/ML Formulation Platforms, 2020-2035
- Figure 183: South Asia Market (USD Million) Forecast, by End User, 2020-2035
- Figure 184: South Asia Market (USD Million) Forecast, by Pharma & Biotech Companies, 2020-2035
- Figure 185: South Asia Market (USD Million) Forecast, by Academic & Research Institutes, 2020-2035
- Figure 186: South Asia Market (USD Million) Forecast, by CDMOs / CROs, 2020-2035
- Figure 187: South Asia Market (USD Million) Forecast, by Reagent Manufacturers & Distributors, 2020-2035
- Figure 188: India Market (USD Million) Forecast, by Product Type, 2020-2035
- Figure 189: India Market (USD Million) Forecast, by Lipid Formulation Kits, 2020-2035
- Figure 190: India Market (USD Million) Forecast, by Application, 2020-2035
- Figure 191: India Market (USD Million) Forecast, by mRNA Vaccines, 2020-2035
- Figure 192: India Market (USD Million) Forecast, by Technology, 2020-2035
- Figure 193: India Market (USD Million) Forecast, by Microfluidic Mixing Systems, 2020-2035
- Figure 194: India Market (USD Million) Forecast, by End User, 2020-2035
- Figure 195: India Market (USD Million) Forecast, by Pharma & Biotech Companies, 2020-2035
- Figure 196: ASEAN Countries Market (USD Million) Forecast, by Product Type, 2020-2035
- Figure 197: ASEAN Countries Market (USD Million) Forecast, by Lipid Formulation Kits, 2020-2035
- Figure 198: ASEAN Countries Market (USD Million) Forecast, by Application, 2020-2035
- Figure 199: ASEAN Countries Market (USD Million) Forecast, by mRNA Vaccines, 2020-2035
- Figure 200: ASEAN Countries Market (USD Million) Forecast, by Technology, 2020-2035
- Figure 201: ASEAN Countries Market (USD Million) Forecast, by Microfluidic Mixing Systems, 2020-2035
- Figure 202: ASEAN Countries Market (USD Million) Forecast, by End User, 2020-2035
- Figure 203: ASEAN Countries Market (USD Million) Forecast, by Pharma & Biotech Companies, 2020-2035
- Figure 204: Australia & New Zealand Market (USD Million) Forecast, by Product Type, 2020-2035
- Figure 205: Australia & New Zealand Market (USD Million) Forecast, by Lipid Formulation Kits, 2020-2035
- Figure 206: Australia & New Zealand Market (USD Million) Forecast, by Application, 2020-2035
- Figure 207: Australia & New Zealand Market (USD Million) Forecast, by mRNA Vaccines, 2020-2035
- Figure 208: Australia & New Zealand Market (USD Million) Forecast, by Technology, 2020-2035
- Figure 209: Australia & New Zealand Market (USD Million) Forecast, by Microfluidic Mixing Systems, 2020-2035
- Figure 210: Australia & New Zealand Market (USD Million) Forecast, by End User, 2020-2035
- Figure 211: Australia & New Zealand Market (USD Million) Forecast, by Pharma & Biotech Companies, 2020-2035
- Figure 212: Rest of South Asia Market (USD Million) Forecast, by Product Type, 2020-2035
- Figure 213: Rest of South Asia Market (USD Million) Forecast, by Lipid Formulation Kits, 2020-2035
- Figure 214: Rest of South Asia Market (USD Million) Forecast, by Application, 2020-2035
- Figure 215: Rest of South Asia Market (USD Million) Forecast, by mRNA Vaccines, 2020-2035
- Figure 216: Rest of South Asia Market (USD Million) Forecast, by Technology, 2020-2035
- Figure 217: Rest of South Asia Market (USD Million) Forecast, by Microfluidic Mixing Systems, 2020-2035
- Figure 218: Rest of South Asia Market (USD Million) Forecast, by End User, 2020-2035
- Figure 219: Rest of South Asia Market (USD Million) Forecast, by Pharma & Biotech Companies, 2020-2035
- Figure 220: Western Europe Market (USD Million) Forecast, by Product Type, 2020-2035
- Figure 221: Western Europe Market (USD Million) Forecast, by Lipid Formulation Kits, 2020-2035
- Figure 222: Western Europe Market (USD Million) Forecast, by Nucleic Acid Encapsulation Reagents, 2020-2035
- Figure 223: Western Europe Market (USD Million) Forecast, by Stabilization & Cryoprotectant Kits, 2020-2035
- Figure 224: Western Europe Market (USD Million) Forecast, by Surface Functionalization Reagents, 2020-2035
- Figure 225: Western Europe Market (USD Million) Forecast, by Purification & Characterization Kits, 2020-2035
- Figure 226: Western Europe Market (USD Million) Forecast, by Application, 2020-2035
- Figure 227: Western Europe Market (USD Million) Forecast, by mRNA Vaccines, 2020-2035
- Figure 228: Western Europe Market (USD Million) Forecast, by Gene Therapy & siRNA Delivery, 2020-2035
- Figure 229: Western Europe Market (USD Million) Forecast, by Drug Delivery Research, 2020-2035
- Figure 230: Western Europe Market (USD Million) Forecast, by Cell Therapy Manufacturing, 2020-2035
- Figure 231: Western Europe Market (USD Million) Forecast, by Protein Replacement Therapy, 2020-2035
- Figure 232: Western Europe Market (USD Million) Forecast, by Technology, 2020-2035
- Figure 233: Western Europe Market (USD Million) Forecast, by Microfluidic Mixing Systems, 2020-2035
- Figure 234: Western Europe Market (USD Million) Forecast, by Ethanol Injection / Solvent Exchange, 2020-2035
- Figure 235: Western Europe Market (USD Million) Forecast, by pH Gradient-Based Assembly, 2020-2035
- Figure 236: Western Europe Market (USD Million) Forecast, by TFF & Size-Exclusion Purification Kits, 2020-2035
- Figure 237: Western Europe Market (USD Million) Forecast, by Hybrid AI/ML Formulation Platforms, 2020-2035
- Figure 238: Western Europe Market (USD Million) Forecast, by End User, 2020-2035
- Figure 239: Western Europe Market (USD Million) Forecast, by Pharma & Biotech Companies, 2020-2035
- Figure 240: Western Europe Market (USD Million) Forecast, by Academic & Research Institutes, 2020-2035
- Figure 241: Western Europe Market (USD Million) Forecast, by CDMOs / CROs, 2020-2035
- Figure 242: Western Europe Market (USD Million) Forecast, by Reagent Manufacturers & Distributors, 2020-2035
- Figure 243: Germany Market (USD Million) Forecast, by Product Type, 2020-2035
- Figure 244: Germany Market (USD Million) Forecast, by Lipid Formulation Kits, 2020-2035
- Figure 245: Germany Market (USD Million) Forecast, by Application, 2020-2035
- Figure 246: Germany Market (USD Million) Forecast, by mRNA Vaccines, 2020-2035
- Figure 247: Germany Market (USD Million) Forecast, by Technology, 2020-2035
- Figure 248: Germany Market (USD Million) Forecast, by Microfluidic Mixing Systems, 2020-2035
- Figure 249: Germany Market (USD Million) Forecast, by End User, 2020-2035
- Figure 250: Germany Market (USD Million) Forecast, by Pharma & Biotech Companies, 2020-2035
- Figure 251: UK Market (USD Million) Forecast, by Product Type, 2020-2035
- Figure 252: UK Market (USD Million) Forecast, by Lipid Formulation Kits, 2020-2035
- Figure 253: UK Market (USD Million) Forecast, by Application, 2020-2035
- Figure 254: UK Market (USD Million) Forecast, by mRNA Vaccines, 2020-2035
- Figure 255: UK Market (USD Million) Forecast, by Technology, 2020-2035
- Figure 256: UK Market (USD Million) Forecast, by Microfluidic Mixing Systems, 2020-2035
- Figure 257: UK Market (USD Million) Forecast, by End User, 2020-2035
- Figure 258: UK Market (USD Million) Forecast, by Pharma & Biotech Companies, 2020-2035
- Figure 259: France Market (USD Million) Forecast, by Product Type, 2020-2035
- Figure 260: France Market (USD Million) Forecast, by Lipid Formulation Kits, 2020-2035
- Figure 261: France Market (USD Million) Forecast, by Application, 2020-2035
- Figure 262: France Market (USD Million) Forecast, by mRNA Vaccines, 2020-2035
- Figure 263: France Market (USD Million) Forecast, by Technology, 2020-2035
- Figure 264: France Market (USD Million) Forecast, by Microfluidic Mixing Systems, 2020-2035
- Figure 265: France Market (USD Million) Forecast, by End User, 2020-2035
- Figure 266: France Market (USD Million) Forecast, by Pharma & Biotech Companies, 2020-2035
- Figure 267: Italy Market (USD Million) Forecast, by Product Type, 2020-2035
- Figure 268: Italy Market (USD Million) Forecast, by Lipid Formulation Kits, 2020-2035
- Figure 269: Italy Market (USD Million) Forecast, by Application, 2020-2035
- Figure 270: Italy Market (USD Million) Forecast, by mRNA Vaccines, 2020-2035
- Figure 271: Italy Market (USD Million) Forecast, by Technology, 2020-2035
- Figure 272: Italy Market (USD Million) Forecast, by Microfluidic Mixing Systems, 2020-2035
- Figure 273: Italy Market (USD Million) Forecast, by End User, 2020-2035
- Figure 274: Italy Market (USD Million) Forecast, by Pharma & Biotech Companies, 2020-2035
- Figure 275: Spain Market (USD Million) Forecast, by Product Type, 2020-2035
- Figure 276: Spain Market (USD Million) Forecast, by Lipid Formulation Kits, 2020-2035
- Figure 277: Spain Market (USD Million) Forecast, by Application, 2020-2035
- Figure 278: Spain Market (USD Million) Forecast, by mRNA Vaccines, 2020-2035
- Figure 279: Spain Market (USD Million) Forecast, by Technology, 2020-2035
- Figure 280: Spain Market (USD Million) Forecast, by Microfluidic Mixing Systems, 2020-2035
- Figure 281: Spain Market (USD Million) Forecast, by End User, 2020-2035
- Figure 282: Spain Market (USD Million) Forecast, by Pharma & Biotech Companies, 2020-2035
- Figure 283: BENELUX Market (USD Million) Forecast, by Product Type, 2020-2035
- Figure 284: BENELUX Market (USD Million) Forecast, by Lipid Formulation Kits, 2020-2035
- Figure 285: BENELUX Market (USD Million) Forecast, by Application, 2020-2035
- Figure 286: BENELUX Market (USD Million) Forecast, by mRNA Vaccines, 2020-2035
- Figure 287: BENELUX Market (USD Million) Forecast, by Technology, 2020-2035
- Figure 288: BENELUX Market (USD Million) Forecast, by Microfluidic Mixing Systems, 2020-2035
- Figure 289: BENELUX Market (USD Million) Forecast, by End User, 2020-2035
- Figure 290: BENELUX Market (USD Million) Forecast, by Pharma & Biotech Companies, 2020-2035
- Figure 291: Nordic Countries Market (USD Million) Forecast, by Product Type, 2020-2035
- Figure 292: Nordic Countries Market (USD Million) Forecast, by Lipid Formulation Kits, 2020-2035
- Figure 293: Nordic Countries Market (USD Million) Forecast, by Application, 2020-2035
- Figure 294: Nordic Countries Market (USD Million) Forecast, by mRNA Vaccines, 2020-2035
- Figure 295: Nordic Countries Market (USD Million) Forecast, by Technology, 2020-2035
- Figure 296: Nordic Countries Market (USD Million) Forecast, by Microfluidic Mixing Systems, 2020-2035
- Figure 297: Nordic Countries Market (USD Million) Forecast, by End User, 2020-2035
- Figure 298: Nordic Countries Market (USD Million) Forecast, by Pharma & Biotech Companies, 2020-2035
- Figure 299: Rest of Western Europe Market (USD Million) Forecast, by Product Type, 2020-2035
- Figure 300: Rest of Western Europe Market (USD Million) Forecast, by Lipid Formulation Kits, 2020-2035
- Figure 301: Rest of Western Europe Market (USD Million) Forecast, by Application, 2020-2035
- Figure 302: Rest of Western Europe Market (USD Million) Forecast, by mRNA Vaccines, 2020-2035
- Figure 303: Rest of Western Europe Market (USD Million) Forecast, by Technology, 2020-2035
- Figure 304: Rest of Western Europe Market (USD Million) Forecast, by Microfluidic Mixing Systems, 2020-2035
- Figure 305: Rest of Western Europe Market (USD Million) Forecast, by End User, 2020-2035
- Figure 306: Rest of Western Europe Market (USD Million) Forecast, by Pharma & Biotech Companies, 2020-2035
- Figure 307: Eastern Europe Market (USD Million) Forecast, by Product Type, 2020-2035
- Figure 308: Eastern Europe Market (USD Million) Forecast, by Lipid Formulation Kits, 2020-2035
- Figure 309: Eastern Europe Market (USD Million) Forecast, by Nucleic Acid Encapsulation Reagents, 2020-2035
- Figure 310: Eastern Europe Market (USD Million) Forecast, by Stabilization & Cryoprotectant Kits, 2020-2035
- Figure 311: Eastern Europe Market (USD Million) Forecast, by Surface Functionalization Reagents, 2020-2035
- Figure 312: Eastern Europe Market (USD Million) Forecast, by Purification & Characterization Kits, 2020-2035
- Figure 313: Eastern Europe Market (USD Million) Forecast, by Application, 2020-2035
- Figure 314: Eastern Europe Market (USD Million) Forecast, by mRNA Vaccines, 2020-2035
- Figure 315: Eastern Europe Market (USD Million) Forecast, by Gene Therapy & siRNA Delivery, 2020-2035
- Figure 316: Eastern Europe Market (USD Million) Forecast, by Drug Delivery Research, 2020-2035
- Figure 317: Eastern Europe Market (USD Million) Forecast, by Cell Therapy Manufacturing, 2020-2035
- Figure 318: Eastern Europe Market (USD Million) Forecast, by Protein Replacement Therapy, 2020-2035
- Figure 319: Eastern Europe Market (USD Million) Forecast, by Technology, 2020-2035
- Figure 320: Eastern Europe Market (USD Million) Forecast, by Microfluidic Mixing Systems, 2020-2035
- Figure 321: Eastern Europe Market (USD Million) Forecast, by Ethanol Injection / Solvent Exchange, 2020-2035
- Figure 322: Eastern Europe Market (USD Million) Forecast, by pH Gradient-Based Assembly, 2020-2035
- Figure 323: Eastern Europe Market (USD Million) Forecast, by TFF & Size-Exclusion Purification Kits, 2020-2035
- Figure 324: Eastern Europe Market (USD Million) Forecast, by Hybrid AI/ML Formulation Platforms, 2020-2035
- Figure 325: Eastern Europe Market (USD Million) Forecast, by End User, 2020-2035
- Figure 326: Eastern Europe Market (USD Million) Forecast, by Pharma & Biotech Companies, 2020-2035
- Figure 327: Eastern Europe Market (USD Million) Forecast, by Academic & Research Institutes, 2020-2035
- Figure 328: Eastern Europe Market (USD Million) Forecast, by CDMOs / CROs, 2020-2035
- Figure 329: Eastern Europe Market (USD Million) Forecast, by Reagent Manufacturers & Distributors, 2020-2035
- Figure 330: Russia Market (USD Million) Forecast, by Product Type, 2020-2035
- Figure 331: Russia Market (USD Million) Forecast, by Lipid Formulation Kits, 2020-2035
- Figure 332: Russia Market (USD Million) Forecast, by Application, 2020-2035
- Figure 333: Russia Market (USD Million) Forecast, by mRNA Vaccines, 2020-2035
- Figure 334: Russia Market (USD Million) Forecast, by Technology, 2020-2035
- Figure 335: Russia Market (USD Million) Forecast, by Microfluidic Mixing Systems, 2020-2035
- Figure 336: Russia Market (USD Million) Forecast, by End User, 2020-2035
- Figure 337: Russia Market (USD Million) Forecast, by Pharma & Biotech Companies, 2020-2035
- Figure 338: Hungary Market (USD Million) Forecast, by Product Type, 2020-2035
- Figure 339: Hungary Market (USD Million) Forecast, by Lipid Formulation Kits, 2020-2035
- Figure 340: Hungary Market (USD Million) Forecast, by Application, 2020-2035
- Figure 341: Hungary Market (USD Million) Forecast, by mRNA Vaccines, 2020-2035
- Figure 342: Hungary Market (USD Million) Forecast, by Technology, 2020-2035
- Figure 343: Hungary Market (USD Million) Forecast, by Microfluidic Mixing Systems, 2020-2035
- Figure 344: Hungary Market (USD Million) Forecast, by End User, 2020-2035
- Figure 345: Hungary Market (USD Million) Forecast, by Pharma & Biotech Companies, 2020-2035
- Figure 346: Poland Market (USD Million) Forecast, by Product Type, 2020-2035
- Figure 347: Poland Market (USD Million) Forecast, by Lipid Formulation Kits, 2020-2035
- Figure 348: Poland Market (USD Million) Forecast, by Application, 2020-2035
- Figure 349: Poland Market (USD Million) Forecast, by mRNA Vaccines, 2020-2035
- Figure 350: Poland Market (USD Million) Forecast, by Technology, 2020-2035
- Figure 351: Poland Market (USD Million) Forecast, by Microfluidic Mixing Systems, 2020-2035
- Figure 352: Poland Market (USD Million) Forecast, by End User, 2020-2035
- Figure 353: Poland Market (USD Million) Forecast, by Pharma & Biotech Companies, 2020-2035
- Figure 354: Rest of Eastern Europe Market (USD Million) Forecast, by Product Type, 2020-2035
- Figure 355: Rest of Eastern Europe Market (USD Million) Forecast, by Lipid Formulation Kits, 2020-2035
- Figure 356: Rest of Eastern Europe Market (USD Million) Forecast, by Application, 2020-2035
- Figure 357: Rest of Eastern Europe Market (USD Million) Forecast, by mRNA Vaccines, 2020-2035
- Figure 358: Rest of Eastern Europe Market (USD Million) Forecast, by Technology, 2020-2035
- Figure 359: Rest of Eastern Europe Market (USD Million) Forecast, by Microfluidic Mixing Systems, 2020-2035
- Figure 360: Rest of Eastern Europe Market (USD Million) Forecast, by End User, 2020-2035
- Figure 361: Rest of Eastern Europe Market (USD Million) Forecast, by Pharma & Biotech Companies, 2020-2035
- Figure 362: Middle East & Africa Market (USD Million) Forecast, by Product Type, 2020-2035
- Figure 363: Middle East & Africa Market (USD Million) Forecast, by Lipid Formulation Kits, 2020-2035
- Figure 364: Middle East & Africa Market (USD Million) Forecast, by Nucleic Acid Encapsulation Reagents, 2020-2035
- Figure 365: Middle East & Africa Market (USD Million) Forecast, by Stabilization & Cryoprotectant Kits, 2020-2035
- Figure 366: Middle East & Africa Market (USD Million) Forecast, by Surface Functionalization Reagents, 2020-2035
- Figure 367: Middle East & Africa Market (USD Million) Forecast, by Purification & Characterization Kits, 2020-2035
- Figure 368: Middle East & Africa Market (USD Million) Forecast, by Application, 2020-2035
- Figure 369: Middle East & Africa Market (USD Million) Forecast, by mRNA Vaccines, 2020-2035
- Figure 370: Middle East & Africa Market (USD Million) Forecast, by Gene Therapy & siRNA Delivery, 2020-2035
- Figure 371: Middle East & Africa Market (USD Million) Forecast, by Drug Delivery Research, 2020-2035
- Figure 372: Middle East & Africa Market (USD Million) Forecast, by Cell Therapy Manufacturing, 2020-2035
- Figure 373: Middle East & Africa Market (USD Million) Forecast, by Protein Replacement Therapy, 2020-2035
- Figure 374: Middle East & Africa Market (USD Million) Forecast, by Technology, 2020-2035
- Figure 375: Middle East & Africa Market (USD Million) Forecast, by Microfluidic Mixing Systems, 2020-2035
- Figure 376: Middle East & Africa Market (USD Million) Forecast, by Ethanol Injection / Solvent Exchange, 2020-2035
- Figure 377: Middle East & Africa Market (USD Million) Forecast, by pH Gradient-Based Assembly, 2020-2035
- Figure 378: Middle East & Africa Market (USD Million) Forecast, by TFF & Size-Exclusion Purification Kits, 2020-2035
- Figure 379: Middle East & Africa Market (USD Million) Forecast, by Hybrid AI/ML Formulation Platforms, 2020-2035
- Figure 380: Middle East & Africa Market (USD Million) Forecast, by End User, 2020-2035
- Figure 381: Middle East & Africa Market (USD Million) Forecast, by Pharma & Biotech Companies, 2020-2035
- Figure 382: Middle East & Africa Market (USD Million) Forecast, by Academic & Research Institutes, 2020-2035
- Figure 383: Middle East & Africa Market (USD Million) Forecast, by CDMOs / CROs, 2020-2035
- Figure 384: Middle East & Africa Market (USD Million) Forecast, by Reagent Manufacturers & Distributors, 2020-2035
- Figure 385: Saudi Arabia Market (USD Million) Forecast, by Product Type, 2020-2035
- Figure 386: Saudi Arabia Market (USD Million) Forecast, by Lipid Formulation Kits, 2020-2035
- Figure 387: Saudi Arabia Market (USD Million) Forecast, by Application, 2020-2035
- Figure 388: Saudi Arabia Market (USD Million) Forecast, by mRNA Vaccines, 2020-2035
- Figure 389: Saudi Arabia Market (USD Million) Forecast, by Technology, 2020-2035
- Figure 390: Saudi Arabia Market (USD Million) Forecast, by Microfluidic Mixing Systems, 2020-2035
- Figure 391: Saudi Arabia Market (USD Million) Forecast, by End User, 2020-2035
- Figure 392: Saudi Arabia Market (USD Million) Forecast, by Pharma & Biotech Companies, 2020-2035
- Figure 393: Other GCC Countries Market (USD Million) Forecast, by Product Type, 2020-2035
- Figure 394: Other GCC Countries Market (USD Million) Forecast, by Lipid Formulation Kits, 2020-2035
- Figure 395: Other GCC Countries Market (USD Million) Forecast, by Application, 2020-2035
- Figure 396: Other GCC Countries Market (USD Million) Forecast, by mRNA Vaccines, 2020-2035
- Figure 397: Other GCC Countries Market (USD Million) Forecast, by Technology, 2020-2035
- Figure 398: Other GCC Countries Market (USD Million) Forecast, by Microfluidic Mixing Systems, 2020-2035
- Figure 399: Other GCC Countries Market (USD Million) Forecast, by End User, 2020-2035
- Figure 400: Other GCC Countries Market (USD Million) Forecast, by Pharma & Biotech Companies, 2020-2035
- Figure 401: Türkiye Market (USD Million) Forecast, by Product Type, 2020-2035
- Figure 402: Türkiye Market (USD Million) Forecast, by Lipid Formulation Kits, 2020-2035
- Figure 403: Türkiye Market (USD Million) Forecast, by Application, 2020-2035
- Figure 404: Türkiye Market (USD Million) Forecast, by mRNA Vaccines, 2020-2035
- Figure 405: Türkiye Market (USD Million) Forecast, by Technology, 2020-2035
- Figure 406: Türkiye Market (USD Million) Forecast, by Microfluidic Mixing Systems, 2020-2035
- Figure 407: Türkiye Market (USD Million) Forecast, by End User, 2020-2035
- Figure 408: Türkiye Market (USD Million) Forecast, by Pharma & Biotech Companies, 2020-2035
- Figure 409: South Africa Market (USD Million) Forecast, by Product Type, 2020-2035
- Figure 410: South Africa Market (USD Million) Forecast, by Lipid Formulation Kits, 2020-2035
- Figure 411: South Africa Market (USD Million) Forecast, by Application, 2020-2035
- Figure 412: South Africa Market (USD Million) Forecast, by mRNA Vaccines, 2020-2035
- Figure 413: South Africa Market (USD Million) Forecast, by Technology, 2020-2035
- Figure 414: South Africa Market (USD Million) Forecast, by Microfluidic Mixing Systems, 2020-2035
- Figure 415: South Africa Market (USD Million) Forecast, by End User, 2020-2035
- Figure 416: South Africa Market (USD Million) Forecast, by Pharma & Biotech Companies, 2020-2035
- Figure 417: Rest of MEA Market (USD Million) Forecast, by Product Type, 2020-2035
- Figure 418: Rest of MEA Market (USD Million) Forecast, by Lipid Formulation Kits, 2020-2035
- Figure 419: Rest of MEA Market (USD Million) Forecast, by Application, 2020-2035
- Figure 420: Rest of MEA Market (USD Million) Forecast, by mRNA Vaccines, 2020-2035
- Figure 421: Rest of MEA Market (USD Million) Forecast, by Technology, 2020-2035
- Figure 422: Rest of MEA Market (USD Million) Forecast, by Microfluidic Mixing Systems, 2020-2035
- Figure 423: Rest of MEA Market (USD Million) Forecast, by End User, 2020-2035
- Figure 424: Rest of MEA Market (USD Million) Forecast, by Pharma & Biotech Companies, 2020-2035
- Figure 425: Precision NanoSystems (Danaher) - Company Snapshot
- Figure 426: Precision NanoSystems (Danaher) - SWOT Analysis
- Figure 427: Evonik Industries AG - Company Snapshot
- Figure 428: Evonik Industries AG - SWOT Analysis
- Figure 429: Polymun Scientific - Company Snapshot
- Figure 430: Polymun Scientific - SWOT Analysis
- Figure 431: Acuitas Therapeutics - Company Snapshot
- Figure 432: Acuitas Therapeutics - SWOT Analysis
- Figure 433: Merck KGaA - Company Snapshot
- Figure 434: Merck KGaA - SWOT Analysis
- Figure 435: Avanti Polar Lipids (Croda) - Company Snapshot
- Figure 436: Avanti Polar Lipids (Croda) - SWOT Analysis
- Figure 437: CordenPharma International - Company Snapshot
- Figure 438: CordenPharma International - SWOT Analysis
- Figure 439: BioNTech Manufacturing Services - Company Snapshot
- Figure 440: BioNTech Manufacturing Services - SWOT Analysis
- Figure 441: Nitto Denko / Reprocell - Company Snapshot
- Figure 442: Nitto Denko / Reprocell - SWOT Analysis

