Large Molecule Bioanalytical Testing Services Market
Large Molecule Bioanalytical Testing Services Market is segmented by Phase (Clinical, Preclinical, With Antibody, Without Antibody), Service Category (Pharmacokinetics, ADA, Others), Test Type (Bioavailability and Bioequivalence, ADME, PK, PD, Other Tests), and Region. Forecast for 2026 to 2036.
Large Molecule Bioanalytical Testing Services Market Size, Market Forecast and Outlook By FMI
Summary of the Large Molecule Bioanalytical Testing Services Market
- Demand and Growth Drivers
- Expansion of biologic and biosimilar drug development pipelines is increasing demand for specialized bioanalytical testing services, including pharmacokinetic, immunogenicity, and biomarker analysis.
- Regulatory requirements for anti-drug antibody (ADA) testing and immunogenicity assessment are creating compliance-driven demand across all phases of biologic drug development.
- Outsourcing of bioanalytical testing by pharmaceutical companies to contract research organizations is accelerating, driven by the specialized capabilities and capital equipment requirements of large molecule analysis.
- Product and Segment View
- Clinical is expected to hold 61.3% of the Phase segment in 2026, reflecting growth in biologic drug development pipelines.
- Pharmacokinetics is expected to hold 57.8% of the Service Category segment in 2026, reflecting growth in biologic drug development pipelines.
- Bioavailability and Bioequivalence is expected to hold 46.0% of the Test Type segment in 2026, reflecting growth in biologic drug development pipelines.
- Geography and Competitive Outlook
- South Korea is expected to lead growth at 12.2%, supported by biosimilar manufacturing growth and clinical trial activity.
- USA follows at 12.0%, driven by the largest biologic drug development pipeline and CRO infrastructure globally.
- Labcorp Drug Development and other established players compete on assay portfolio and regulatory expertise.
- Analyst Opinion
- The Large Molecule Bioanalytical Testing Services Market is growing in line with the global biologic drug pipeline, with immunogenicity testing requirements creating compliance-driven demand that is less sensitive to discretionary R&D budgets.
- Outsourcing trends are accelerating as pharmaceutical companies recognize the capital and expertise requirements of maintaining in-house large molecule bioanalytical capabilities.
- Biosimilar development activity is creating an additional demand layer, with comparative bioanalytical studies required to demonstrate similarity to reference biologic products.
- CROs that combine validated assay portfolios with regulatory submission expertise and geographic flexibility are expected to capture a growing share of testing outsourcing budgets.
Large Molecule Bioanalytical Testing Services Market Definition
The Large Molecule Bioanalytical Testing Services Market covers outsourced and in-house laboratory testing services for biologic drug development, including pharmacokinetic analysis, anti-drug antibody testing, biomarker analysis, and bioavailability studies across preclinical and clinical development phases.
Large Molecule Bioanalytical Testing Services Market Inclusions
Market scope covers all commercially available products and services categorized by phase (Phase, Clinical, Preclinical, With Antibody, Without Antibody), service category (Service Category, Pharmacokinetics, ADA, Others), test type (Test Type, Bioavailability and Bioequivalence, ADME, PK, PD, Other Tests), therapeutic area (Therapeutic Area, Oncology, Infectious Diseases, Cardiology, Neurology, Others), end user (End User, Large Pharmaceutical Companies, Small and Medium Enterprises, Contract Research Organizations, Sponsor Organizations), distribution channel (Distribution Channel, Direct Contract Research Services, Third Party Laboratory Networks, Online Service Portals). Revenue coverage spans the 2026 to 2036 forecast period.
Large Molecule Bioanalytical Testing Services Market Exclusions
The scope does not include small molecule bioanalytical testing, analytical chemistry services for chemical drugs, clinical laboratory diagnostics for patient care, or manufacturing quality control testing.
Large Molecule Bioanalytical Testing Services Market Research Methodology
- Primary Research: Directors of bioanalytical laboratories, executives in pharmaceutical R&D, CRO business development managers, and regulatory affairs experts in important markets were interviewed by FMI analysts.
- Desk research: Integrated information from regulatory guidance documents, pharmaceutical pipeline trackers, clinical trial databases, and CRO financial disclosures.
- Market sizing and forecasting: Adoption velocity modeling, cross-validation against industry benchmarks, and bottom-up aggregation across sectors and geographies.
- Data validation: Quarterly cross-checked against CRO financial reports, pharmaceutical R&D spending surveys, and clinical trial initiation data.
Why is the Large Molecule Bioanalytical Testing Services Market Growing?
- Monoclonal antibodies, ADCs, and cell treatments necessitate specific bioanalytical characterisation, which generates a structured demand for testing services as the biologic drug development pipeline expands internationally.
- ADA screening and confirmatory assays are now required as part of development processes due to regulatory bodies like the FDA and EMA raising immunogenicity testing requirements for biologic submissions.
- Growth in biosimilar production and clinical trial activity are the main drivers of South Korea's 12.2% growth.
As biologic drug development activity rises worldwide, the market for large molecule bioanalytical testing services is growing. 61.3% of the phase segment is made up of clinical-phase testing, which reflects the number of ongoing biologic clinical studies that call for pharmacokinetic, immunogenicity, and biomarker analysis. Since drug concentration analysis and absorption studies are necessary at every stage of biologic development, pharmacokinetic testing tops the service category at 61.3%. The concentration of monoclonal antibody and ADC development programs aimed at solid tumors and hematological malignancies makes oncology the leading therapeutic area at 61.3%. Additional testing volume across several biologic modalities is contributed by neurology, cardiology, and infectious illnesses. 61.3% of end-user demand comes from large pharmaceutical companies, with the remaining portion coming from biotech startups and contract research firms. 61.3% of the test type segment is made up of bioavailability and bioequivalency testing, which reflects the expanding pipeline for biosimilar development that necessitates comparable analytical research. 61.3% of distribution is made up of direct contract research services, whereas long-term and project-based contracts give testing service providers financial stability.
Market Segmentation Analysis
- Clinical holds 61.3% of the Phase segment in 2026.
- Pharmacokinetics accounts for 57.8% of the Service Category segment.
The Large Molecule Bioanalytical Testing Services Market is organized across 6 segmentation dimensions: Phase, Service Category, Test Type, Therapeutic Area, End User, Distribution Channel. Procurement patterns reflect pharmaceutical R&D outsourcing decisions, regulatory testing requirements, and CRO selection criteria.
Insights into the Clinical Phase Segment
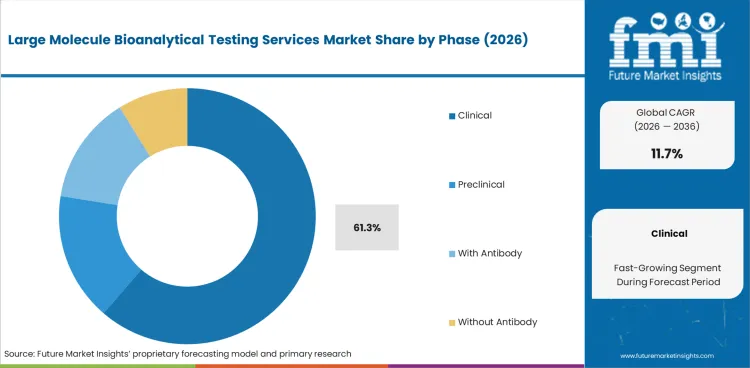
In 2026, Clinical is expected to account for 61.3% of the Phase segment. Key sub-categories include Phase I, Phase II, Phase III, each serving specific application requirements.
Clinical holds 61.3% of the segment, contributing additional demand through specialized applications. Preclinical is also contributing to segment growth as the market diversifies across product categories and application requirements.
Insights into the Bioavailability and Bioequivalence Test Type Segment
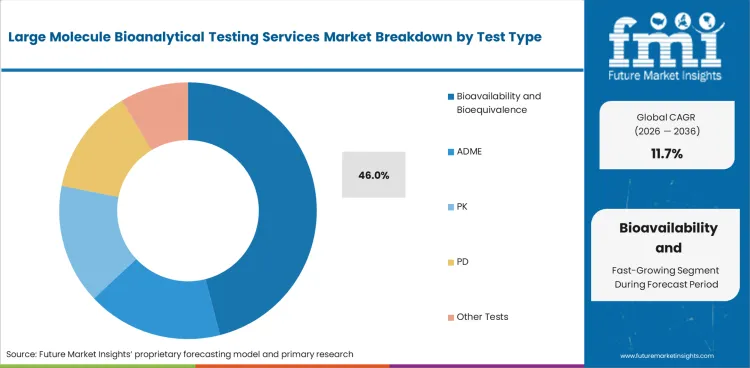
It is anticipated that bioavailability and bioequivalency will make up 46.0% of the Test Type segment in 2026. Regulatory studies and comparative studies are important subcategories that each meet particular application needs. With specialized applications, bioavailability and bioequivalence account for 46.0% of the market. As the industry diversifies across product categories and application needs, ADME is also supporting segment growth.
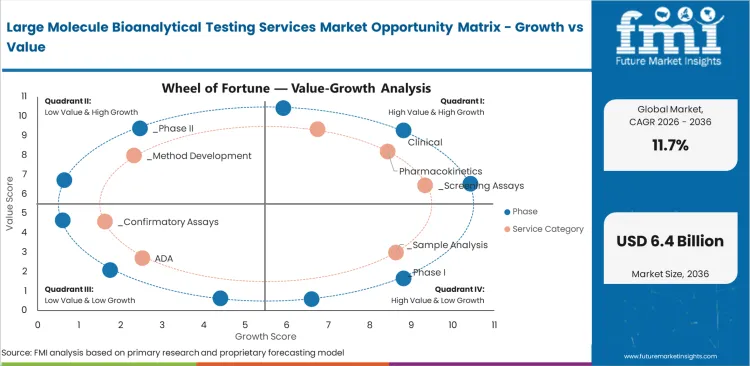
Large Molecule Bioanalytical Testing Services Market Drivers, Restraints, and Opportunities
- Biologic Pipeline Expansion: Monoclonal antibodies, antibody-drug conjugates, bispecific antibodies, and cell treatments account for a growing portion of clinical development activities in the world's biologic drug pipeline.
- For biologic submissions, regulatory bodies need thorough anti-drug antibody screening and characterisation.
- Bioanalytical testing is increasingly being outsourced by pharmaceutical companies to CROs that have made investments in the specialized tools, approved procedures, and regulatory know-how needed for large molecule analysis.
The Large Molecule Bioanalytical Testing Services Market is shaped by biologic pipeline expansion, regulatory immunogenicity testing requirements, outsourcing trends, and biosimilar development activity. Growth reflects the balance between clinical or commercial demand drivers and adoption barriers.
Biologic Pipeline Expansion
The global biologic drug pipeline continues to grow, with monoclonal antibodies, antibody-drug conjugates, bispecific antibodies, and cell therapies representing an increasing share of clinical development activity. Each biologic candidate requires specialized bioanalytical testing, creating volume growth that is tied to pipeline expansion rather than discretionary spending.
Immunogenicity Testing Requirements
Regulatory agencies require comprehensive anti-drug antibody screening and characterization for biologic submissions. ADA testing, neutralizing antibody assays, and immunogenicity risk assessment are mandatory components that create compliance-driven demand for specialized testing laboratories.
Outsourcing and Capacity Constraints
Pharmaceutical companies are increasingly outsourcing bioanalytical testing to CROs that have invested in the specialized equipment, validated methods, and regulatory expertise required for large molecule analysis. In-house capacity constraints and the capital cost of maintaining advanced analytical platforms are accelerating this outsourcing trend.
Biosimilar Development Activity
The growing biosimilar development pipeline requires comparative bioanalytical studies to demonstrate bioequivalence with reference products. This creates additional testing demand that is linked to patent cliff timelines and regulatory pathway developments across jurisdictions.
Analysis of Large Molecule Bioanalytical Testing Services Market By Key Countries
.webp)
| Country | CAGR |
|---|---|
| USA | 12.0% |
| UK | 11.3% |
| Japan | 10.9% |
| EU | 11.5% |
| South Korea | 12.2% |
Source: FMI analysis based on primary research and proprietary forecasting model
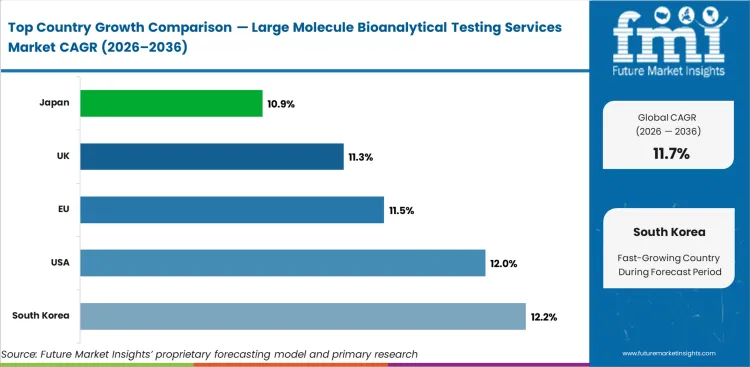
- South Korea at 12.2% is supported by biosimilar manufacturing growth and clinical trial activity.
- USA at 12.0% is supported by the largest biologic drug development pipeline and CRO infrastructure globally.
- EU at 11.5% is supported by harmonized biologic regulatory frameworks and biosimilar development activity.
- UK at 11.3% is supported by biotech cluster activity and regulatory science expertise.
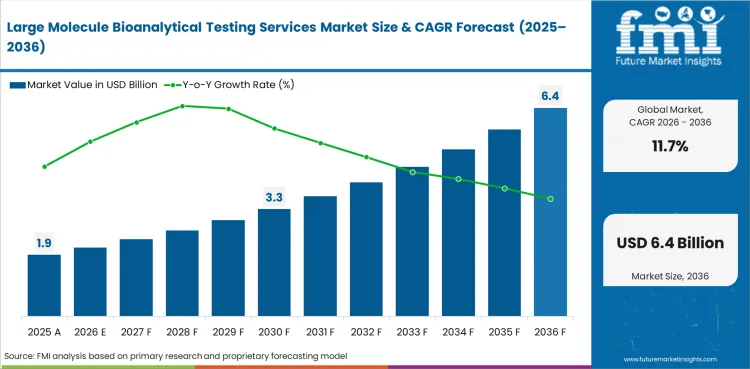
The global Large Molecule Bioanalytical Testing Services Market is projected to expand at a CAGR of 11.7% from 2026 to 2036. The analysis covers more than 30 countries, with distinct growth profiles based on biologic pipeline expansion, regulatory immunogenicity testing requirements, outsourcing trends, and biosimilar development activity.
Demand Outlook for Large Molecule Bioanalytical Testing Services Market in the United States
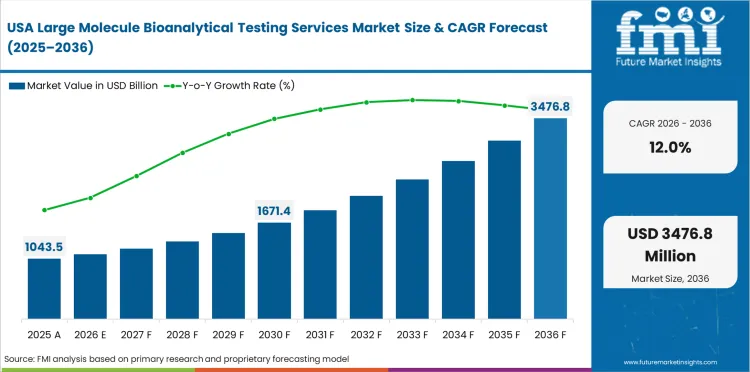
The United States is projected to grow at 12.0% through 2036. The largest biologic drug development pipeline and CRO infrastructure globally are the primary factors shaping demand.
- The largest biologic drug development pipeline and CRO infrastructure globally supports demand growth.
- Expanding demand base contributes to market expansion.
- Regulatory and institutional frameworks are supporting market development.
Future Outlook for Large Molecule Bioanalytical Testing Services Market in the United Kingdom
The United Kingdom is projected to grow at 11.3% through 2036. Biotech cluster activity and regulatory science expertise are the primary factors shaping demand.
- Biotech cluster activity and regulatory science expertise supports demand growth.
- Expanding demand base contributes to market expansion.
- Regulatory and institutional frameworks are supporting market development.
Opportunity Analysis of Large Molecule Bioanalytical Testing Services Market in Japan
Japan is projected to grow at 10.9% through 2036. Advanced pharmaceutical R&D infrastructure and biologic development programs are the primary factors shaping demand.
- Advanced pharmaceutical R&D infrastructure and biologic development programs supports demand growth.
- Expanding demand base contributes to market expansion.
- Regulatory and institutional frameworks are supporting market development.
In-depth Analysis of Large Molecule Bioanalytical Testing Services Market in the European Union
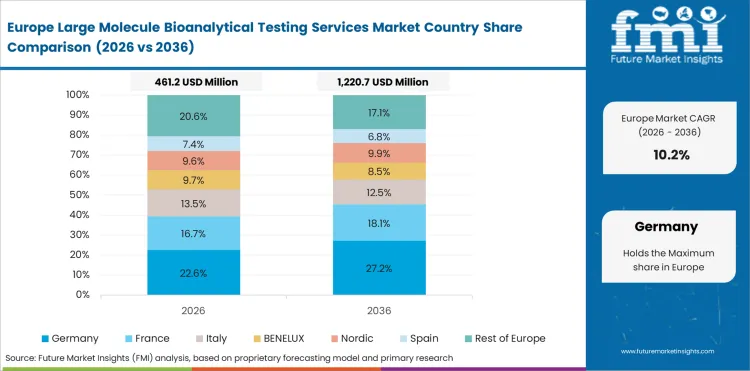
The European Union is projected to grow at 11.5% through 2036. Harmonized biologic regulatory frameworks and biosimilar development activity are the primary factors shaping demand.
- Harmonized biologic regulatory frameworks and biosimilar development activity supports demand growth.
- Expanding demand base contributes to market expansion.
- Regulatory and institutional frameworks are supporting market development.
Sales Analysis of Large Molecule Bioanalytical Testing Services Market in South Korea
South Korea is projected to grow at 12.2% through 2036. Biosimilar manufacturing growth and clinical trial activity are the primary factors shaping demand.
- Biosimilar manufacturing growth and clinical trial activity supports demand growth.
- Expanding demand base contributes to market expansion.
- Regulatory and institutional frameworks are supporting market development.
Competitive Landscape and Strategic Positioning
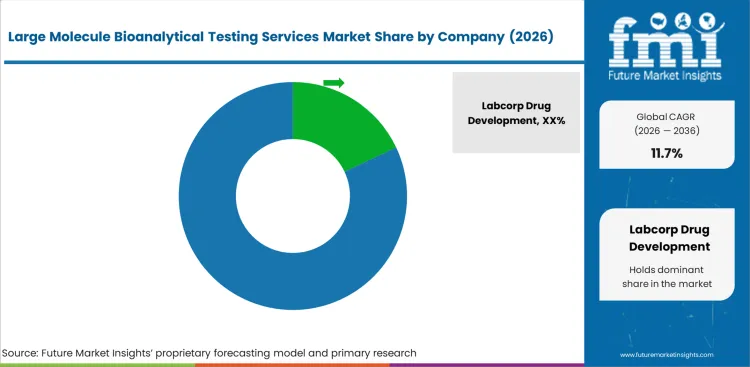
- Labcorp Drug Development holds a leading position based on assay portfolio and regulatory expertise.
- Established players compete on assay portfolio, regulatory expertise, capacity scale, geographic footprint.
- Emerging players are focusing on specialized capabilities and niche market positioning.
Labcorp Drug Development holds a leading market position through its established assay portfolio and regulatory expertise. Charles River Laboratories maintains a strong position through complementary capabilities and capacity scale.
ICON plc, Syneos Health, WuXi AppTec compete on specialized capabilities and geographic footprint. These companies serve distinct segments of the market through targeted product and service strategies.
Market entry requires investment in assay portfolio, regulatory compliance, and distribution infrastructure. Established companies benefit from customer relationships and track records that take years to develop.
Key Companies in the Large Molecule Bioanalytical Testing Services Market
Key global companies leading the large molecule bioanalytical testing services market include:
- Labcorp Drug Development, Charles River Laboratories, ICON plc, Syneos Health are the established market leaders with broad capabilities across assay portfolio and regulatory expertise.
- WuXi AppTec, Eurofins Scientific, PPD (Thermo Fisher Scientific) have built regional positions through specialized capabilities and targeted market strategies.
- Frontage Laboratories, BioAgilytix Labs, Intertek Group plc are entering through niche specialization and technology-driven differentiation.
Competitive Benchmarking: Large Molecule Bioanalytical Testing Services Market
| Company | Assay Portfolio | Regulatory Expertise | Capacity Scale | Geographic Footprint |
|---|---|---|---|---|
| Labcorp Drug Development | High | High | Strong | Global |
| Charles River Laboratories | High | High | Strong | Global |
| ICON plc | High | High | Strong | Global |
| Syneos Health | Medium | High | Moderate | Regional |
| WuXi AppTec | Medium | High | Moderate | Regional |
| Eurofins Scientific | Medium | High | Moderate | Regional |
| PPD (Thermo Fisher Scientific) | Medium | Medium | Low | Regional |
| Frontage Laboratories | Medium | Medium | Low | Regional |
| BioAgilytix Labs | Medium | Medium | Low | Regional |
| Intertek Group plc | Medium | Medium | Low | Regional |
Source: Future Market Insights competitive analysis, 2026.
Key Developments in Large Molecule Bioanalytical Testing Services Market
- In March 2025, Labcorp acquired BioReference Health’s oncology assets and Incyte Diagnostics to expand precision medicine and USA presence.
- In May 2025, Charles River partnered with Singapore General Hospital for cGMP master cell banking and NGS services for CAR-T cells.
Key Players in the Large Molecule Bioanalytical Testing Services Market:
Major Global Players:
- Labcorp Drug Development
- Charles River Laboratories
- ICON plc
- Syneos Health
- WuXi AppTec
- Eurofins Scientific
- PPD (Thermo Fisher Scientific)
Emerging Players/Startups
- Frontage Laboratories
- BioAgilytix Labs
- Intertek Group plc
Report Scope and Coverage
| Parameter | Details |
|---|---|
| Quantitative Units | USD 2.11 billion to USD 6.37 billion, at a CAGR of 11.7% |
| Market Definition | The Large Molecule Bioanalytical Testing Services Market covers outsourced and in-house laboratory testing services for biologic drug development, including pharmacokinetic analysis, anti-drug antibody testing, biomarker analysis, and bioavailability studies across preclinical and clinical development phases. |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia and Pacific, Middle East and Africa |
| Countries Covered | USA, UK, Japan, EU, South Korea, Italy, South Korea, 30 plus countries |
| Key Companies Profiled | Labcorp Drug Development, Charles River Laboratories, ICON plc, Syneos Health, WuXi AppTec, Eurofins Scientific, PPD (Thermo Fisher Scientific), Frontage Laboratories, BioAgilytix Labs, Intertek Group plc |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid bottom-up and top-down methodology starting with verified data, projecting adoption velocity across segments and regions. |
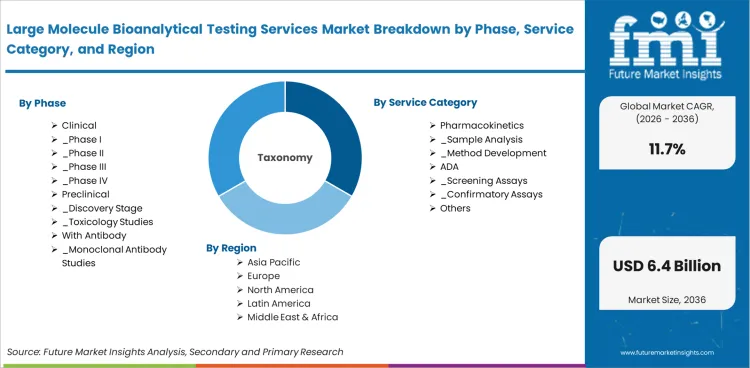
Large Molecule Bioanalytical Testing Services Market Segmentation
Large Molecule Bioanalytical Testing Services Market Market Segmented by Phase:
- Clinical
- Phase I
- Phase II
- Phase III
- Phase IV
- Preclinical
- Discovery Stage
- Toxicology Studies
- With Antibody
- Monoclonal Antibody Studies
- Without Antibody
- Non Antibody Biologics
Large Molecule Bioanalytical Testing Services Market Market Segmented by Service Category:
- Pharmacokinetics
- Sample Analysis
- Method Development
- ADA
- Screening Assays
- Confirmatory Assays
- Others
Large Molecule Bioanalytical Testing Services Market Market Segmented by Test Type:
- Bioavailability and Bioequivalence
- Comparative Studies
- Regulatory Studies
- ADME
- Absorption Studies
- Metabolism Studies
- PK
- Drug Concentration Analysis
- PD
- Biomarker Analysis
- Other Tests
Large Molecule Bioanalytical Testing Services Market Market Segmented by Therapeutic Area:
- Oncology
- Solid Tumors
- Hematological Malignancies
- Infectious Diseases
- Viral Infections
- Bacterial Infections
- Cardiology
- Heart Failure
- Neurology
- Neurodegenerative Disorders
- Others
Large Molecule Bioanalytical Testing Services Market Market Segmented by End User:
- Large Pharmaceutical Companies
- Global Pharma Firms
- Biologics Developers
- Small and Medium Enterprises
- Biotech Startups
- Contract Research Organizations
- Full Service CROs
- Sponsor Organizations
- Academic Sponsors
Large Molecule Bioanalytical Testing Services Market Market Segmented by Distribution Channel:
- Direct Contract Research Services
- Long Term Contracts
- Project Based Contracts
- Third Party Laboratory Networks
- Partner Laboratories
- Online Service Portals
- Digital Service Platforms
Large Molecule Bioanalytical Testing Services Market Market by Region:
- North America
- USA
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Western Europe
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- Eastern Europe
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- East Asia
- China
- Japan
- South Korea
- South Asia and Pacific
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
Research Sources and Bibliography
- USA Food and Drug Administration. (2024). Guidance for Industry: Immunogenicity Testing of Therapeutic Protein Products. FDA.
- European Medicines Agency. (2024). Guideline on Immunogenicity Assessment of Biotechnology-Derived Therapeutic Proteins. EMA.
- International Council for Harmonization. (2025). ICH M10: Bioanalytical Method Validation and Study Sample Analysis. ICH.
- World Health Organization. (2024). WHO Guidelines on Biosimilar Biotherapeutic Products. WHO.
- Organization for Economic Co-operation and Development. (2024). OECD Pharmaceutical R&D Expenditure and Pipeline Analysis. OECD.
- National Institutes of Health. (2024). NIH Clinical Trials Registry: Biologic Drug Development Statistics. NIH.
This bibliography is provided for reader reference. The full Future Market Insights report contains the complete reference list with publication dates, URLs, and supporting data for all cited works.
This Report Answers
- Estimating the size of the market and revenue from 2026 to 2036.
- Segmentation by Phase, Service Category, Test Type, Therapeutic Area, End User, Distribution Channel, and region.
- Insights about more than 30 markets across regions.
- Technology and treatment landscape analysis.
- Assessment of the competitive landscape.
- Finding investment opportunities across segments and regions.
- Supply chain and distribution analysis.
- Delivery of data in PDF and Excel formats.
Frequently Asked Questions
What is the global market demand for Large Molecule Bioanalytical Testing Services in 2026?
In 2026, the global large molecule bioanalytical testing services market is expected to be worth USD 2.11 billion.
How big will the Large Molecule Bioanalytical Testing Services Market be in 2036?
By 2036, the large molecule bioanalytical testing services market is expected to be worth USD 6.37 billion.
How much is demand expected to grow between 2026 and 2036?
Between 2026 and 2036, demand is expected to grow at a CAGR of 11.7%.
Which Phase segment is expected to lead in 2026?
Clinical is expected to account for 61.3% of the Phase segment in 2026, driven by growth in biologic drug development pipelines and regulatory requirements for immunogenicity assessment.
What is driving demand in South Korea?
South Korea is projected to grow at 12.2% through 2036, supported by biosimilar manufacturing growth and clinical trial activity.
What is driving demand in USA?
USA is projected to grow at 12.0% through 2036, supported by the largest biologic drug development pipeline and CRO infrastructure globally.
What does this report mean by 'Large Molecule Bioanalytical Testing Services Market definition'?
The Large Molecule Bioanalytical Testing Services Market covers outsourced and in-house laboratory testing services for biologic drug development, including pharmacokinetic analysis, anti-drug antibody testing, biomarker analysis, and bioavailability studies across preclinical and clinical development phases.
How does FMI forecast and validate the Large Molecule Bioanalytical Testing Services data?
Forecasting models use a hybrid bottom-up and top-down approach, starting with verified data and cross-checking against clinical trial initiation data, CRO financial reports, and pharmaceutical R&D spending surveys.
What is the global market demand for Large Molecule Bioanalytical Testing Services in 2026?
In 2026, the global large molecule bioanalytical testing services market is expected to be worth USD 2.11 billion.
How big will the Large Molecule Bioanalytical Testing Services Market be in 2036?
By 2036, the large molecule bioanalytical testing services market is expected to be worth USD 6.37 billion.
How much is demand expected to grow between 2026 and 2036?
Between 2026 and 2036, demand is expected to grow at a CAGR of 11.7%.
Which Phase segment is expected to lead in 2026?
Clinical is expected to account for 61.3% of the Phase segment in 2026, driven by growth in biologic drug development pipelines and regulatory requirements for immunogenicity assessment.
What is driving demand in South Korea?
South Korea is projected to grow at 12.2% through 2036, supported by biosimilar manufacturing growth and clinical trial activity.
What is driving demand in USA?
USA is projected to grow at 12.0% through 2036, supported by the largest biologic drug development pipeline and CRO infrastructure globally. What does "Large Molecule Bioanalytical Testing Services Market definition" mean in this report? Pharmacokinetic analysis, anti-drug antibody testing, biomarker analysis, and bioavailability studies during the preclinical and clinical stages of biologic drug development are all included in the market for large molecule bioanalytical testing services. How is the data from Large Molecule Bioanalytical Testing Services forecasted and validated by FMI? Pharmaceutical R&D spending surveys, CRO financial reports, clinical trial initiation data, and validated data are the beginning points for forecasting models, which employ a hybrid bottom-up and top-down methodology.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Phase
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Phase , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Phase , 2026 to 2036
- Clinical
- Phase I
- Phase II
- Phase III
- Phase IV
- Preclinical
- Discovery Stage
- Toxicology Studies
- With Antibody
- Monoclonal Antibody Studies
- Without Antibody
- Non Antibody Biologics
- Clinical
- Y to o to Y Growth Trend Analysis By Phase , 2021 to 2025
- Absolute $ Opportunity Analysis By Phase , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Service Category
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Service Category, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Service Category, 2026 to 2036
- Pharmacokinetics
- Sample Analysis
- Method Development
- ADA
- Screening Assays
- Confirmatory Assays
- Others
- Pharmacokinetics
- Y to o to Y Growth Trend Analysis By Service Category, 2021 to 2025
- Absolute $ Opportunity Analysis By Service Category, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Test Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Test Type, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Test Type, 2026 to 2036
- Bioavailability and Bioequivalence
- Comparative Studies
- Regulatory Studies
- ADME
- Absorption Studies
- Metabolism Studies
- PK
- Drug Concentration Analysis
- PD
- Biomarker Analysis
- Other Tests
- Bioavailability and Bioequivalence
- Y to o to Y Growth Trend Analysis By Test Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Test Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Therapeutic Area
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Therapeutic Area, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Therapeutic Area, 2026 to 2036
- Oncology
- Solid Tumors
- Hematological Malignancies
- Infectious Diseases
- Viral Infections
- Bacterial Infections
- Cardiology
- Heart Failure
- Neurology
- Neurodegenerative Disorders
- Others
- Oncology
- Y to o to Y Growth Trend Analysis By Therapeutic Area, 2021 to 2025
- Absolute $ Opportunity Analysis By Therapeutic Area, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User, 2026 to 2036
- Large Pharmaceutical Companies
- Global Pharma Firms
- Biologics Developers
- Small and Medium Enterprises
- Biotech Startups
- Contract Research Organizations
- Full Service CROs
- Sponsor Organizations
- Academic Sponsors
- Large Pharmaceutical Companies
- Y to o to Y Growth Trend Analysis By End User, 2021 to 2025
- Absolute $ Opportunity Analysis By End User, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Distribution Channel
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Distribution Channel, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Distribution Channel, 2026 to 2036
- Direct Contract Research Services
- Long Term Contracts
- Project Based Contracts
- Third Party Laboratory Networks
- Partner Laboratories
- Online Service Portals
- Digital Service Platforms
- Direct Contract Research Services
- Y to o to Y Growth Trend Analysis By Distribution Channel, 2021 to 2025
- Absolute $ Opportunity Analysis By Distribution Channel, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Phase
- By Service Category
- By Test Type
- By Therapeutic Area
- By End User
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Phase
- By Service Category
- By Test Type
- By Therapeutic Area
- By End User
- By Distribution Channel
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Phase
- By Service Category
- By Test Type
- By Therapeutic Area
- By End User
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Phase
- By Service Category
- By Test Type
- By Therapeutic Area
- By End User
- By Distribution Channel
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Phase
- By Service Category
- By Test Type
- By Therapeutic Area
- By End User
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Phase
- By Service Category
- By Test Type
- By Therapeutic Area
- By End User
- By Distribution Channel
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Phase
- By Service Category
- By Test Type
- By Therapeutic Area
- By End User
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Phase
- By Service Category
- By Test Type
- By Therapeutic Area
- By End User
- By Distribution Channel
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Phase
- By Service Category
- By Test Type
- By Therapeutic Area
- By End User
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Phase
- By Service Category
- By Test Type
- By Therapeutic Area
- By End User
- By Distribution Channel
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Phase
- By Service Category
- By Test Type
- By Therapeutic Area
- By End User
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Phase
- By Service Category
- By Test Type
- By Therapeutic Area
- By End User
- By Distribution Channel
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Phase
- By Service Category
- By Test Type
- By Therapeutic Area
- By End User
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Phase
- By Service Category
- By Test Type
- By Therapeutic Area
- By End User
- By Distribution Channel
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Phase
- By Service Category
- By Test Type
- By Therapeutic Area
- By End User
- By Distribution Channel
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Phase
- By Service Category
- By Test Type
- By Therapeutic Area
- By End User
- By Distribution Channel
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Phase
- By Service Category
- By Test Type
- By Therapeutic Area
- By End User
- By Distribution Channel
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Phase
- By Service Category
- By Test Type
- By Therapeutic Area
- By End User
- By Distribution Channel
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Phase
- By Service Category
- By Test Type
- By Therapeutic Area
- By End User
- By Distribution Channel
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Phase
- By Service Category
- By Test Type
- By Therapeutic Area
- By End User
- By Distribution Channel
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Phase
- By Service Category
- By Test Type
- By Therapeutic Area
- By End User
- By Distribution Channel
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Phase
- By Service Category
- By Test Type
- By Therapeutic Area
- By End User
- By Distribution Channel
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Phase
- By Service Category
- By Test Type
- By Therapeutic Area
- By End User
- By Distribution Channel
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Phase
- By Service Category
- By Test Type
- By Therapeutic Area
- By End User
- By Distribution Channel
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Phase
- By Service Category
- By Test Type
- By Therapeutic Area
- By End User
- By Distribution Channel
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Phase
- By Service Category
- By Test Type
- By Therapeutic Area
- By End User
- By Distribution Channel
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Phase
- By Service Category
- By Test Type
- By Therapeutic Area
- By End User
- By Distribution Channel
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Phase
- By Service Category
- By Test Type
- By Therapeutic Area
- By End User
- By Distribution Channel
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Phase
- By Service Category
- By Test Type
- By Therapeutic Area
- By End User
- By Distribution Channel
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Phase
- By Service Category
- By Test Type
- By Therapeutic Area
- By End User
- By Distribution Channel
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Phase
- By Service Category
- By Test Type
- By Therapeutic Area
- By End User
- By Distribution Channel
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Phase
- By Service Category
- By Test Type
- By Therapeutic Area
- By End User
- By Distribution Channel
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Phase
- By Service Category
- By Test Type
- By Therapeutic Area
- By End User
- By Distribution Channel
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Phase
- By Service Category
- By Test Type
- By Therapeutic Area
- By End User
- By Distribution Channel
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Phase
- By Service Category
- By Test Type
- By Therapeutic Area
- By End User
- By Distribution Channel
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Phase
- By Service Category
- By Test Type
- By Therapeutic Area
- By End User
- By Distribution Channel
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Phase
- By Service Category
- By Test Type
- By Therapeutic Area
- By End User
- By Distribution Channel
- Competition Analysis
- Competition Deep Dive
- Labcorp Drug Development
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Charles River Laboratories
- ICON plc
- Syneos Health
- WuXi AppTec
- Eurofins Scientific
- PPD (Thermo Fisher Scientific)
- Frontage Laboratories
- BioAgilytix Labs
- Intertek Group plc
- Labcorp Drug Development
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Phase , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Service Category, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Test Type, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by Therapeutic Area, 2021 to 2036
- Table 6: Global Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 7: Global Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Phase , 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by Service Category, 2021 to 2036
- Table 11: North America Market Value (USD Million) Forecast by Test Type, 2021 to 2036
- Table 12: North America Market Value (USD Million) Forecast by Therapeutic Area, 2021 to 2036
- Table 13: North America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 14: North America Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 16: Latin America Market Value (USD Million) Forecast by Phase , 2021 to 2036
- Table 17: Latin America Market Value (USD Million) Forecast by Service Category, 2021 to 2036
- Table 18: Latin America Market Value (USD Million) Forecast by Test Type, 2021 to 2036
- Table 19: Latin America Market Value (USD Million) Forecast by Therapeutic Area, 2021 to 2036
- Table 20: Latin America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 21: Latin America Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 22: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Western Europe Market Value (USD Million) Forecast by Phase , 2021 to 2036
- Table 24: Western Europe Market Value (USD Million) Forecast by Service Category, 2021 to 2036
- Table 25: Western Europe Market Value (USD Million) Forecast by Test Type, 2021 to 2036
- Table 26: Western Europe Market Value (USD Million) Forecast by Therapeutic Area, 2021 to 2036
- Table 27: Western Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 28: Western Europe Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 29: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 30: Eastern Europe Market Value (USD Million) Forecast by Phase , 2021 to 2036
- Table 31: Eastern Europe Market Value (USD Million) Forecast by Service Category, 2021 to 2036
- Table 32: Eastern Europe Market Value (USD Million) Forecast by Test Type, 2021 to 2036
- Table 33: Eastern Europe Market Value (USD Million) Forecast by Therapeutic Area, 2021 to 2036
- Table 34: Eastern Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 35: Eastern Europe Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 36: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 37: East Asia Market Value (USD Million) Forecast by Phase , 2021 to 2036
- Table 38: East Asia Market Value (USD Million) Forecast by Service Category, 2021 to 2036
- Table 39: East Asia Market Value (USD Million) Forecast by Test Type, 2021 to 2036
- Table 40: East Asia Market Value (USD Million) Forecast by Therapeutic Area, 2021 to 2036
- Table 41: East Asia Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 42: East Asia Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 43: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 44: South Asia and Pacific Market Value (USD Million) Forecast by Phase , 2021 to 2036
- Table 45: South Asia and Pacific Market Value (USD Million) Forecast by Service Category, 2021 to 2036
- Table 46: South Asia and Pacific Market Value (USD Million) Forecast by Test Type, 2021 to 2036
- Table 47: South Asia and Pacific Market Value (USD Million) Forecast by Therapeutic Area, 2021 to 2036
- Table 48: South Asia and Pacific Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 49: South Asia and Pacific Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 50: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 51: Middle East & Africa Market Value (USD Million) Forecast by Phase , 2021 to 2036
- Table 52: Middle East & Africa Market Value (USD Million) Forecast by Service Category, 2021 to 2036
- Table 53: Middle East & Africa Market Value (USD Million) Forecast by Test Type, 2021 to 2036
- Table 54: Middle East & Africa Market Value (USD Million) Forecast by Therapeutic Area, 2021 to 2036
- Table 55: Middle East & Africa Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 56: Middle East & Africa Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Phase , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Phase , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Phase
- Figure 6: Global Market Value Share and BPS Analysis by Service Category, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Service Category, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Service Category
- Figure 9: Global Market Value Share and BPS Analysis by Test Type, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Test Type, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Test Type
- Figure 12: Global Market Value Share and BPS Analysis by Therapeutic Area, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Therapeutic Area, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Therapeutic Area
- Figure 15: Global Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 17: Global Market Attractiveness Analysis by End User
- Figure 18: Global Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 19: Global Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 20: Global Market Attractiveness Analysis by Distribution Channel
- Figure 21: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 22: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 23: Global Market Attractiveness Analysis by Region
- Figure 24: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 25: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 26: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 27: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 28: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 29: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 30: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 31: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 32: North America Market Value Share and BPS Analysis by Phase , 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Phase , 2026-2036
- Figure 34: North America Market Attractiveness Analysis by Phase
- Figure 35: North America Market Value Share and BPS Analysis by Service Category, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by Service Category, 2026-2036
- Figure 37: North America Market Attractiveness Analysis by Service Category
- Figure 38: North America Market Value Share and BPS Analysis by Test Type, 2026 and 2036
- Figure 39: North America Market Y-o-Y Growth Comparison by Test Type, 2026-2036
- Figure 40: North America Market Attractiveness Analysis by Test Type
- Figure 41: North America Market Value Share and BPS Analysis by Therapeutic Area, 2026 and 2036
- Figure 42: North America Market Y-o-Y Growth Comparison by Therapeutic Area, 2026-2036
- Figure 43: North America Market Attractiveness Analysis by Therapeutic Area
- Figure 44: North America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 45: North America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 46: North America Market Attractiveness Analysis by End User
- Figure 47: North America Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 48: North America Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 49: North America Market Attractiveness Analysis by Distribution Channel
- Figure 50: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 51: Latin America Market Value Share and BPS Analysis by Phase , 2026 and 2036
- Figure 52: Latin America Market Y-o-Y Growth Comparison by Phase , 2026-2036
- Figure 53: Latin America Market Attractiveness Analysis by Phase
- Figure 54: Latin America Market Value Share and BPS Analysis by Service Category, 2026 and 2036
- Figure 55: Latin America Market Y-o-Y Growth Comparison by Service Category, 2026-2036
- Figure 56: Latin America Market Attractiveness Analysis by Service Category
- Figure 57: Latin America Market Value Share and BPS Analysis by Test Type, 2026 and 2036
- Figure 58: Latin America Market Y-o-Y Growth Comparison by Test Type, 2026-2036
- Figure 59: Latin America Market Attractiveness Analysis by Test Type
- Figure 60: Latin America Market Value Share and BPS Analysis by Therapeutic Area, 2026 and 2036
- Figure 61: Latin America Market Y-o-Y Growth Comparison by Therapeutic Area, 2026-2036
- Figure 62: Latin America Market Attractiveness Analysis by Therapeutic Area
- Figure 63: Latin America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 64: Latin America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 65: Latin America Market Attractiveness Analysis by End User
- Figure 66: Latin America Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 67: Latin America Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 68: Latin America Market Attractiveness Analysis by Distribution Channel
- Figure 69: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 70: Western Europe Market Value Share and BPS Analysis by Phase , 2026 and 2036
- Figure 71: Western Europe Market Y-o-Y Growth Comparison by Phase , 2026-2036
- Figure 72: Western Europe Market Attractiveness Analysis by Phase
- Figure 73: Western Europe Market Value Share and BPS Analysis by Service Category, 2026 and 2036
- Figure 74: Western Europe Market Y-o-Y Growth Comparison by Service Category, 2026-2036
- Figure 75: Western Europe Market Attractiveness Analysis by Service Category
- Figure 76: Western Europe Market Value Share and BPS Analysis by Test Type, 2026 and 2036
- Figure 77: Western Europe Market Y-o-Y Growth Comparison by Test Type, 2026-2036
- Figure 78: Western Europe Market Attractiveness Analysis by Test Type
- Figure 79: Western Europe Market Value Share and BPS Analysis by Therapeutic Area, 2026 and 2036
- Figure 80: Western Europe Market Y-o-Y Growth Comparison by Therapeutic Area, 2026-2036
- Figure 81: Western Europe Market Attractiveness Analysis by Therapeutic Area
- Figure 82: Western Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 83: Western Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 84: Western Europe Market Attractiveness Analysis by End User
- Figure 85: Western Europe Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 86: Western Europe Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 87: Western Europe Market Attractiveness Analysis by Distribution Channel
- Figure 88: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 89: Eastern Europe Market Value Share and BPS Analysis by Phase , 2026 and 2036
- Figure 90: Eastern Europe Market Y-o-Y Growth Comparison by Phase , 2026-2036
- Figure 91: Eastern Europe Market Attractiveness Analysis by Phase
- Figure 92: Eastern Europe Market Value Share and BPS Analysis by Service Category, 2026 and 2036
- Figure 93: Eastern Europe Market Y-o-Y Growth Comparison by Service Category, 2026-2036
- Figure 94: Eastern Europe Market Attractiveness Analysis by Service Category
- Figure 95: Eastern Europe Market Value Share and BPS Analysis by Test Type, 2026 and 2036
- Figure 96: Eastern Europe Market Y-o-Y Growth Comparison by Test Type, 2026-2036
- Figure 97: Eastern Europe Market Attractiveness Analysis by Test Type
- Figure 98: Eastern Europe Market Value Share and BPS Analysis by Therapeutic Area, 2026 and 2036
- Figure 99: Eastern Europe Market Y-o-Y Growth Comparison by Therapeutic Area, 2026-2036
- Figure 100: Eastern Europe Market Attractiveness Analysis by Therapeutic Area
- Figure 101: Eastern Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 102: Eastern Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 103: Eastern Europe Market Attractiveness Analysis by End User
- Figure 104: Eastern Europe Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 105: Eastern Europe Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 106: Eastern Europe Market Attractiveness Analysis by Distribution Channel
- Figure 107: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 108: East Asia Market Value Share and BPS Analysis by Phase , 2026 and 2036
- Figure 109: East Asia Market Y-o-Y Growth Comparison by Phase , 2026-2036
- Figure 110: East Asia Market Attractiveness Analysis by Phase
- Figure 111: East Asia Market Value Share and BPS Analysis by Service Category, 2026 and 2036
- Figure 112: East Asia Market Y-o-Y Growth Comparison by Service Category, 2026-2036
- Figure 113: East Asia Market Attractiveness Analysis by Service Category
- Figure 114: East Asia Market Value Share and BPS Analysis by Test Type, 2026 and 2036
- Figure 115: East Asia Market Y-o-Y Growth Comparison by Test Type, 2026-2036
- Figure 116: East Asia Market Attractiveness Analysis by Test Type
- Figure 117: East Asia Market Value Share and BPS Analysis by Therapeutic Area, 2026 and 2036
- Figure 118: East Asia Market Y-o-Y Growth Comparison by Therapeutic Area, 2026-2036
- Figure 119: East Asia Market Attractiveness Analysis by Therapeutic Area
- Figure 120: East Asia Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 121: East Asia Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 122: East Asia Market Attractiveness Analysis by End User
- Figure 123: East Asia Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 124: East Asia Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 125: East Asia Market Attractiveness Analysis by Distribution Channel
- Figure 126: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 127: South Asia and Pacific Market Value Share and BPS Analysis by Phase , 2026 and 2036
- Figure 128: South Asia and Pacific Market Y-o-Y Growth Comparison by Phase , 2026-2036
- Figure 129: South Asia and Pacific Market Attractiveness Analysis by Phase
- Figure 130: South Asia and Pacific Market Value Share and BPS Analysis by Service Category, 2026 and 2036
- Figure 131: South Asia and Pacific Market Y-o-Y Growth Comparison by Service Category, 2026-2036
- Figure 132: South Asia and Pacific Market Attractiveness Analysis by Service Category
- Figure 133: South Asia and Pacific Market Value Share and BPS Analysis by Test Type, 2026 and 2036
- Figure 134: South Asia and Pacific Market Y-o-Y Growth Comparison by Test Type, 2026-2036
- Figure 135: South Asia and Pacific Market Attractiveness Analysis by Test Type
- Figure 136: South Asia and Pacific Market Value Share and BPS Analysis by Therapeutic Area, 2026 and 2036
- Figure 137: South Asia and Pacific Market Y-o-Y Growth Comparison by Therapeutic Area, 2026-2036
- Figure 138: South Asia and Pacific Market Attractiveness Analysis by Therapeutic Area
- Figure 139: South Asia and Pacific Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 140: South Asia and Pacific Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 141: South Asia and Pacific Market Attractiveness Analysis by End User
- Figure 142: South Asia and Pacific Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 143: South Asia and Pacific Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 144: South Asia and Pacific Market Attractiveness Analysis by Distribution Channel
- Figure 145: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 146: Middle East & Africa Market Value Share and BPS Analysis by Phase , 2026 and 2036
- Figure 147: Middle East & Africa Market Y-o-Y Growth Comparison by Phase , 2026-2036
- Figure 148: Middle East & Africa Market Attractiveness Analysis by Phase
- Figure 149: Middle East & Africa Market Value Share and BPS Analysis by Service Category, 2026 and 2036
- Figure 150: Middle East & Africa Market Y-o-Y Growth Comparison by Service Category, 2026-2036
- Figure 151: Middle East & Africa Market Attractiveness Analysis by Service Category
- Figure 152: Middle East & Africa Market Value Share and BPS Analysis by Test Type, 2026 and 2036
- Figure 153: Middle East & Africa Market Y-o-Y Growth Comparison by Test Type, 2026-2036
- Figure 154: Middle East & Africa Market Attractiveness Analysis by Test Type
- Figure 155: Middle East & Africa Market Value Share and BPS Analysis by Therapeutic Area, 2026 and 2036
- Figure 156: Middle East & Africa Market Y-o-Y Growth Comparison by Therapeutic Area, 2026-2036
- Figure 157: Middle East & Africa Market Attractiveness Analysis by Therapeutic Area
- Figure 158: Middle East & Africa Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 159: Middle East & Africa Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 160: Middle East & Africa Market Attractiveness Analysis by End User
- Figure 161: Middle East & Africa Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 162: Middle East & Africa Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 163: Middle East & Africa Market Attractiveness Analysis by Distribution Channel
- Figure 164: Global Market - Tier Structure Analysis
- Figure 165: Global Market - Company Share Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

