Small Molecule Innovator CDMO Market : Global Industry Analysis 2015 - 2024 and Opportunity Assessment 2025 - 2035
Small Molecule Innovator CDMO Market Size and Share Forecast Outlook 2025 to 2035
Historical Data Covered: 2015 - 2024 | Base Year: 2024 | Estimated Year: 2025 | Forecast Period: 2025 - 2035
Small Molecule Innovator CDMO Market Size and Share Forecast Outlook 2025 to 2035
The Small Molecule Innovator CDMO Market is estimated to be valued at USD 54.4 billion in 2025 and is projected to reach USD 101.2 billion by 2035, registering a compound annual growth rate (CAGR) of 6.4% over the forecast period.
Quick Stats for Small Molecule Innovator CDMO Market
- Small Molecule Innovator CDMO Market Industry Value (2025): USD 54.4 billion
- Small Molecule Innovator CDMO Market Forecast Value (2035): USD 101.2 billion
- Small Molecule Innovator CDMO Market Forecast CAGR: 6.4%
- Leading Segment in Small Molecule Innovator CDMO Market in 2025: Small Molecule API (57.3%)
- Key Growth Region in Small Molecule Innovator CDMO Market: North America, Asia-Pacific, Europe
- Top Key Players in Small Molecule Innovator CDMO Market: Piramal Pharma Solutions, CordenPharma International, Wuxi AppTec, Cambrex Corporation, Recipharm AB, Thermo Fisher Scientific, Lonza, Catalent Inc., Siegfried Holding AG, Boehringer Ingelheim
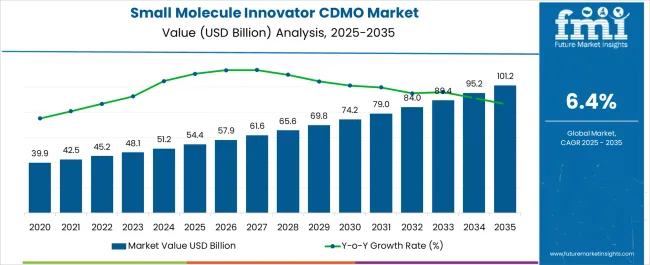
| Metric | Value |
|---|---|
| Small Molecule Innovator CDMO Market Estimated Value in (2025 E) | USD 54.4 billion |
| Small Molecule Innovator CDMO Market Forecast Value in (2035 F) | USD 101.2 billion |
| Forecast CAGR (2025 to 2035) | 6.4% |
Rationale for Segmental Growth in the Small Molecule Innovator CDMO Market
The small molecule innovator CDMO market is expanding steadily as pharmaceutical and biotechnology companies increasingly rely on outsourcing to accelerate drug development and reduce operational costs. Rising complexity of active pharmaceutical ingredient synthesis, coupled with the growing demand for specialized expertise in formulation and regulatory compliance, has heightened the role of CDMOs in the value chain.
Strategic partnerships and long term contracts between drug developers and CDMOs are becoming more common, driven by the need for scalability and global supply reliability. Technological advancements in continuous manufacturing, green chemistry, and analytical development are further supporting growth in this market.
The outlook remains positive as innovators continue to focus on accelerating time to market, optimizing cost structures, and leveraging CDMO capabilities for both niche therapies and high volume production requirements.
Segmental Analysis
Insights into the Small Molecule API Product Segment
The small molecule API product segment is projected to contribute 57.30% of total revenue by 2025 within the product category, making it the leading segment. Growth in this segment is being supported by rising demand for outsourced API development, the complexity of chemical synthesis, and the need for scalable production facilities.
CDMOs have been increasingly relied upon for cost efficiency, quality assurance, and regulatory compliance across multiple geographies.
Their ability to handle large scale production while meeting stringent safety standards has reinforced the dominance of this segment in the overall market.
Insights into the Preclinical Stage Type Segment
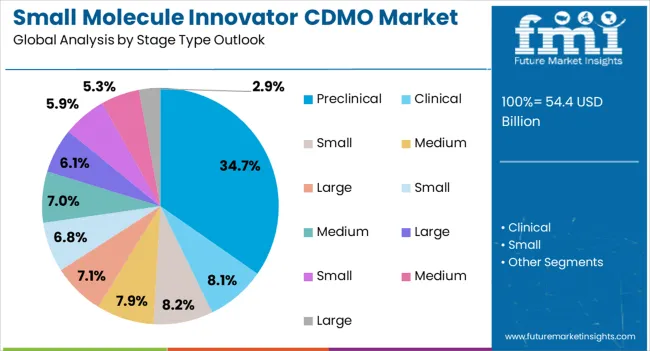
The preclinical stage type segment is expected to hold 34.70% of total revenue by 2025, establishing it as the most significant stage category. This dominance is being driven by growing investments in early drug discovery, rising demand for preclinical safety and efficacy data, and increased outsourcing of non core R&D functions by pharmaceutical companies.
CDMOs offering integrated services including preformulation, analytical method development, and toxicology support have gained preference.
The emphasis on faster progression from laboratory to clinical testing has further reinforced reliance on CDMOs at this stage.
Insights into the Pharmaceutical Customer Type Segment
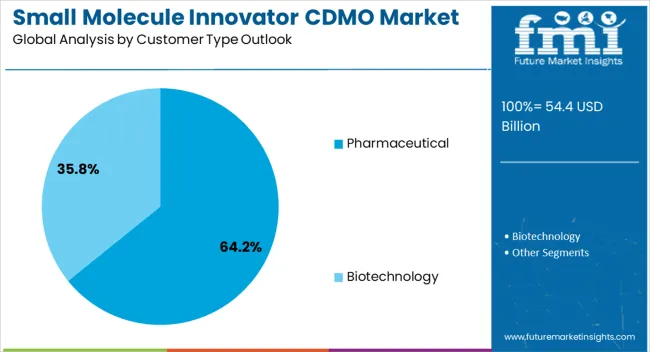
The pharmaceutical customer type segment is projected to account for 64.20% of the overall market revenue by 2025, positioning it as the leading customer category. This leadership is attributed to the increasing outsourcing strategies adopted by pharmaceutical companies seeking to optimize resources, expand global reach, and enhance operational efficiency.
CDMOs have been trusted partners for handling complex synthesis, ensuring regulatory compliance, and providing flexible capacity for both large volume and specialty drugs.
The strong presence of established pharmaceutical players in outsourcing partnerships has consolidated this segment’s dominance within the customer type category.
2020 to 2024 Small Molecule Innovator CDMO Market Outlook Compared to Forecast from 2025 to 2035
The Small Molecule Innovator CDMO market was valued at USD 54.4 billion in 2025, with a substantial growth trajectory of 6.1%% between the analysis period of 2020 to 2025.
The small molecule innovator CDMO market is a growing segment of the pharmaceutical industry that provides contract services for developing and manufacturing small molecule drugs. The market is driven by increasing demand for small-molecule drugs, rising R&D costs, and the need for specialized expertise in drug development and manufacturing.
Future Market Insights expects the small molecule innovator CDMO market to grow at a CAGR of 6.4% from 2025 to 2035. The growth can be attributed to the following reasons
The market is driven by increasing demand for small-molecule drugs, which effectively treat various diseases and disorders. The trend of outsourcing drug development and manufacturing is also driving the market as companies look to reduce costs and gain access to specialized expertise. Additionally, the need for innovative and efficient drug development and manufacturing processes is driving the market.
Overall, the small molecule innovator CDMO market is poised for continued growth, driven by increasing demand for small molecule drugs and the outsourcing of drug development and manufacturing. Companies in this space have to need to stay abreast of new technologies and regulatory requirements to remain competitive in the market.
Considering the factors mentioned above, FMI opines the small molecule innovator CDMO market is more likely to witness a market value of USD 101.2 billion by the end of 2035.
Strategic Overview of the Small Molecule Innovator CDMO Industry
- Growing Demand for Small Molecule Drugs: The demand for small molecule drugs is on the rise due to their effectiveness in treating a wide range of diseases and disorders. Small molecule drugs are also easier to manufacture and have a lower cost of goods, making them attractive to pharmaceutical companies. This has LED to an increased demand for small molecule innovator CDMO services.
- Increasing Outsourcing of Drug Development and Manufacturing: The trend of outsourcing drug development and manufacturing has been gaining momentum in recent years due to rising R&D costs and the need for specialized expertise. Small molecule innovator CDMOs are well-positioned to provide these services, as they have the necessary infrastructure, technology, and expertise to support the development and manufacturing of small molecule drugs.
- Focus on Quality and Compliance: The small molecule innovator CDMO market is highly regulated, and companies in this space must comply with strict quality and safety standards. As a result, there is a strong focus on quality and compliance, with companies investing in technology and processes to ensure that they meet regulatory requirements.
- Consolidation in the Market: The small molecule innovator CDMO market is highly fragmented, with a large number of players offering similar services. However, there has been a trend of consolidation in the market, with larger companies acquiring smaller ones to expand their capabilities and geographic reach.
- Emergence of New Technologies: The small molecule innovator CDMO market is constantly evolving, with new technologies and processes emerging to improve drug development and manufacturing. For example, advances in synthetic biology and biocatalysis are enabling the production of complex small molecules more efficiently and sustainably.
Small Molecule Innovator CDMO Industry Analysis by Category
In 2025, the clinical stage segment is expected to have a prominent share of 55.1%.
The market is divided into three stages: preclinical, clinical, and commercial. A large number of small-molecule drugs are in the clinical stage. Aside from that, drug research and development spending is constantly increasing. For example, according to World Preview, total research and development spending on drugs accounted for USD 54.4.0 billion in 2025, and total R&D spending on drugs is expected to reach USD 69.8.0 billion by 2029. The increase in R&D spending is expected to boost clinical segment growth.
The biotechnology sector is expected to grow at a 4.8% annual rate during the forecast period.
The rapid advancement of biopharmaceutical research and technology creates new opportunities for developing innovative and creative small-molecule drugs. Advances in prediction, structure-based design, and imaging, as well as automation, artificial intelligence, and machine learning, have all become major enablers for small molecule-led optimization to speed up and improve success rates in recent years.
The oncology segment held a prominent share of 41.8% in 2025 and is expected to record a profitable CAGR of more than 7.0% during the forecast period.
The market is divided into therapeutic areas such as cardiovascular diseases, oncology, respiratory disorders, neurology, metabolic disorders, infectious diseases, and others. Other factors driving segment growth include an increase in cancer cases worldwide, significant government reimbursement policies, and an increase in funding for the development of small-molecule oncology therapies.
Top Regions Supporting the Growth of the Small Molecule Innovator CDMO Market
North America is expected to record a CAGR of 5.8% through 2035
North America is a significant contributor to the growth of the Contract Development & Manufacturing Organization (CDMO) industry for small molecule API innovators.
North America is one of the significant markets for small molecule innovator CDMOs, accounting for a significant share of the global market. The region is home to many large pharmaceutical companies and has a well-established healthcare infrastructure, which drives demand for small-molecule drugs. Additionally, the trend of outsourcing drug development and manufacturing is also driving the market in the region.
European region has a well-developed healthcare system, and the authority is supportive of the pharmaceutical industry's development
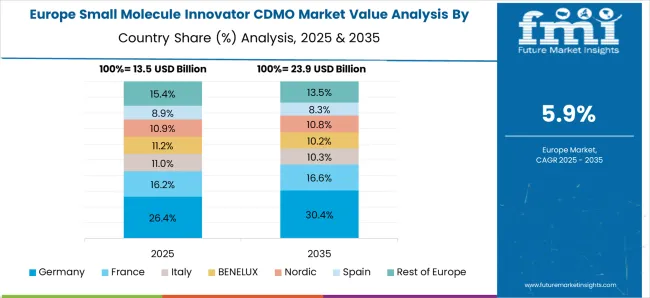
Europe is another potential market for small molecule innovator CDMOs, with a well-established pharmaceutical industry and a strong focus on research and development. The region is also home to many contract manufacturing organizations (CMOs) that provide small molecule innovator CDMO services.
In 2025, Asia Pacific had the Prominent Revenue Share of 42.1%.
Technological advancements, low service costs, and the availability of skilled labor are expected to drive regional market growth.
Asia Pacific region is expected to be the highly progressive market for small molecule innovator CDMOs, driven by increasing demand for small molecule drugs and a growing pharmaceutical industry. The region is home to many contract manufacturing organizations, particularly in countries such as India and China, which offer cost-effective services.
Given the presence of a large number of pharmaceutical and biotechnology companies, the Latin American small molecule innovator CDMO market is expected to grow at a moderate rate.
Latin America is a growing market for small molecule innovator CDMOs, driven by increasing demand for affordable healthcare and rising investment in the pharmaceutical industry. Brazil and Mexico are the potential markets in the region.
Middle East and Africa
The Middle East and Africa region is a relatively small market for small molecule innovator CDMOs. However, the region is experiencing significant growth in the pharmaceutical industry, driven by rising demand for affordable healthcare and increasing investment in healthcare infrastructure.
Leading Companies in the Small Molecule Innovator CDMO Market
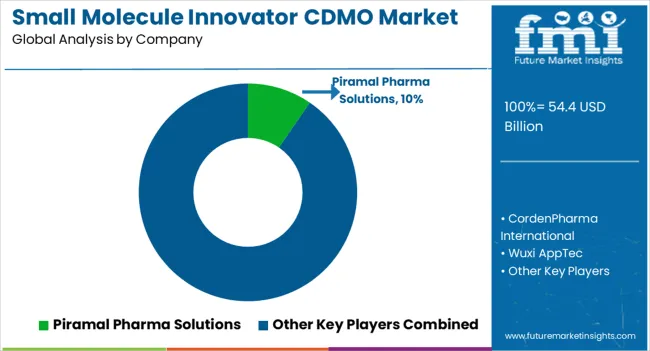
The small molecule innovator CDMO market is highly competitive, with several key players dominating the market. The market is characterized by a few large players and many smaller players, with some companies offering a full suite of services while others focus on a specific aspect of drug development or manufacturing. Here is an overview of the competitive landscape in the market:
Profiles of Key Players in the Small Molecule Innovator CDMO Industry
- Lonza Group: Lonza Group is a leading player in the Small Molecule Innovator CDMO Market, offering a wide range of services, including drug development, process development, and commercial manufacturing. The company operates in North America, Europe, and Asia-Pacific, and serves both pharmaceutical and biotech companies.
- Catalent, Inc.: Catalent, Inc. is another significant player in the market, offering services in drug development, process development, analytical testing, and commercial manufacturing. The company operates in North America, Europe, and Asia-Pacific, and serves both branded and generic small molecule drugs.
- Thermo Fisher Scientific, Inc.: Thermo Fisher Scientific, Inc. is a global leader in the life sciences industry and offers a wide range of services for small molecule drug development and manufacturing. The company operates in North America, Europe, and Asia-Pacific, and serves both pharmaceutical and biotech companies.
- Patheon N.V.: Patheon N.V. is a leading contract development and manufacturing organization, offering services in drug development, process development, and commercial manufacturing. The company operates in North America, Europe, and Asia-Pacific, and serves both branded and generic small molecule drugs.
- Cambrex Corporation: Cambrex Corporation is a global leader in the small molecule innovator CDMO market, offering services in drug development, process development, and commercial manufacturing. The company operates in North America, Europe, and Asia-Pacific, and serves both pharmaceutical and biotech companies.
Other key players in the market include WuXi AppTec Co., Ltd., Piramal Pharma Solutions, Jubilant Life Sciences Ltd., Recipharm AB, and Sterling Pharma Solutions.
Overall, the small molecule innovator CDMO market is highly competitive, with several large players dominating the market. These companies offer a wide range of services and operate globally, serving both pharmaceutical and biotech companies. The market is also characterized by many smaller players, particularly in the Asia-Pacific region, which offer cost-effective services. Companies in the space will need to navigate intense competition, changing customer demands, and evolving regulatory requirements to remain competitive.
Recent Developments
- Catalent, a global provider and distributor of superior medicines, announced the completion of a USD 2.2 million expansion to its clinical supply facility in Singapore, resulting in a 31,000 square-foot increase in the site's footprint, allowing for the installation of 35 additional new ultra-low temperature (ULT) freezers. The facility is now better equipped to handle biopharmaceuticals and advanced modalities such as mRNA-based vaccines, cell and gene therapies, and larger packaging campaigns as a result of the investment.
- December 2025: Thermo Fisher Scientific, a global leader in scientific services, recently opened a new facility in Hangzhou, China, as part of a global initiative to help businesses deliver treatments to patients more quickly.
Scope of the Small Molecule Innovator CDMO Report
| Attribute | Details |
|---|---|
| Growth Rate | CAGR of 6.4% from 2025 to 2035 |
| Base Year of Estimation | 2025 |
| Historical Data | 2020 to 2025 |
| Forecast Period | 2025 to 2035 |
| Quantitative Units | Revenue in USD Billion and Volume in Units and F-CAGR from 2025 to 2035 |
| Report Coverage | Revenue Forecast, Volume Forecast, Company Ranking, Competitive Landscape, growth factors, Trends and Pricing Analysis |
| Key Segments Covered | By Therapeutic Area, By Product, By Stage Type, By Customer Type, By Region |
| Regions Covered | North America; Latin America; Europe; East Asia; South Asia; Middle East & Africa; Oceania |
| Key Countries Profiled | USA, Canada, Brazil, Mexico, Germany, Italy, France, United kingdom, Spain, Russia, China, Japan, India, GCC Countries, Australia |
| Key Companies Profiled | Piramal Pharma Solutions; CordenPharma International; Wuxi AppTec; Cambrex Corporation; Recipharm AB; Pantheon (Thermo Fisher Scientific); Lonza; Catalent Inc.; Siegfried Holding AG; Boehringer Ingelheim; Labcorp Drug Development |
| Customization & Pricing | Available upon Request |
Top Investment Segments in the Small Molecule Innovator CDMO Market
Product Outlook:
- Small Molecule API
- Small Molecule Drug Product
- Oral solid dose
- Semi-Solid Dose
- Liquid Dose
- Others
Stage Type Outlook:
- Preclinical
- Clinical
- Phase I
- Small
- Medium
- Large
- Phase II
- Small
- Medium
- Large
- Phase III
- Small
- Medium
- Large
- Commercial
Customer Type Outlook:
- Pharmaceutical
- Small
- Medium
- Large
- Biotechnology
Therapeutic Area Outlook:
- Cardiovascular disease
- Oncology
- Respiratory disorders
- Neurology
- Metabolic disorders
- Infectious disease
- Others
By Region:
- North America
- Latin America
- Western Europe
- Eastern Europe
- Asia Pacific excluding Japan (APEJ)
- Japan
- Middle East & Africa (MEA)
Frequently Asked Questions
How big is the small molecule innovator CDMO market in 2025?
The global small molecule innovator CDMO market is estimated to be valued at USD 54.4 billion in 2025.
What will be the size of small molecule innovator CDMO market in 2035?
The market size for the small molecule innovator CDMO market is projected to reach USD 101.2 billion by 2035.
How much will be the small molecule innovator CDMO market growth between 2025 and 2035?
The small molecule innovator CDMO market is expected to grow at a 6.4% CAGR between 2025 and 2035.
What are the key product types in the small molecule innovator CDMO market?
The key product types in small molecule innovator CDMO market are small molecule api, _small molecule drug product, _oral solid dose, _semi-solid dose, _liquid dose and _others.
Which stage type outlook segment to contribute significant share in the small molecule innovator CDMO market in 2025?
In terms of stage type outlook, preclinical segment to command 34.7% share in the small molecule innovator CDMO market in 2025.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand-side Trends
- Supply-side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter's Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2020 to 2024 and Forecast, 2025 to 2035
- Historical Market Size Value (USD Million) Analysis, 2020 to 2024
- Current and Future Market Size Value (USD Million) Projections, 2025 to 2035
- Y-o-Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2020 to 2024 and Forecast 2025 to 2035
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Product Outlook
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product Outlook , 2020 to 2024
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product Outlook , 2025 to 2035
- Small Molecule API
- Small Molecule Drug Product
- Oral solid dose
- Semi-Solid Dose
- Liquid Dose
- Others
- Small Molecule API
- Y-o-Y Growth Trend Analysis By Product Outlook , 2020 to 2024
- Absolute $ Opportunity Analysis By Product Outlook , 2025 to 2035
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Stage Type Outlook
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Stage Type Outlook, 2020 to 2024
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Stage Type Outlook, 2025 to 2035
- Preclinical
- Clinical
- Phase I
- Small
- Medium
- Large
- Phase II
- Small
- Medium
- Large
- Phase III
- Small
- Medium
- Large
- Y-o-Y Growth Trend Analysis By Stage Type Outlook, 2020 to 2024
- Absolute $ Opportunity Analysis By Stage Type Outlook, 2025 to 2035
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Customer Type Outlook
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Customer Type Outlook, 2020 to 2024
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Customer Type Outlook, 2025 to 2035
- Pharmaceutical
- Small
- Medium
- Large
- Biotechnology
- Pharmaceutical
- Y-o-Y Growth Trend Analysis By Customer Type Outlook, 2020 to 2024
- Absolute $ Opportunity Analysis By Customer Type Outlook, 2025 to 2035
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Therapeutic Area Outlook
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Therapeutic Area Outlook, 2020 to 2024
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Therapeutic Area Outlook, 2025 to 2035
- Cardiovascular disease
- Oncology
- Respiratory disorders
- Neurology
- Metabolic disorders
- Infectious disease
- Others
- Y-o-Y Growth Trend Analysis By Therapeutic Area Outlook, 2020 to 2024
- Absolute $ Opportunity Analysis By Therapeutic Area Outlook, 2025 to 2035
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2020 to 2024
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2025 to 2035
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- USA
- Canada
- Mexico
- By Product Outlook
- By Stage Type Outlook
- By Customer Type Outlook
- By Therapeutic Area Outlook
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Outlook
- By Stage Type Outlook
- By Customer Type Outlook
- By Therapeutic Area Outlook
- Key Takeaways
- Latin America Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product Outlook
- By Stage Type Outlook
- By Customer Type Outlook
- By Therapeutic Area Outlook
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Outlook
- By Stage Type Outlook
- By Customer Type Outlook
- By Therapeutic Area Outlook
- Key Takeaways
- Western Europe Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product Outlook
- By Stage Type Outlook
- By Customer Type Outlook
- By Therapeutic Area Outlook
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Outlook
- By Stage Type Outlook
- By Customer Type Outlook
- By Therapeutic Area Outlook
- Key Takeaways
- Eastern Europe Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product Outlook
- By Stage Type Outlook
- By Customer Type Outlook
- By Therapeutic Area Outlook
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Outlook
- By Stage Type Outlook
- By Customer Type Outlook
- By Therapeutic Area Outlook
- Key Takeaways
- East Asia Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- China
- Japan
- South Korea
- By Product Outlook
- By Stage Type Outlook
- By Customer Type Outlook
- By Therapeutic Area Outlook
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Outlook
- By Stage Type Outlook
- By Customer Type Outlook
- By Therapeutic Area Outlook
- Key Takeaways
- South Asia and Pacific Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product Outlook
- By Stage Type Outlook
- By Customer Type Outlook
- By Therapeutic Area Outlook
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Outlook
- By Stage Type Outlook
- By Customer Type Outlook
- By Therapeutic Area Outlook
- Key Takeaways
- Middle East & Africa Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product Outlook
- By Stage Type Outlook
- By Customer Type Outlook
- By Therapeutic Area Outlook
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Outlook
- By Stage Type Outlook
- By Customer Type Outlook
- By Therapeutic Area Outlook
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2024
- By Product Outlook
- By Stage Type Outlook
- By Customer Type Outlook
- By Therapeutic Area Outlook
- Canada
- Pricing Analysis
- Market Share Analysis, 2024
- By Product Outlook
- By Stage Type Outlook
- By Customer Type Outlook
- By Therapeutic Area Outlook
- Mexico
- Pricing Analysis
- Market Share Analysis, 2024
- By Product Outlook
- By Stage Type Outlook
- By Customer Type Outlook
- By Therapeutic Area Outlook
- Brazil
- Pricing Analysis
- Market Share Analysis, 2024
- By Product Outlook
- By Stage Type Outlook
- By Customer Type Outlook
- By Therapeutic Area Outlook
- Chile
- Pricing Analysis
- Market Share Analysis, 2024
- By Product Outlook
- By Stage Type Outlook
- By Customer Type Outlook
- By Therapeutic Area Outlook
- Germany
- Pricing Analysis
- Market Share Analysis, 2024
- By Product Outlook
- By Stage Type Outlook
- By Customer Type Outlook
- By Therapeutic Area Outlook
- UK
- Pricing Analysis
- Market Share Analysis, 2024
- By Product Outlook
- By Stage Type Outlook
- By Customer Type Outlook
- By Therapeutic Area Outlook
- Italy
- Pricing Analysis
- Market Share Analysis, 2024
- By Product Outlook
- By Stage Type Outlook
- By Customer Type Outlook
- By Therapeutic Area Outlook
- Spain
- Pricing Analysis
- Market Share Analysis, 2024
- By Product Outlook
- By Stage Type Outlook
- By Customer Type Outlook
- By Therapeutic Area Outlook
- France
- Pricing Analysis
- Market Share Analysis, 2024
- By Product Outlook
- By Stage Type Outlook
- By Customer Type Outlook
- By Therapeutic Area Outlook
- India
- Pricing Analysis
- Market Share Analysis, 2024
- By Product Outlook
- By Stage Type Outlook
- By Customer Type Outlook
- By Therapeutic Area Outlook
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2024
- By Product Outlook
- By Stage Type Outlook
- By Customer Type Outlook
- By Therapeutic Area Outlook
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2024
- By Product Outlook
- By Stage Type Outlook
- By Customer Type Outlook
- By Therapeutic Area Outlook
- China
- Pricing Analysis
- Market Share Analysis, 2024
- By Product Outlook
- By Stage Type Outlook
- By Customer Type Outlook
- By Therapeutic Area Outlook
- Japan
- Pricing Analysis
- Market Share Analysis, 2024
- By Product Outlook
- By Stage Type Outlook
- By Customer Type Outlook
- By Therapeutic Area Outlook
- South Korea
- Pricing Analysis
- Market Share Analysis, 2024
- By Product Outlook
- By Stage Type Outlook
- By Customer Type Outlook
- By Therapeutic Area Outlook
- Russia
- Pricing Analysis
- Market Share Analysis, 2024
- By Product Outlook
- By Stage Type Outlook
- By Customer Type Outlook
- By Therapeutic Area Outlook
- Poland
- Pricing Analysis
- Market Share Analysis, 2024
- By Product Outlook
- By Stage Type Outlook
- By Customer Type Outlook
- By Therapeutic Area Outlook
- Hungary
- Pricing Analysis
- Market Share Analysis, 2024
- By Product Outlook
- By Stage Type Outlook
- By Customer Type Outlook
- By Therapeutic Area Outlook
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2024
- By Product Outlook
- By Stage Type Outlook
- By Customer Type Outlook
- By Therapeutic Area Outlook
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2024
- By Product Outlook
- By Stage Type Outlook
- By Customer Type Outlook
- By Therapeutic Area Outlook
- South Africa
- Pricing Analysis
- Market Share Analysis, 2024
- By Product Outlook
- By Stage Type Outlook
- By Customer Type Outlook
- By Therapeutic Area Outlook
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product Outlook
- By Stage Type Outlook
- By Customer Type Outlook
- By Therapeutic Area Outlook
- Competition Analysis
- Competition Deep Dive
- Piramal Pharma Solutions
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- CordenPharma International
- Wuxi AppTec
- Cambrex Corporation
- Recipharm AB
- Thermo Fisher Scientific
- Lonza
- Catalent Inc.
- Siegfried Holding AG
- Boehringer Ingelheim
- Piramal Pharma Solutions
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2020-2035
- Table 2: Global Market Value (USD Million) Forecast by Product Outlook , 2020-2035
- Table 3: Global Market Value (USD Million) Forecast by Stage Type Outlook, 2020-2035
- Table 4: Global Market Value (USD Million) Forecast by Customer Type Outlook, 2020-2035
- Table 5: Global Market Value (USD Million) Forecast by Therapeutic Area Outlook, 2020-2035
- Table 6: North America Market Value (USD Million) Forecast by Country, 2020-2035
- Table 7: North America Market Value (USD Million) Forecast by Product Outlook , 2020-2035
- Table 8: North America Market Value (USD Million) Forecast by Stage Type Outlook, 2020-2035
- Table 9: North America Market Value (USD Million) Forecast by Customer Type Outlook, 2020-2035
- Table 10: North America Market Value (USD Million) Forecast by Therapeutic Area Outlook, 2020-2035
- Table 11: Latin America Market Value (USD Million) Forecast by Country, 2020-2035
- Table 12: Latin America Market Value (USD Million) Forecast by Product Outlook , 2020-2035
- Table 13: Latin America Market Value (USD Million) Forecast by Stage Type Outlook, 2020-2035
- Table 14: Latin America Market Value (USD Million) Forecast by Customer Type Outlook, 2020-2035
- Table 15: Latin America Market Value (USD Million) Forecast by Therapeutic Area Outlook, 2020-2035
- Table 16: Western Europe Market Value (USD Million) Forecast by Country, 2020-2035
- Table 17: Western Europe Market Value (USD Million) Forecast by Product Outlook , 2020-2035
- Table 18: Western Europe Market Value (USD Million) Forecast by Stage Type Outlook, 2020-2035
- Table 19: Western Europe Market Value (USD Million) Forecast by Customer Type Outlook, 2020-2035
- Table 20: Western Europe Market Value (USD Million) Forecast by Therapeutic Area Outlook, 2020-2035
- Table 21: Eastern Europe Market Value (USD Million) Forecast by Country, 2020-2035
- Table 22: Eastern Europe Market Value (USD Million) Forecast by Product Outlook , 2020-2035
- Table 23: Eastern Europe Market Value (USD Million) Forecast by Stage Type Outlook, 2020-2035
- Table 24: Eastern Europe Market Value (USD Million) Forecast by Customer Type Outlook, 2020-2035
- Table 25: Eastern Europe Market Value (USD Million) Forecast by Therapeutic Area Outlook, 2020-2035
- Table 26: East Asia Market Value (USD Million) Forecast by Country, 2020-2035
- Table 27: East Asia Market Value (USD Million) Forecast by Product Outlook , 2020-2035
- Table 28: East Asia Market Value (USD Million) Forecast by Stage Type Outlook, 2020-2035
- Table 29: East Asia Market Value (USD Million) Forecast by Customer Type Outlook, 2020-2035
- Table 30: East Asia Market Value (USD Million) Forecast by Therapeutic Area Outlook, 2020-2035
- Table 31: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2020-2035
- Table 32: South Asia and Pacific Market Value (USD Million) Forecast by Product Outlook , 2020-2035
- Table 33: South Asia and Pacific Market Value (USD Million) Forecast by Stage Type Outlook, 2020-2035
- Table 34: South Asia and Pacific Market Value (USD Million) Forecast by Customer Type Outlook, 2020-2035
- Table 35: South Asia and Pacific Market Value (USD Million) Forecast by Therapeutic Area Outlook, 2020-2035
- Table 36: Middle East & Africa Market Value (USD Million) Forecast by Country, 2020-2035
- Table 37: Middle East & Africa Market Value (USD Million) Forecast by Product Outlook , 2020-2035
- Table 38: Middle East & Africa Market Value (USD Million) Forecast by Stage Type Outlook, 2020-2035
- Table 39: Middle East & Africa Market Value (USD Million) Forecast by Customer Type Outlook, 2020-2035
- Table 40: Middle East & Africa Market Value (USD Million) Forecast by Therapeutic Area Outlook, 2020-2035
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2020-2035
- Figure 3: Global Market Value (USD Million) Share and BPS Analysis by Region, 2025 and 2035
- Figure 4: Global Market Y-o-Y Growth Comparison by Region, 2025-2035
- Figure 5: Global Market Attractiveness Analysis by Region
- Figure 6: North America Market Incremental Dollar Opportunity, 2025-2035
- Figure 7: Latin America Market Incremental Dollar Opportunity, 2025-2035
- Figure 8: Western Europe Market Incremental Dollar Opportunity, 2025-2035
- Figure 9: Eastern Europe Market Incremental Dollar Opportunity, 2025-2035
- Figure 10: East Asia Market Incremental Dollar Opportunity, 2025-2035
- Figure 11: South Asia and Pacific Market Incremental Dollar Opportunity, 2025-2035
- Figure 12: Middle East & Africa Market Incremental Dollar Opportunity, 2025-2035
- Figure 13: North America Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 14: Latin America Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 15: Western Europe Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 16: Eastern Europe Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 17: East Asia Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 18: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 19: Middle East & Africa Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 20: Global Market - Tier Structure Analysis
- Figure 21: Global Market - Company Share Analysis

