Small Bore Connectors Market : Global Industry Analysis 2015 - 2024 and Opportunity Assessment 2025 - 2035
The small bore connectors market is segmented by product and application. Product covers liquid bore connectors and gas bore connectors. Applications include respiratory and enteral use. Urology and neuraxial anesthesia are included. End users cover hospitals and clinics. Ambulatory surgical centers and home care settings are included. Forecast for 2026 to 2036.
Historical Data Covered: 2015 - 2024 | Base Year: 2024 | Estimated Year: 2025 | Forecast Period: 2025 - 2035
Small Bore Connectors Market Size, Market Forecast and Outlook By FMI
The small bore connectors market is projected at USD 660.7 million in 2026. Revenue is forecast to total USD 964.7 million by 2036. Sales are anticipated to expand at 4.3% CAGR from 2026 to 2036. Liquid bore connectors are likely to lead with 56.4% product share. Hospitals are projected to account for 48.4% end-user demand in 2026.
Summary of the Small Bore Connectors Market
- Demand and Growth Drivers
- Patient safety rules may support wider use of standardized medical connectors.
- Minimally invasive procedures are likely to lift connector demand in fluid systems.
- ISO compliance is anticipated to guide product selection across healthcare facilities.
- Product and Segment View
- Liquid bore connectors are likely to lead through infusion and dialysis applications.
- Hospitals may stay ahead because many procedures depend on secure fluid transfer.
- Gas bore connectors are forecast to gain value in respiratory and anesthesia systems.
- Geography and Competitive Outlook
- North America is likely to retain value through advanced medical device adoption.
- Asia may grow faster as healthcare production and hospital investment increase.
- Europe is forecast to support steady demand through safety standards and device compliance.
- Analyst Opinion
- Sabyasachi Ghosh, Principal Consultant for Healthcare at FMI, suggests, “Small bore connector demand is likely to favor manufacturers with strong ISO compliance and precise product design. Hospitals will prefer connectors that reduce misconnection risk and support safer fluid delivery.”
- Small Bore Connectors Market Value Analysis
- Small bore connectors are moving from basic fittings into safety-critical medical components.
- Liquid connectors hold value because fluid delivery needs accuracy and leak resistance.
- Hospital use keeps demand stable through IV therapy and procedural care.
- Suppliers with compliant designs and custom connector capability will gain stronger healthcare contracts.
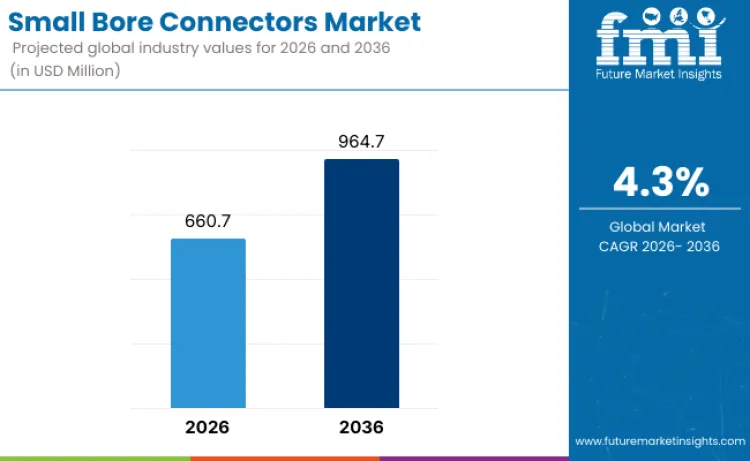
Small Bore Connectors Market Key Takeaways
| Metric | Value |
|---|---|
| Market Value (2026) | USD 660.7 million |
| Market Forecast Value (2036) | USD 964.7 million |
| Forecast CAGR (2026 to 2036) | 4.3% |
How Are the Segments Classified in the Small Bore Connectors Market?
The small bore connectors market is categorized by product, end use, and region. By product, the industry is divided into liquid bore connectors, gas bore connectors, and other types. The liquid bore connectors segment is expected to grow significantly, driven by demand in medical applications. By end use, the industry includes hospitals, pharmaceutical manufacturing, healthcare devices, and industrial sectors. Regionally, the industry spans North America, Latin America, Western Europe, Eastern Europe, East Asia, South Asia & Pacific, and the Middle East & Africa. This segmentation highlights the various applications of small bore connectors across different industries, with medical and healthcare applications dominating.
Which Product Segment Accounts for the Largest Share in the Small Bore Connectors Market?
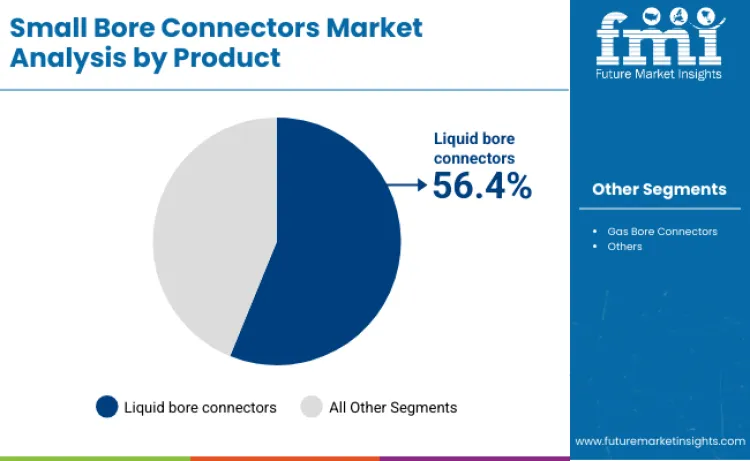
Liquid bore connectors lead the small bore connectors market with a 56.4% share. This dominance is primarily due to their widespread use in critical medical devices such as IV systems, infusion pumps, dialysis equipment, and other fluid delivery systems. With the rise in chronic diseases and an aging population, demand for these connectors is increasing rapidly. Additionally, their use in pharmaceutical manufacturing and laboratory settings further expands their market scope. Liquid bore connectors are highly reliable, adhere to strict safety standards, and ensure accurate and safe fluid transfer, making them indispensable in healthcare and related sectors.
Which End Use Segment Represents the Largest Share in the Small Bore Connectors Market?
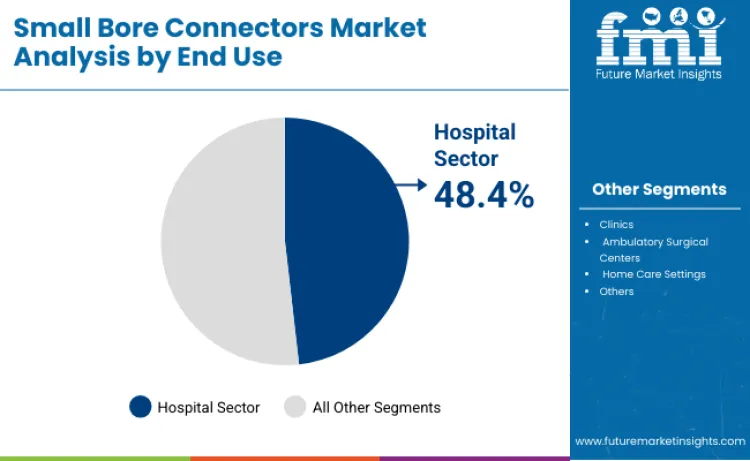
The hospital sector holds the largest share of 48.4% in the small bore connectors market, with a projected CAGR of 3.4% by 2036. This segment's dominance is due to the critical role small bore connectors play in various medical procedures such as intravenous fluid administration, dialysis, anesthesia delivery, and minimally invasive surgeries. As the demand for high-quality medical devices and instruments increases, especially in the face of rising chronic disease rates, hospitals require connectors that meet strict safety and regulatory standards. The growing prevalence of surgeries and medical procedures further supports the hospital sector's growth in this market.
What Are the Key Dynamics in the Small Bore Connectors Market?
The small bore connectors market is driven by increasing demand for reliable, precise, and safe fluid and gas transfer systems in medical devices. The emphasis on regulatory compliance and patient safety is pushing the adoption of advanced connectors. Manufacturers are focusing on developing new, safer alternatives to traditional welding methods for creating secure connections in medical devices. As small bore connectors offer superior performance, reliability, and ease of use, they are rapidly replacing older methods, leading to growth in the industry. The shift towards safer and more efficient technologies is expected to continue to drive demand across healthcare applications.
How Do Custom-Designed Small Bore Connectors Offer Growth Opportunities?
Custom-designed small bore connectors present significant growth opportunities for manufacturers, enabling them to meet specific needs in healthcare, pharmaceuticals, and industrial applications. These connectors can be tailored to optimize fluid or gas transfer, enhance safety, and reduce leakage risks, especially in critical medical devices like ventilators, infusion pumps, and anesthesia systems. Customization helps manufacturers target niche markets, creating differentiation and increasing market share. As demand grows for next-generation medical technologies and portable healthcare devices, manufacturers offering personalized solutions will gain a competitive edge. Furthermore, custom-designed connectors can lead to long-term customer relationships and recurring revenue, as industries increasingly seek specialized, high-performance solutions.
How Are Regulatory Standards and Safety Concerns Shaping Small Bore Connector Adoption?
Regulatory bodies, such as ISO, play a significant role in shaping the small bore connectors market by enforcing strict safety standards, including the ISO 80369 series. These standards are designed to prevent misconnections between incompatible medical systems, ensuring patient safety and reliability in healthcare applications. Manufacturers are adopting advanced materials and designs to comply with these regulations, which has fostered innovation in the industry. As a result, the industry is seeing the development of connectors with better leak resistance, greater durability, and improved compatibility. Regulatory initiatives are driving widespread adoption across different regions and healthcare systems, further bolstering the industry’s growth.
What Are the Challenges Facing the Small Bore Connectors Market?
Despite their advantages, small bore connectors face competition from traditional connection methods such as welding, soldering, and flanged connections. These conventional methods are often perceived as more cost-effective, reliable, and easier to implement, with lower initial costs. Moreover, there is skepticism about the long-term performance of small bore connectors, especially in high-pressure or high-temperature environments. Many industries are reluctant to transition to new connector technologies, fearing potential risks and operational challenges. Additionally, manufacturers offering alternative solutions that achieve similar benefits without the complexities of small bore connectors may slow market penetration. Overcoming these barriers will require continued education, improved performance, and cost-effective solutions.
How Are Key Countries Driving Growth in the Small Bore Connectors Market?
The small bore connectors market is experiencing steady growth across various countries, influenced by regional dynamics and specific demand drivers. In North America, the United States leads the industry with a dominant share, largely driven by advancements in medical technology and a shift toward minimally invasive procedures. Meanwhile, in Asia Pacific, South Korea is expected to show significant growth due to its increasing demand for high-precision medical and industrial applications. Other regions, including Europe and the GCC, are also contributing to the industry's expansion through investments in healthcare infrastructure and industrial modernization. Each country brings its own set of factors, creating a diverse and evolving market landscape.
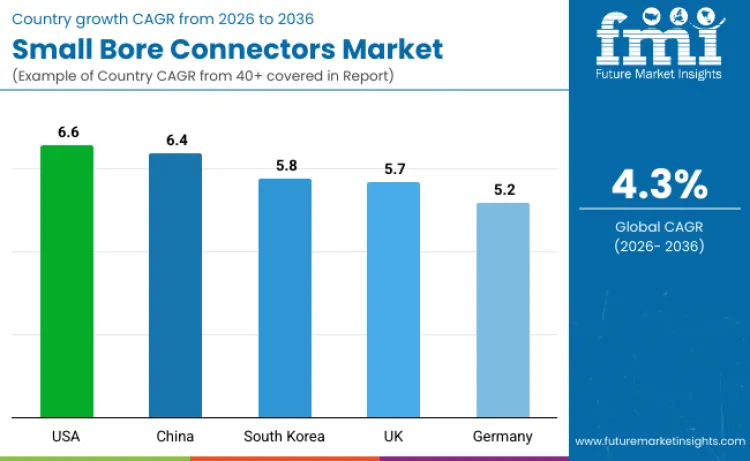
| Country | CAGR |
|---|---|
| USA | 6.6% |
| China | 6.4% |
| South Korea | 5.8% |
| UK | 5.7% |
| Germany | 5.2% |
How is the Small Bore Connectors Market Evolving in the USA?
The small bore connectors market in the USA is set to grow at a CAGR of 6.6% from 2026 to 2036, reaching over USD 80.1 million by 2036. The rising preference for minimally invasive medical procedures is a key driver of this growth, with small-bore connectors being vital for devices like catheters, endoscopes, and infusion pumps. Their compact design and ability to provide reliable fluid and gas transfer make them essential in modern medical applications. As more patients opt for procedures with quicker recovery times, the demand for these connectors will continue to rise, further propelling market expansion in the USA.
What is Fueling China's Small Bore Connectors Market Growth?
China's small bore connectors market is anticipated to grow significantly from 2026 to 2036, with a projected CAGR of 6.4%. The nation's strong manufacturing capabilities are driving this growth, enabling local producers to offer cost-effective, high-quality connectors. Government policies focused on enhancing the medical device industry and improving production standards are contributing to the industry's development. By producing connectors domestically, China is reducing reliance on imports, which stabilizes the supply chain and makes small-bore connectors more accessible across medical and industrial sectors. These factors, combined with domestic innovation, are fueling the industry's growth.
Why is Germany Poised for Steady Small Bore Connectors Market Growth?
Germany’s small bore connectors market is expected to grow at a steady CAGR of 5.2% from 2026 to 2036. Known for its precision engineering and compliance with global safety standards, Germany continues to be a leader in producing high-quality small bore connectors. The country’s exports are gaining traction, particularly in emerging markets investing heavily in healthcare infrastructure. As Germany’s manufacturing sector adapts to international demands, the industry will benefit from its reputation for innovation, safety, and reliability, further solidifying its position in the global market for small-bore connectors.
How Will South Korea’s Market for Small Bore Connectors Evolve?
South Korea’s small bore connectors market is forecast to grow at a CAGR of 5.8% from 2026 to 2036. The country is focusing on improving both its medical and industrial sectors, with increasing demand for small-bore connectors in high-precision applications. The government’s continued investment in healthcare infrastructure, coupled with a growing need for more efficient energy solutions, supports this growth. As South Korea continues to innovate and expand its production capabilities, the industry for small bore connectors will be driven by the rising demand for medical devices and high-performance industrial solutions.
What Trends Are Shaping the Small Bore Connectors Market in the UK?
The UK is projected to see steady growth in the small bore connectors market with a CAGR of 5.7% from 2026 to 2036. Despite challenges like Brexit, the UK’s strong healthcare sector and focus on medical technology innovation are key drivers of demand. Small-bore connectors are crucial in the UK’s medical devices industry, particularly for their reliability and compliance with safety regulations. As more healthcare providers look to localize production and develop innovative solutions, the industry for small bore connectors will continue to grow, especially as demand for high-quality, safe products remains high.
What Is the Competitive Landscape of the Small Bore Connectors Market in 2026 to 2036?
The small bore connectors market is expected to experience robust competition between 2026 and 2036, driven by key players investing in product innovation, regulatory compliance, and strategic partnerships. Leading manufacturers will continue to focus on expanding their product portfolios to meet the growing demand for safe and standardized medical connectors. As the industry matures, companies will increasingly collaborate with healthcare providers and regulatory bodies to ensure compliance with evolving standards like ISO 80369-3, which is crucial for patient safety. These partnerships will enable market leaders to enhance their offerings and expand their global reach, especially in emerging markets.
Technological advancements in connector designs, such as the integration of smart sensors and advanced materials, will intensify competition. Companies that successfully combine durability, ease of use, and enhanced functionality will have a competitive edge. As healthcare institutions place a stronger emphasis on patient safety and efficient workflows, manufacturers who can offer connectors that minimize the risk of misconnections while improving ease of use and performance will thrive. Strategic mergers and acquisitions are likely, enabling smaller players to scale up and gain access to global markets, further fueling the competitive dynamics of the industry.
Key Players of Small Bore Connectors Industry
- Baxter International Inc.
- B. Braun
- BD
- Smiths Medical - Smiths Group Company
- ICU Medical Inc
- Merit Medical Systems, Inc.
- Nordson Corporation
- Elcam Medical
- CardioMed Supplies Inc. (Nipro Corporation)
Scope of Report
| Report Attribute | Details |
|---|---|
| Current Market Size (2026) | USD 660.7 million |
| Projected Market Size (2036) | USD 964.7 million |
| CAGR (2026 to 2036) | 4.3% |
| Market Analysis Parameter | Revenue in USD million |
| Product Types Analyzed | Liquid Bore Connectors, Gas Bore Connectors |
| End-Use Applications | Respiratory, Enteral, Urology, Limb cuff inflation, Neuraxial Anesthesia, Intravascular (Luer) |
| End-User Segmentation | Hospitals, Clinics, Ambulatory Surgical Centers, Home Care Settings, Others |
| Regions Covered | North America, Latin America, East Asia, South Asia & Pacific, Western Europe, Eastern Europe, Middle East & Africa |
| Countries Covered | United States, Japan, Germany, India, United Kingdom, France, Italy, Brazil, Canada, South Korea, Australia, Spain, Netherlands, Saudi Arabia, Switzerland |
| Key Players in the industry | Baxter International Inc., B. Braun, BD, Smiths Medical (Smiths Group), ICU Medical Inc., Merit Medical Systems, Inc., Nordson Corporation, Elcam Medical, CardioMed Supplies Inc. (Nipro Corporation) |
| Additional Attributes | Technological advancements, Government policies and incentives, Environmental impact considerations, Market trends, Health and wellness trends |
Key Segments of the Small Bore Connectors Industry
By Product:
- Liquid Bore Connectors
- Gas Bore Connectors
By Application:
- Respiratory
- Enteral
- Urology
- Limb Cuff Inflation
- Neuraxial Anesthesia
- Intravascular (Luer)
By End-User:
- Hospitals
- Clinics
- Ambulatory Surgical Centers
- Home Care Settings
- Others
By Region:
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia & Pacific
- Middle East & Africa (MEA)
Frequently Asked Questions
What is the future of the global small bore connectors industry?
The industry is projected to grow at a CAGR of 4.3% from 2026 to 2036.
What was the worth of the global small bore connectors industry in 2024?
The industry was valued at USD 607.0 million in 2024.
What will the worth of the global small bore connectors industry be by 2036?
The industry is expected to reach USD 924.8 million by 2036.
Which region will showcase the highest CAGR during the forecast period?
China is expected to record the highest CAGR of 6.4% during 2026 to 2036.
Who are the key manufacturers in the global small bore connectors industry?
Key players include Baxter International Inc., B. Braun, BD, Smiths Medical, ICU Medical Inc., Merit Medical Systems, Inc., Nordson Corporation, Elcam Medical, and CardioMed Supplies Inc. (Nipro Corporation).
Table of Content
- Executive Summary
- Industry Overview
- Small Bore Connectors Industry Introduction
- Industry Size and Forecast Overview 2026 to 2036
- Industry Value Chain Overview
- Industry Pricing Overview
- Industry Background
- Industry Definition and Scope
- Industry Segmentation Framework
- Industry Ecosystem Analysis
- Industry Trends and Adoption Patterns
- Small Bore Connectors Market Analysis
- Small Bore Connectors Industry Size and Forecast 2026 to 2036
- Small Bore Connectors Demand Drivers
- Small Bore Connectors Market Segmentation
- Small Bore Connectors Regional Analysis
- Small Bore Connectors Key Market Dynamics
- Industry Pricing Analysis
- Global Pricing Trends
- Regional Pricing Comparison
- Material Cost Impact Assessment
- Industry Structure Analysis
- Industry Cost Structure
- Industry Value Chain Analysis
- Market Share Analysis of Key Players
- Industry Entry Barrier Assessment
- Competition Analysis
- Industry Competitive Landscape
- Strategic Developments and Expansion Trends
- Product Portfolio Comparison
- Competitive Positioning of Market Leaders
- Assumptions and Acronyms Used
- Research Methodology
List of Tables
- Table 1: Small Bore Connectors Market Size 2026 to 2036
- Table 2: Small Bore Connectors Industry Growth Rate 2026 to 2036
- Table 3: Small Bore Connectors Value Share by Product Type 2026
- Table 4: Small Bore Connectors Market Share by End-Use Sector 2026
- Table 5: Small Bore Connectors Regional Demand Breakdown 2026
- Table 6: Competitive Benchmarking of Leading Players
- Table 7: Growth Drivers for Small Bore Connectors Market
- Table 8: Impact of Regulatory Standards on Small Bore Connectors Market
- Table 9: Technological Innovations in Small Bore Connectors
- Table 10: Country-wise Growth Projections
List of Figures
- Figure 1: Small Bore Connectors Market Value Share by Product Type 2026
- Figure 2: Small Bore Connectors Market Growth Trend 2026 to 2036
- Figure 3: Small Bore Connectors Market Share by End-Use Sector 2026
- Figure 4: Small Bore Connectors Regional Distribution
- Figure 5: Liquid Bore Connectors Demand Growth
- Figure 6: Hospital Sector Demand Growth for Small Bore Connectors
- Figure 7: Custom-Designed Small Bore Connectors Market Trends
- Figure 8: Country-wise Demand Growth for Small Bore Connectors
- Figure 9: Regulatory Standards Impact on Adoption
- Figure 10: Competitive Positioning of Small Bore Connector Manufacturers

