Severe to profound sensorineural hearing loss represents the primary clinical indication for cochlear implant candidacy. Patients demonstrating hearing thresholds worse than 70 to 90 decibels across speech frequencies and limited benefit from conventional hearing aids meet basic screening criteria. Audiological testing confirms insufficient speech recognition scores with optimally fitted hearing aids, typically defined as less than 50 percent sentence recognition in aided conditions. This narrow eligibility window excludes mild to moderate hearing loss populations, concentrating demand among patients with profound auditory nerve damage or cochlear dysfunction.
Congenital deafness in paediatric populations drives substantial implant volumes in developed healthcare systems with established newborn hearing screening programs. Early identification through universal screening enables intervention during critical language development periods. Clinical guidelines increasingly recommend implantation before 12 months of age to maximise auditory cortex plasticity and speech development outcomes. Bilateral implantation gains acceptance for paediatric candidates based on evidence supporting superior spatial hearing and language acquisition compared with unilateral approaches. Paediatric pathways involve multidisciplinary assessment including audiologists, speech therapists, psychologists and surgeons before proceeding to implantation.
Acquired deafness in adult populations presents distinct clinical and economic considerations compared with paediatric cases. Post lingual hearing loss following meningitis, ototoxic medication exposure, sudden sensorineural hearing loss or progressive age related decline creates adult candidate pools. These patients retain auditory memory and speech production capability, often achieving faster rehabilitation progress than prelingually deaf children. However, duration of deafness negatively correlates with outcomes as prolonged auditory deprivation leads to neural pathway degradation. Candidate evaluation emphasises realistic expectation setting regarding speech perception recovery timelines.
Bone conduction hearing implants address conductive hearing loss, mixed hearing loss and single sided deafness in populations where cochlear implants prove inappropriate. Conductive losses from chronic ear infections, congenital aural atresia or ossicular chain abnormalities prevent sound transmission to functional cochleae. Bone conduction systems bypass damaged middle ear structures by transmitting vibrations directly through skull bone to the inner ear. Single sided deafness following acoustic neuroma surgery or sudden hearing loss represents expanding indication segments as evidence supporting quality of life improvements accumulates.
Geographic and socioeconomic factors create substantial candidacy disparities beyond pure clinical criteria. Developed nations with comprehensive hearing screening and referral infrastructure identify candidates systematically through healthcare systems. Developing regions with limited audiological services underdiagnose severe hearing loss, leaving large unmet clinical need despite technical candidacy. Urban concentration of specialist services creates rural access barriers even in developed economies. These structural factors constrain demand realisation below epidemiological prevalence estimates.
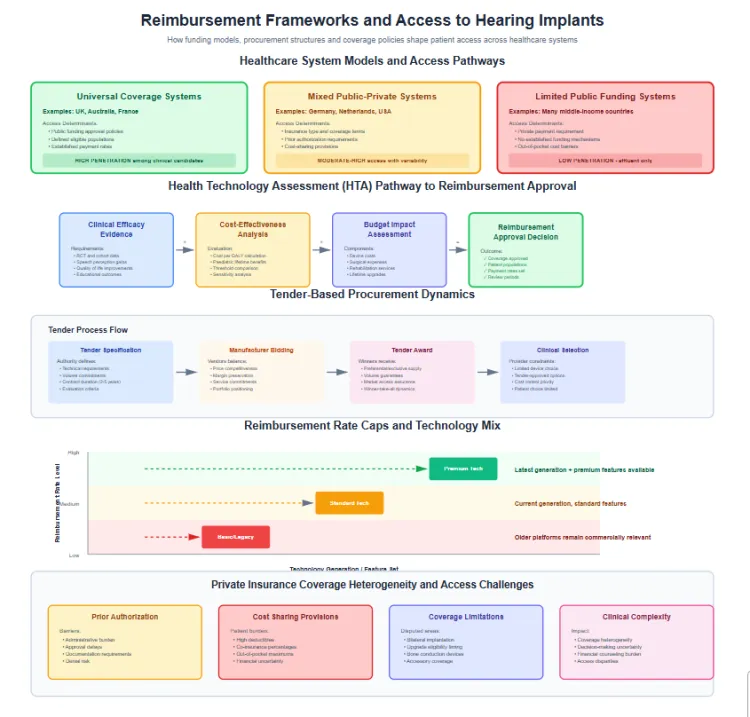
National health systems in countries with universal coverage determine hearing implant access primarily through funding approval policies rather than clinical eligibility alone. Public reimbursement decisions establish whether cochlear implants receive coverage, define eligible patient populations and set payment rates for procedures and devices. Countries including the United Kingdom, Australia and France provide comprehensive cochlear implant coverage for approved indications, driving high penetration among clinical candidates. Systems without established funding mechanisms leave patients dependent on private payment, effectively restricting access to affluent populations despite clinical suitability.
Reimbursement approval processes involve health technology assessment evaluating clinical efficacy, cost effectiveness and budget impact. Payers assess evidence from randomised trials and cohort studies demonstrating speech perception improvements, quality of life gains and educational outcome enhancements. Economic evaluations calculate cost per quality adjusted life year, with paediatric implantation demonstrating favourable ratios due to lifetime benefit accrual. Budget impact analyses account for device costs, surgical expenses, rehabilitation services and processor upgrades over patient lifetimes. Positive reimbursement decisions require evidence meeting established thresholds across these evaluation dimensions.
Tender based procurement in public systems creates competitive pricing pressure while establishing approved device lists. National or regional health authorities issue tenders specifying technical requirements, volume commitments and contract duration. Manufacturers submit bids balancing price competitiveness against margin preservation. Winning tenders provide preferential or exclusive supply arrangements for contract periods. This procurement model concentrates volume among selected vendors, creating winner take all dynamics in specific geographies. Hospitals and surgeons typically select from tender approved options, limiting patient choice but controlling costs.
Reimbursement rate caps influence device mix and technology tier availability within funded programs. Payers establish maximum reimbursement amounts covering device, surgical and rehabilitation costs. When caps fall below premium device pricing, healthcare providers absorb shortfalls, creating preference for cost effective options meeting minimum specifications. Manufacturers respond by offering product portfolios at different price points, with premium features reserved for private pay or higher reimbursement settings. Rate adequacy directly affects technology generation availability as older platforms remain commercially relevant when reimbursement levels prohibit latest generation adoption.
Private insurance coverage varies substantially across countries and policy types, creating access disparities even in predominantly private healthcare systems. United States insurers increasingly cover cochlear implants following Medicare precedent, but prior authorisation requirements, coverage limitations and cost sharing provisions affect accessibility. High deductible plans impose significant out of pocket costs despite technical coverage. Disputes over bilateral implantation, upgrade eligibility and bone conduction device coverage generate administrative burden and access uncertainty. Insurance coverage heterogeneity complicates clinical decision making and financial counselling during candidacy evaluation.
Electrode array design represents fundamental technical differentiation affecting surgical approach and hearing outcome potential. Straight electrode arrays minimise cochlear trauma during insertion, preserving residual hearing for hybrid acoustic electric stimulation strategies. Perimodilar arrays curve toward the modiolus positioning contacts closer to spiral ganglion neurons, potentially improving channel selectivity and power efficiency. Array length, contact number and spacing influence spectral resolution and frequency mapping flexibility. Surgeons develop preferences based on anatomical considerations, insertion success rates and historical outcomes experience.
Signal processing algorithms distinguish manufacturer platforms through approaches to sound encoding and noise reduction. Current steering strategies create virtual channels between physical electrode contacts, increasing spectral resolution beyond hardware channel limits. Adaptive processing adjusts stimulation parameters based on acoustic environment characteristics, optimising speech intelligibility in noise. Frequency allocation tables map acoustic frequencies to electrode positions, with different philosophies regarding spectral compression versus place based mapping. Audiologists evaluate processing strategies based on patient specific outcomes during mapping sessions, with some platforms offering multiple algorithm options.
MRI compatibility specifications influence device selection particularly for paediatric candidates facing lifetime imaging needs. Conventional cochlear implants require magnet removal before MRI scanning or limit field strength to 1.5 Tesla with positioning restrictions. Advanced systems incorporate removable or rotating magnet designs enabling 3.0 Tesla scanning without surgical intervention. This capability proves valuable for monitoring neurological conditions, investigating non auditory pathology and managing traumatic injuries. Parents and patients prioritise MRI compatibility to avoid future surgical procedures or diagnostic limitations.
Implant longevity and reliability specifications affect total ownership cost and revision surgery risk. Internal implants require surgical replacement upon device failure, creating patient burden and healthcare system costs. Manufacturers provide failure rate data and warranty coverage establishing reliability expectations. Hermeticity testing, impact resistance specifications and historical survival analyses inform surgeon confidence in long term device performance. Higher reliability devices command pricing premiums justified by reduced revision probability and associated surgical risks.
External processor performance and upgrade flexibility create ongoing differentiation beyond initial implantation. Processor generations improve every 3 to 7 years, offering enhanced signal processing, connectivity features and form factor refinements. Upgrade pathways allowing processor replacement without implant surgery provide patient value and manufacturer recurring revenue. Wireless streaming capability, rechargeable battery options, water resistance ratings and physical durability affect daily use experience. Patients increasingly weigh processor ecosystem evolution potential during initial device selection.
ENT surgeon expertise requirements limit procedure scalability as cochlear implantation demands subspecialised otology training beyond general ENT scope. Surgical complexity involves mastoidectomy, facial recess approach, cochleostomy and electrode insertion requiring microsurgical precision to preserve residual hearing and avoid complications. Training pathways extend 1 to 2 years beyond standard ENT residency through fellowship programs at high volume centres. Surgeon availability concentrates in academic medical centres and urban regions, creating geographic access barriers. Developing healthcare systems face severe specialist shortages preventing program establishment despite clinical demand.
Audiological support infrastructure proves equally critical as surgical capacity for sustainable hearing implant programs. Preoperative candidacy evaluation requires comprehensive audiometric testing, speech perception assessment and aided hearing aid trial documentation. Postoperative device activation, programming and mapping demand specialised audiologists trained in manufacturer specific software platforms. Initial programming typically involves multiple sessions over several months adjusting stimulation levels and electrode configurations. Ongoing management includes troubleshooting, reprogramming as hearing changes and processor upgrades. Programs require dedicated audiologist time that competes with conventional hearing aid services in resource constrained settings.
Rehabilitation services including speech therapy, auditory verbal therapy and educational support determine functional outcomes particularly for paediatric implant recipients. Children receiving implants require intensive therapy integrating auditory input with speech production and language development. Family training ensures consistent auditory stimulation and communication strategies supporting neural pathway development. School based accommodations and specialised educational resources optimise academic progress. Geographic regions lacking rehabilitation infrastructure achieve suboptimal outcomes despite successful surgical implantation, undermining clinical value proposition and potentially affecting future referrals.
Hospital infrastructure requirements include surgical equipment, imaging capability and device inventory management systems. Operating rooms require surgical microscopes, facial nerve monitors, intraoperative imaging and specialised instrument sets. Preoperative CT and MRI scanning assess cochlear anatomy and surgical approach feasibility. Device inventory management tracks implant serial numbers, maintains backup units and coordinates with manufacturers on special orders. These infrastructure investments create fixed costs limiting program viability below minimum procedure volumes. Centralisation at regional hubs achieves operational efficiency but reduces geographic access.
Geographic disparities in specialist distribution create pronounced urban rural access gaps. Metropolitan areas host multiple implant programs with competitive referral networks, while rural regions may require travel exceeding several hundred kilometres to reach qualified centres. Telemedicine enables some remote audiological support but cannot substitute for surgical access or hands on mapping expertise. These disparities disproportionately affect disadvantaged populations already experiencing higher hearing loss prevalence due to occupational exposures and limited preventive care. Addressing geographic barriers requires workforce development, regional training programs and potentially mobile service delivery models.
External processor upgrades represent recurring revenue opportunities exceeding initial implant system pricing over device lifetimes. Processor generations advance every 3 to 7 years incorporating improved signal processing, enhanced connectivity and refined ergonomics. Patients desire upgrades accessing new features and performance improvements despite functional existing processors. Manufacturer pricing strategies balance upgrade accessibility against revenue optimisation, typically offering trade in programs, insurance coverage navigation and instalment payment options. Reimbursement policies vary regarding upgrade coverage, with some systems funding replacement after 5 to 7 years while others restrict upgrades to device failure situations.
Software updates and remote programming capabilities increasingly differentiate service models and patient satisfaction. Over the air firmware updates deliver new processing algorithms, feature enhancements and bug fixes without clinical visits. Remote programming enables audiologists to adjust device settings via telehealth platforms, reducing appointment burden and improving access for geographically distant patients. These capabilities create ongoing manufacturer patient touchpoints supporting brand loyalty and upgrade conversion. Subscription models for premium features may emerge as manufacturers explore recurring revenue beyond hardware sales.
Service contracts and warranty extensions provide predictable cost structures while generating manufacturer margin. Standard warranties cover manufacturing defects for 2 to 5 years, after which extended coverage becomes optional purchase. Service agreements include device replacement for failures, damage protection, loaner units during repairs and priority technical support. These programs prove particularly valuable for paediatric patients facing decades of device dependence. Predictable service costs enable families and payers to budget for long term expenses, while manufacturers secure steady revenue streams with high margins on service offerings.
Audiological mapping services create ongoing clinical engagement and patient retention mechanisms throughout implant lifetimes. Programming appointments occur most frequently during initial activation and first year, then transition to annual or as needed schedules. Manufacturers provide audiologist training, software platforms and technical support enabling these services. Some systems employ manufacturer audiologists for direct patient care particularly in emerging regions lacking independent specialists. Mapping service quality affects patient outcomes and satisfaction, influencing brand perception and upgrade decisions. Integrated service models strengthening audiologist manufacturer relationships create competitive moats.
Patient retention economics favour manufacturers maintaining existing user bases through upgrade cycles rather than acquiring new implant recipients. Implant platform compatibility limits processor upgradeability to same manufacturer products, creating natural switching barriers. Established audiologist familiarity with specific platforms and programming software creates preference inertia. Patient adaptation to particular sound processing approaches makes cross platform migration challenging despite technical feasibility. These retention factors enable manufacturers to preserve pricing power for upgrades and accessories even as competitive pressure intensifies for initial implantations.

Cochlear implants address severe to profound sensorineural hearing loss with functional cochleae but damaged hair cells or auditory nerves. Bone conduction implants serve conductive or mixed hearing loss where middle ear pathology prevents sound transmission to functional inner ears, plus single sided deafness. Clinical differentiation depends on audiometric testing, imaging and specific anatomical pathology.
Internal cochlear implants demonstrate reliability exceeding 95 percent at 10 years, with many functioning beyond 20 years before requiring surgical replacement. External processors typically upgrade every 5 to 10 years for technology improvements rather than failure. Bone conduction implants show similar internal component longevity with processor replacement following comparable cycles.
Hearing implant systems cost 30,000 to 100,000 dollars including device, surgery and rehabilitation, exceeding affordability for most patients without insurance coverage. Public reimbursement determines access for majority of candidates in universal healthcare systems, while private insurance coverage varies substantially affecting accessibility in other contexts.
Patients typically upgrade external processors every 5 to 10 years balancing technology advancement benefits against costs. Reimbursement policies influence upgrade frequency, with some systems funding replacement after specified intervals while others require demonstrated device failure. Manufacturer product cycles releasing new generations every 3 to 7 years create upgrade opportunities.
Surgeons prioritise electrode insertion success, reliability data, MRI compatibility and institutional procurement contracts. Audiologists value programming software intuitiveness, processing algorithm flexibility, patient outcome history and manufacturer technical support quality. Institutional tender agreements often constrain individual preferences by limiting available device options.
The report offers an in-depth examination of the demand for hearing implant in usa market, covering market valuation, forward-looking revenue analysis, competitive benchmarking, demand trends, principal growth factors, constraints, Product Type outlook, End User outlook, supply chain analysis, and strategic prospects.
This study provides a comprehensive evaluation of the demand for hearing implant in japan market, encompassing market size estimation, revenue projections, competitive dynamics, demand analysis, key growth catalysts, market challenges, Product Type developments, End User developments, supply chain insights, and emerging opportunities for stakeholders.
Demand for Hearing Implants in UK Size and Share Forecast Outlook 2026 to 2036
Demand for Hearing Implant in South Korea Size and Share Forecast Outlook 2026 to 2036
Implantable Neurostimulation Device EMI and Safety Test Systems Market is segmented by Test Focus (EMI and EMC, Electrical Safety, MRI Safety, Battery Reliability, Telemetry Validation), Device Type (Spinal Cord, Deep Brain, Sacral Nerve, Vagus Nerve, Peripheral Nerve), End User (OEMs, Contract Labs, Design Firms, Academic Centers, Hospital Research), System Format (Integrated Benches, Modular Racks, Shielded Chambers, Software Suites, MRI Phantoms), Sales Channel (Direct Sales, Channel Partners, Turnkey Projects, Validation Services, Service Contracts), and Region. Forecast for 2026 to 2036.