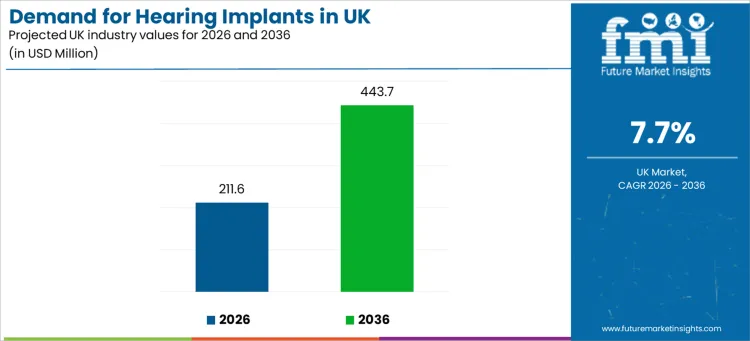
Demand for hearing implants in the UK is projected at USD 211.6 million in 2026 and is expected to reach USD 443.7 million by 2036, expanding at a 7.7% CAGR. This growth trajectory is anchored in rising diagnosis rates of sensorineural hearing loss, broader clinical acceptance of implantable auditory devices, and expanding candidacy criteria across both adult and pediatric populations.
Hearing implants are positioned as long-term auditory rehabilitation solutions where conventional amplification no longer delivers functional benefit. Buyers evaluate these systems through speech recognition outcomes, long-term neural compatibility, processor reliability, and post-implant rehabilitation efficiency. These factors make them central to treatment pathways for severe to profound hearing impairment and complex auditory nerve conditions.
For hospital administrators and clinical directors, adoption is closely tied to procedural throughput, post-surgical outcomes, and reimbursement alignment. Implant programs that demonstrate predictable rehabilitation trajectories and stable device performance support institutional reputation and long-term patient follow-up engagement.
Procurement teams also view hearing implants as part of a wider otology and neurotology equipment ecosystem, aligned with broader auditory device investment themes and implantable medical technology planning frameworks that shape long-range capital allocation decisions. Category planners often align choices with adjacent hearing restoration platforms and cochlear device system roadmaps when structuring long-term service line development.
| Metric | Value |
|---|---|
| Industry Value (2026) | USD 211.6 million |
| Industry Forecast Value (2036) | USD 443.7 million |
| Forecast CAGR 2026 to 2036 | 7.7% |
Demand is being shaped by demographic aging, higher survival rates among premature infants, and improved early screening protocols for congenital hearing loss. These trends are expanding the candidate pool for implantable auditory devices beyond traditionally narrow clinical definitions.
Clinical confidence in cochlear and neural interface technologies has strengthened as long-term outcome data continues to validate speech recognition improvements and quality-of-life gains. ENT specialists increasingly view implantation as a durable intervention rather than a last-resort therapy, particularly for progressive hearing deterioration.
Rehabilitation infrastructure development also supports adoption. Audiology clinics and hospital departments are investing in structured post-implant therapy programs, remote processor tuning platforms, and speech rehabilitation tools that reduce long-term care burden while improving patient adherence.
Category planners often align hearing implant strategies with adjacent themes tied to auditory prosthetics and implantable neuro-devices, while broader capital planning reflects overlaps with cochlear device platforms and hearing restoration system roadmaps used in tertiary care centers. Service line design teams often benchmark progress against wider cochlear implant system expansion and auditory prosthetics deployment themes when prioritizing long-range investment cycles.
This segmentation reflects differences in neural interface complexity, surgical pathway requirements, and post-implant rehabilitation intensity. It also mirrors procurement realities, since implant programs depend on specialist training pipelines, long-term service support, and structured patient monitoring systems.
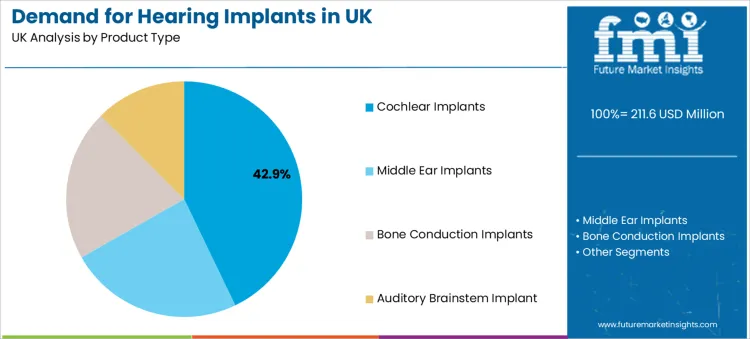
Cochlear implants hold a 42.9% share, making them the leading product type. This leadership reflects their established clinical validation for severe to profound sensorineural hearing loss and their compatibility with structured auditory rehabilitation programs.
Buyers value their predictable speech recognition outcomes, expanding processor upgrade pathways, and growing suitability for bilateral implantation protocols. Manufacturers benefit from long device life cycles and recurring revenue streams tied to processor upgrades and accessory ecosystems.
Middle ear implants and bone conduction implants are increasingly used for patients with conductive or mixed hearing loss who are unsuitable for conventional hearing aids. These systems support anatomical-specific rehabilitation strategies and are often selected where external device intolerance or chronic ear conditions limit amplification use.
Auditory brainstem implants remain specialized tools for patients with cochlear nerve absence or severe inner ear malformations, reinforcing their role as tertiary-level intervention technologies.
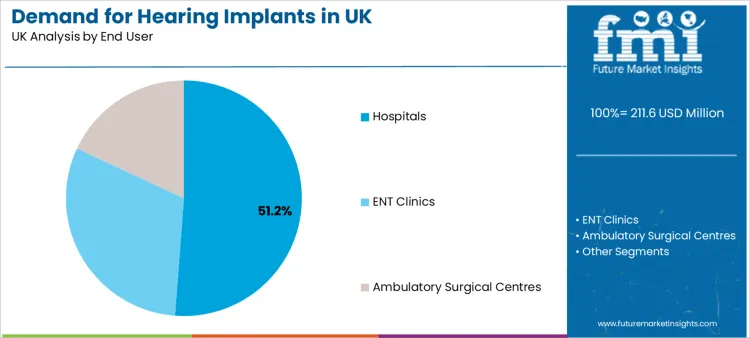
Hospitals account for 51.2% of total demand, positioning them as the dominant end user. Implantation procedures require surgical theaters, neurotology expertise, and post-operative audiological calibration, all of which concentrate within hospital settings.
Hospitals also act as long-term patient follow-up hubs, managing rehabilitation protocols, device upgrades, and outcome monitoring. ENT clinics and ambulatory surgical centers participate in growing implantation volumes, particularly for bone conduction systems and selected middle ear implant procedures.
Rising clinical acceptance, improved speech processor performance, and expanding pediatric implantation programs are central growth drivers. Public health screening initiatives and improved referral pathways are accelerating diagnosis-to-implantation timelines.
Another driver is system-level integration between implant hardware, rehabilitation software, and remote fitting tools, which improves long-term patient engagement and outcome consistency.
High upfront device and surgical costs can delay procurement decisions, particularly in budget-constrained hospital trusts. Surgical workforce capacity and specialist training availability also influence implantation throughput.
Patient hesitancy related to surgical intervention and long rehabilitation commitments can slow conversion rates among borderline candidates.
Opportunities rise in bilateral implantation expansion, adult late-onset hearing loss treatment, and processor upgrade programs. Suppliers that support clinician training, outcome documentation, and patient engagement tools can strengthen institutional loyalty.
There is also room for expanded service packaging that integrates surgery, rehabilitation, and long-term monitoring under unified care frameworks.
Regulatory changes affecting reimbursement policies or device approval timelines can alter procurement cycles. Competitive pressure from next-generation non-implant auditory technologies could also reshape patient decision pathways.
| Region | CAGR 2026 to 2036 |
|---|---|
| England | 8.5% |
| Scotland | 7.5% |
| Wales | 7.0% |
| Northern Ireland | 6.1% |
England grows at 8.5%, supported by the dense concentration of tertiary ENT and neurotology centers, structured pediatric hearing screening coverage, and higher overall procedural throughput. Major teaching hospitals and regional implant hubs act as referral anchors, drawing patient volumes from surrounding counties. This centralization allows implant programs to operate at scale, improving surgeon proficiency, rehabilitation consistency, and long-term outcome tracking.
Procurement teams in England benefit from established reimbursement pathways and predictable commissioning volumes, which support long-term supply contracts and processor upgrade planning. The presence of advanced audiology networks enables faster diagnosis-to-surgery conversion and strengthens follow-up adherence, reinforcing repeat procedural momentum. Regional planners often align service expansion with broader cochlear device deployment frameworks used across high-volume neurotology programs.
Scotland advances at 7.5%, reflecting centralized implant centers serving wide geographic catchment areas. Public health support for hearing restoration interventions remains consistent, and referral protocols emphasize early intervention for pediatric and progressive adult hearing loss.
Climatic exposure patterns and higher prevalence of otitis media complications in certain regions contribute to sustained diagnostic volumes. Buyers prioritize long-term service reliability, remote fitting capability, and stable processor upgrade access due to longer travel distances for follow-up care. These factors strengthen preference for suppliers with strong post-implant service infrastructure.
Wales increases at 7.0%, driven by improved diagnostic coverage, expansion of regional ENT surgical capacity, and growing clinician confidence in implantable auditory technologies. Investments in audiology staffing and post-surgical rehabilitation resources are shortening waiting lists and supporting higher annual implantation volumes.
Procurement teams emphasize predictable delivery schedules, standardized rehabilitation workflows, and training support for local clinicians. Adoption is strongest where implant programs are integrated into broader hearing restoration service lines linked to community audiology networks and referral coordination platforms.
Northern Ireland grows at 6.1%, shaped by a smaller clinical base and more selective patient referral pathways. Implantation volumes remain concentrated in a limited number of specialist centers, and adoption rises where surgical expertise and post-implant rehabilitation capacity align.
Buyers place weight on system durability, long-term processor availability, and training continuity due to tighter clinical staffing pools. Growth is incremental, supported by pediatric implantation programs and gradual expansion of adult candidacy criteria. Service planners often benchmark progress against broader auditory prosthetics deployment themes used in smaller regional health systems.
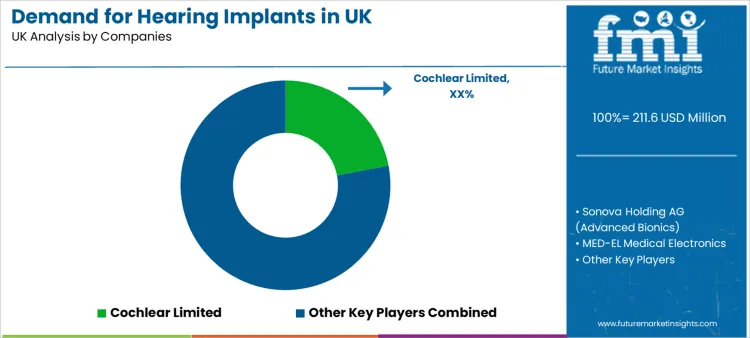
Competition is shaped by clinical performance outcomes, processor upgrade roadmaps, rehabilitation integration, and long-term service reliability. Buyers evaluate suppliers based on speech recognition results, device longevity, surgical compatibility, and availability of clinician training support.
Cochlear Limited competes through established cochlear implant platforms and global clinical validation. Sonova Holding AG (Advanced Bionics) competes through processor innovation and integrated audiology ecosystem alignment. MED-EL Medical Electronics competes through electrode design diversity and anatomical adaptability. Oticon Medical (Demant A/S) competes through system compatibility and auditory rehabilitation tools. WS Audiology (Signia, Widex) competes through auditory processing algorithms and audiology service integration depth.
Stakeholders balancing implant technology choices often align decisions with adjacent themes tied to cochlear device systems and broader hearing restoration platforms used across neurotology programs.
| Items | Values |
|---|---|
| Quantitative Units | USD Million |
| Product Type | Cochlear Implants; Middle Ear Implants; Bone Conduction Implants; Auditory Brainstem Implant |
| End User | Hospitals; ENT Clinics; Ambulatory Surgical Centres |
| Regions Covered | England; Scotland; Wales; Northern Ireland |
| Key Companies Profiled | Cochlear Limited; Sonova Holding AG (Advanced Bionics); MED-EL Medical Electronics; Oticon Medical (Demant A/S); WS Audiology (Signia, Widex) |
How big is the demand for hearing implants in uk in 2026?
The demand for hearing implants in uk is estimated to be valued at USD 211.6 million in 2026.
What will be the size of hearing implants in uk in 2036?
The market size for the hearing implants in uk is projected to reach USD 443.7 million by 2036.
How much will be the demand for hearing implants in uk growth between 2026 and 2036?
The demand for hearing implants in uk is expected to grow at a 7.7% CAGR between 2026 and 2036.
What are the key product types in the hearing implants in uk?
The key product types in hearing implants in uk are cochlear implants, middle ear implants, bone conduction implants and auditory brainstem implant.
Which end user segment is expected to contribute significant share in the hearing implants in uk in 2026?
In terms of end user, hospitals segment is expected to command 51.2% share in the hearing implants in uk in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.