Implantable Neurostimulation Device EMI and Safety Test Systems Market Size, Market Forecast and Outlook By FMI
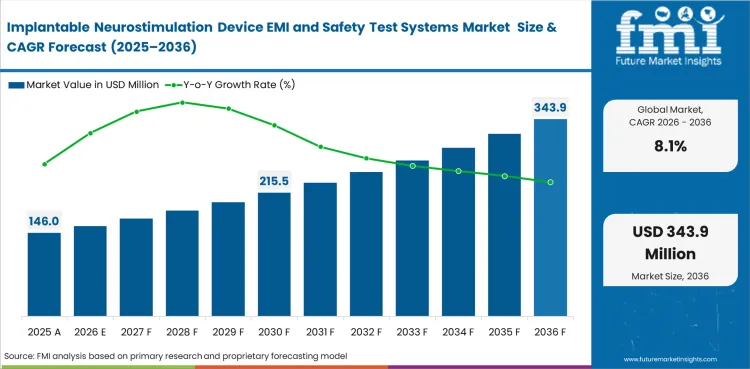
The implantable neurostimulation device EMI and safety test systems market was valued at USD 135.1 million in 2025. It is expected to reach USD 146.0 million by the end of 2026 and expand at a CAGR of 8.1% from 2026 to 2036 to reach USD 318.1 million by 2036. EMI and EMC systems are projected to lead with a 34.0% share of test focus demand in 2026, while spinal cord device programs are set to account for 38.0% of device type demand.
Summary of the Implantable Neurostimulation Device EMI and Safety Test Systems Market
- Demand and Growth Drivers
- Higher verification spending is expected as implant developers add rechargeability, sensing, telemetry, and MRI-related claims to neurostimulation platforms.
- Repeat testing demand is likely to stay firm as label updates and design modifications keep creating EMI and documentation work.
- Product and Segment View
- EMI and EMC systems are likely to lead the test focus segment, since interference screening stays central to active implant qualification and later design change work. Spinal cord device programs are projected to lead device-type demand.
- Integrated benches should continue to attract steady demand as developers look for one workflow that combines monitoring and protocol control. Related progress in smart implant telemetry and reliability test equipment points to stronger demand for coordinated validation setups across implant programs.
- Geography and Competitive Outlook
- China is expected to stay one of the faster-growing markets, supported by local device development and expanding regulatory activity. India is also projected to post strong growth as domestic compliance capability improves.
- Competition is likely to stay centered on technical depth rather than simple equipment pricing. Intertek, TÜV SÜD, UL Solutions, Element, SGS, Eurofins, and Rohde & Schwarz are positioned around accredited testing and service support. Adjacent momentum in neuromodulation closed-loop brain-computer interfaces supports longer-term demand for more advanced implant validation programs.
- Analyst Opinion
- , Principal Consultant at FMI, says, “Companies that can support implant developers with accredited EMI, MRI, and electrical safety validation are likely to strengthen their position over the forecast period, as neurostimulation platforms move toward more complex sensing, telemetry, and software-controlled architectures.”
- Implantable Neurostimulation Device EMI and Safety Test Systems Market Value Analysis
- The market is moving from a narrow compliance equipment niche into a more specialized validation category as implant developers spend more on EMI, MRI, telemetry, and electrical safety workflows.
- Demand is being supported by wider development of MRI-compatible implant platforms, and the rise of MRI-safe neurostimulation systems is increasing the need for higher-specification safety and compatibility verification.
- The market is benefiting from continued development of implant hardware, since progress in the implantable neurostimulator electrode market adds pressure for more detailed qualification across active neurostimulation programs.
- Demand is further supported by the shift toward adaptive and software-linked implant systems, while growth in neuromodulation closed-loop brain-computer interfaces points to a broader need for repeatable and more advanced validation setups over the forecast period.
Implantable Neurostimulation Device EMI and Safety Test Systems Market Definition
This market covers capital equipment, configured test platforms, validation rigs, measurement instruments, chamber based setups, software environments, and associated services used to assess implantable neurostimulation devices for electromagnetic compatibility, electrical safety, MRI related interactions, battery behavior under fault conditions, and wireless or telemetry stability.
Implantable Neurostimulation Device EMI and Safety Test Systems Market Inclusions
Included within scope are EMI and EMC benches, immunity and emissions generators, coupling and monitoring accessories, implant simulation fixtures, MRI phantom based test arrangements, leakage and dielectric safety analyzers, battery stress setups, environmental supports that are part of safety validation, data capture software used during regulated test execution.
Implantable Neurostimulation Device EMI and Safety Test Systems Market Exclusions
Excluded from scope are the implantable neurostimulation devices themselves, standard surgical tools, programmer handsets sold for routine therapy management, hospital diagnostic imaging systems, generic laboratory equipment without neurostimulation specific validation use, and broad outsourced clinical research services.
Implantable Neurostimulation Device EMI and Safety Test Systems Market Research Methodology
- Primary Research: Assessment combined official regulatory records, government and foundation health statistics, standards recognition databases, and supplier capability evidence to map demand drivers and likely procurement behavior.
- Desk Research: Desk review covered FDA PMA pages, WHO and national health datasets, national reimbursement or tariff sources, and company or regulator development announcements issued after January 2024.
- Market Sizing and Forecasting: Values were modelled using program count assumptions, expected validation spend per major platform, outsourcing intensity, and the effect of MRI, telemetry, and EMC complexity on average system revenue.
- Data Validation and Update Cycle: Forecast assumptions were cross checked against device approval trends, burden of neurological disease, country healthcare demand signals, and competitive activity across accredited testing and instrumentation suppliers.
Why is the Implantable Neurostimulation Device EMI and Safety Test Systems Market Growing?
- Neurostimulation platforms now carry more software, sensing, rechargeability, and wireless features that widen validation scope before release.
- MRI conditional claims and label updates keep creating repeat safety testing demand long after a core implant platform is first approved.
- Growing chronic pain and neurological disease burdens enlarge the addressable base for implant therapies and therefore for supporting qualification systems.
Disease burden remains a broad demand driver for this category. CDC data show that 24.3% of U.S. adults experienced chronic pain in 2023. WHO states that around 50 million people worldwide have epilepsy, while the Parkinson’s Foundation estimates that more than 10 million people worldwide are living with Parkinson’s disease. These disease populations support continued development activity across neuromodulation and related device programs.
Market Segmentation Analysis
- EMI and EMC is expected to account for 34.0% of the market in 2026, reflecting the central role of interference testing in active implant validation.
- Spinal cord device programs are projected to lead demand by device type with a 38.0% share in 2026.
- OEMs are expected to remain the largest end user group with a 42.0% share, as manufacturers continue to keep a meaningful part of validation work in-house before shifting selected studies to outside labs.
- Integrated benches are likely to hold 31.0% of the system format segment in 2026 while Direct sales are projected to lead the sales channel segment with a 58.0% share.
The market is segmented by test focus into EMI and EMC, Electrical Safety, MRI Safety, Battery Reliability, and Telemetry Validation. By device type, the market includes Spinal Cord, Deep Brain, Sacral Nerve, Vagus Nerve, and Peripheral Nerve systems. By end user, it covers OEMs, Contract Labs, Design Firms, Academic Centers, and Hospital Research. By system format, the market is divided into Integrated Benches, Modular Racks, Shielded Chambers, Software Suites, and MRI Phantoms. By sales channel, it includes Direct Sales, Channel Partners, Turnkey Projects, Validation Services, and Service Contracts.
Insights into the test focus segment
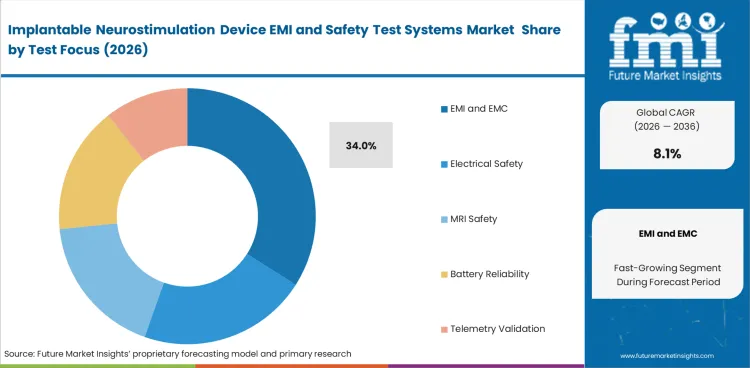
- EMI and EMC is expected to account for 34.0% share in 2026. Every active implantable neurostimulation program must prove stable function in the presence of emissions from chargers and hospital equipment, including MRI related environments or telemetry workflows.
- MRI safety and telemetry validation are growing faster in value per project, although they come behind core EMI work in installed system count.
Insights into the device type segment
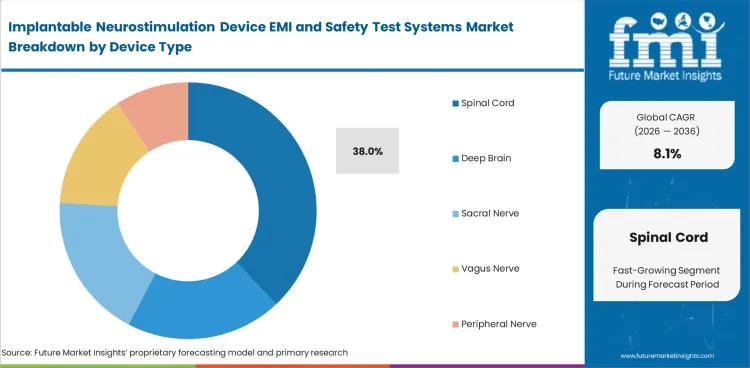
- In 2026 spinal cord device programs are projected to contribute 38.0% of total market share. Spinal cord stimulation sits at the center of this niche because pain management is the largest implantable neurostimulation application.
- Spinal cord stimulators shows how scrutiny on effectiveness and use conditions continues after commercialization.
Insights into the end user segment
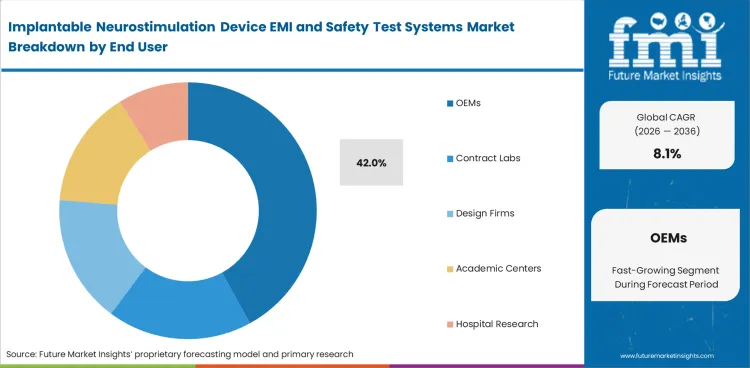
- OEMs are anticipated to represent 42.0% of the market in 2026. Internal engineering and quality groups usually conduct early risk screening and portions of formal verification before selected studies are repeated or completed at an accredited outside laboratory.
- Academic centers and hospital research groups participate mainly in translational or investigator programs, so their system demand is smaller and more grant linked.
Insights into the system format segment
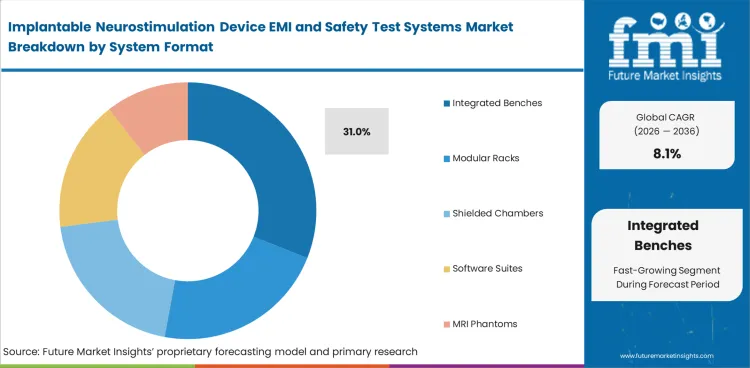
- Integrated benches are likely to secure 31.0% share in 2026. Developers prefer a compact setup that combines data capture and scripted execution since regulated implant programs generate large documentation loads and repeated protocol runs.
- Modular racks hold a strong place in larger OEM laboratories that need flexibility across product lines. Shielded chambers account for higher-value systems tied to full compliance workflows or outsourced service operations.
Insights into the sales channel segment
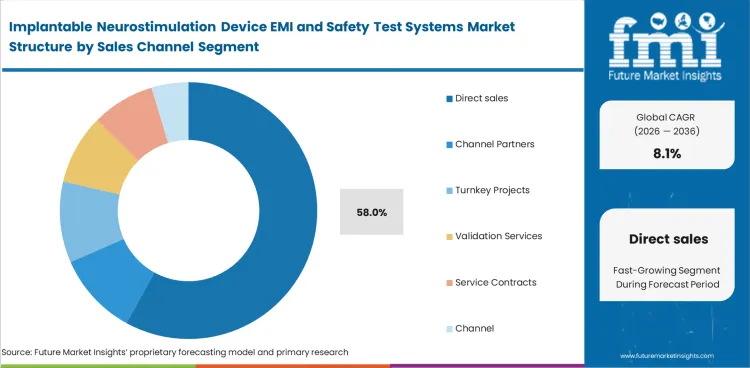
- Direct sales is set to make up 58.0% of the market in 2026. Sales staff and application engineers are important because implant developers usually select these systems through direct specification and review, not a standard catalogue.
- Channel partners work better in smaller geographies and in less customized instrument packages. Turnkey projects are chosen for large site expansions or greenfield validation labs.
Implantable Neurostimulation Device EMI and Safety Test Systems Market Drivers, Restraints, and Opportunities
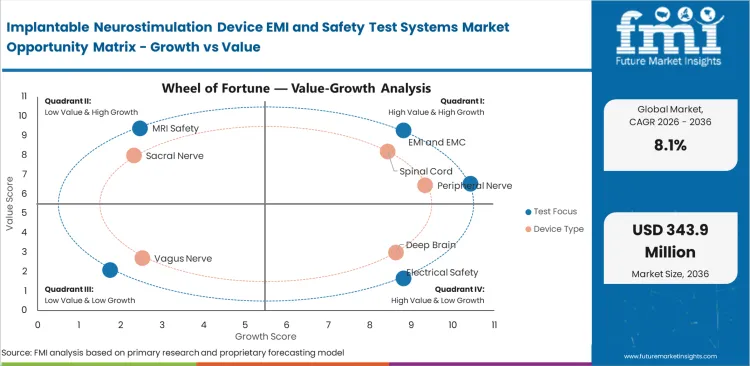
- MRI labeling work and connected implant features are raising the amount of verification carried by each program.
- Customer concentration remains high because only a small number of neurostimulation developers buy these systems in volume.
- Customers are shifting toward combined hardware and software platforms that reduce reconfiguration between EMC, safety, and telemetry tasks.
Market direction depends more on validation content attached to each implantable program than raw unit volumes. A single implant refresh can involve changes in firmware, charge behavior, or MRI conditions that require fresh protocol work even when therapy indications stay unchanged. This creates a favorable revenue mix for suppliers with flexible benches and strong logging software. Narrow customer concentration and long purchase cycles keep the business disciplined and technically selective.
Testing and validation needs
Recent FDA records show how repeat changes keep the installed base of test systems active. Boston Scientific obtained a late-2025 labeling update that supported MRI scan eligibility for mixed system use, while Medtronic received several 2025 to 2026 supplements across Altaviva and separate spinal cord stimulation platforms including Vanta, Intellis, and Inceptiv. Each type of change drives fresh work across emissions, immunity, safety, compatibility, and risk documentation.
Certification burden
Compliance requirements can slow the sales cycle. Active implantable devices face a heavier evidence burden than many external electronics because interference events can affect therapy delivery, sensing accuracy, charging, or MRI access. FDA documentation for adaptive DBS already highlights EMI related risks tied to sensing function, which means the test scope must reflect both device safety and therapeutic performance.
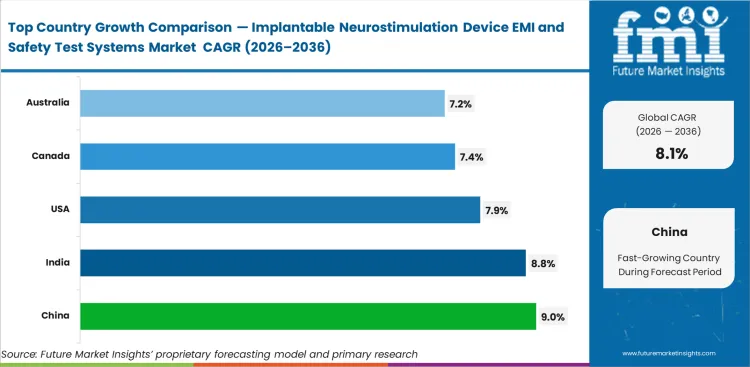
Analysis of Implantable Neurostimulation Device EMI and Safety Test Systems Market by Key Countries
.webp)
| Country |
CAGR |
| China |
9.0% |
| India |
8.8% |
| United States |
7.9% |
| Canada |
7.4% |
| Australia |
7.2% |
Source: FMI analysis based on primary research and proprietary forecasting model
Implantable Neurostimulation Device EMI and Safety Test Systems Market CAGR Analysis By Country
- China leads the country outlook with a projected 9.0% CAGR through 2036.
- India follows at 8.8%, with growth backed by improving domestic compliance capability and rising interest in local testing capacity.
- The United States is expected to expand at 7.9% CAGR, reflecting its large OEM base, repeat PMA supplement activity, and high-value verification budgets.
- Canada is projected to grow at 7.4%, while Australia is forecast at 7.2%, with both markets benefiting from specialist neuro care pathways and steady demand for advanced validation work.
- The United Kingdom is set to post 6.8% CAGR, and Germany is projected at 6.6%, showing a more measured pattern shaped by mature compliance infrastructure and disciplined capital spending.
- Across the full market, the global industry is expected to advance at a CAGR of 8.1% from 2026 to 2036.
Demand Outlook for Implantable Neurostimulation Device EMI and Safety Test Systems Market in China
China is forecast to register the fastest growth, with a CAGR of 9.0% through 2036. Demand is driven by expanding domestic device development and wider regulatory activity around implant stability under EMI and MRI-related conditions. Support from implantable neurostimulator electrode market development is also adding weight to more detailed qualification requirements across local programs.
- Domestic development activity is widening the testing base.
- Regulatory progress is improving the case for local validation spending.
- MRI and telemetry verification remain strong areas of demand.
Demand Outlook for Implantable Neurostimulation Device EMI and Safety Test Systems Market in India
India is expected to expand at a CAGR of 8.8% through 2036. Growth is supported by improving medtech lab capability, rising interest in cost-conscious modular systems, and gradual strengthening of domestic compliance infrastructure. Demand connected to neuromodulation closed-loop brain-computer interfaces points to broader long-term need for advanced implant testing and repeat validation workflows.
- Cost discipline supports modular and service-backed systems.
- OEM outsourcing and local lab build-out can advance together.
- Compliance expectations are becoming more structured.
Demand Outlook for Implantable Neurostimulation Device EMI and Safety Test Systems Market in United States
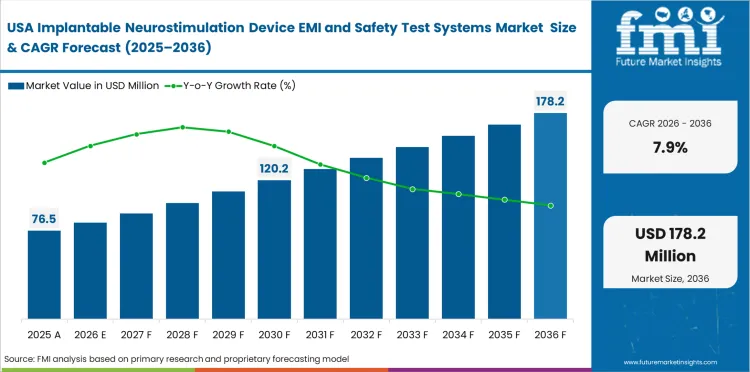
The U.S. market is projected to expand at a CAGR of 7.9% through 2036. Demand stays firm due to a high volume of FDA-led submission work and repeat supplement activity. Growth in preclinical medical device testing services supports testing demand by strengthening outsourced and in-house verification capacity across regulated implant programs.
- Repeat PMA supplements keep validation demand active.
- OEMs continue to invest in mixed in-house and outsourced testing models.
- Software traceability and documentation remain key buying factors.
Demand Outlook for Implantable Neurostimulation Device EMI and Safety Test Systems Market in Canada
Canada is projected to grow at a CAGR of 7.4% through 2036. Demand comes from specialist neurology care pathways and cross-border technology flow with the United States. Expansion in preclinical medical device testing services strengthens the wider testing base that supports regulated device qualification work in the country.
- Cross-border sourcing influences vendor selection.
- Specialist centers support advanced neurology-related testing demand.
- Absolute market volume remains moderate despite healthy technical demand.
Demand Outlook for Implantable Neurostimulation Device EMI and Safety Test Systems Market in Australia
Australia is expected to record a CAGR of 7.2% through 2036. Demand is supported by visible use of implant therapies and payer scrutiny around evidence quality. Broader progress in MRI-safe neurostimulation systems is helping sustain demand for more rigorous compatibility and safety verification in the market.
- Payer review keeps evidence standards visible.
- Spinal cord therapy use supports validation demand.
- Compact specialist networks favor service-led suppliers.
Demand Outlook for Implantable Neurostimulation Device EMI and Safety Test Systems Market in United Kingdom
The U.K. market is expected to rise at a CAGR of 6.8% through 2036. Demand depends on a mature compliance environment and steady use of specialist external testing services. Activity linked to MRI-safe neurostimulation systems is supporting demand for higher-specification EMI, safety, and compatibility verification across implant development programs.
- Hospital budget discipline favors selective capital purchases.
- Outsourced testing remains important for recognized reports and documentation.
- Mature implant programs support steady replacement and upgrade demand.
Demand Outlook for Implantable Neurostimulation Device EMI and Safety Test Systems Market in Germany

Germany is projected to grow at a CAGR of 6.6% through 2036. The market benefits from a strong engineering base, disciplined qualification practices, and a preference for proven testing suppliers with reliable documentation support. Demand tied to smart implant telemetry and reliability test equipment reflects the broader push toward more coordinated implant validation systems in the country.
- Engineering depth supports demand for premium systems.
- Replacement demand is more visible than greenfield build-outs.
- Buyers prefer established suppliers with strong application support.
Competitive landscape of Implantable Neurostimulation Device EMI and Safety Test Systems Market
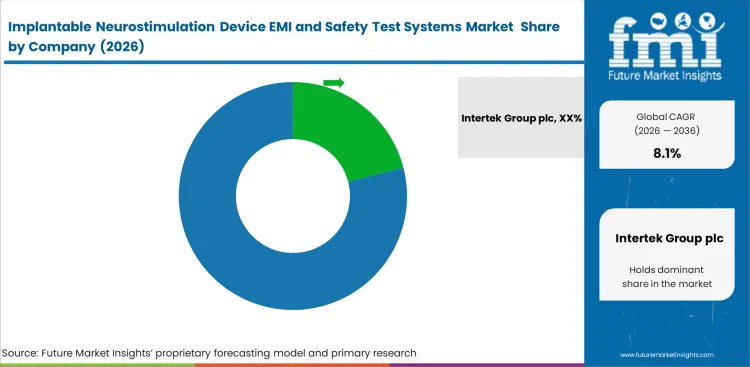
- Competition is moderately concentrated because large accredited labs and global instrument suppliers hold most complex projects.
- Leading companies compete on accreditation depth, implant specific know how, documentation quality, and the ability to combine hardware with services.
- Specialists still find room where MRI, telemetry, or custom fixture work is too narrow for broad line providers to prioritize.
Competition centers on technical credibility more than on simple list price. Manufacturers need proof that a supplier can handle active implantable workflows, understand recognized standards, and provide data formats that support internal design control or outside submissions. Intertek, TÜV SÜD, UL Solutions, Element, SGS, Eurofins, and Rohde and Schwarz are near the core of the field because they offer some mix of accredited testing, global reach, instrumentation depth, or medical device application support. Entry is difficult since new suppliers must build both technical infrastructure and trust with regulatory and quality teams that tend to be conservative.
Independent laboratories gain advantage from recognized reports and the ability to absorb overflow from OEM programs close to submission. Instrument vendors gain advantage where manufacturers want more work in house and need configurable generators, receivers, chambers, and software without depending fully on outside labs. The best positioned firms can bridge both needs through consulting, protocol design, equipment supply, and recurring service. This blend is important because customer relationships often begin with a single program and later expand into calibration, upgrades, or support for later supplements.
Major Industry Players
- Intertek Group plc, TÜV SÜD, and UL Solutions fit this tier because they combine broad regulatory coverage, recognized medical device test capability and the ability to handle complex EMC and safety workflows across multiple geographies and submission pathways.
- Element Materials Technology, SGS SA, and Eurofins Scientific operate with strong laboratory depth and broad life sciences reach.
- Rohde & Schwarz, HLL Lifecare EMC laboratory, and specialist fixture or software providers gain relevance where OEMs are building more internal capability and need modular instruments, local technical support, or narrow implant specific validation tools rather than a full outsourced compliance package.
Competitive Benchmarking: Implantable Neurostimulation Device EMI and Safety Test Systems Market
| Company |
EMC Depth |
Implant Application Support |
Service Breadth |
Geographic Footprint |
| Intertek Group plc |
High |
High |
Strong |
Global |
| TÜV SÜD |
High |
High |
Strong |
Global |
| UL Solutions |
High |
Medium |
Strong |
Global |
| Element Materials Technology |
High |
Medium |
Moderate |
Global |
| SGS SA |
Medium |
Medium |
Moderate |
Global |
| Eurofins Scientific |
Medium |
Medium |
Moderate |
Multi-region |
| Rohde & Schwarz |
High |
Low |
Moderate |
Global |
| AMETEK CTS |
Medium |
Low |
Moderate |
Multi-region |
| ETS-Lindgren |
Medium |
Low |
Low |
Multi-region |
| HLL Lifecare EMC Lab |
Low |
Medium |
Low |
India |
Source: Future Market Insights competitive analysis, 2026.
Key Developments in Implantable Neurostimulation Device EMI and Safety Test Systems Market
- In November 2025, Intertek recorded an acquisition event as the company acquired Suplilab, a Costa Rica-based provider of food safety and medical devices testing services.
- In December 2025, Element Materials Technology recorded an acquisition event as the company announced the acquisition of CETECOM Inc. in the United States.
Key Players in the Implantable Neurostimulation Device EMI and Safety Test Systems Market
Major global players
- Intertek Group plc
- TÜV SÜD
- UL Solutions
- Element Materials Technology
- SGS SA
Key emerging players/startups
- AMETEK CTS
- ETS-Lindgren
- HLL Lifecare EMC Lab
- IAQ-EMF Consulting
Scope of the Report
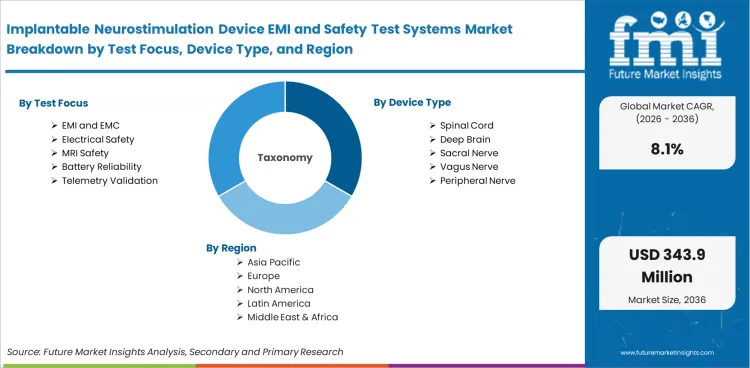
| Metric |
Value |
| Quantitative Units |
USD 135.1 million to USD 318.1 million, at a CAGR of 8.1% from 2026 to 2036 |
| Market Definition |
Capital systems, configured benches, software, and related services used to verify implantable neurostimulation devices against EMI, EMC, MRI, and electrical safety requirements. |
| Regions Covered |
North America, Europe, Asia Pacific |
| Countries Covered |
China, India, United States, Canada, Australia, United Kingdom, Germany |
| Key Companies Profiled |
Intertek, TÜV SÜD, UL Solutions, Element, SGS, Eurofins, Rohde & Schwarz |
| Forecast Period |
2026 to 2036 |
| Approach |
Analyst model built from regulatory records, disease burden indicators, standards recognition, and supplier capability evidence. |
Implantable Neurostimulation Device EMI and Safety Test Systems Market by Segments
Implantable Neurostimulation Device EMI and Safety Test Systems Market segmented as Test Focus
- EMI and EMC
- Electrical Safety
- MRI Safety
- Battery Reliability
- Telemetry Validation
Implantable Neurostimulation Device EMI and Safety Test Systems Market segmented as Device Type
- Spinal Cord
- Deep Brain
- Sacral Nerve
- Vagus Nerve
- Peripheral Nerve
Implantable Neurostimulation Device EMI and Safety Test Systems Market segmented as End User
- OEMs
- Contract Labs
- Design Firms
- Academic Centers
- Hospital Research
Implantable Neurostimulation Device EMI and Safety Test Systems Market segmented as System Format
- Integrated Benches
- Modular Racks
- Shielded Chambers
- Software Suites
- MRI Phantoms
Implantable Neurostimulation Device EMI and Safety Test Systems Market segmented as Sales Channel
- Direct Sales
- Channel Partners
- Turnkey Projects
- Validation Services
- Service Contracts
Implantable Neurostimulation Device EMI and Safety Test Systems Market by Region and Country
- North America
- Europe
- Asia Pacific
Bibliography
- Intertek Group plc. (2025, November 3). Intertek expands ATIC footprint in Central America with acquisition of leading Costa Rican testing business Suplilab.
- Element Materials Technology. (2025, December 8). Element announces strategic acquisition of CETECOM Inc.
- Centers for Disease Control and Prevention. (2024, November). Chronic pain and high-impact chronic pain in U.S. adults, 2023.
- Digital NHS. (2026, January 27). Health Survey for England 2024: Chronic pain.
- World Health Organization. (2024, February 7). Epilepsy.
This bibliography is provided for reader reference.
This Report Addresses
- Market sizing and forecast for implantable neurostimulation device EMI and safety test systems from 2026 to 2036.
- Demand structure across test focus, device type, end user, system format, and sales channel.
- Country level outlook across China, India, the United States, Canada, Australia, the United Kingdom, and Germany.
- Competitive assessment of accredited labs, instrumentation vendors, and specialist validation suppliers.
- Regulatory and standards factors that influence EMC, safety, MRI, and telemetry verification demand.
- Investment themes around integrated benches, modular racks, chamber infrastructure, and validation software.
- Value chain coverage from device development activity to outsourced compliance and post change support.
- Manufacturers questions around pricing discipline, replacement timing, and service intensity in a narrow customer base.
Frequently Asked Questions
What is the estimated market size in 2026?
The market is estimated at USD 146.0 million in 2026 for implantable neurostimulation device EMI and safety test systems.
What value is projected for 2036?
Forecast value is estimated at USD 318.1 million by 2036 as validation scope and software content continue to increase.
What CAGR is expected through 2036?
The market is projected to expand at 8.1% CAGR between 2026 and 2036 under the base case forecast.
Which segment leads test focus?
EMI and EMC systems lead test focus with an estimated 34.0% share in 2026 due to mandatory interference qualification work.
Which device area leads demand?
Spinal cord stimulation programs lead demand with an estimated 38.0% share of device type related spending in 2026.
Which sales channel is strongest?
Direct sales lead the channel mix with 58.0% share because buyers usually need configured systems and application support.







.webp)





