The neuromodulation closed-loop brain-computer interfaces market is segmented by Device modality (Implantable responsive neurostimulation systems, Adaptive deep brain stimulation systems, Endovascular / minimally invasive implantable BCI systems, High-bandwidth cortical interface systems, Research-only closed-loop neural interface platforms), Signal and control architecture (Sensing + adaptive stimulation closed-loop systems, Decoding-only implantable BCI systems, Biomarker-triggered stimulation systems, AI-assisted neural decoding and control systems, Cloud-linked neurodata and software layers), Indication (Drug-resistant epilepsy, Parkinson’s disease and movement disorders, Severe motor impairment / communication restoration, Stroke neurorehabilitation, Refractory depression and psychiatric applications), End user (Tertiary neurology and neurosurgery centers, Academic medical centers, Specialty epilepsy centers, Neurorehabilitation institutes, Translational research hospitals), Commercialization stage (Commercial therapy platforms, Early feasibility systems, Clinical trial platforms, Preclinical development systems, Software / algorithm support revenue), and Region. Forecast for 2026 to 2036.
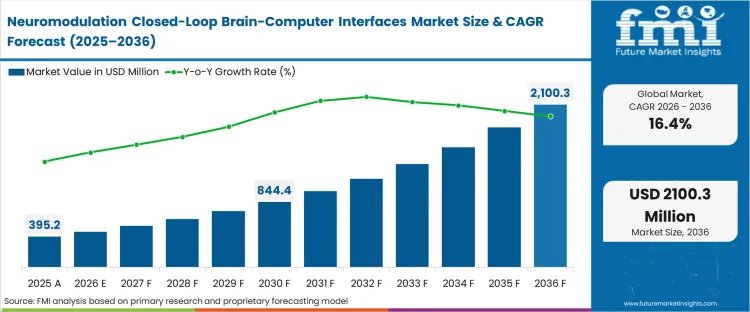
The neuromodulation closed-loop brain-computer interfaces market was valued at USD 390.0 million in 2025. The industry is set to cross USD 460.0 million in 2026 at a CAGR of 16.40% during the forecast period. Ongoing funding fuels the invasive BCI market size to USD 2,100 million through 2036 as real-time adaptive neurostimulation switches from clinical observation tools to real-time therapeutic triggers capable of autonomous modulation.
Neurology department heads and surgical directors are no longer evaluating hardware durability in isolation; they are being forced to assess longitudinal algorithm upgradeability. The decision leads to a transition from purchasing a static stimulation device to selecting a proprietary data ecosystem that can interpret biomarker telemetry over a patient's lifetime. Hospitals that lock into closed architectures risk stranding their surgical patients on legacy brain computer interface platforms that cannot integrate next-generation predictive AI updates. The cost of replacing an implanted hardware suite due to software obsolescence alters procurement math.
Before adoption scales beyond tertiary academic centers, the industry must clear a specific reimbursement threshold: the creation of discrete billing codes for algorithm-driven therapy adjustment. Currently, clinical programming sessions require intensive epileptologist or neurologist oversight. Once autonomous closed-loop neurostimulation market protocols achieve distinct payer coverage, the staffing bottleneck limiting patient throughput will dissolve, accelerating commercial volume.
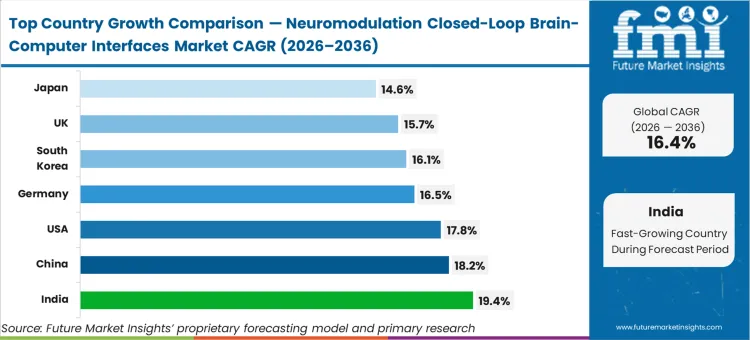
India and China are anticipated to expand at 19.4% and 18.2% respectively, driven by rapid capacity additions in neurosurgical infrastructure from a smaller installed base. The United States is poised to track at 17.8%, maintaining its position as the primary commercialisation anchor due to dominant domestic revenue concentrations for early adaptive systems. South Korea is estimated to advance at 16.1%, while Germany and the United Kingdom are set to record 16.5% and 15.7% respectively. Japan follows at 14.6%. The divergence across these markets reflects the varying speeds at which national healthcare systems convert early feasibility clinical trial sites into broad commercial therapy centers.
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 460.0 million |
| Industry Value (2036) | USD 2,100 million |
| CAGR (2026-2036) | 16.40% |
The neuromodulation closed-loop BCI market encompasses active medical devices and associated software that continuously record neural activity, decode physiological biomarkers in real-time, and autonomously deliver targeted electrical stimulation to alter neurological function. Unlike traditional open-loop pacemakers, these sensing and stimulation neural interface systems form a bidirectional communication loop with the nervous system, adapting their therapeutic output dynamically based on the patient's immediate neurophysiological state.
Scope incorporates implantable responsive neurostimulation arrays, adaptive deep brain stimulation market hardware with sensing capabilities, endovascular brain-computer interface arrays, and high-density cortical interface implant market systems intended for therapeutic neuromodulation or severe motor impairment communication. Associated telemetry hardware, external patient programmers, physician control interfaces, and the recurring software licensing integral to brain computer interface implant functionality are included in the valuation.
The market explicitly excludes traditional open-loop deep brain stimulators, vagus nerve stimulators without concurrent biosignal recording, and purely diagnostic electroencephalography (EEG) mapping arrays. External devices are excluded when comparing an invasive vs non-invasive brain-computer interface because non-invasive options lack the high-fidelity, internal telemetry required for autonomous, microsecond-level closed-loop therapeutic adjustment. Furthermore, thought-controlled implants utilized strictly for non-medical consumer applications or cognitive enhancement fall outside the clinical scope of this analysis.

The reason implantable responsive neurostimulation systems hold 41.0% of this market is rooted in the physiological requirements of therapy triggering. External, non-invasive sensing cannot deliver the single-neuron or localised field potential resolution required to identify a pre-ictal state before clinical manifestation. According to FMI's estimates, functional neurosurgeons rely on these systems because they offer the only verified method for intervening at the microsecond timescale required to abort a focal seizure. The decision process for these buyers centers heavily on the longevity of the implantable power source and the density of the recording array. Hospitals that select systems with inferior battery chemistry force their patients into premature battery replacement surgeries, exposing the institution to increased surgical risk and patient dissatisfaction. By prioritizing high-density, low-power continuous recording, modern neurostimulation hardware reduces the necessity for invasive revision procedures.
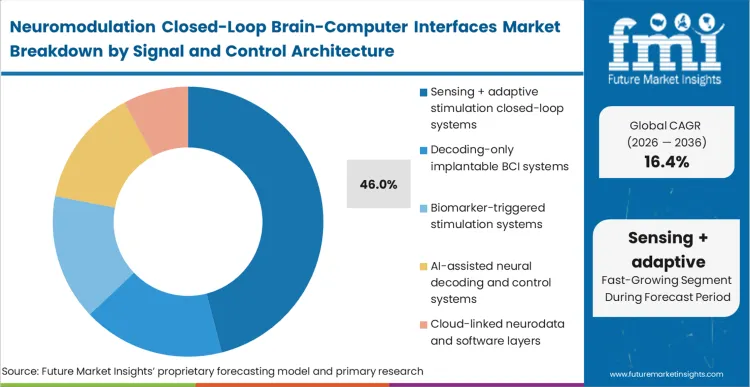
Sensing + adaptive stimulation closed-loop systems capture 46.0% share by changing what happens during the patient's daily life, switching therapy from a static prescription to a dynamic physiological response. Legacy continuous stimulation forces a compromise: deliver enough energy to suppress symptoms during peak severity, which inevitably causes dysarthria or battery drain during periods of low disease activity. Based on FMI's assessment, adaptive architectures solve this by throttling energy output based on real-time beta band activity or other specific neural markers. Movement disorder specialists mandate this architecture because it provides an objective, quantified measurement of disease state rather than relying on subjective patient diaries. If a clinical team delays transitioning to biomarker-triggered neurostimulation architectures, they commit their practice to high-frequency, manual programming adjustments that consume valuable outpatient clinic time.
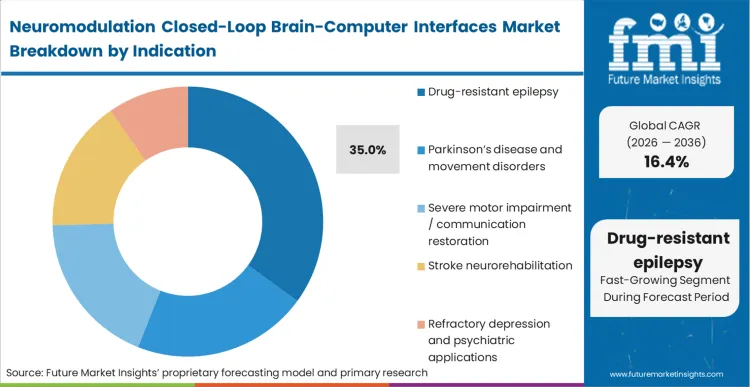
Drug-resistant epilepsy represents 35.0% of the market because it possesses the most established diagnostic pathway leading directly to a surgical intervention decision. When a patient fails two appropriately chosen and tolerated antiseizure medications, the clinical protocol definitively moves from pharmaceutical management to surgical evaluation. FMI analysts opine that this clear procedural trigger creates a predictable patient funnel that BCI for Parkinson’s symptom control and psychiatric indications have not yet fully replicated. Epileptologists utilize closed-loop systems not merely to stop seizures, but to map the epileptogenic zone chronically, gathering data that informs long-term care. Clinical centers that delay the integration of brain-computer interface for epilepsy therapies often lose referrals to comprehensive level 4 epilepsy centers that offer the complete spectrum of advanced interventions within the epilepsy device market.
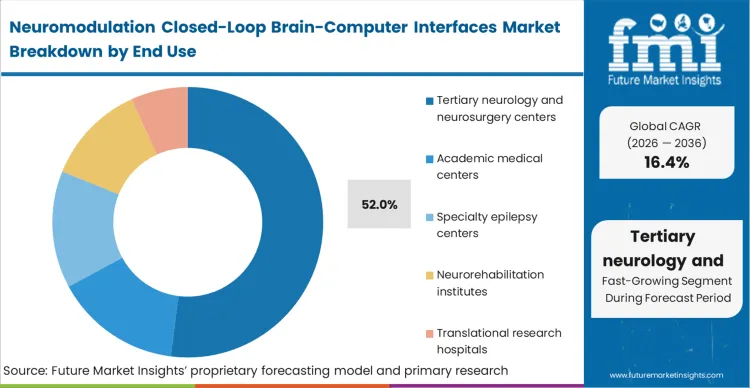
Tertiary neurology and neurosurgery centers hold 52.0% of the market because the deployment of closed-loop interfaces requires a multidisciplinary infrastructure that smaller hospitals cannot sustain. The placement of high-bandwidth cortical arrays or adaptive depth electrodes demands not only stereotactic surgical precision but also dedicated neurophysiologists capable of interpreting real-time data during the procedure. In FMI's view, the capital required to maintain functional neurosurgery suites, combined with the specialized clinical staff needed for post-operative algorithmic tuning, naturally concentrates adoption at the apex of the healthcare system. Community hospitals attempting to adopt these non-invasive brain stimulation system alternatives often lack the necessary follow-up infrastructure, leading to suboptimal patient outcomes. Institutions failing to centralize this expertise risk compromising their status as regional neurological hubs within the neurology device market.
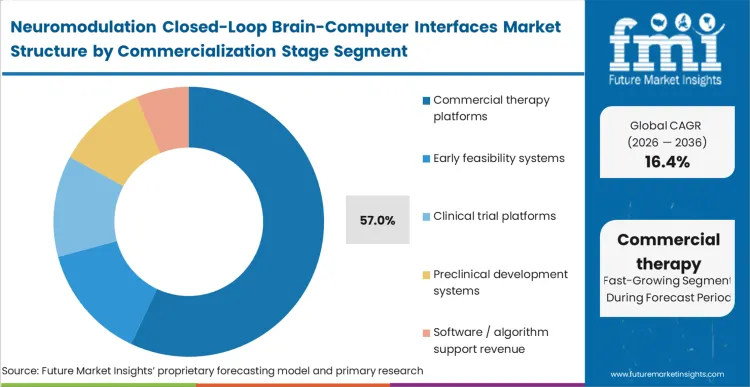
The dominance of commercial therapy platforms at 57.0% share underscores the transition of adaptive neurotechnology from experimental validation to standard-of-care implementation. As per FMI's projection, hospital procurement committees are prioritizing systems with established reimbursement codes and long-term safety profiles over early-stage, theoretical platforms. While early feasibility systems targeting BCI for paralysis communication garner significant attention, the reliable revenue streams are generated by platforms addressing prevalent conditions like Parkinson's and epilepsy through established surgical pathways. Vendors offering purely experimental platforms struggle to penetrate standard hospital purchasing cycles until they can demonstrate definitive health economic advantages over existing therapies. Delaying the transition from clinical trial status to full commercial deployment starves emerging manufacturers of the capital necessary to fund subsequent software iterations.
The accumulation of chronic neural data forces clinical algorithm developers and medical device manufacturers to deploy machine learning updates that refine stimulation parameters without requiring surgical hardware replacement. This pressure requires software engineering teams to design implantable platforms with significant excess processing capacity and secure, bidirectional telemetry. Hospitals investing in these systems are essentially purchasing a forward-compatible therapy platform, moving their capital expenditure from static hardware to dynamic, evolving algorithms. Institutions that fail to secure upgradable systems face the commercial penalty of performing costly, invasive revision surgeries to deliver next-generation therapeutic benefits to their patients.
The single biggest operational friction slowing adoption is the clinical workflow burden of reviewing and acting upon massive volumes of raw neural telemetry. The sheer density of data generated by continuous, high-bandwidth implantable neurostimulator electrode arrays overwhelms the standard 15-minute outpatient neurology consult. This is a barrier because physician reimbursement models are currently built around episodic evaluation, not continuous data monitoring. While manufacturers are introducing AI-assisted summary dashboards, these tools remain rudimentary and often lack the specific diagnostic validation required to entirely replace physician review, creating a persistent bottleneck in clinical scaling.
Opportunities in the Neuromodulation Closed-Loop Brain-Computer Interfaces Market
Regional evaluation places the Neuromodulation Closed-Loop Brain-Computer Interfaces market into four key areas, North America, Europe, Asia Pacific, and the rest of the world, encompassing more than 40 countries.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| India | 19.4% |
| China | 18.2% |
| United States | 17.8% |
| Germany | 16.5% |
| South Korea | 16.1% |
| United Kingdom | 15.7% |
| Japan | 14.6% |
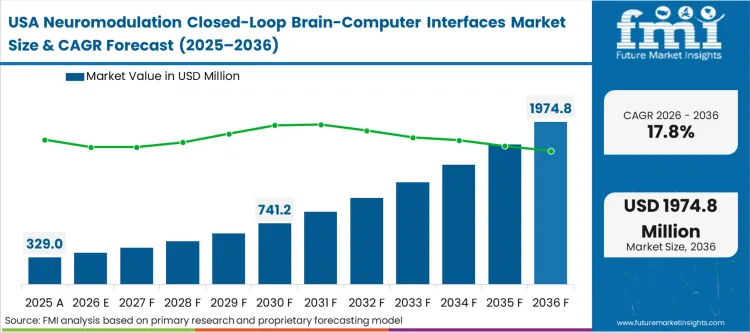
The regulatory environment in North America, specifically the established pathways for breakthrough device designation and premarket approval, defines the commercial trajectory of closed-loop systems. According to FMI's estimates, the presence of dominant domestic manufacturers ensures that the region serves as the primary launchpad for early adaptive technologies. The ecosystem is characterized by a strong alignment between venture capital funding, translational academic research, and clear, albeit complex, reimbursement frameworks. This alignment allows brain monitoring system innovations to transition from academic laboratories to commercial surgical suites more rapidly than in regions requiring extensive, parallel national health technology assessments.
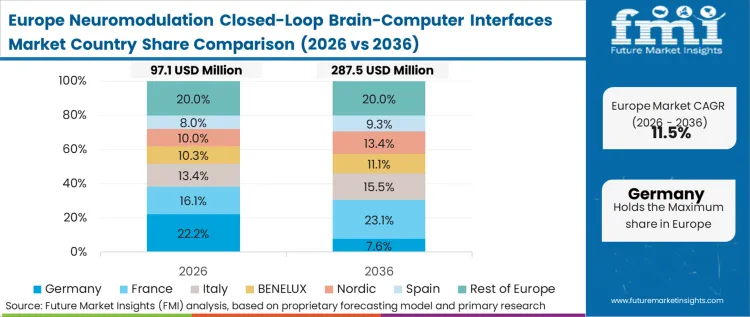
Cost structures and national health technology assessments dominate the adoption logic in the European market. Unlike systems that rapidly adopt new technologies based solely on clinical efficacy, European procurement bodies require robust health economic data proving long-term cost savings. As per FMI's projection, this forces neurotech manufacturers to design extensive post-market clinical follow-up studies to maintain their CE mark and secure national funding. The stringent requirements of the Medical Device Regulation (MDR) have slowed the introduction of novel neurotech devices, inadvertently extending the lifecycle of legacy systems while hospitals wait for next-generation platforms to navigate the certification process.
Infrastructure readiness and the rapid expansion of specialized neurosurgical capacity shape the adoption pattern across the Asia Pacific region. Historically reliant on imported technologies, the region is experiencing a surge in domestic investment aimed at establishing sovereign capabilities in neural engineering and advanced bioelectronics. In FMI's view, the vast patient populations suffering from neurological disorders outpace the available clinical specialist capacity, creating an acute need for highly autonomous systems within the neuroprosthetics market that require minimal ongoing physician intervention. The challenge lies in building the stereotactic surgical infrastructure necessary to safely deploy these complex implants outside of a few major metropolitan hubs.
FMI's report includes extensive analysis of emerging clinical trial hubs across Latin America and the Middle East, capturing 40 plus countries. The expansion of regulatory harmonization across these regions is gradually lowering the barrier for entry, allowing manufacturers to deploy mature commercial platforms without replicating extensive local clinical trials.
The concentrated nature of the closed-loop neuromodulation market stems from the immense capital requirements and specialized engineering expertise required to cross the regulatory threshold for Class III active implantable devices. Buyers, specifically hospital procurement committees and surgical directors, do not differentiate brain implant companies based on minor hardware specifications; they evaluate based on the maturity and clinical validation of the decoding algorithms. Medtronic and NeuroPace dominate because they possess the largest proprietary datasets of chronic neural recordings, giving them an insurmountable lead in training the machine learning models that dictate therapeutic efficacy.
For challenger implantable BCI manufacturers like Synchron or Precision Neuroscience to replicate this advantage, they must build capabilities in ultra-high-density recording and minimally invasive deployment. It is not sufficient to simply manufacture a functioning electrode array; a challenger must engineer a complete, secure data pipeline that handles the massive telemetry output of smart implants while meeting stringent cybersecurity standards. They must develop novel surgical delivery mechanisms to fundamentally alter the risk-benefit calculation for the implanting neurosurgeon evaluating the best implantable BCI companies.
As the market approaches 2036, the tension lies between large hospital networks seeking interoperable data standards and dominant closed-loop neurotechnology vendors fighting to maintain closed, proprietary ecosystems. Hospitals are increasingly resistant to vendor lock-in that traps patient data within specific proprietary portals, complicating longitudinal research and population health management. However, the market is likely to remain highly concentrated, as the immense regulatory burden of proving safety for next-generation, AI-driven autonomous stimulation will force smaller brain-computer interface technology partners to seek acquisition by established incumbents rather than attempting independent commercialization as standalone closed-loop BCI suppliers.
| Metric | Value |
|---|---|
| Quantitative Units | USD 460.0 million to USD 2,100 million, at a CAGR of 16.40% |
| Market Definition | This market encompasses active medical devices and associated software that continuously record neural activity, decode physiological biomarkers in real-time, and autonomously deliver targeted electrical stimulation to alter neurological function. |
| Device Modality Segmentation | Implantable responsive neurostimulation systems, Adaptive deep brain stimulation systems, Endovascular / minimally invasive implantable BCI systems, High-bandwidth cortical interface systems, Research-only closed-loop neural interface platforms |
| Signal and Control Architecture Segmentation | Sensing + adaptive stimulation closed-loop systems, Decoding-only implantable BCI systems, Biomarker-triggered stimulation systems, AI-assisted neural decoding and control systems, Cloud-linked neurodata and software layers |
| Indication Segmentation | Drug-resistant epilepsy, Parkinson’s disease and movement disorders, Severe motor impairment / communication restoration, Stroke neurorehabilitation, Refractory depression and psychiatric applications |
| End User Segmentation | Tertiary neurology and neurosurgery centers, Academic medical centers, Specialty epilepsy centers, Neurorehabilitation institutes, Translational research hospitals |
| Regions Covered | North America, Europe, Asia Pacific, Rest of the World |
| Countries Covered | United States, Germany, United Kingdom, Japan, China, India, South Korea, and 40 plus countries |
| Key Companies Profiled | Medtronic, NeuroPace, Synchron, Paradromics, Precision Neuroscience, Blackrock Neurotech, CorTec |
| Forecast Period | 2026 to 2036 |
| Approach | FMI engaged functional neurosurgeons and clinical trial directors to understand the shift toward adaptive therapies. The baseline sizing anchors to the verified commercial placement volumes and average selling prices of current FDA-approved responsive systems. Forecasts are validated against the scale-up capacities of major neurotech manufacturers and the anticipated expansion of specific payer reimbursement codes. |
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary source documentation.
What is a closed-loop brain computer interface
A closed-loop interface is a bidirectional system that continuously records neural activity, decodes physiological biomarkers, and autonomously delivers targeted electrical stimulation. It signals the transition of neurotechnology from passive stimulation devices to active, physiological response systems.
What is the growth outlook for implantable BCIs through 2036
The valuation reaches USD 2,100 million by 2036. This expansion signals the point where closed-loop decoding algorithms switch from being premium add-ons to mandatory requirements for functional neurosurgery departments managing chronic movement and psychiatric disorders.
Explain the closed-loop BCI market for investors
The market tracks a 16.40% CAGR through the forecast period. This rate is constrained not by technological innovation, but by the speed at which specialized surgical centers can train multidisciplinary teams to manage complex post-operative data ecosystems and secure discrete payer billing codes.
Which device modality segment leads?
Implantable responsive neurostimulation systems lead the market. Their dominance stems from their entrenched position in the established surgical pathway for drug-resistant epilepsy, where localized field potential resolution is critical for intervening at the microsecond timescale required to abort a focal seizure.
Which signal and control architecture leads?
Sensing and adaptive stimulation closed-loop systems capture the largest share. Movement disorder specialists dictate this procurement choice because it provides objective, quantified disease measurements that eliminate the reliance on subjective patient symptom diaries.
What diseases can closed-loop BCIs treat
Drug-resistant epilepsy leads the clinical indications. The definitive failure of dual-pharmaceutical management acts as a rigid, predictable forcing function that pushes patients predictably toward surgical evaluation, ensuring a steady funnel for specialized centers. Other applications include brain-computer interface for Parkinson’s disease, severe motor impairment, and emerging psychiatric conditions.
What drives rapid growth?
Growth is driven by the clinical need to prevent neural habituation. The continuous delivery of open-loop energy causes off-target side effects; adaptive systems solve this by throttling energy output dynamically based on real-time neural signatures, forcing hospitals to upgrade their surgical hardware.
What is the primary restraint?
The sheer volume of raw neural telemetry overwhelms current clinical workflows. Because physician reimbursement is largely tied to episodic evaluation rather than continuous data monitoring, the lack of automated, validated diagnostic dashboards creates a bottleneck in scaling patient capacity.
Which country grows fastest?
India expands at 19.4%, outpacing China's 18.2%. The difference lies in India's rapid, private-capital-driven modernization of tertiary surgical suites, allowing institutions to leapfrog legacy technologies directly to advanced sensing architectures to attract medical tourism.
How does adaptive DBS work, and how is it different from conventional DBS?
Legacy continuous DBS delivers a static prescription, which can cause dysarthria or battery drain when symptoms are naturally low. Adaptive DBS solves this by actively sensing real-time beta band activity and throttling energy output based on the immediate physiological need, reducing the cognitive burden on programming clinicians.
Are closed-loop BCIs FDA approved, and summarize FDA progress in implanted brain computer interfaces?
Yes, specific platforms are commercially approved. The FDA has granted premarket approval (PMA) for early-generation responsive systems and adaptive deep brain stimulators for epilepsy and Parkinson's. Investigational device exemptions (IDEs) are currently accelerating for more advanced BCI for speech restoration and severe motor impairment applications.
Which companies make implantable BCIs, and which closed-loop BCI companies are commercially active?
Dominant incumbents hold massive, proprietary datasets of chronic neural recordings. Medtronic and NeuroPace lead the commercial therapy spaces. Smaller innovators and neuromodulation BCI companies like Synchron, CorTec, and Precision Neuroscience are advancing through clinical trial phases, engineering novel delivery mechanisms like endovascular navigation.
What is responsive neurostimulation and why is it the most established segment?
The localized field potential resolution provided by direct cortical arrays is critical for identifying a pre-ictal state before clinical manifestation. This established capability cannot currently be replicated by external sensing, making it the only verified method for arresting focal seizures at the necessary microsecond timescale.
How much does a brain implant system cost?
The capital expenditure for these systems moves the focus from static hardware to dynamic, evolving software platforms. While the upfront hardware procurement cost remains high, hospital procurement committees evaluate the total lifecycle cost, factoring in the avoidance of invasive battery replacement surgeries and the value of over-the-air firmware upgrades.
Cortical implant vs endovascular BCI: what dictates the choice?
While endovascular approaches bypass craniotomy risks, they face challenges in achieving the single-neuron spatial resolution possible with direct cortical arrays. Their adoption is gated by the necessity to prove long-term signal fidelity through the vessel wall without causing thrombosis, primarily targeting BCI for paralysis communication and severe motor impairment.
Why is cloud telemetry integration critical for scalability?
The gigabytes of neural data generated daily cannot be efficiently stored or processed on local hospital servers. Secure, compliant cloud infrastructure allows off-board algorithm refinement, which is essential for neurologists attempting to manage expanding panels of chronically implanted patients.
How does South Korea's digital health infrastructure impact its market?
South Korea leverages its deep domestic digital maturity to integrate continuous neural data streams seamlessly into standard electronic health records. This efficiency reduces the friction of chronic monitoring, streamlining patient management compared to regions with fragmented IT ecosystems.
What forces the transition to AI-assisted neural decoding?
The clinical reality that a physician cannot manually review months of high-density continuous electrocorticography data. AI summarization is necessary to parse the telemetry into actionable insights, bridging the gap between hardware capabilities and human cognitive limits in the clinic.
Compare adaptive DBS and responsive neurostimulation: what drives the transition?
Legacy continuous stimulation often induces dysarthria or rapidly drains implant batteries during periods of low symptom severity. By explicitly tracking beta band oscillations and delivering energy only when that biomarker crosses a specific threshold, adaptive systems preserve battery life and minimize side effects.
How close are BCIs to mainstream clinical use?
Preclinical platforms and early feasibility trials supply the foundational hardware for BCI platform for clinical trials testing novel algorithmic approaches. While broad mainstream consumer use remains restricted, implantable medical neurotechnology has already crossed into standard commercial procurement for specific drug-resistant neurological conditions.
How do reimbursement codes specifically restrict market expansion?
Currently, clinical programming sessions demand intensive epileptologist oversight without corresponding autonomous therapy management codes. Until payers establish distinct billing pathways that compensate for algorithmic, off-board therapy adjustment, hospitals cannot financially justify expanding their implant volumes.
Alternatives to closed-loop neurostimulation: what are the limitations?
Unlike the distinct electrophysiological signatures of a focal seizure or a Parkinsonian tremor, the biomarkers for refractory depression are diffuse and less definitively mapped. The market remains constrained until large-scale feasibility studies can validate reliable, patient-specific neural triggers over traditional pharmaceutical management or open-loop alternatives.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.