The implantable neurostimulator electrode market is estimated at USD 1.3 billion in 2026 and is projected to reach USD 2.6 billion by 2036, growing at a CAGR of 7.0%. Directional leads hold 40% of the product segment, with applications dominated by deep brain stimulation (DBS) at 40%. Hospitals represent the primary end users at 65%, while outpatient clinics and distributors support additional adoption. Key players include Medtronic, Abbott, Boston Scientific, Nevro, and LivaNova.
Market growth is driven by increasing prevalence of neurological disorders, rising demand for targeted neuromodulation, and technological improvements in electrode design. Directional and cylindrical leads are expanding adoption due to precision stimulation and reduced side effects. Continuous innovation in percutaneous systems, minimally invasive delivery methods, and integrated accessory support enhances procedural efficiency. Strategic partnerships with healthcare providers and distribution networks further enable widespread market penetration across developed and emerging regions over the forecast period.
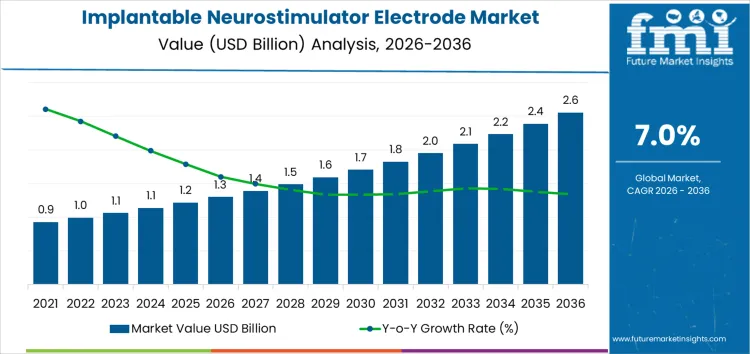
From 2026 to 2031, the implantable neurostimulator electrode market grows from USD 1.3 billion to approximately USD 1.7 billion, representing the early adoption phase. Annual increments increase from USD 0.1 billion to USD 0.14 billion during this period. Growth is driven by adoption in neuromodulation therapies for chronic pain, Parkinson’s disease, and other neurological disorders. North America and Europe account for roughly 65% of early market value, with uptake concentrated in advanced hospital systems and specialty clinics. Early adoption is influenced by clinical validation, regulatory approvals, and integration with established neurostimulation devices.
Between 2031 and 2036, the market expands from roughly USD 1.7 billion to USD 2.6 billion, illustrating the late-stage adoption and breakpoint where demand begins shifting from selective high-value deployments to broader portfolio-wide integration. Annual increments increase to USD 0.16–0.18 billion, driven by expansion in emerging markets, repeat implant procedures, and scaling across multi-site hospital networks. Growth is reinforced by technological improvements in electrode design, longevity, and compatibility with next-generation neurostimulators. Early growth relied on pilot clinical programs, whereas later expansion reflects structural adoption across global neuromodulation treatment centers.
| Metric | Value |
|---|---|
| Market Value (2026) | USD 1.3 billion |
| Forecast Value (2036) | USD 2.6 billion |
| Forecast CAGR (2026-2036) | 7.0% |
Demand for implantable neurostimulator electrodes developed from clinical limitations in pharmacological treatment of chronic neurological conditions. Patients with Parkinson’s disease, epilepsy, chronic pain, and movement disorders often experienced diminishing drug efficacy or intolerable side effects over time. Neurostimulation offered an alternative by directly modulating neural circuits, but early electrode designs faced challenges with signal precision, tissue response, and long term stability. Clinicians and device developers refined electrode geometry, materials, and placement strategies to improve targeting and reduce inflammatory response. Historical demand was driven by clinical need in severe cases, guided by specialist centres rather than broad neurological practice.
Future demand reflects changes in therapeutic strategy and device expectations rather than expanded diagnosis alone. Neurostimulation is increasingly considered earlier in treatment pathways, supported by improved imaging, patient selection, and outcome data. Electrodes are being designed for higher channel counts, directional stimulation, and compatibility with closed loop systems that adjust output based on neural feedback. Reliability over extended implant lifetimes is now a core requirement, not a secondary consideration. Demand growth will follow clinical protocol updates, reimbursement acceptance, and evidence supporting broader use across neurological and psychiatric indications.
The implantable neurostimulator electrode market is shaped by advances in neuromodulation therapy, expanding clinical indications, and demand for precise neural targeting. Growth is concentrated in electrode technologies that improve stimulation control, reduce side effects, and support long term therapy effectiveness. Clinical adoption is driven by outcomes in movement disorders and chronic pain management, where electrode performance directly influences patient response. Market leadership reflects therapeutic precision, clinician familiarity, and compatibility with established implantable neurostimulation systems rather than procedural volume growth alone.
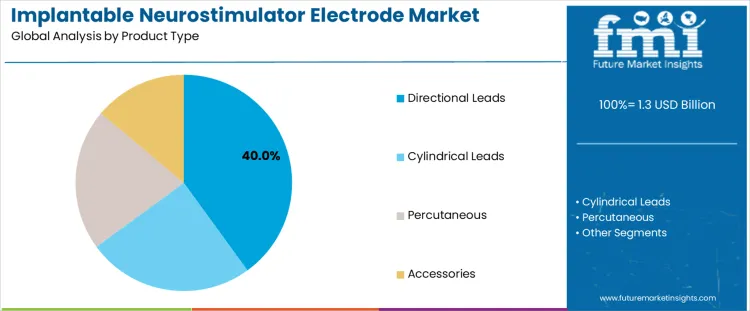
Directional leads account for about 40% of demand because they allow targeted stimulation of specific neural pathways while minimizing off target effects. These leads enable clinicians to shape electric fields, improving symptom control and reducing adverse effects such as speech or balance disturbances. Their use is strongest in complex neurological conditions where precision is critical. Cylindrical leads remain widely used due to familiarity, ease of implantation, and compatibility with existing systems. Percutaneous leads support less invasive procedures but offer limited directional control. Accessories support implantation and maintenance but do not influence therapeutic outcomes directly.
Other product types contribute to market breadth but do not displace directional leads. Cylindrical leads continue to be selected for standard cases with predictable anatomy. Percutaneous options are favored in temporary or trial settings. Accessories support system reliability and replacement cycles rather than driving primary demand. Product type leadership therefore reflects clinical demand for controllable stimulation and improved patient outcomes rather than cost minimization or procedural simplicity alone.
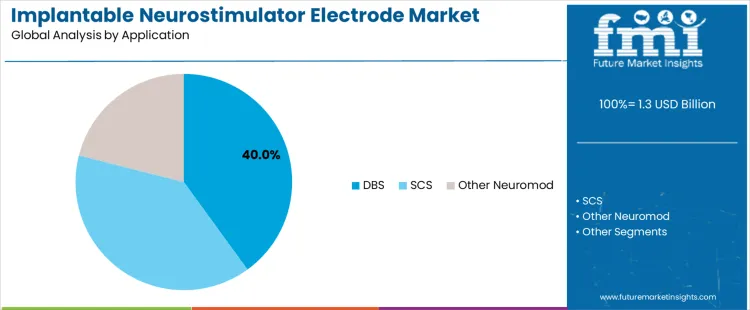
Deep brain stimulation accounts for about 40% of demand because it is an established therapy for movement disorders such as Parkinson’s disease and essential tremor. DBS requires precise electrode placement and stimulation control, making it closely aligned with advanced electrode technologies. Clinical evidence supporting long term symptom improvement sustains adoption. Spinal cord stimulation represents a significant application for chronic pain management but relies on broader stimulation fields. Other neuromodulation applications remain smaller due to limited indications and patient populations.
DBS maintains leadership due to strong clinical outcomes, reimbursement support, and specialist expertise. SCS adoption grows steadily but emphasizes pain coverage rather than precision targeting. Other neuromodulation uses remain selective and indication specific. Application leadership therefore reflects maturity of clinical evidence and dependence on electrode precision rather than expansion of experimental neuromodulation therapies.
Use appears in medical treatments where targeted electrical stimulation of neural tissue addresses chronic pain, movement disorders, or neurological conditions. Hospitals and specialized clinics implant electrodes in the spinal cord, peripheral nerves, or brain to modulate neural activity in conditions such as Parkinson’s disease, epilepsy, or chronic back pain. Manufacturers integrate electrodes with pulse generators and programming systems to allow personalized therapy adjustments. These applications reflect operational and clinical priorities rather than aesthetic considerations, with adoption driven by treatment efficacy, precision targeting, and patient outcomes.
What Operational and Material Conditions Support Adoption of Implantable Neurostimulator Electrodes?
Selection aligns with biocompatible, corrosion-resistant materials capable of long-term implantation. Electrodes are optimized for electrical conductivity, flexibility, and durability under physiological conditions. Surgical teams rely on precise placement and imaging guidance to ensure efficacy and minimize tissue damage. Device manufacturers integrate electrodes with pulse generators, telemetry systems, and programming software to provide adjustable stimulation. These conditions emerge from operational reliability, patient safety, and therapeutic effectiveness priorities in structured neuromodulation workflows.
Which Practical Considerations Limit Wider Use of Implantable Neurostimulator Electrodes?
High device and procedural costs can restrict adoption to specialized centers. Surgical implantation requires trained neurosurgeons and advanced imaging support. Patient selection and preoperative planning are critical for efficacy. Regulatory approvals for each electrode design and application vary by region. Device longevity and potential need for replacement or revision influence long-term adoption. These factors lead to selective deployment where clinical benefits, precision therapy, and patient outcomes justify incremental cost and operational complexity.
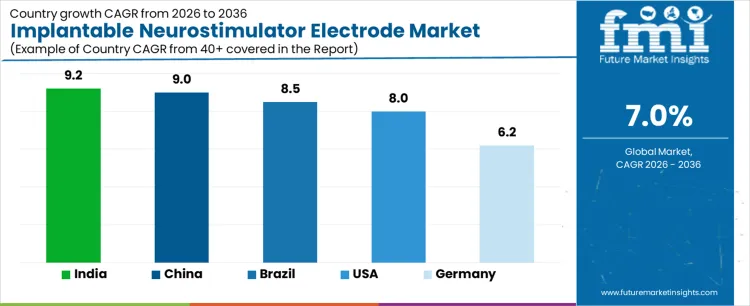
| Country | CAGR (%) |
|---|---|
| USA | 8.0% |
| Brazil | 8.5% |
| China | 9.0% |
| India | 9.2% |
| Germany | 6.2% |
The demand for implantable neurostimulator electrodes varies across countries, driven by neurological disorder prevalence, advanced healthcare infrastructure, and adoption of neurostimulation therapies. India leads with a 9.2% CAGR, supported by increasing cases of neurological disorders, growing awareness of implantable neurostimulation solutions, and expansion of healthcare facilities. China follows at 9.0%, driven by rising adoption of neurostimulation devices and investment in advanced medical technologies. Brazil records 8.5%, shaped by improving healthcare access and adoption of minimally invasive neuro therapies. The USA grows at 8.0%, reflecting established healthcare infrastructure and consistent adoption of neurostimulation devices. Germany posts 6.2%, supported by mature healthcare systems and steady device utilization.
In the United States, revenue from the implantable neurostimulator electrode market is expanding at a CAGR of 8% through 2036, driven by rising adoption of neuromodulation therapies for chronic pain, movement disorders, and neurological conditions. Hospitals, specialized clinics, and research institutions are integrating implantable electrodes with advanced stimulation systems to improve treatment precision and patient outcomes. Demand is concentrated in neurology centers, pain management clinics, and academic hospitals. Domestic suppliers are providing high-performance, biocompatible electrodes compatible with various neurostimulator platforms. Increasing prevalence of neurological disorders, recurring device implantation, and regulatory approvals are sustaining predictable adoption nationwide.
In Brazil, the market is growing at a CAGR of 8.5% through 2036, supported by expanding neurology and pain management services. Manufacturers are integrating implantable electrodes to deliver precise neurostimulation for chronic pain, Parkinson’s disease, and other neurological disorders. Demand is concentrated in hospitals, specialized clinics, and rehabilitation centers. Local suppliers provide high-quality, biocompatible electrodes suitable for implantable neurostimulators. Rising awareness of neuromodulation therapies, expanding healthcare access, and recurring device usage are sustaining adoption nationwide.
China is witnessing growth at a CAGR of 9% through 2036, driven by rapid expansion of neurology clinics, hospitals, and research centers adopting implantable neurostimulator electrodes. Manufacturers are integrating electrodes with advanced stimulation devices to improve clinical outcomes and treatment efficiency. Demand is concentrated in urban hospitals, specialized neurology centers, and rehabilitation facilities. Domestic suppliers are scaling production of high-performance, biocompatible electrodes compatible with multiple neurostimulator platforms. Increasing prevalence of neurological disorders, infrastructure expansion, and recurring therapy cycles are sustaining rapid adoption nationwide.
In India, revenue from the implantable neurostimulator electrode market is expanding at a CAGR of 9.2% through 2036, supported by increasing adoption of neuromodulation therapies in hospitals and specialty clinics. Manufacturers are integrating implantable electrodes for chronic pain management, movement disorder treatment, and neurological therapy enhancement. Demand is concentrated in neurology centers, pain clinics, and academic hospitals. Domestic suppliers provide high-performance, biocompatible electrodes suitable for various neurostimulator platforms. Rising awareness of neuromodulation, healthcare infrastructure development, and recurring device implantation are sustaining robust adoption nationwide.
Germany is witnessing growth at a CAGR of 6.2% through 2036, driven by adoption of implantable neurostimulator electrodes in hospitals and specialized clinics for neuromodulation therapies. Manufacturers are integrating electrodes to provide precise stimulation for chronic pain, Parkinson’s disease, and other neurological conditions. Demand is concentrated in academic hospitals, neurology centers, and specialized pain management clinics. Domestic suppliers provide high-quality, biocompatible electrodes compatible with advanced neurostimulator platforms. Compliance with healthcare standards, recurring device use, and clinical adoption are sustaining consistent procurement nationwide.
Leading companies in the implantable neurostimulator electrode market compete primarily through platform integration, clinical evidence generation, and long term physician alignment rather than frequent product redesign. Medtronic, Abbott, and Boston Scientific pursue strategies centered on maintaining closed system compatibility between electrodes, pulse generators, and programming software. This approach reduces switching risk for clinicians and reinforces procedural familiarity. These companies invest heavily in multiyear clinical studies to demonstrate durability, consistent stimulation coverage, and reduced lead migration. Incremental improvements in contact geometry, insulation materials, and lead flexibility are introduced cautiously to preserve backward compatibility with installed platforms. Commercial strategy emphasizes surgeon training, procedural support, and long term service relationships rather than aggressive price competition or rapid technology cycles.
Specialist players differentiate through indication specific strategies rather than scale. Nevro focuses on electrode designs optimized for high frequency stimulation, aligning product development tightly with proprietary therapy protocols rather than broad platform expansion. LivaNova emphasizes precision electrode placement and durability for epilepsy and psychiatric indications, where outcome consistency and safety drive adoption. Across the market, key strategies include reinforcing physician preference through training programs, supporting investigator led research, and prioritizing regulatory stability over rapid iteration. Competitive advantage is built through long approval cycles, clinical trust, and predictable performance over the full implant lifespan rather than short term technical differentiation.
| Items | Values |
|---|---|
| Quantitative Units (2026) | USD billion |
| Product Type | Directional Leads, Cylindrical Leads, Percutaneous, Accessories |
| Application | DBS, SCS, Other Neuromodulation |
| End-Use Industry | Hospitals, Outpatient Clinics, Distributors, Others |
| Region | Asia Pacific (China, Japan, South Korea, India, Australia & New Zealand, ASEAN, Rest of Asia Pacific), Europe (Germany, United Kingdom, France, Italy, Spain, Nordic, BENELUX, Rest of Europe), North America (United States, Canada, Mexico), Latin America (Brazil, Chile, Rest of Latin America), Middle East & Africa (KSA, Other GCC Countries, Turkey, South Africa, Other African Union, Rest of Middle East & Africa) |
| Key Companies Profiled | Medtronic, Abbott, Boston Scientific, Nevro, LivaNova, Others |
| Additional Attributes | Dollar by sales by product type, Dollar by sales by application, Dollar by sales by end-user, Dollar by sales by region, Clinical efficacy and precision targeting, Electrode design and channel count, Compatibility with pulse generators, Closed-loop system support, Surgical workflow integration, Patient outcome improvements, Minimally invasive implantation adoption, Regulatory compliance and approval, Recurring device replacement cycles, Training and physician support programs, Emerging market penetration, Adoption in specialized neurology and pain clinics |
How big is the implantable neurostimulator electrode market in 2026?
The global implantable neurostimulator electrode market is estimated to be valued at USD 1.3 billion in 2026.
What will be the size of implantable neurostimulator electrode market in 2036?
The market size for the implantable neurostimulator electrode market is projected to reach USD 2.6 billion by 2036.
How much will be the implantable neurostimulator electrode market growth between 2026 and 2036?
The implantable neurostimulator electrode market is expected to grow at a 7.0% CAGR between 2026 and 2036.
What are the key product types in the implantable neurostimulator electrode market?
The key product types in implantable neurostimulator electrode market are directional leads, cylindrical leads, percutaneous and accessories.
Which application segment to contribute significant share in the implantable neurostimulator electrode market in 2026?
In terms of application, dbs segment to command 40.0% share in the implantable neurostimulator electrode market in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.