The Urology Electrodes market is estimated at USD 1.4 billion in 2026 and is expected to reach USD 2.7 billion by 2036, reflecting a CAGR of 6.8%. Monopolar electrodes remain widely used, supporting BPH procedures, while bipolar and specialty electrodes address bladder cancer and other urological interventions. Hospitals are the leading end users, followed by ambulatory surgery centers and clinics. Key players include Olympus, Karl Storz, Boston Scientific, Richard Wolf, and Stryker. Growth is driven by rising urological disorder prevalence and increasing adoption of minimally invasive procedures.
Market expansion is fueled by technological improvements enhancing precision, patient safety, and procedure efficiency. Increasing investments in urology departments, coupled with surgeon training programs, improve electrode utilization. The growing shift toward outpatient treatments and advanced procedural methods supports market growth. Continuous innovation in electrode design and integration with imaging systems ensures robust adoption across hospitals and specialized centers, sustaining long-term growth through 2036.
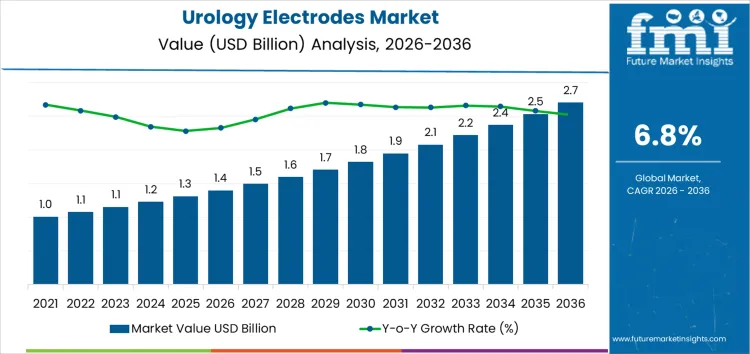
From 2026 to 2031, the urology electrodes market grows from USD 1.4 billion to approximately USD 1.9 billion, representing the early to mid-stage adoption phase of the market maturity curve. Annual increments increase from USD 0.08 billion in 2026 to USD 0.12 billion in 2031. Growth is driven by hospitals, outpatient urology clinics, and specialty care centers adopting electrodes for minimally invasive procedures, diagnostic urodynamics, and neuromodulation therapies. Early adoption is concentrated in North America and Europe, accounting for roughly 65% of market value, with uptake focused on high-volume facilities validating product performance, reliability, and procedural integration.
Between 2031 and 2036, the market expands from roughly USD 1.9 billion to USD 2.7 billion, illustrating the late-stage adoption and market maturity curve. Annual increments rise steadily from USD 0.12 billion to USD 0.18 billion, driven by portfolio-wide deployment, multi-site installations, and expansion into emerging markets in Asia Pacific and Latin America. Market maturity is reinforced by structural adoption across hospitals, repeat procurement cycles, and integration with advanced urology devices. Early-stage growth relied on pilot programs and high-value institutions, while later expansion reflects full-scale adoption and normalized integration across global urology care settings.
| Metric | Value |
|---|---|
| Market Value (2026) | USD 1.4 billion |
| Forecast Value (2036) | USD 2.7 billion |
| Forecast CAGR (2026-2036) | 6.8% |
Growth in the urology electrodes market has been influenced by expanding use of minimally invasive and endoscopic procedures to diagnose and treat urinary tract disorders. Traditional open surgical approaches carried higher morbidity, longer recovery, and greater risk of complications. Electrosurgical and energy-based electrodes enabled precise cutting, coagulation, and ablation in transurethral resection of the prostate, bladder tumour removal, and urethral strictures. As hospitals and surgical centres shifted practice toward less invasive techniques, demand for specialised electrodes increased. Rising incidence of benign prostatic hyperplasia, bladder cancer, and urinary stones further contributed to procedural frequency. Historical uptake reflected clinician preference for devices that improved procedural control and reduced operative time.
Future market growth will be shaped by procedural innovation, technology integration, and evolving clinical standards rather than simple increases in caseloads. Newer electrode designs focus on energy modulation, improved tissue selectivity, and compatibility with image-guided systems to enhance precision and safety. Integration with digital platforms that provide feedback on tissue response is influencing device selection in high-acuity settings. Regulatory expectations for device performance and safety reporting are guiding procurement toward products with documented clinical outcomes. Urology service lines are standardising workflows and specifying electrodes that support reproducible results, reduced complication rates, and shorter hospital stays. Demand will align with evidence-based practice patterns, reimbursement frameworks that favour minimally invasive options, and clinician adoption of advanced energy technologies rather than reliance on legacy instruments alone.
The urology electrodes market is driven by high procedure volumes, aging populations, and continued reliance on electrosurgical techniques in minimally invasive urology. Demand centers on electrode types that align with established surgical workflows, generator compatibility, and predictable outcomes. Market structure reflects routine procedural needs rather than rapid technological shifts, with purchasing decisions shaped by installed base, training continuity, and cost control across hospitals and ambulatory settings.
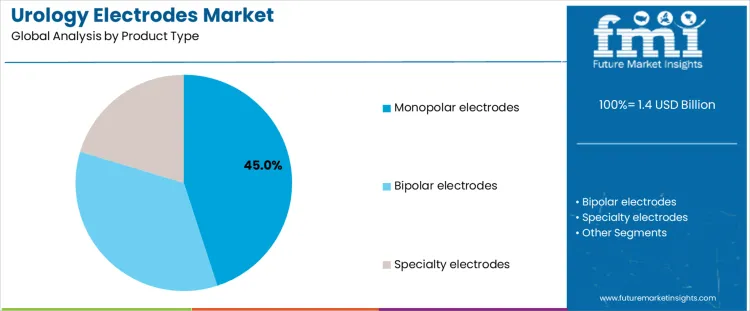
Monopolar electrodes account for about 45% of demand because they remain compatible with most installed electrosurgical generators used in urology suites. Surgeons rely on predictable cutting and coagulation behavior during transurethral procedures, where workflow familiarity reduces operative risk. Lower unit cost supports high volume use across routine cases. Monopolar systems also integrate easily with legacy resectoscopes, limiting capital upgrades. These factors anchor monopolar electrodes as default tools in everyday urologic resections despite incremental advances in alternative energy modalities.
Bipolar electrodes and specialty designs expand clinical options but serve narrower roles within urology practice. Bipolar systems reduce thermal spread and irrigation risks, supporting safety in select patients, yet require dedicated generators and higher acquisition cost. Specialty electrodes address anatomical challenges or niche techniques rather than standard workflows. These subsegments complement monopolar use instead of replacing it, reflecting procurement decisions driven by installed base compatibility, training continuity, and predictable outcomes rather than marginal performance gains alone today globally.
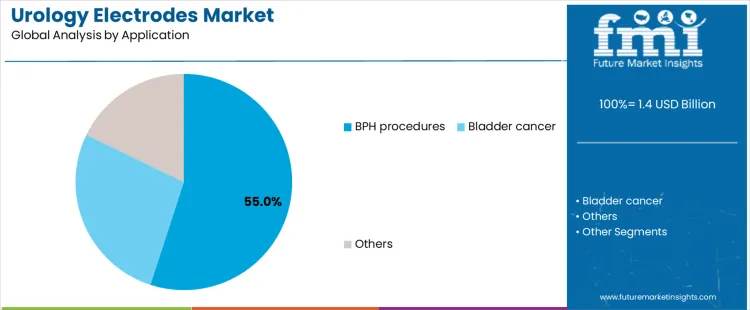
BPH procedures represent about 55% of demand because benign prostatic hyperplasia is highly prevalent and frequently managed with electrosurgical intervention. Transurethral resections require reliable electrodes for tissue removal and hemostasis, creating consistent consumption volumes. Aging populations and earlier diagnosis sustain procedure frequency. Electrode selection prioritizes control and efficiency during repetitive cases, reinforcing routine use. This concentration makes BPH the primary application shaping purchasing volumes across hospitals and ambulatory urology centers worldwide today across healthcare systems networks globally.
Bladder cancer procedures form a secondary application, relying on electrodes for tumor resection and coagulation during episodic interventions. Volumes are lower than BPH because treatment frequency depends on disease stage and recurrence patterns. Other uses include stone management and diagnostic access, each contributing limited demand. These applications require reliable performance but occur less often, keeping overall share smaller. Application leadership therefore reflects routine procedural frequency rather than severity or technical complexity within modern urology practice worldwide today broadly.
Use appears in hospitals, outpatient clinics, and specialized urology centers where electrical stimulation or ablation is required for diagnosis and treatment. Surgeons use electrodes in procedures such as transurethral resection of the prostate (TURP), bladder tumor ablation, and nerve stimulation for urinary incontinence. Electrode designs vary for monopolar or bipolar energy delivery, targeting precise tissue removal or modulation while minimizing collateral damage. These applications reflect operational and clinical priorities rather than aesthetic purposes, with adoption driven by procedural accuracy, patient safety, and therapeutic efficacy.
What Operational and Material Conditions Support Adoption of Urology Electrodes?
Selection aligns with sterilizable, biocompatible materials that maintain conductivity and mechanical integrity during procedures. Electrodes are optimized for energy delivery, surface area, and geometry to achieve consistent tissue effect. Surgical teams require compatibility with cautery, laser, or stimulation generators and imaging guidance systems. Hospitals implement protocols for safe handling, sterilization, and storage. These conditions emerge from operational reliability, procedural safety, and clinical performance priorities in structured urology workflows.
Which Practical Considerations Limit Wider Use of Urology Electrodes?
High device cost and the need for specialized generators can limit adoption in smaller facilities. Electrode selection must match specific procedures, patient anatomy, and energy settings. Regulatory approvals for safety and efficacy vary by region. Reuse or reprocessing requires strict adherence to sterilization and handling protocols. These factors lead to selective deployment where precision, patient safety, and clinical outcomes justify incremental cost and operational management.
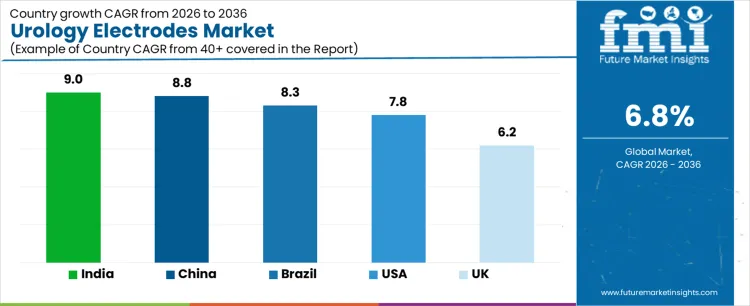
| Country | CAGR (%) |
|---|---|
| USA | 7.8% |
| Brazil | 8.3% |
| China | 8.8% |
| India | 9.0% |
| UK | 6.2% |
The demand for urology electrodes varies across countries, driven by growing urological procedures, hospital infrastructure expansion, and adoption of advanced medical devices. India leads with a 9.0% CAGR, supported by increasing urology procedures, expanding healthcare access, and rising adoption of minimally invasive techniques. China follows at 8.8%, driven by large patient population, hospital modernization, and growing demand for advanced urological equipment. Brazil records 8.3%, shaped by expanding healthcare services and adoption of specialized devices. The USA grows at 7.8%, reflecting established hospital networks and consistent procedural volumes. The UK posts 6.2%, supported by mature healthcare infrastructure and steady demand for urology electrodes.
In the United States, the Urology Electrodes Market is expanding at a CAGR of 7.8% through 2036, driven by rising adoption of minimally invasive procedures for prostate, bladder, and urinary tract interventions. Hospitals, specialty urology centers, and academic medical facilities are integrating high-performance electrodes to improve procedural accuracy, reduce operative time, and enhance patient outcomes. Demand is concentrated in surgical units, outpatient clinics, and high-volume hospitals. Domestic suppliers are providing biocompatible electrodes compatible with multiple neurostimulation and surgical platforms. Regulatory standards, procedural volume growth, and recurring surgeries are sustaining steady adoption nationwide.
In Brazil, revenue is growing at a CAGR of 8.3% through 2036, supported by increased hospital capacity and expansion of private surgical centers. Hospitals and specialty clinics are employing high-performance electrodes to enhance procedural precision, improve patient safety, and reduce complications in urological surgeries. Demand is concentrated in urban hospitals, outpatient clinics, and private medical facilities. Local suppliers provide biocompatible electrodes that meet regulatory requirements and clinical standards. Growth in urology care services, rising patient awareness, and increasing minimally invasive procedure volumes are driving adoption. Recurring surgical cycles are supporting stable procurement and long-term market expansion nationwide.
In China, the market is expanding at a CAGR of 8.8% through 2036, driven by rapid adoption of advanced surgical technologies and hospital infrastructure expansion. Hospitals, specialty surgical centers, and research facilities are integrating electrodes to improve surgical precision, reduce complications, and optimize patient outcomes in bladder and prostate procedures. Demand is concentrated in tertiary hospitals, urban medical centers, and high-volume specialty clinics. Domestic suppliers are scaling production of high-performance, biocompatible electrodes compatible with various neurostimulation and surgical platforms. Government support, rising procedural volumes, and expanding healthcare infrastructure are sustaining rapid adoption and long-term market growth across the country.
In India, revenue from the Urology Electrodes Market is expanding at a CAGR of 9% through 2036, supported by increasing outpatient surgeries, expansion of specialty urology clinics, and rising healthcare infrastructure. Hospitals and teaching hospitals are adopting high-performance electrodes to enhance procedural accuracy, improve patient safety, and optimize outcomes in urological interventions. Demand is concentrated in urban hospitals, specialty clinics, and academic medical facilities. Domestic suppliers provide clinically validated, biocompatible electrodes compatible with multiple surgical platforms. Growing healthcare investment, increasing procedure volumes, and recurring surgeries are sustaining robust adoption and contributing to market growth across the country.
In the United Kingdom, the market is growing at a CAGR of 6.2% through 2036, driven by adoption of high-performance electrodes in hospitals, outpatient clinics, and specialty urology centers. Surgeons are using electrodes to improve precision in minimally invasive procedures, reduce complications, and optimize patient outcomes. Demand is concentrated in academic hospitals, urology units, and surgical centers. Domestic suppliers provide biocompatible, clinically validated electrodes compatible with standard surgical platforms. Regulatory standards, rising surgical volumes, and recurring procedures are supporting consistent adoption. Increasing integration of advanced technologies in clinical workflows is also contributing to sustainable growth in the UK market.
Competition in the Urology Electrodes Market is shaped by how electrodes integrate with endoscopic platforms, energy generators, and procedural preferences rather than by standalone component innovation. Olympus and Karl Storz pursue strategies centered on tight compatibility between electrodes, resectoscopes, and visualization systems, reinforcing closed ecosystem adoption in transurethral and endoscopic urology procedures. Boston Scientific aligns electrodes with its broader urology portfolio, linking energy delivery to stone management and benign prostatic hyperplasia treatment pathways. Richard Wolf emphasizes modularity and precision, targeting clinicians who prioritize flexibility across resection, coagulation, and vaporization procedures. Stryker participates selectively through energy based surgical platforms adapted for urologic use, focusing on performance consistency and durability.
Strategic differentiation increasingly reflects service support, training, and lifecycle management. Leading suppliers prioritize incremental electrode design refinements that preserve generator compatibility and regulatory approvals, limiting disruption to hospital inventories. Physician training programs, standardized electrode kits, and procedure specific configurations are used to reinforce repeat adoption. Smaller and regional players tend to compete through niche electrode geometries or localized distribution rather than platform scale. Across the market, competitive advantage depends on procedural reliability, ease of setup, and alignment with surgeon technique. Long qualification cycles, installed base protection, and clinician familiarity constrain rapid substitution, making continuity and workflow fit more decisive than frequent technological change.
| Items | Values |
|---|---|
| Quantitative Units (2026) | USD billion |
| Product Type | Monopolar electrodes, Bipolar electrodes, Specialty electrodes |
| Application | BPH procedures, Bladder cancer, Others |
| End-Use Industry | Hospitals, ASCs, Clinics |
| Region | Asia Pacific (China, Japan, South Korea, India, Australia & New Zealand, ASEAN, Rest of Asia Pacific), Europe (Germany, UK, France, Italy, Spain, Nordic, BENELUX, Rest of Europe), North America (United States, Canada, Mexico), Latin America (Brazil, Chile, Rest of Latin America), Middle East & Africa (KSA, Other GCC, Turkey, South Africa, Other African Union, Rest of MEA) |
| Key Companies Profiled | Olympus, Karl Storz, Boston Scientific, Richard Wolf, Stryker, Others |
| Additional Attributes | Dollar by sales by product type, Dollar by sales by application, Dollar by sales by end-user, Dollar by sales by region, Annual procurement cycles, Procedural volume coverage, Device precision and tissue selectivity, Electrosurgical and energy delivery performance, Biocompatibility and sterilization compatibility, Integration with imaging and endoscopic systems, Surgeon training and handling programs, Repeat procurement frequency, Compliance with surgical standards, Domestic vs. imported device distribution, Supply chain reliability, Regulatory adherence (FDA, CE, ISO), Adoption by high-volume urology units, Operational efficiency and procedure time metrics |
How big is the urology electrodes market in 2026?
The global urology electrodes market is estimated to be valued at USD 1.4 billion in 2026.
What will be the size of urology electrodes market in 2036?
The market size for the urology electrodes market is projected to reach USD 2.7 billion by 2036.
How much will be the urology electrodes market growth between 2026 and 2036?
The urology electrodes market is expected to grow at a 6.8% CAGR between 2026 and 2036.
What are the key product types in the urology electrodes market?
The key product types in urology electrodes market are monopolar electrodes, bipolar electrodes and specialty electrodes.
Which application segment to contribute significant share in the urology electrodes market in 2026?
In terms of application, bph procedures segment to command 55.0% share in the urology electrodes market in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.