The global implantable cardiac fluorine-free resins market is valued at USD 1.6 billion in 2026 and is projected to reach USD 4.4 billion by 2036, growing at a CAGR of 10.7%. Regional demand for fluorine-free resins is concentrated in developed markets such as North America and Europe, where stricter regulations on medical device safety and sustainability are driving adoption. Adoption rates are uneven, with some regions lagging due to slower regulatory progress or cost barriers.
In countries like China and South Korea, rapid growth is fueled by expanding healthcare infrastructure and increasing demand for biocompatible materials in implantable cardiac devices. The cost advantages in regions with established manufacturing hubs, like Germany and the USA, enable competitive pricing and faster production scaling. As global regulations tighten, the market is likely to experience significant regional shifts, with higher adoption rates in emerging markets expected to accelerate growth.
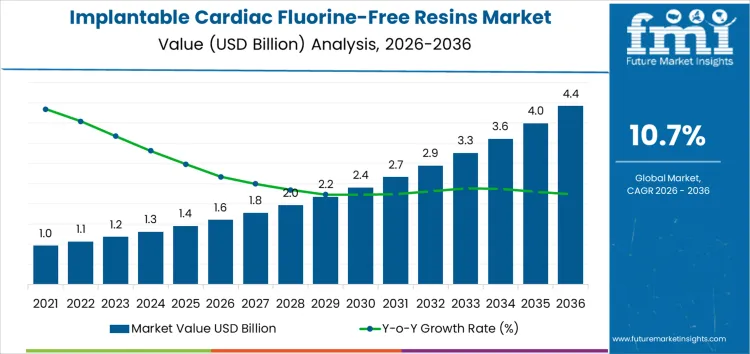
| Metric | Value |
|---|---|
| Industry Value (2026) | USD 1.6 Billion |
| Forecast Value (2036) | USD 4.4 Billion |
| Forecast CAGR 2026 to 2036 | 10.7% |
The global implantable cardiac fluorine-free resins market is experiencing substantial growth, driven by increasing demand for safer and more biocompatible materials in the medical device industry. Fluorine-free resins, specifically designed for use in implantable cardiac devices, offer an alternative to traditional fluorinated resins, which can present concerns regarding toxicity and biocompatibility. These resins are essential for manufacturing components such as pacemakers, defibrillators, and other cardiac implants, where material safety and performance are critical for patient outcomes.
A major driver for this market is the growing emphasis on developing medical materials that offer enhanced biocompatibility, lower toxicity, and improved long-term performance. Fluorine-free resins are gaining favor in the cardiac implant industry because they reduce the risk of adverse reactions and ensure that devices are more compatible with the human body. As the aging population increases and the prevalence of cardiovascular diseases rises, the demand for implantable cardiac devices is expanding, which directly boosts the need for high-performance, fluorine-free resins.
Technological advancements in resin formulations and processing techniques are also contributing to the growth of the market. Newer fluorine-free resins are being engineered to meet the demanding requirements of implantable cardiac devices, offering improved durability, flexibility, and resistance to degradation over time. As the healthcare industry moves toward safer and more sustainable solutions, these resins are seen as a crucial component in enhancing the safety and reliability of cardiac implants. As a result, the implantable cardiac fluorine-free resins market is set to grow rapidly, offering a vital solution for the medical device sector’s shift toward more biocompatible materials.
The implantable cardiac fluorine-free resins market is segmented by resin type and end-user. Medical-grade fluorine-free polymers lead the resin type segment with a 41% share, followed by Polyether Ether Ketone (PEEK) alternatives, bio-derived high-performance resins, acrylic & styrenic resins, and other types of resins. Medical-grade fluorine-free polymers are preferred for implantable cardiac devices due to their biocompatibility and high performance. In terms of end-users, hospitals & cardiac clinics account for the largest share at 45%, followed by medical device OEMs, research & academic institutions, and other users.
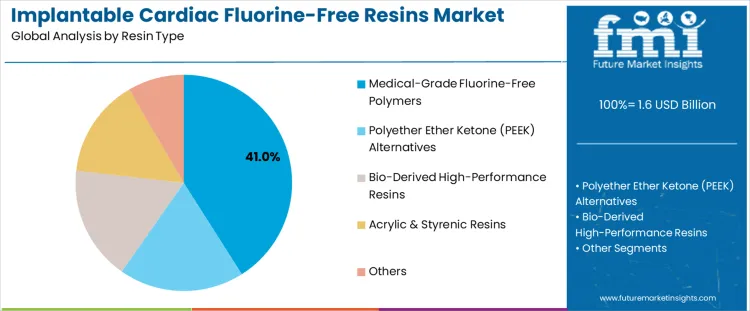
Medical-grade fluorine-free polymers lead the implantable cardiac fluorine-free resins market with a 41% share due to their essential role in creating biocompatible, durable, and safe materials for implantable cardiac devices. These polymers meet the stringent regulatory and safety standards required for medical devices, particularly those used in cardiac applications. With increasing awareness about the need for safer, more sustainable materials in healthcare, fluorine-free alternatives are becoming the preferred choice. The growing demand for high-performance, biocompatible polymers that provide enhanced patient safety and environmental benefits drives their adoption in the medical sector. As the healthcare industry focuses more on patient well-being and sustainability, these polymers remain the dominant option for implantable cardiac devices.
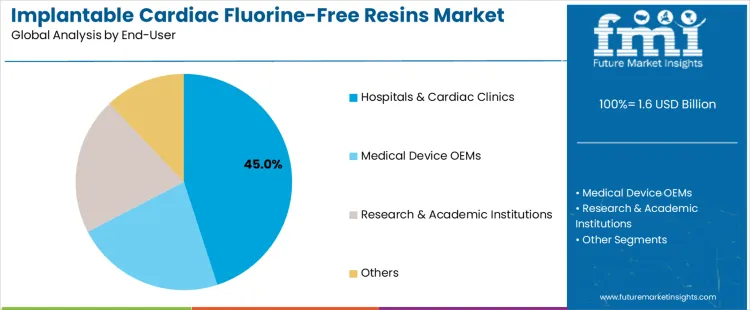
Hospitals & cardiac clinics account for 45% of the end-user segment in the implantable cardiac fluorine-free resins market. These healthcare providers are the primary adopters of implantable cardiac devices, such as pacemakers, defibrillators, and stents, which require materials that are safe, biocompatible, and durable. The growing demand for minimally invasive procedures, coupled with the need for longer-lasting and safer implants, is increasing the reliance on fluorine-free resins in the production of these devices. As healthcare providers focus on providing high-quality, effective treatments, hospitals and cardiac clinics drive the demand for innovative, sustainable materials in cardiac implants, thus promoting the widespread use of fluorine-free resins in medical applications.
Fluorine-free resins are gaining traction as they offer lower environmental and health risks compared to fluorine-based resins, which can release toxic compounds. These resins are designed to provide enhanced durability, flexibility, and compatibility with human tissue, essential for long-lasting cardiac implants. The growing prevalence of cardiovascular diseases, combined with advancements in medical device technologies and stricter regulations on material safety, is propelling the adoption of fluorine-free resins in the cardiac implant sector.
The market for implantable cardiac fluorine-free resins is growing due to increasing concerns over the environmental and health risks associated with fluorinated compounds used in medical devices. As regulatory bodies tighten safety standards for medical materials, particularly for implantable devices, the demand for safer alternatives like fluorine-free resins is rising. The increasing number of cardiac surgeries and the growing prevalence of heart disease globally are contributing to the growth of this market. The desire for materials that enhance the biocompatibility, performance, and longevity of implants is also pushing the adoption of fluorine-free resins in cardiac applications.
Key drivers shaping the implantable cardiac fluorine-free resins market include the rising global incidence of cardiovascular diseases, driving the need for innovative and safer cardiac implants. As regulatory pressures increase to limit the use of harmful chemicals, particularly in medical devices, manufacturers are turning to fluorine-free resins that offer improved biocompatibility and lower environmental impact. The advancements in material science and the development of high-performance, durable resins with tailored properties for cardiac implants are also boosting market demand. The growing focus on patient safety and the preference for non-toxic, eco-friendly materials are accelerating market growth.
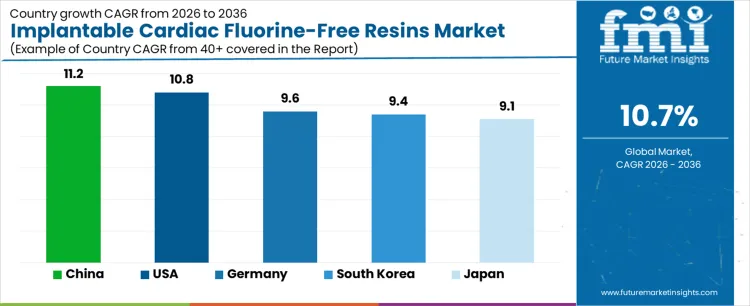
| Country | CAGR (%) |
|---|---|
| China | 11.2% |
| USA | 10.8% |
| South Korea | 9.4% |
| Germany | 9.6% |
| Japan | 9.1% |
The implantable cardiac fluorine-free resins market is expanding across key regions, with China leading at 11.2%, driven by the growing demand for eco-friendly and safe materials in the medical device sector. The USA follows at 10.8%, supported by stringent regulatory standards and increasing consumer demand for sustainable medical solutions. South Korea’s market grows at 9.4%, fueled by a focus on technological innovation and eco-friendly healthcare materials. Germany’s market expands at 9.6%, driven by a strong medical device industry and a commitment to sustainability. Japan grows at 9.1%, supported by a rising aging population and the demand for high-performance, biocompatible materials in cardiac implants.
China’s implantable cardiac fluorine-free resins market is growing at 11.2%, driven by the increasing demand for high-quality, safe materials in the medical devices industry. As China’s healthcare infrastructure improves and the adoption of advanced medical technologies increases, there is growing demand for implantable cardiac devices, such as pacemakers and defibrillators. Fluorine-free resins are gaining popularity in the medical device industry due to their safety, reduced toxicity, and eco-friendliness compared to fluorine-based alternatives. China’s focus on improving its medical device manufacturing capabilities and adhering to international regulatory standards for biocompatibility is driving the market for these advanced materials. As the country’s aging population increases, the demand for cardiac implantable devices and materials like fluorine-free resins will continue to rise.
The implantable cardiac fluorine-free resins market in the USA is growing at 10.8%, driven by strong demand for safe, reliable, and biocompatible materials used in cardiac implants. The USA, as a global leader in healthcare innovation, places high emphasis on medical device safety, which has led to increased adoption of fluorine-free resins for cardiac applications. These materials offer reduced environmental impact, lower toxicity, and superior performance, particularly for long-term implantable devices like pacemakers and defibrillators. The USA’s regulatory environment, which emphasizes stringent testing and compliance for medical device materials, is further contributing to the demand for fluorine-free resins. The aging population and increasing prevalence of heart disease in the country drive the need for advanced cardiac implants, fueling the market for these high-performance, sustainable resins.
South Korea’s implantable cardiac fluorine-free resins market is growing at 9.4%, supported by the country’s strong focus on healthcare innovation and sustainability. South Korea is investing heavily in the medical device sector, with a growing emphasis on biocompatible, eco-friendly materials for cardiac implants. Fluorine-free resins are becoming increasingly popular due to their reduced environmental impact, lower toxicity, and compliance with international medical device regulations. The growing demand for implantable cardiac devices in South Korea, particularly due to an aging population and rising heart disease rates, is driving the need for safer, more sustainable materials in medical applications. South Korea’s focus on technological advancements and the development of high-quality, sustainable healthcare solutions continues to support the growth of this market.
Germany’s implantable cardiac fluorine-free resins market is growing at 9.6%, driven by its advanced medical device industry and strong emphasis on sustainability and safety. Germany has a well-established healthcare system, with increasing demand for advanced cardiac implantable devices such as pacemakers, stents, and defibrillators. Fluorine-free resins are becoming a preferred choice in the German market due to their lower environmental impact and non-toxic properties, making them more suitable for long-term implantation. Germany’s regulatory framework, which ensures that medical devices meet stringent safety standards, is also contributing to the increased use of these biocompatible materials in cardiac devices. The country’s continued focus on innovation, combined with the rise in cardiovascular diseases, supports the growth of the fluorine-free resins market in implantable cardiac applications.
Japan’s implantable cardiac fluorine-free resins market is growing at 9.1%, supported by the country’s strong healthcare infrastructure and aging population. Japan’s emphasis on patient safety and the environmental impact of medical devices has led to an increased adoption of fluorine-free resins, particularly in the production of implantable cardiac devices like pacemakers and defibrillators. The country’s well-developed medical device industry is increasingly focused on using eco-friendly, non-toxic materials to meet international regulatory standards and consumer preferences for sustainable healthcare solutions. As Japan’s aging population faces higher rates of heart disease, the demand for cardiac implants, and thus fluorine-free resins, is expected to continue growing. Japan’s commitment to advancing medical technology, combined with a rising need for biocompatible materials, further drives the expansion of this market.
Competition in the implantable cardiac fluorine free resins market is driven by biocompatibility, mechanical reliability in dynamic environments, long term chemical stability, manufacturing readiness, and regulatory support for implantable medical applications. Solvay SA positions its fluorine free resin solutions with a strong emphasis on predictable in body performance and dimensional stability. Its product materials highlight consistent mechanical integrity under cardiac motion and safety across sterilization cycles, helping medical OEMs meet stringent regulatory and clinical benchmarks.
Evonik Industries competes through specialty biomedical polymers designed for implantable device compatibility and chemical resistance, with collateral focusing on controlled polymer properties that support predictable tissue interaction and minimal leachables. Röchling Medical differentiates with tailored resin grades engineered for medical manufacturing, and its documentation underscores injection molding and extrusion readiness, as well as quality systems that align with ISO 13485 and other implantable standards.
Quadrant AG offers fluorine free structural resins crafted for fatigue resistance and dimensional precision, marketed with emphasis on material traceability and manufacturing support that help device designers streamline part qualification. Synthomer plc adds value with custom resin chemistries engineered to balance flexural strength and implanted durability, and its product materials highlight controlled polymer architecture and reproducible property profiles.
Others in the market contribute with emerging resin formulations and niche compounding expertise aimed at specific cardiac device classes or manufacturing pathways. Across all vendors, product literature focuses on biocompatibility evidence, mechanical performance under cyclic loading, processing compatibility, and alignment with implant safety requirements as key differentiators that help cardiac device developers choose materials suited for long term, fluorine free implant applications.
| Attributes | Description |
|---|---|
| Quantitative Unit (2026) | USD Billion |
| Resin Type | Medical Grade Fluorine Free Polymers, Polyether Ether Ketone (PEEK) Alternatives, Bio Derived High Performance Resins, Acrylic & Styrenic Resins, Others |
| End-User | Hospitals & Cardiac Clinics, Medical Device OEMs, Research & Academic Institutions, Others |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Countries Covered | China, Japan, South Korea, India, Australia & New Zealand, ASEAN, Germany, United Kingdom, France, Italy, Spain, Nordic, BENELUX, United States, Canada, Mexico, Brazil, Chile, Turkey, South Africa, Other GCC Countries |
| Key Companies Profiled | Solvay SA, Evonik Industries, Röchling Medical, Quadrant AG, Synthomer plc, Others |
| Additional Attributes | Dollar sales by resin type and end-user; regional CAGR and growth outlook for the implantable cardiac fluorine-free resins market. |
How big is the implantable cardiac fluorine-free resins market in 2026?
The global implantable cardiac fluorine-free resins market is estimated to be valued at USD 1.6 billion in 2026.
What will be the size of implantable cardiac fluorine-free resins market in 2036?
The market size for the implantable cardiac fluorine-free resins market is projected to reach USD 4.4 billion by 2036.
How much will be the implantable cardiac fluorine-free resins market growth between 2026 and 2036?
The implantable cardiac fluorine-free resins market is expected to grow at a 10.7% CAGR between 2026 and 2036.
What are the key product types in the implantable cardiac fluorine-free resins market?
The key product types in implantable cardiac fluorine-free resins market are medical‑grade fluorine‑free polymers, polyether ether ketone (peek) alternatives, bio‑derived high‑performance resins, acrylic & styrenic resins and others.
Which end‑user segment to contribute significant share in the implantable cardiac fluorine-free resins market in 2026?
In terms of end‑user, hospitals & cardiac clinics segment to command 45.0% share in the implantable cardiac fluorine-free resins market in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.