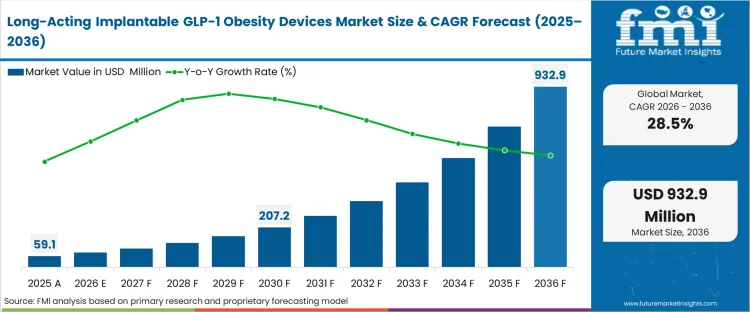
The long-acting implantable GLP-1 obesity devices market was valued at USD 59.1 million in 2025. The sector is poised to hit USD 76.0 million in 2026 at a CAGR of 28.50% during the forecast period. Steady investment advances the obesity drug implant market forecast valuation to USD 932.9 million through 2036 as developers target the massive drop-off rate in real-world patient adherence by extending therapy persistence beyond the limits of weekly injectables.
Payers and clinical providers are switching from evaluating GLP-1s purely on peak weight-loss efficacy to demanding 12-month persistence guarantees, cementing the obesity medication adherence implant as a necessity. This transition forces obesity-care networks to rethink how therapy is administered, moving away from pharmacy-dispensed self-administration toward procedural, clinic-based models. Delays in adopting these longer-duration formats expose health systems to the wasted pharmacoeconomic spend associated with patients who abandon treatment within the first three months. FMI analysts suggest that integrating obesity glp-1 formulations into solid, retrievable subdermal matrices fundamentally changes the commercial calculus for managed care organizations.
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 76.0 Million |
| Industry Value (2036) | USD 932.9 Million |
| CAGR (2026-2036) | 28.50% |
Sustained growth in this precommercial segment depends on demonstrating that solid implants can match the steady‑state tolerability of weekly injectables without inducing localized tissue damage. Direct developers mapping the obesity implant clinical pipeline are currently triggering this inflection through first-in-human trial data. Once this safety and release-rate parity is established, the obesity implant regulatory pathway for subsequent drug delivery solutions shortens considerably, allowing biomaterial CDMOs to rapidly scale manufacturing.
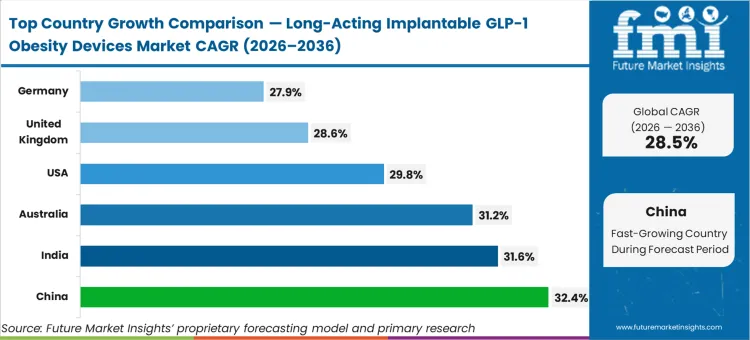
China is anticipated to expand at 32.4%, followed by India at 31.6%, Australia at 31.2%, the United States at 29.8%, the United Kingdom at 28.6%, Germany at 27.9%, and Japan at 24.7%. Australia's trajectory reflects its selection as a primary beachhead for first-in-human obesity implant studies, positioning its clinical infrastructure ahead of larger markets. The divergence across this range stems directly from how each healthcare system manages its adult overweight populations, markets with centralized chronic care procurement frameworks are moving faster to qualify single-intervention glp-1 diabetes treatment drugs than those relying on fragmented, patient-driven retail pharmacy refills.
The long-acting implantable GLP-1 obesity devices market encompasses the development, biomaterial manufacturing, and clinical deployment of subdermal and matrix-based systems designed to release glucagon-like peptide-1 receptor agonists continuously over multiple months. Analytically, these implantable weight loss GLP-1 devices exclude the underlying raw pharmaceutical active ingredients and focus entirely on the proprietary delivery architectures, polymer platforms, and integrated device-drug combinations that enable extended therapeutic persistence for metabolic management.
Scope includes solid subdermal implants, biodegradable polymer matrices, passive drug-eluting reservoirs, and the associated application or insertion tools required for clinical placement. It covers contract manufacturing revenues for implantable GLP-1 delivery systems, upfront technology licensing fees for delivery platforms, and clinical-stage value creation within the obesity management sector. Revenues generated by biomaterial partners supplying medical-grade absorbable polymers specifically for incretin delivery are also included.
The market explicitly excludes traditional weekly or daily GLP-1 subcutaneous injectables, oral formulations, and standard wearable auto-injectors. These are excluded because they rely on patient-driven adherence and distinct, short-term pharmacokinetic profiles, making a direct GLP-1 implant vs weekly injection cost comparison highly asymmetrical. General-purpose surgical implants without an active pharmaceutical ingredient, and implantable pumps requiring mechanical or electronic propulsion (such as insulin pumps), are also outside scope due to their entirely different regulatory classifications and manufacturing complexities.
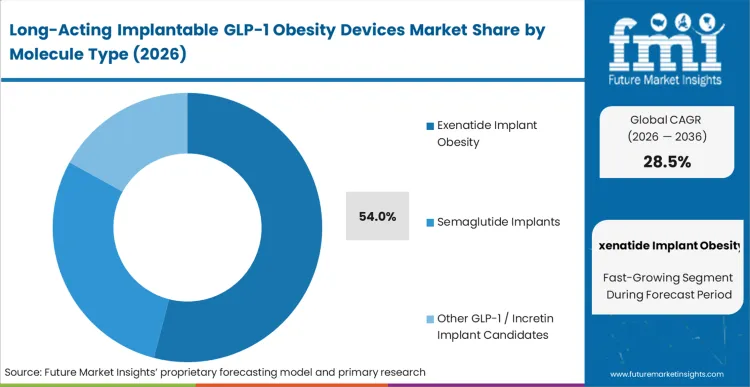
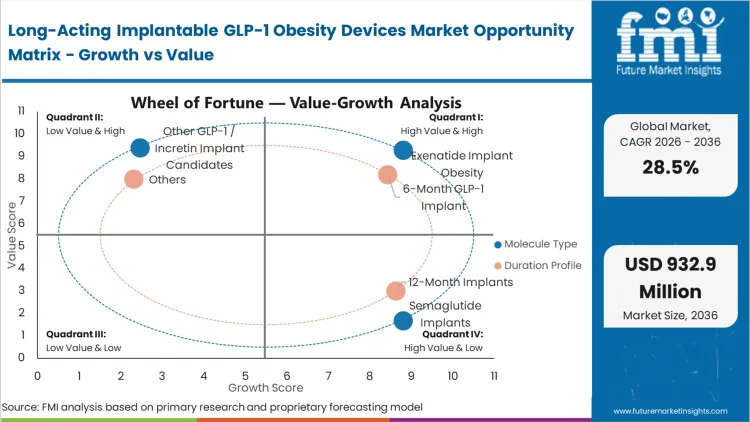
Legacy glp-1 receptor agonist injectables failed to solve the fundamental adherence gap, forcing developers to look backward at established molecules to validate new delivery architectures. Expanding the exenatide implant obesity market, these platforms command a 54.0% share because clinical sponsors require a thoroughly understood, off-patent active ingredient to prove the safety of their novel subdermal platforms. By de-risking the mechanical release profile first, developers isolate the regulatory variables, avoiding the compounding risk of testing a new delivery system with a novel biologic. This approach allows clinical investigators to focus entirely on tissue tolerability and zero-order release kinetics rather than unexpected molecular toxicities. Formulators who attempt to skip this step and immediately target the semaglutide implant obesity market encounter severe regulatory holds. According to FMI's estimates, this systematic validation sequence will eventually support glp-1 nutritional support combinations in later generations.
The six-month GLP-1 implant for obesity holds a 58.0% share due to the way how medical billing and patient monitoring cycles are currently designed. This duration perfectly overlays existing bi-annual endocrinology checkup intervals, allowing clinics to bill for the procedure without disrupting established patient flow. Rather than attempting to push the biological limits of polymer degradation toward a theoretical once-yearly semaglutide implant, platform engineers optimized for this specific commercial window, balancing adequate payload capacity with device miniaturization. A six-month window also mitigates the risk of long-term tissue encapsulation that can unpredictably alter diffusion rates in longer-lasting formats. In FMI's view, the integration of glp-1 obesity home delivery injectables for breakthrough cravings remains an option, but the baseline therapy is locked. Procurement teams evaluating lifecycle economics heavily favor this duration as the optimal compromise between intervention frequency and clinical safety.
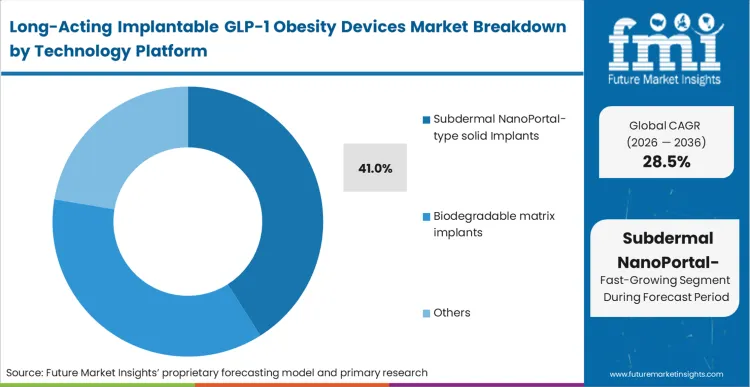
The clinical outcome of choosing an unpredictable degradation matrix is often severe localized inflammation and erratic dose dumping, which is why buyers strictly favor the reversible subdermal GLP-1 implant. Holding a 41.0% share, this Subdermal NanoPortal-type solid architecture utilizes precise, mechanically defined channels that guarantee a consistent release rate regardless of the patient's subcutaneous pH or metabolic fluctuations. The non-degrading nature of the titanium or stable polymer casing ensures the device remains intact, allowing for immediate retrieval if the patient experiences adverse gastrointestinal reactions. Based on FMI's assessment, the ability to halt therapy instantly is a mandatory safety requirement for broad regulatory approval in non-diabetic oral obesity therapies. Developers attempting to push fully resorbable matrices face extended clinical holds precisely because their systems lack this emergency reversal capability.
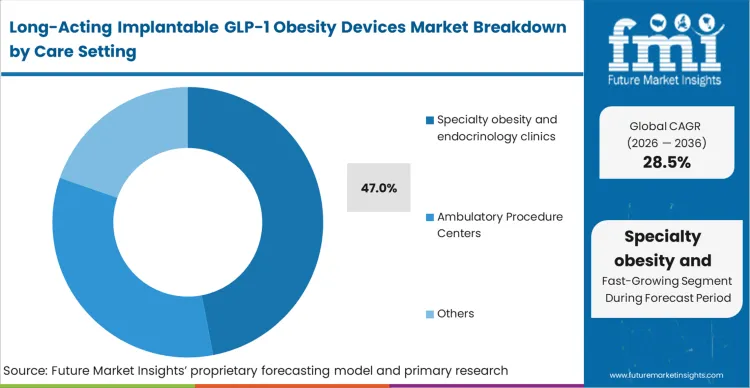
Administrators at specialty obesity and endocrinology clinics are currently deciding whether to adapt their facilities for minor surgical procedures or remain entirely prescription focused. This segment captures 47.0% share because these clinics already possess the specialized patient monitoring infrastructure required for long-acting metabolic interventions. They manage the specific complications associated with aggressive weight loss, and their staff is trained in sterile field protocols for subdermal insertions. Unlike general practitioners, these specialists understand how to transition patients from traditional obesity diabetes drugs to continuous-release systems without triggering overlapping toxicity. As per FMI's projection, the clinics that move first to qualify as certified implant centers capture a captive patient base that must return bi-annually. Facilities that delay this operational upgrade will rapidly lose their highest-value chronic care patients to competing procedural networks.
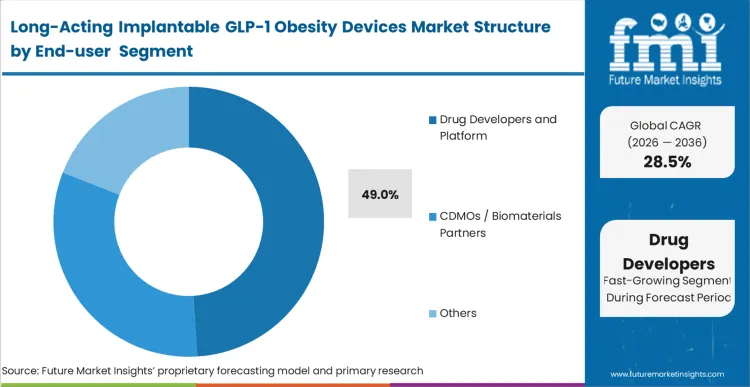
Drug developers and platform owners hold a commanding 49.0% share by aggressively securing the foundational intellectual property around continuous peptide diffusion. These long-acting GLP-1 implant manufacturers are not merely licensing technology; they are running the expensive first-in-human clinical trials that legally validate the implant's pharmacokinetic profile. Their dominance is a function of the high capital barrier required to merge advanced biomaterial science with complex controlled release drug delivery technology. By capturing the regulatory approvals, these platform owners dictate terms to GLP-1 implant CDMO partners and downstream clinical networks, locking in long-term value before a single commercial device is sold. FMI analysts opine that this concentration will persist until the first generation of patents expires. Entities failing to secure proprietary delivery mechanisms are relegated to low-margin contract manufacturing roles.
The pressure of massive real-world therapy abandonment compels managed care organizations and clinical networks to fundamentally rewrite their weight management coverage policies. Payers are no longer willing to underwrite the cost of high-frequency injectables when claims data proves that a majority of patients discontinue treatment before achieving clinically meaningful outcomes. This forces procurement directors to demand delivery formats that entirely remove the patient from the dosing equation. By transitioning the therapy from a patient-controlled pharmacy benefit to a physician-controlled procedural benefit, payers secure the 12-month persistence necessary to justify the GLP-1 implant reimbursement outlook, penalizing manufacturers who cannot offer continuous-delivery solutions.
The primary operational friction slowing adoption is the clinical workflow transition from prescription writing to minor surgical intervention. This friction is not about drug efficacy; it is a bottleneck caused by the lack of sterile procedural training and dedicated minor-surgery time slots within standard endocrinology practices. While manufacturers are attempting to solve this by developing highly automated, pre-loaded trocar systems that mimic the simplicity of large volume injectors, the requirement for local anesthesia and post-insertion site monitoring remains. Until billing codes fully compensate physicians for this additional procedural time, broad adoption outside of highly specialized metabolic clinics will remain constrained.
Regionally, the long‑acting implantable GLP‑1 obesity devices market is divided across North America, Europe, and Asia Pacific, with coverage extending to more than 40 countries.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| China | 32.4% |
| India | 31.6% |
| Australia | 31.2% |
| United States | 29.8% |
| United Kingdom | 28.6% |
| Germany | 27.9% |
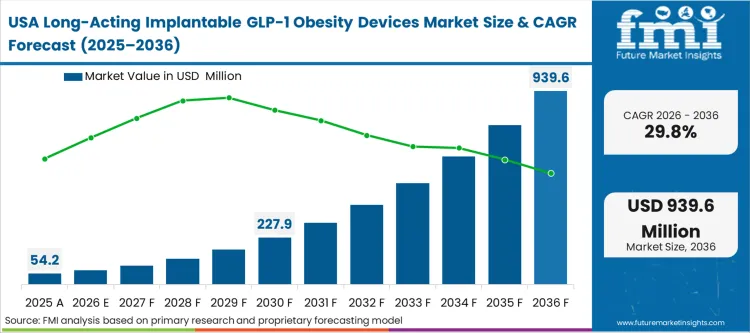
The regulatory environment in the United States fundamentally shapes how developers approach device approval, requiring simultaneous clearance of both the complex peptide and the novel delivery mechanism. FMI analysts opine that this dual-jurisdiction review process forces developers to conduct highly structured, phased trials that prioritize mechanical safety data over rapid commercialization. Unlike markets that view implants as simple reformulations, North American regulators treat them as entirely new therapeutic entities, demanding exhaustive proof of zero-order kinetics and fail-safe retrievability. While the integration of connected drug delivery devices for adherence tracking is popular in other sectors, the FDA focuses purely on the physical reliability of the subdermal platform.
FMI's report includes detailed analysis of the Canadian market. Canadian provincial health authorities are observing US clinical trials closely, preparing parallel procurement guidelines that prioritize single-intervention formats to reduce the burden on their publicly funded endocrinology infrastructure.
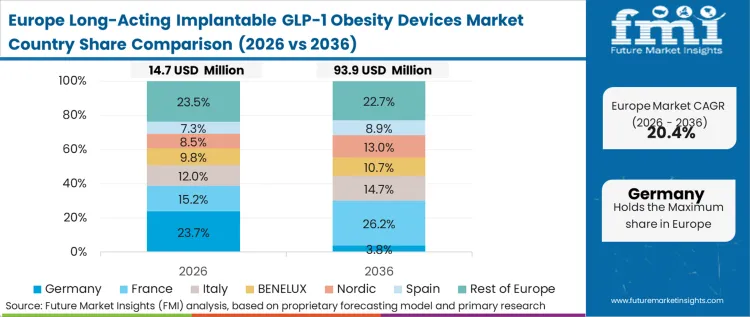
Europe's trajectory is defined by the maturity of its existing minor-surgical and specialized clinical infrastructure, which readily supports the transition from pharmacy-dispensed to physician-administered therapies. The physical layout and operational protocols of European metabolic centers are already optimized for minor interventions, meaning the bottleneck of training general practitioners is significantly reduced. Based on FMI's assessment, procurement frameworks in this region heavily emphasize total lifecycle costs, evaluating drug delivery technology based on its ability to reduce secondary clinical interventions and specialist visits.
FMI's report includes analysis of France, Italy, and Spain. These markets exhibit similar infrastructure advantages, with regional health authorities actively establishing standardized reimbursement codes specifically for minor subdermal drug delivery insertions to accelerate adoption.
Cost structures and massive patient-to-physician ratios dictate the adoption patterns across the Asia Pacific region, fundamentally separating it from Western regulatory-led dynamics. Public health systems here cannot mathematically support millions of adults requiring weekly monitoring and continuous pharmacy interventions. FMI estimates the region relies on the deployment of highly efficient implantable drug eluting devices to manage the sheer scale of the metabolic crisis. Single-intervention therapies are prioritized not as a premium convenience, but as a critical infrastructural necessity.
FMI's report includes analysis of South Korea and Southeast Asian hubs. These markets leverage their sophisticated medical tourism infrastructures to rapidly adopt and market these advanced metabolic procedures to regional patients seeking premium care.
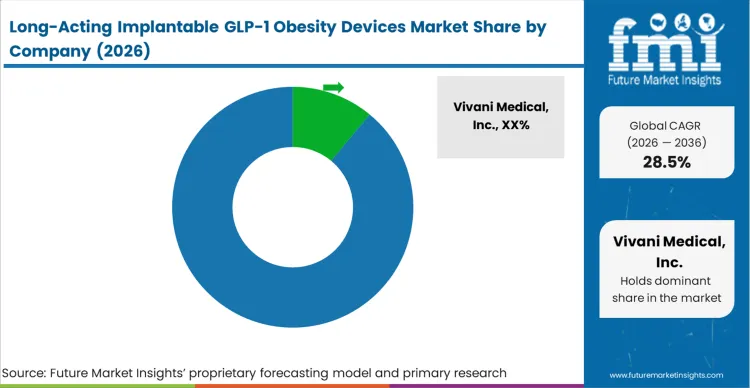
The competition in the sector is highly concentrated because the capital and technical barriers to fusing polymer science, mechanical engineering, and peptide stabilization are immense. When evaluating who is developing obesity GLP-1 implants, Vivani Medical, Inc., DelSiTech Ltd., and Delpor, Inc. dominate early positioning because buyers, in this case, clinical trial sponsors and eventual large-scale procurement networks, valuate vendors strictly on validated in-vivo pharmacokinetic data, not theoretical capabilities. Market participants cannot simply iterate on existing auto-injectors; they must prove zero-order release and tissue safety in human trials, a hurdle that immediately disqualifies generic delivery manufacturers from entering the space.
Incumbents hold a distinct advantage in their established cGMP biomaterial supply chains and proprietary channel architectures. Companies like Evonik Health Care, Corbion, and Ashland Inc. act as the premier semaglutide implant development partner for clinical-stage entities, maintaining precise control over the ultra-pure, absorbable polymers required to construct these matrices safely. For a challenger to disrupt this, they must build a completely parallel, bio-compatible materials ecosystem that navigates identical regulatory scrutiny without infringing on heavily guarded long acting implant and ocular drug delivery polymer systems patents. This reliance on highly specialized material science ensures that early leaders retain their competitive moat through the initial commercialization waves.
Through 2036, the tension between large biopharmaceutical buyers and independent platform owners will force a wave of vertical integration. Major drug developers seeking to protect their incretin franchises from generic competition will aggressively acquire the underlying delivery technologies rather than license them. The market will become increasingly consolidated as the line between wearable injectors and fully integrated, proprietary implant ecosystems blurs, preventing massive procurement networks from playing independent delivery providers against each other for pricing leverage.

| Metric | Value |
|---|---|
| Quantitative Units | USD 76.0 Million to USD 932.9 Million, at a CAGR of 28.50% |
| Market Definition | The long-acting implantable GLP-1 obesity devices market covers the proprietary subdermal delivery architectures, biomaterial platforms, and integrated device-drug combinations designed to release incretin therapies continuously over multiple months. |
| Molecule Type Segmentation | Exenatide implants, Semaglutide implants, Other GLP-1 / incretin implant candidates |
| Duration Profile Segmentation | 6-month implants, 12-month implants, Other multi-month implant formats |
| Technology Platform Segmentation | Subdermal NanoPortal-type solid implants, Biodegradable matrix implants, Passive reservoir-based implants |
| Care Setting Segmentation | Specialty obesity and endocrinology clinics, Ambulatory procedure centers, Hospital metabolic clinics |
| Regions Covered | North America, Europe, Asia Pacific |
| Countries Covered | China, India, Australia, United States, United Kingdom, Germany, Japan, and 40 plus countries |
| Key Companies Profiled | Vivani Medical, Inc., DelSiTech Ltd., Delpor, Inc., Evonik Health Care, Corbion, Ashland Inc., Poly-Med, Inc. |
| Forecast Period | 2026 to 2036 |
| Approach | Primary interviews targeted chief scientific officers and biomaterial engineering leads managing continuous-release platforms. The valuation baseline anchors directly to verified pure-play developer R&D expenditures. Independent validation cross-checked this data against massive real-world claims tracking current generation therapy abandonment rates. |
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary source documentation.
How large is the long-acting implantable GLP-1 obesity devices market in 2026?
The market is valued at USD 76.0 million in 2026. This relatively small initial figure signals that the sector is entirely in its precommercial, clinical-validation phase, driven by pipeline R&D expenditures rather than broad commercial sales.
What will it be valued at by 2036?
The valuation reaches USD 932.9 million by 2036. This massive expansion reflects the anticipated regulatory clearance and subsequent rapid commercial scaling of these devices into high-volume metabolic clinics.
What CAGR is projected?
A CAGR of 28.50% is projected from 2026 to 2036. This premium rate outpaces the broader metabolic drug sector because it is tied to the critical need to solve real-world adherence drop-offs, making it highly attractive to cost-conscious payers.
Which Molecule Type segment leads?
Exenatide implants lead the market by capturing 54.0% share. Developers rely on this deeply understood, legacy peptide to isolate and prove the mechanical safety of their novel subdermal delivery platforms before risking more complex incretins.
Which Duration Profile segment leads?
The 6-month implants segment leads with a 58.0% share. This duration is strictly aligned with the existing bi-annual endocrinology checkup and billing cycles, allowing clinics to integrate the procedure seamlessly without overhauling their patient flow.
Which Technology Platform segment leads?
Subdermal NanoPortal-type solid implants dominate with a 41.0% share. Buyers heavily favor these systems because their rigid structure guarantees predictable zero-order release kinetics and ensures the device can be easily retrieved during adverse events.
What drives rapid growth?
Growth is catalyzed by massive real-world therapy abandonment that destroys payer pharmacoeconomic models. Procurement directors are forcing the transition to these continuous-delivery systems to secure guaranteed 12-month persistence and eliminate the cost of wasted injectable pens.
What is the primary restraint?
The main bottleneck is the necessary workflow change from writing standard prescriptions to performing sterile, minor surgical interventions. Broad adoption is constrained by the lack of dedicated procedure time and specific billing codes within general endocrinology practices.
Which country grows fastest?
China expands at 32.4%, closely followed by India at 31.6%. The difference is that China's centralized public health apparatus views single-intervention formats as a systemic necessity to manage its sheer patient volume, whereas India's growth is driven by competitive differentiation among premium, out-of-pocket urban clinics.
Why are hospital metabolic clinics adopting these devices slower than specialty centers?
Hospital clinics operate under rigid, multi-departmental committee approvals that require exhaustive in-house cost-benefit analyses before introducing new surgical codes. Specialty outpatient centers possess the administrative agility to immediately redesign workflows and train staff for minor trocar insertions.
How does the solid titanium casing impact the regulatory pathway?
The solid, non-degrading casing dramatically shortens the FDA combination-product review cycle compared to fully resorbable matrices. Regulators prioritize the ability to halt therapy instantly, which is only possible when the intact device can be located and removed via a simple superficial incision.
Why is CDMO biomaterial integration so critical to competitive positioning?
Direct developers must prove their platforms do not cause severe localized tissue necrosis, which requires access to ultra-pure, cGMP-grade absorbable polymers. Companies that secure exclusive supply agreements for these highly specific materials effectively block generic competitors from replicating their delivery architecture.
How does the Australian regulatory environment shape its trajectory?
Australia possesses a highly streamlined clinical trial infrastructure that made it the optimal beachhead for first-in-human incretin implant studies. This early exposure ensures that Australian clinical networks will already have established insertion protocols and deep procedural familiarity when commercialization begins.
When could semaglutide implants launch broadly into the commercial market?
While exenatide trials are establishing the initial mechanical safety profiles, fully commercialized semaglutide equivalents are largely tracking toward the latter half of the forecast period once zero-order release kinetics are perfected for highly potent macro-molecules.
Why are wearable auto-injectors excluded from this scope?
Wearable auto-injectors rely on external mechanical or electronic propulsion and remain fundamentally patient-administered, short-term solutions. This report focuses strictly on subdermal, continuous-release architectures that change the treatment paradigm to physician-controlled, multi-month procedural interventions.
What happens if a patient experiences adverse gastrointestinal reactions to a biodegradable matrix implant?
Because fully degrading matrices lack a stable casing, locating and removing the active payload mid-cycle is exceptionally difficult and often surgically invasive. This residual risk is precisely why clinical sponsors currently favor solid, retrievable platforms for early commercialization.
How do German ambulatory centers impact regional adoption?
Germany's advanced ambulatory surgical infrastructure provides the exact physical environment needed to scale minor interventions rapidly outside of major hospitals. Clinic administrators use these specific procedural billing codes to capture recurring revenue streams, driving the region's 27.9% growth rate.
What drives the change from pharmacy benefit to procedural reimbursement?
Managed care actuaries recognize that relying on retail pharmacy refills for GLP-1s results in massive financial waste due to patient discontinuation. Transitioning to upfront procedural reimbursements ensures the payer actually receives the 12-month metabolic outcomes they are underwriting.
How do formulators use solid matrix stability to their advantage?
The lack of an aqueous environment in a solid matrix prevents fragile macro-molecules from degrading prematurely. Formulators exploit this physical property to engineer advanced, dual-action incretin combinations that would be chemically impossible to maintain in a traditional liquid pen.
What constraint defines the Japanese market's moderate 24.7% CAGR?
Japan operates with a materially lower baseline adult obesity burden compared to Western markets, completely removing the systemic urgency for population-wide procedural interventions. The market remains constrained to a highly profitable but low-volume niche catering to premium executive health clients and medical tourism.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.