GLP-1 Obesity Home Delivery Injectables Market
GLP-1 Obesity Home Delivery Injectables Market Size and Share Forecast Outlook 2026 to 2036
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
GLP-1 Obesity Home Delivery Injectables Market Forecast and Outlook 2026 to 2036
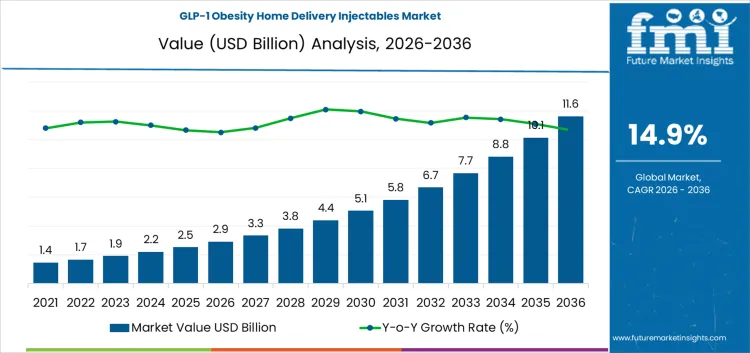
The GLP-1 obesity home delivery injectables market is likely to reach USD 2.9 billion in 2026 and is projected to reach USD 11.1 billion by 2036, expanding at a CAGR of 14.90% over the forecast period. Regulatory oversight plays a decisive role in shaping adoption patterns, as GLP-1 injectables are regulated biologics requiring strict compliance across prescribing, dispensing, storage, and home delivery channels. Vendors must navigate varying national frameworks governing cold-chain handling, remote prescribing, patient verification, and pharmacovigilance, which increases operational complexity and cost.
Compliance investments in temperature-controlled logistics, validated packaging, digital traceability, and patient training programs are becoming baseline requirements rather than differentiators. Certification complexity favors manufacturers and pharmacy partners with established regulatory expertise and scalable compliance systems, reinforcing the position of large pharmaceutical players and licensed specialty pharmacies. Smaller entrants face higher barriers due to audit intensity, documentation burdens, and cross-border regulatory fragmentation. As home delivery volumes grow, regulatory credibility increasingly determines market access, partnership eligibility, and long-term competitive positioning.
Quick Stats for GLP-1 Obesity Home Delivery Injectables Market
- GLP-1 Obesity Home Delivery Injectables Market Value (2026): USD 2.9 Billion
- GLP-1 Obesity Home Delivery Injectables Market Forecast Value (2036): USD 11.1 Billion
- GLP-1 Obesity Home Delivery Injectables Market Forecast CAGR 2026 to 2036: 14.90%
- Leading Molecule in GLP-1 Obesity Home Delivery Injectables Market: Semaglutide
- Leading Patient Type in GLP-1 Obesity Home Delivery Injectables Market: Chronic Obesity
- Key Growth Regions in GLP-1 Obesity Home Delivery Injectables Market: North America, Europe, Asia Pacific
- Top Key Players in GLP-1 Obesity Home Delivery Injectables Market: Novo Nordisk, Eli Lilly, AstraZeneca, Pfizer, Amgen, Roche
Metric
| Metric | Value |
|---|---|
| Industry Value (2026) | USD 2.9 Billion |
| Forecast Value (2036) | USD 11.1 Billion |
| Forecast CAGR 2026 to 2036 | 14.90% |
How Is the Global GLP-1 Obesity Home Delivery Injectables Market Advancing?
The global GLP-1 obesity home delivery injectables market is advancing rapidly, driven by the rising prevalence of obesity and the growing adoption of GLP-1 receptor agonists as long-term weight management therapies. Home delivery models for injectable GLP-1 treatments are gaining traction as patients increasingly seek convenient, consistent access to prescription therapies without frequent clinic visits.
A key factor supporting market growth is the expansion of digital healthcare platforms and pharmacy logistics networks that enable direct-to-patient delivery of temperature-sensitive injectable medications. These models improve treatment adherence by ensuring regular supply schedules and reducing barriers associated with in-clinic dispensing. In parallel, healthcare providers are increasingly integrating remote monitoring and teleconsultation services to support long-term GLP-1 therapy management in home settings.
Ongoing advancements in cold-chain packaging, last-mile delivery systems, and patient-friendly injectable devices are further strengthening market adoption. Improvements in prefilled pen designs, dosing accuracy, and storage stability are making home administration more accessible for a broader patient population. As obesity treatment increasingly shifts toward chronic care models supported by digital health infrastructure, the GLP-1 obesity home delivery injectables market is expected to maintain strong growth over the forecast period.
What Is the Segment-Wise Overview of the GLP-1 Obesity Home Delivery Injectables Market?
The GLP-1 obesity home delivery injectables market is shaped by molecule preference and patient treatment profiles. By molecule, semaglutide leads with a 42% share, supported by strong clinical efficacy, convenient dosing schedules, and wide physician adoption for weight management. Tirzepatide, liraglutide, and next-generation GLP-1s follow, addressing differentiated efficacy and metabolic profiles. By patient type, chronic obesity accounts for 54% of demand, reflecting long-term treatment adoption among patients requiring sustained weight control. Patients with combined obesity and diabetes, as well as diabetes overlap cases, contribute significantly to demand as GLP-1 therapies increasingly serve multi-indication management. The shift toward home delivery models further supports adherence and market expansion.
Why Does Semaglutide Lead the GLP-1 Obesity Home Delivery Injectables Market?
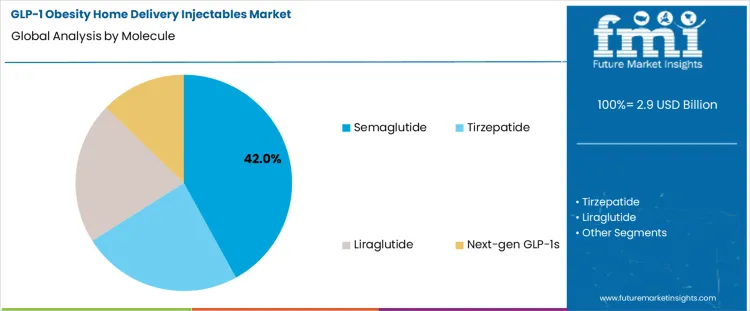
Semaglutide holds a 42% share of the GLP-1 Obesity Home Delivery Injectables Market due to its strong clinical performance, broad regulatory approvals, and patient-friendly dosing regimen. Its once-weekly injectable format supports higher adherence levels compared to older daily formulations, making it well suited for long-term obesity management in home-delivery settings. Semaglutide demonstrates consistent weight reduction outcomes, which has driven high physician confidence and patient acceptance. The molecule’s compatibility with prefilled pens and auto-injectors further supports efficient distribution through home delivery channels. In addition, strong manufacturing scale-up and supply chain investment by developers have improved availability across key regions. These combined factors position semaglutide as the preferred GLP-1 therapy for home-administered obesity treatment.
Why Do Chronic Obesity Patients Dominate the GLP-1 Obesity Home Delivery Injectables Market?
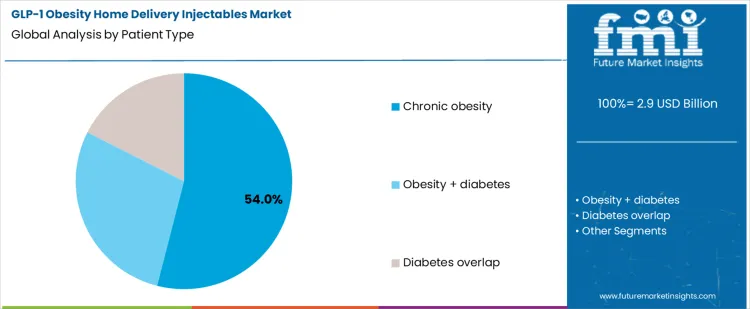
Chronic obesity patients account for 54% of total demand in the GLP-1 Obesity Home Delivery Injectables Market, driven by the need for sustained, long-term pharmacological management. Obesity is increasingly recognized as a chronic condition requiring continuous therapy rather than short-term intervention. GLP-1 injectables support appetite regulation and metabolic control over extended treatment periods, making them suitable for chronic patient populations. Home delivery models align well with these patients’ needs by ensuring consistent medication access, reducing clinic visits, and supporting adherence. As awareness of long-term obesity management grows among healthcare providers and patients, demand from chronic obesity populations continues to expand, reinforcing this segment’s leading position.
What Are the Key Dynamics for the GLP-1 Obesity Home Delivery Injectables Market?
The GLP-1 obesity home delivery injectables market is driven by increasing prevalence of obesity and related metabolic disorders, coupled with growing acceptance of glucagon-like peptide-1 (GLP-1) receptor agonist therapies as effective pharmacological treatment options. These injectables offer clinicians and patients a targeted approach to weight management by regulating appetite and improving glycemic control. Key market dynamics include rising demand for convenient home delivery healthcare solutions, supportive healthcare policies promoting chronic disease management outside of clinical settings, and enhanced patient adherence associated with at-home treatment regimens. As healthcare systems shift toward decentralized care models, demand for GLP-1 therapies delivered directly to patients’ residences continues to rise.
How Are the Key Drivers Shaping the GLP-1 Obesity Home Delivery Injectables Market?
Key drivers shaping the market include heightened clinical awareness of the benefits of GLP-1 receptor agonists in weight management and cardiometabolic risk reduction. Improvements in distribution logistics, supply chain integration, and temperature-controlled delivery solutions are enabling reliable home delivery services. Growing patient preference for at-home care options, driven by convenience and comfort, is motivating providers and pharmacies to develop robust delivery programs. Partnerships among pharmaceutical manufacturers, specialty pharmacies, and telehealth service providers are enhancing patient access and support services.
What Are the Challenges Impacting the GLP-1 Obesity Home Delivery Injectables Market?
Challenges in the market include high treatment costs, which can create barriers to access and limit widespread adoption, especially in regions with constrained healthcare spending. Complex cold-chain logistics and stringent storage requirements for biologic injectables present operational hurdles for home delivery networks. Patient education and training for safe self-administration are critical, and gaps in support may impact adherence and outcomes. Regulatory requirements for prescribing and dispensing controlled biologic therapies across different jurisdictions can also complicate delivery models. Insurance coverage variability and reimbursement limitations in certain markets may slow overall growth.
What Is the Growth Outlook for the GLP-1 Obesity Home Delivery Injectables Market by Country?
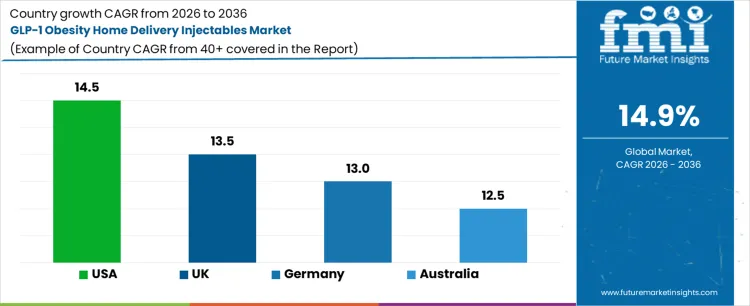
| Country | CAGR (%) |
|---|---|
| USA | 14.5% |
| UK | 13.5% |
| Germany | 13.0% |
| Australia | 12.5% |
The GLP-1 obesity home delivery injectables market is expanding rapidly as patients and healthcare providers adopt convenient, home-based treatment models. The USA leads at 14.5%, driven by high obesity prevalence, strong digital health integration, and mature specialty pharmacy networks. The UK follows at 13.5%, supported by online prescribing platforms and growing private weight-management services. Germany grows at 13.0%, benefiting from reliable digital pharmacy infrastructure and structured chronic care models. Australia expands at 12.5%, driven by telehealth adoption and demand for improved access across remote regions. As long-term obesity treatment increasingly relies on injectable therapies, home delivery models are expected to play a critical role in market expansion.
What Is Driving the Growth of the GLP-1 Obesity Home Delivery Injectables Market in the USA?
The GLP-1 obesity home delivery injectables market in the USA is growing at a CAGR of 14.5%, driven by strong demand for prescription weight-management therapies and rapid expansion of home-based treatment models. High obesity prevalence, increasing physician adoption of GLP-1 therapies, and broad patient acceptance of injectable self-administration are supporting market growth. Home delivery services are gaining traction as patients seek convenience, privacy, and uninterrupted therapy access without frequent clinic visits. The presence of large pharmaceutical manufacturers, specialty pharmacies, and digital health platforms enables efficient cold-chain distribution and prescription fulfillment. In addition, insurance coverage expansion and employer-sponsored health programs are improving therapy accessibility. Telehealth integration with prescription management further supports recurring home delivery models. As long-term obesity management increasingly shifts toward chronic therapy supported by digital care pathways, demand for home-delivered GLP-1 injectables in the USA is expected to remain strong.
How Is the GLP-1 Obesity Home Delivery Injectables Market Expanding in the UK?
The GLP-1 obesity home delivery injectables market in the UK is expanding at a CAGR of 13.5%, supported by rising obesity awareness and increasing use of pharmacological weight-management therapies. Patients are increasingly opting for home delivery services to simplify treatment adherence and reduce dependence on in-person pharmacy visits. Growth of online prescribing platforms and private weight-management clinics is improving access to GLP-1 therapies outside traditional hospital settings. In parallel, expanding digital health adoption is enabling remote consultations, prescription renewals, and doorstep delivery of injectable medications. The UK’s well-developed pharmacy distribution infrastructure supports reliable temperature-controlled delivery, ensuring product integrity. Increasing focus on preventive healthcare and long-term weight management is also driving physician confidence in GLP-1 therapy use. As healthcare delivery models continue to evolve toward patient-centric access, demand for home-delivered GLP-1 injectables in the UK is expected to grow steadily.
What Is Supporting Growth of the GLP-1 Obesity Home Delivery Injectables Market in Germany?
The GLP-1 obesity home delivery injectables market in Germany is growing at a CAGR of 13.0%, driven by rising prescription use of GLP-1 therapies and increasing acceptance of home-based medication delivery. German patients value structured, reliable healthcare services, and home delivery injectables align well with long-term obesity treatment plans requiring consistent dosing. Expansion of digital prescription services and licensed online pharmacies is making home delivery more accessible. Strong regulatory oversight ensures high standards in cold-chain logistics, supporting safe distribution of injectable therapies. Growing physician focus on metabolic disease management and preventative care is increasing GLP-1 therapy uptake. Germany’s aging population and increasing obesity-related health risks further support demand. As digital pharmacy services expand and patients seek convenience without compromising care quality, the home delivery GLP-1 injectables segment is expected to see sustained growth.
How Is the GLP-1 Obesity Home Delivery Injectables Market Developing in Australia?
The GLP-1 obesity home delivery injectables market in Australia is expanding at a CAGR of 12.5%, supported by increasing obesity rates and rising adoption of prescription-based weight-management therapies. Australia’s geographically dispersed population makes home delivery an attractive solution for improving therapy access, particularly outside major urban centers. Growth of telemedicine and digital prescribing platforms is enabling remote consultations and direct-to-home delivery of injectable treatments. Patients increasingly prefer home administration for long-term therapies due to convenience and reduced travel requirements. Australia’s well-established pharmaceutical distribution and cold-chain logistics networks support safe delivery of temperature-sensitive injectables. In addition, private healthcare providers and specialty pharmacies are expanding obesity-focused service offerings. As demand for long-term weight management solutions continues to rise, home delivery of GLP-1 injectables is expected to gain further traction across Australia.
How Are Companies Competing in the GLP-1 Obesity Home Delivery Injectables Market?
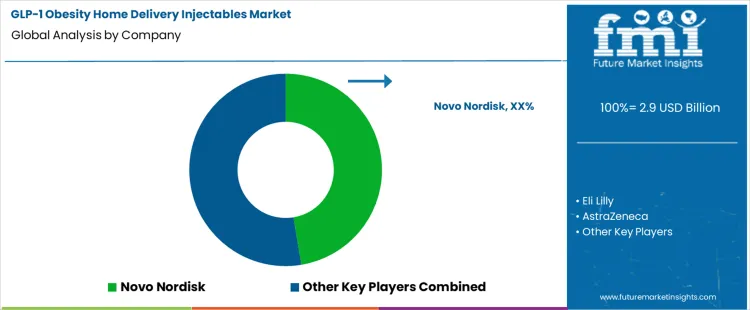
Competition in the GLP-1 obesity home delivery injectables market is driven by therapeutic efficacy, dosing convenience, supply reliability, and patient adherence in at-home use settings. As demand for GLP-1-based obesity therapies expands beyond clinic administration, manufacturers increasingly focus on prefilled pens, auto-injectors, and packaging formats that support safe, simple self-administration. Cold-chain integrity, consistent supply availability, and digital support for adherence and refill management have become critical differentiators as therapies scale into direct-to-patient delivery models.
Novo Nordisk and Eli Lilly lead competition through strong clinical efficacy profiles and advanced injection device ecosystems. Novo Nordisk emphasizes user-friendly pen designs, dose-flexibility, and large-scale manufacturing capacity to support widespread home delivery. Eli Lilly competes by pairing high-potency GLP-1 or dual-agonist formulations with streamlined auto-injector systems and expanding fill-finish capabilities to address global demand. AstraZeneca and Pfizer compete by leveraging broad pharmaceutical portfolios, formulation expertise, and global distribution networks to advance GLP-1 pipelines with an emphasis on long-term safety, differentiated dosing regimens, and scalable home-use formats.
Amgen and Roche compete through innovation in next-generation biologics and delivery optimization. Amgen focuses on extended-half-life and differentiated peptide or biologic approaches that may reduce injection frequency, improving adherence in home settings. Roche leverages its biologics manufacturing depth and diagnostics-driven care models to support patient monitoring and personalized treatment pathways. Across the market, competitive advantage is increasingly shaped by ease of self-administration, supply chain resilience, device reliability, and integrated patient-support ecosystems that sustain long-term treatment adherence in home-based obesity care.
Key Players in GLP-1 Obesity Home Delivery Injectables Market
- Novo Nordisk
- Eli Lilly
- AstraZeneca
- Pfizer
- Amgen
- Roche
Scope of Report
| Attributes | Description |
|---|---|
| Quantitative Unit (2026) | USD Billion |
| Molecule | Semaglutide, Tirzepatide, Liraglutide, Next-Gen GLP-1s |
| Patient Type | Chronic Obesity, Obesity + Diabetes, Diabetes Overlap |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Countries Covered | China, Japan, South Korea, India, Australia & New Zealand, ASEAN, Rest of Asia Pacific, Germany, United Kingdom, France, Italy, Spain, Nordic, BENELUX, Rest of Europe, United States, Canada, Mexico, Brazil, Chile, Rest of Latin America, Kingdom of Saudi Arabia, Other GCC Countries, Turkey, South Africa, Other African Union, Rest of Middle East & Africa |
| Key Companies Profiled | Novo Nordisk, Eli Lilly, AstraZeneca, Pfizer, Amgen, Roche |
| Additional Attributes | Dollar sales by molecule and patient type; regional market size and forecast analysis; growth outlook across major regions; adoption trends for home-delivered GLP-1 injectables; assessment of prescription expansion, patient adherence patterns, and demand dynamics across chronic obesity and obesity-diabetes overlap segments. |
GLP-1 Obesity Home Delivery Injectables Market by Key Segments
Molecule
- Semaglutide
- Tirzepatide
- Liraglutide
- Next-gen GLP-1s
Patient Type
- Chronic obesity
- Obesity + diabetes
- Diabetes overlap
Region
- Asia Pacific
- China
- Japan
- South Korea
- India
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Nordic
- BENELUX
- Rest of Europe
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Other African Union
- Rest of Middle East & Africa
Bibiliography
Therapeutic Goods Administration. (2024, September 4). AusPAR: Wegovy (semaglutide): Product information (PI) [PDF]. Australian Government Department of Health and Aged Care.
U.S. Food and Drug Administration, Center for Drug Evaluation and Research. (2023, November 8). NDA approval letter: Zepbound (tirzepatide) injection [PDF].
U.S. Food and Drug Administration, Center for Drug Evaluation and Research. (2024). WEGOVY (semaglutide) injection, for subcutaneous use: Prescribing information [Label].
U.S. Food and Drug Administration, Center for Drug Evaluation and Research. (2024, November 27). Prior approval sNDA letter: Wegovy (semaglutide) injection (labeling updates) [PDF].
U.S. Food and Drug Administration. (2024, July 26). FDA alerts health care providers, compounders, and patients of dosing errors associated with compounded injectable semaglutide products [Web page].
U.S. Food and Drug Administration. (2025, February 21). Declaratory order: Resolution of shortages of semaglutide injection products [PDF]
Frequently Asked Questions
How big is the glp-1 obesity home delivery injectables market in 2026?
The global glp-1 obesity home delivery injectables market is estimated to be valued at USD 2.9 billion in 2026.
What will be the size of glp-1 obesity home delivery injectables market in 2036?
The market size for the glp-1 obesity home delivery injectables market is projected to reach USD 11.6 billion by 2036.
How much will be the glp-1 obesity home delivery injectables market growth between 2026 and 2036?
The glp-1 obesity home delivery injectables market is expected to grow at a 14.9% CAGR between 2026 and 2036.
What are the key product types in the glp-1 obesity home delivery injectables market?
The key product types in glp-1 obesity home delivery injectables market are semaglutide, tirzepatide, liraglutide and next-gen glp-1s.
Which patient type segment to contribute significant share in the glp-1 obesity home delivery injectables market in 2026?
In terms of patient type, chronic obesity segment to command 54.0% share in the glp-1 obesity home delivery injectables market in 2026.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Molecule
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Molecule , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Molecule , 2026 to 2036
- Semaglutide
- Tirzepatide
- Liraglutide
- Next-gen GLP-1s
- Semaglutide
- Y to o to Y Growth Trend Analysis By Molecule , 2021 to 2025
- Absolute $ Opportunity Analysis By Molecule , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Patient Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Patient Type, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Patient Type, 2026 to 2036
- Chronic obesity
- Obesity + diabetes
- Diabetes overlap
- Chronic obesity
- Y to o to Y Growth Trend Analysis By Patient Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Patient Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Molecule
- By Patient Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Molecule
- By Patient Type
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Molecule
- By Patient Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Molecule
- By Patient Type
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Molecule
- By Patient Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Molecule
- By Patient Type
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Molecule
- By Patient Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Molecule
- By Patient Type
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Molecule
- By Patient Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Molecule
- By Patient Type
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Molecule
- By Patient Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Molecule
- By Patient Type
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Molecule
- By Patient Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Molecule
- By Patient Type
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Molecule
- By Patient Type
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Molecule
- By Patient Type
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Molecule
- By Patient Type
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Molecule
- By Patient Type
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Molecule
- By Patient Type
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Molecule
- By Patient Type
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Molecule
- By Patient Type
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Molecule
- By Patient Type
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Molecule
- By Patient Type
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Molecule
- By Patient Type
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Molecule
- By Patient Type
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Molecule
- By Patient Type
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Molecule
- By Patient Type
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Molecule
- By Patient Type
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Molecule
- By Patient Type
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Molecule
- By Patient Type
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Molecule
- By Patient Type
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Molecule
- By Patient Type
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Molecule
- By Patient Type
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Molecule
- By Patient Type
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Molecule
- By Patient Type
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Molecule
- By Patient Type
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Molecule
- By Patient Type
- Competition Analysis
- Competition Deep Dive
- Novo Nordisk
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Eli Lilly
- AstraZeneca
- Pfizer
- Amgen
- Roche
- Novo Nordisk
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Molecule , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Patient Type, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Molecule , 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Patient Type, 2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by Molecule , 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Patient Type, 2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by Molecule , 2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by Patient Type, 2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Molecule , 2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by Patient Type, 2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by Molecule , 2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by Patient Type, 2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Molecule , 2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by Patient Type, 2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Molecule , 2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by Patient Type, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Molecule , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Molecule , 2026 to 2036
- Figure 5: Global Market Attractiveness Analysis by Molecule
- Figure 6: Global Market Value Share and BPS Analysis by Patient Type, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Patient Type, 2026 to 2036
- Figure 8: Global Market Attractiveness Analysis by Patient Type
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Region, 2026 to 2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by Molecule , 2026 and 2036
- Figure 21: North America Market Y-o-Y Growth Comparison by Molecule , 2026 to 2036
- Figure 22: North America Market Attractiveness Analysis by Molecule
- Figure 23: North America Market Value Share and BPS Analysis by Patient Type, 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Patient Type, 2026 to 2036
- Figure 25: North America Market Attractiveness Analysis by Patient Type
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by Molecule , 2026 and 2036
- Figure 28: Latin America Market Y-o-Y Growth Comparison by Molecule , 2026 to 2036
- Figure 29: Latin America Market Attractiveness Analysis by Molecule
- Figure 30: Latin America Market Value Share and BPS Analysis by Patient Type, 2026 and 2036
- Figure 31: Latin America Market Y-o-Y Growth Comparison by Patient Type, 2026 to 2036
- Figure 32: Latin America Market Attractiveness Analysis by Patient Type
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by Molecule , 2026 and 2036
- Figure 35: Western Europe Market Y-o-Y Growth Comparison by Molecule , 2026 to 2036
- Figure 36: Western Europe Market Attractiveness Analysis by Molecule
- Figure 37: Western Europe Market Value Share and BPS Analysis by Patient Type, 2026 and 2036
- Figure 38: Western Europe Market Y-o-Y Growth Comparison by Patient Type, 2026 to 2036
- Figure 39: Western Europe Market Attractiveness Analysis by Patient Type
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Molecule , 2026 and 2036
- Figure 42: Eastern Europe Market Y-o-Y Growth Comparison by Molecule , 2026 to 2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by Molecule
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by Patient Type, 2026 and 2036
- Figure 45: Eastern Europe Market Y-o-Y Growth Comparison by Patient Type, 2026 to 2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by Patient Type
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by Molecule , 2026 and 2036
- Figure 49: East Asia Market Y-o-Y Growth Comparison by Molecule , 2026 to 2036
- Figure 50: East Asia Market Attractiveness Analysis by Molecule
- Figure 51: East Asia Market Value Share and BPS Analysis by Patient Type, 2026 and 2036
- Figure 52: East Asia Market Y-o-Y Growth Comparison by Patient Type, 2026 to 2036
- Figure 53: East Asia Market Attractiveness Analysis by Patient Type
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Molecule , 2026 and 2036
- Figure 56: South Asia and Pacific Market Y-o-Y Growth Comparison by Molecule , 2026 to 2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Molecule
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by Patient Type, 2026 and 2036
- Figure 59: South Asia and Pacific Market Y-o-Y Growth Comparison by Patient Type, 2026 to 2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by Patient Type
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Molecule , 2026 and 2036
- Figure 63: Middle East & Africa Market Y-o-Y Growth Comparison by Molecule , 2026 to 2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Molecule
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by Patient Type, 2026 and 2036
- Figure 66: Middle East & Africa Market Y-o-Y Growth Comparison by Patient Type, 2026 to 2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by Patient Type
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis

