Specialty Pharma Home Delivery and Patient Support Logistics Market
The specialty pharma Home delivery and patient support logistics market is segmented by Service Type (Home delivery, Cold-chain logistics, Nursing support, Refill management, Adherence support), Therapy Area (Oncology, Immunology, Rare disease, Neurology, Pulmonology), Delivery Mode (Direct-to-patient, Provider-office, Home infusion, White-glove, Scheduled refill), Customer Type (Manufacturers, Specialty pharmacies, Health systems, Payers, Providers), Support Function (Hub services, Benefits verification, Prior authorization, Copay assistance, Care coordination), and Region. Forecast for 2026 to 2036.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Specialty Pharma Home Delivery and Patient Support Logistics Market Size, Market Forecast and Outlook By FMI
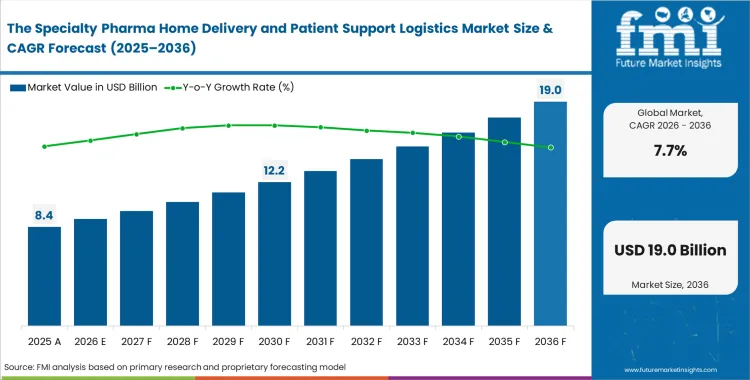
The specialty pharma home delivery and patient support logistics market was valued at USD 7.8 billion in 2025 and is expected to reach USD 8.4 billion in 2026, rising at a CAGR of 7.7% over the forecast period. Continued investment is set to drive total valuation to USD 17.7 billion by 2036, supported by a clear shift in payer strategy to move high-cost therapies out of hospital outpatient settings and into home-based care.
Summary of Specialty Pharma Home Delivery and Patient Support Logistics Market
- Market Snapshot
- The specialty pharma home delivery and patient support logistics market is valued at USD 7.80 billion in 2025 and is projected to reach USD 17.70 billion by 2036.
- The market is expected to grow at a 7.7% CAGR from 2026 to 2036, creating an incremental opportunity of USD 9.30 billion.
- This is a compliance-heavy healthcare services market that covers specialty medication fulfillment, temperature-controlled last-mile delivery, hub coordination, and patient access support rather than the invoice value of the drugs themselves.
- The operating model is shaped by accreditation, reimbursement workflow complexity, cold-chain execution, and coordination among physicians, pharmacies, payers, nurses, and patients.
- Demand and Growth Drivers
- Demand is rising because specialty drugs increasingly require special handling, storage, dispensing, and disease-management workflows, which pushes dispensing toward accredited specialty channels.
- Growth is also supported by home infusion and home IVIG care, where CMS defines delivery, pumps, supplies, nursing, training, and monitoring as linked parts of the service bundle.
- Patient support logistics are becoming more important because over half of new prescriptions for novel medicines go unfilled, increasing the value of benefits investigation, prior authorization support, refill coordination, and adherence programs.
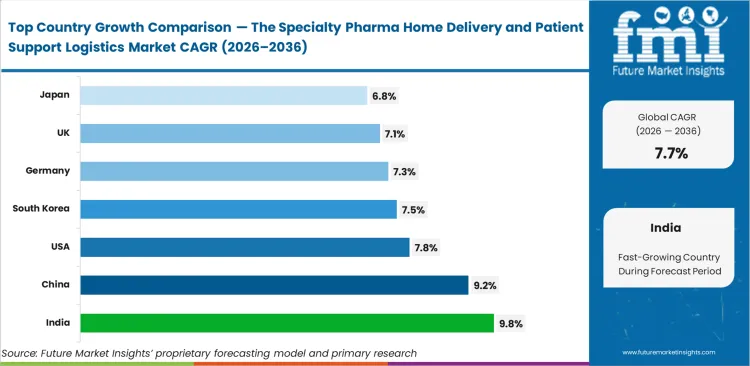
- Among key countries, India leads at 9.8% CAGR, followed by China at 9.2%, the United States at 7.8%, South Korea at 7.5%, Germany at 7.3%, the United Kingdom at 7.1%, and Japan at 6.8%.
- Growth is moderated by payer controls, prior-authorization friction, accreditation costs, temperature-control compliance, and the capital intensity of national home-delivery networks.
- Product and Segment View
- The market covers home delivery, cold-chain handling, nursing support, refill management, adherence support, and hub services for specialty therapies across oncology, immunology, rare disease, neurology, and pulmonology.
- Home delivery leads the Service Type segment with 36.8% share because high-cost specialty therapies require scheduled, traceable delivery to the patient or caregiver.
- Oncology leads the Therapy Area segment with 29.4% share because specialty pharmacy and hub workflows remain especially concentrated around high-cost oncology regimens and support programs.
- Direct-to-patient leads the Delivery Mode segment with 42.1% share because accredited specialty and mail channels increasingly ship directly to patient homes under managed scheduling.
- Manufacturers lead the Customer Type segment with 38.2% share because drug makers fund hub services, access support, and patient onboarding for complex specialty brands.
- Hub services lead the Support Function segment with 33.7% share because patient onboarding, benefits checks, prior authorization, and copay coordination remain central workflow bottlenecks.
- The scope includes specialty pharmacy fulfillment, temperature-controlled last mile, reimbursement support, and care coordination, but excludes the face value of drug sales, hospital inpatient pharmacy operations, and broad retail e-pharmacy transactions.
- Geography and Competitive Outlook
- The fastest-growing national markets are India, China, and South Korea, with the United States being the largest and most mature base of demand since they already integrate home delivery and specialty pharmacy with infusion infrastructure at scale.
- Integrated distribution footprints, payer and PBM relationships, specialty pharmacy accreditation, hub-service capability, and home infusion reach are the determinants of competition.
- Some of the main players in the area of access support, specialty distribution, delivery, and infusion coordination are Cencora, McKesson, CVS Health, The Cigna Group, UnitedHealth Group, Cardinal Health, and Option Care Health. (SEC)
- The market is moderately fragmented because scale matters but no single player fully controls specialty home delivery, hub support, and infusion logistics globally.
Health plan administrators are under increasing pressure to rein in specialty drug costs, prompting a faster transition of infusion therapies from clinical settings to the home. Delaying this shift exposes sponsors to significant facility fees that can severely compress margins, particularly in rare disease treatments. Pharmaceutical logistics is evolving, as the focus which was once laid on bulk wholesale distribution, procurement teams are now prioritizing specialized delivery models. These last-mile platforms are designed to manage stringent chain-of-custody requirements, including ultra-low temperature handling, while also integrating complex processes such as benefits verification.
As pharmacies begin to integrate real-time benefit investigation with scheduling systems, patient onboarding timelines are shrinking considerably, from several weeks to just a matter of hours. Managed care organizations are reinforcing this shift by restricting prior authorizations for infusion therapies unless they are administered at home. Providers that are slow to adapt risk losing access to high-value chronic care contracts.
India is expected to lead expansion at a CAGR of 9.8%, driven by rapid expansion of digital pharmacy infrastructure that is improving access in underserved rural areas. China follows closely at 9.2%, supported by strong policy momentum encouraging the transition from hospital-based care to home settings. United States is projected to garner steady momentum at 7.8%, reflecting its well-established cold-chain capabilities and specialized logistics networks. South Korea is forecast to expand at 7.5%, underpinned by rising demand for specialty care services. Germany and the United Kingdom are expected to rise at 7.3% and 7.1%, respectively, due to aging populations and increasingly stringent payer controls. Japan is projected to expand at 6.8%, with slower reimbursement reform moderating the pace of home-delivery adoption in specialty pharmacy.
Segmental Analysis
Specialty Pharma Home Delivery and Patient Support Logistics Market Analysis by Service Type
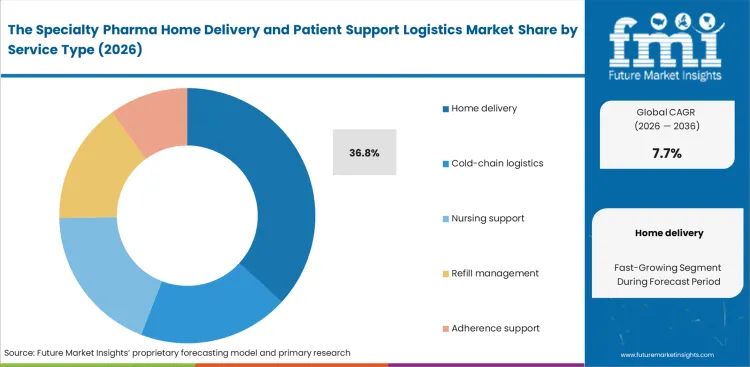
Procurement leaders within specialty pharmacies evaluate execution partners primarily on their ability to minimize therapy abandonment, particularly within the first 30 days of treatment. Home delivery accounts for a 36.8% share of the market, driven less by speed and tightly coordinated clinical drug supply chain execution. In specialty pharma, a delivery that misses the nursing schedule can delay administration, waste product, and put the provider relationship at risk. Health systems are therefore moving away from standalone delivery arrangements and placing more value on contracts that combine distribution with adherence support and patient monitoring.
- Delivery synchronization: Advanced logistics systems coordinate courier arrivals with scheduled nursing visits, helping pharmacy teams avoid missed administrations and costly drug wastage.
- Temperature compliance: Purpose-built pharma cold chain packaging ensures strict temperature control throughout transit, safeguarding high-value biologics from degradation and reducing financial risk.
- Abandonment prevention: Patient engagement teams connect with patients ahead of dispatch to confirm readiness, allowing care coordinators to reduce failed delivery attempts and improve therapy initiation rates.
Specialty Pharma Home Delivery and Patient Support Logistics Market Analysis by Therapy Area
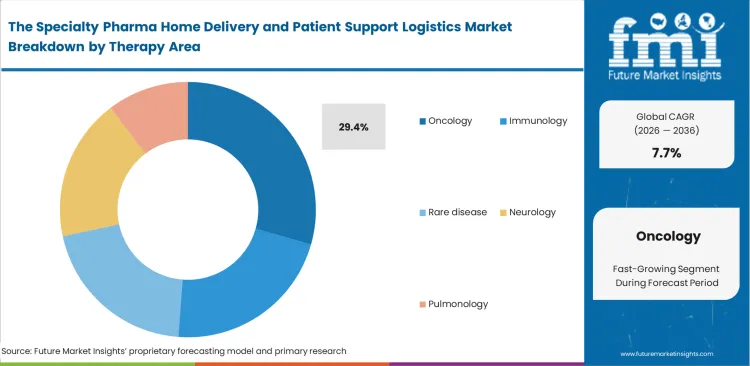
Targeted oral and injectable oncology therapies demand close clinical supervision even when administered outside hospital settings. Managing toxicity and monitoring side effects requires structured and continuous patient engagement, which keeps oncology at the forefront of this market, accounting for a 29.4% share. Specialized oncology nurses play a critical role in reviewing patient blood counts and treatment tolerance before pharmacies release the next cycle of oral therapies. An often overlooked factor is the impact of pharmaceutical packaging on adherence. Packaging design, such as advanced blister packs with embedded tracking, has a greater influence on patient compliance than standard counselling alone. Providers attempting to manage oncology drug delivery at home without robust temperature controlled packaging and integrated adherence systems face challenges in demonstrating outcomes, which can put value based reimbursement agreements at risk.
- Toxicity monitoring: Clinical teams assess patient symptoms and response before approving subsequent treatment cycles, helping to reduce the risk of serious adverse events during home-based care.
- Protocol verification: Automated platforms match laboratory results with prescribing guidelines, ensuring that treatment parameters are clinically appropriate before dispensing.
- Dose adjustment: Pharmacists tailor dispensing volumes based on real-time patient tolerance, allowing care teams to limit wastage while maintaining therapeutic effectiveness.
Specialty Pharma Home Delivery and Patient Support Logistics Market Analysis by Delivery Mode
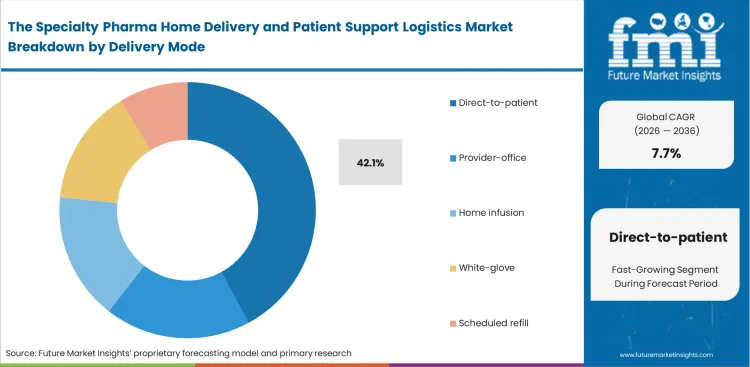
Infection control requirements and patient convenience now play a central role in shaping fulfillment models for chronic care populations. Direct-to-patient delivery continues to pull specialty dispensing away from centralized pharmacy pickup, especially for complex biologics that require tighter handling discipline and more predictable access for patients. FMI’s estimates indicate that this shift is raising operational expectations around transport packaging, with providers needing formats that meet medical compliance standards without creating added difficulty at the patient end.
Home delivery for specialty medications exposes a practical weakness in many current programs. Reusable biologic shippers may offer environmental benefits, yet elderly patients often find them difficult to open and burdensome to return after use. With direct-to-patient delivery expected to represent 42.1% of the market in 2026, those usability gaps become harder to absorb within routine operations. Fulfillment models built around sophisticated reusable systems rather than patient ease of handling often generate more complaints and greater product damage during the return cycle. Programs that fail to account for these patient-level constraints usually become harder to scale as volumes increase.
- Unpacking ergonomics: Engineering teams focus on lightweight packaging with intuitive tear-away features, while patient experience teams work to minimize frustration at the critical moment of first use.
- Return logistics: Integrated systems streamline the retrieval of reusable cooling components, enabling operations teams to recover valuable assets without placing additional burden on patients.
- Chain-of-custody transfer: Digital signature capture ensures verifiable proof of delivery, helping compliance teams meet strict payer and regulatory documentation standards.
Specialty Pharma Home Delivery and Patient Support Logistics Market Analysis by Customer Type
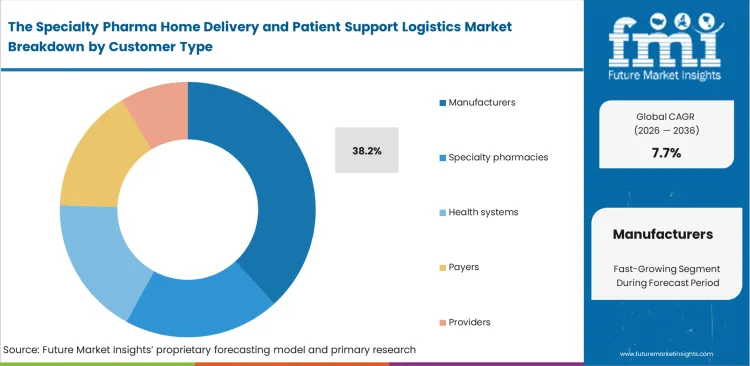
Avoidable logistical barriers carry direct financial consequences for treatment sponsors when patients discontinue rare disease therapies. Manufacturers are estimated to account for 38.2% of the market in 2026, reflecting investment in dedicated patient access networks built to protect treatment continuity and preserve revenue flow. That investment has become more necessary as traditional specialty pharmacy models show clear limits in therapies that rely on home infusion devices and tighter service coordination. Manufacturer-funded hub models therefore operate under a defined compliance constraint, since sponsors can finance access infrastructure but cannot engage patients too directly without raising anti-kickback concerns. Third-party logistics providers fill that operational gap by managing infusion equipment and coordinating clinical services, allowing them to capture a larger share of value across the delivery chain.
- Program funding: Drug sponsors finance end-to-end patient access programs, with brand teams focused on ensuring a smooth and timely therapy start to protect both uptake and brand reputation.
- Compliance firewalling: Independent hub providers administer financial assistance and support services, allowing legal and compliance teams to mitigate regulatory risk while still addressing the needs of underinsured patients.
- Outcome tracking: Specialized platforms collect real-world data from home-based therapy administration, giving market access teams the evidence needed to strengthen formulary negotiations and payer discussions.
Specialty Pharma Home Delivery and Patient Support Logistics Market Analysis by Support Function
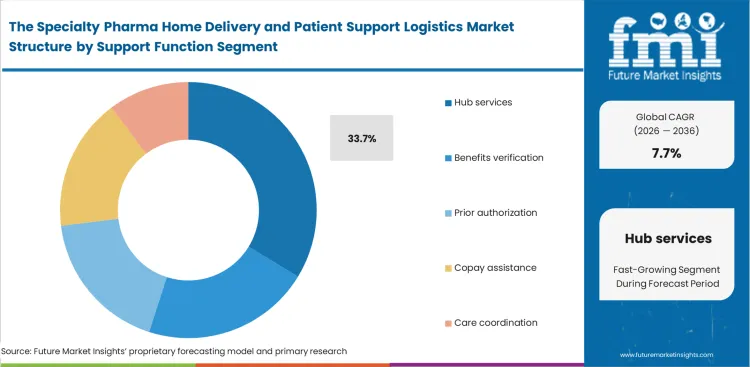
Financial barriers continue to be the most significant obstacle to patient access for prescribed biologics. FMI’s observes that approval for advanced infusion systems often determines the overall pace of treatment initiation. Even well-structured infusion pump rental programs can stall without rigorous benefit verification upfront. While general logistics providers tend to focus on shipment tracking as the core delivery challenge, hub services are predicted to account for a 33.7% share in 2026 by consolidating prior authorization and copay support into a unified process, reflecting where real value is created. Operators within specialty drug hub services understand that payer approval, such as overriding step-therapy requirements, is effectively the true point of delivery. Vendors without strong integration into pharmacy benefit manager systems frequently face delays exceeding twenty days, pushing patients toward alternative treatment options out of necessity.
- Benefit investigation: Automated systems quickly determine patient cost exposure, enabling access teams to move faster through financial clearance.
- Prior authorization: Tailored software aligns clinical documentation with payer-specific criteria, helping reimbursement teams lower denial rates.
- Financial assistance: Platforms direct eligible patients to copay support programs or charitable funding, allowing hub operators to remove financial barriers and support timely treatment initiation.
Specialty Pharma Home Delivery and Patient Support Logistics Market Drivers, Restraints, and Opportunities
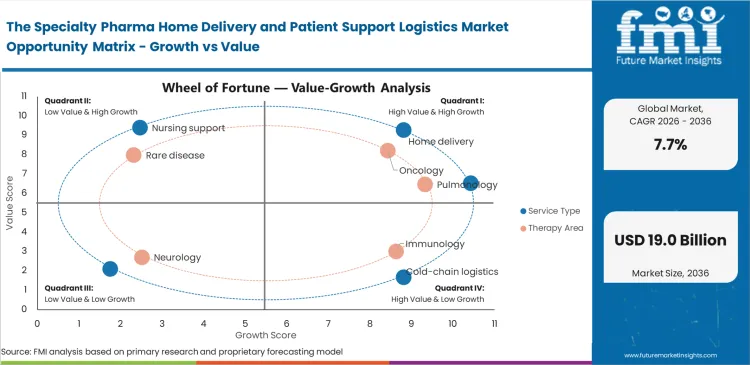
Health plan administrators refuse to authorize high-cost infusion therapies administered in hospital outpatient departments. They demand a transition to domestic settings to eliminate facility fees. This aggressive reimbursement pressure forces specialty pharmacy directors to rapidly construct decentralized clinical execution networks. Delaying this transition results in immediate claim denials and patient leakage to competing home health networks. Providers must integrate physical delivery with specialty pharmacy care coordination to capture this shifting volume. The urgency scales as novel cell and gene therapies receive approval, requiring hyper-coordinated telepharmacy service models to monitor severe adverse events remotely.
Benefit verification friction stalls therapy initiation despite robust physical logistics infrastructure. Medical benefit designs for rare disease therapeutics involve complex algorithms and step-therapy requirements that automated pharmacy benefit manager portals cannot process seamlessly. This structural barrier persists because payers intentionally design approval workflows to verify clinical necessity before releasing high-cost authorizations. Emerging artificial intelligence tools parse clinical notes to support prior authorization support logistics, but inconsistent payer integration limits widespread efficiency gains.
Opportunities in the Specialty Pharma Home Delivery and Patient Support Logistics Market
- Digital intake automation: Software developers build platforms bridging electronic medical records with hub service databases. Patient support hub service vendors cut onboarding time by eliminating manual data entry.
- Integrated epharmacy platforms: Network engineers design unified epharmacy systems linking prescription triage with cold-chain scheduling. Pharmacy directors achieve end-to-end visibility over the fulfillment lifecycle.
- Biometric adherence tracking: Clinical researchers deploy smart packaging that transmits usage data to care teams. Outcomes managers identify non-compliant patients before health deteriorates.
Regional Analysis
Based on regional analysis, Specialty Pharma Home Delivery and Patient Support Logistics Market is segmented into North America, Latin America, Western Europe, Eastern Europe, East Asia, South Asia and Pacific, and Middle East and Africa across 40 plus countries.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| India | 9.8% |
| China | 9.2% |
| United States | 7.8% |
| South Korea | 7.5% |
| Germany | 7.3% |
| United Kingdom | 7.1% |
| Japan | 6.8% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
South Asia and Pacific Specialty Pharma Home Delivery and Patient Support Logistics Market Analysis
Digital health companies are investing heavily in specialized fulfillment infrastructure to overcome the limitations of fragmented rural clinical networks. Urban specialty pharmacies are using these decentralized hubs to extend cold chain access into Tier-2 cities, where the burden of chronic diseases continues to rise. Government-led efforts to expand access to biopharmaceuticals are further encouraging private investment, particularly in last-mile tracking and monitoring capabilities. Companies are placing greater emphasis on mobile-first patient interfaces, favoring ease of access and real-time engagement over more complex desktop-based systems.
- India: Corporate hospital chains across India are tightening control over post-discharge oncology care by mandating in-house specialty pharmacy delivery models. To support this, operations managers are racing to localize automation systems for Indian prescription volumes, pricing structures, and distribution realities. These efforts are reshaping the market, pushing India’s specialty pharmacy logistics industry to expand at a 9.8% CAGR, as hospitals reclaim outpatient revenues that once flowed to fragmented, unorganized chemist networks.
FMI's report includes Australia, New Zealand, Indonesia, Thailand, and Malaysia. Pharmaceutical manufacturers launching orphan drugs across these fragmented geographies must partner with local third-party logistics providers who already possess validated temperature-mapping data for complex inter-island transit routes. Singapore also features prominently in the analysis, emerging as a regional cold-chain hub where cross-border clinical and commercial orphan drug flows are accelerating.
East Asia Specialty Pharma Home Delivery and Patient Support Logistics Market Analysis
The shift from hospital-based care to home-based treatment is reshaping how high-value therapeutics are distributed. Large medical centers are increasingly moving toward direct-to-patient delivery models, supported by regulatory changes that allow specialized couriers to operate from centralized clinical facilities. As these restrictions ease, traditional wholesale distributors are being pushed to develop their own direct delivery capabilities or risk losing relevance in a system that is rapidly decentralizing.
- China: Across China, regional health authorities are deliberately shifting long-term disease management out of overcrowded tertiary hospitals and into community clinics and home-based care models. This transition is supporting an estimated expansion of 9.2% CAGR in China’s specialty pharmacy delivery market by 2036, as logistics networks are reconfigured to handle frequent, low-volume shipments. Distribution systems are under strain, pushing logistics leaders to secure dedicated cold-chain capacity that can support the wider rollout of domestically approved biologics and the ongoing monitoring requirements tied to home-based post‑market surveillance programs.
- South Korea: Policy-driven expansion of national insurance coverage is reshaping how rare disease therapies reach patients, with care increasingly managed outside hospital boundaries. Pharmacy operators have responded by embedding digital adherence tools directly into local courier operations, reducing reliance on in-person pharmacy visits. This operational model is enabling the specialty pharmacy delivery segment in South Korea, which is estimated to rise at 7.5% CAGR through 2036, as providers offering integrated last-mile monitoring gain ground over those dependent on patient pickup.
- Japan: Japan’s pharmaceutical distribution landscape remains shaped by strict handling and documentation standards, which continue to limit the rapid adoption of fully outsourced, courier-led delivery models. Industry in Japan is expanding at an estimated 6.8% CAGR during the forecast period, as operators prioritize compliance and control over speed of scale. Rather than developing independent logistics platforms, many distribution leaders are choosing to work through long-established regional hospital and clinic networks, reinforcing a structural preference for institution-led oversight instead of aggressive third‑party expansion.
FMI's report includes Taiwan, Hong Kong, and Macau. Maturing reimbursement frameworks for advanced therapeutics are forcing logistics operators to closely synchronize physical drug delivery with localized digital health applications. South Korea is also highlighted in the report, as rising adoption of specialty biologics and payer-backed remote monitoring programs increase demand for tightly coordinated last‑mile distribution.
North America Specialty Pharma Home Delivery and Patient Support Logistics Market Analysis
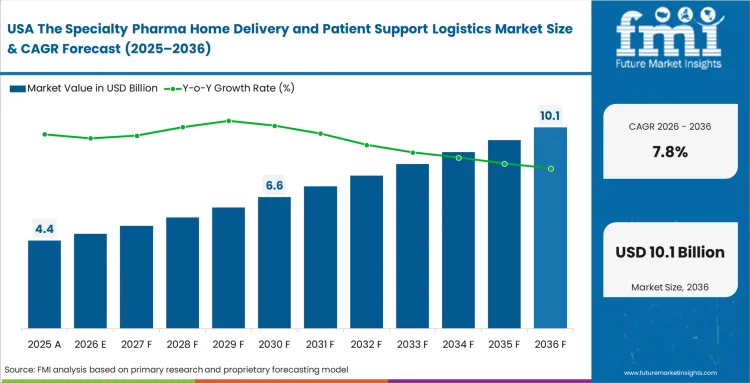
Established specialty pharmacy and home infusion networks are facing sustained margin pressure from the consolidation of pharmacy benefit managers. Growth opportunities are increasingly tied to securing contracts in rare and complex diseases, where service intensity is high and extends well beyond basic logistics. Independent infusion providers are managing to remain competitive by demonstrating stronger clinical outcomes and lower total treatment costs compared to hospital outpatient settings.
- United States: Escalating reimbursement scrutiny from payers is accelerating the move of immunology and neurology treatments out of hospital settings and into structured home‑infusion programs. This shift has expanded the role of care coordinators, who now operate at the center of benefit verification, scheduling, and last‑mile fulfillment. USA specialty pharmaceutical home delivery market is poised to advance at a 7.8% CAGR by 2036, supported by providers that deploy tightly integrated authorization and workflow platforms. By consistently securing approvals faster than hospital systems, these operators are locking in preferred and often exclusive distribution arrangements with therapy manufacturers.
FMI's report includes Canada and Mexico. Cross-border procurement strategies remain highly restricted due to divergent federal regulatory standards governing biologic chain‑of‑custody documentation. Germany also features in the assessment, where tightening serialization requirements and hospital-driven contracting models are reshaping specialty logistics partnerships.
Western Europe Specialty Pharma Home Delivery and Patient Support Logistics Market Analysis
Demographic and economic pressures are further accelerating this transition. Aging populations are driving demand for home-based care, while national healthcare systems continue to enforce tighter controls on specialty drug spending. In response, regional authorities are gradually approving decentralized fulfillment models for selected chronic conditions. Operators that succeed in this environment are those that can manage complex regulatory requirements across borders while ensuring uninterrupted access to therapies, particularly for patients relying on orphan drugs.
- Germany: In Germany, tight reimbursement rules for home-based therapies place a heavy documentation burden on providers, particularly around proof of adherence and handling integrity. To navigate this environment, logistics operators are embedding temperature validation and audit trails directly into physicians electronic medical record systems. Germany’s specialty pharmacy logistics sector is expected to record roughly 7.3% CAGR, as seamless data transfer shortens claims cycles and reduces payer friction for complex biologic therapies.
- United Kingdom: NHS policy is steadily redirecting long-duration treatment regimens away from acute hospital wards and into community care settings. This realignment is supporting a 7.1% CAGR in the UK specialty pharmacy delivery market, as fulfillment success increasingly depends on coordination rather than speed. By tightly matching delivery windows to district nurse visit schedules, providers are producing measurable service outcomes that strengthen their position in highly competitive regional tender processes.
FMI's report includes France, Italy, Spain, BENELUX, and the Nordics. The European Medicines Agency strictly enforces serialization requirements that complicate decentralized direct‑to‑patient distribution models. Poland is also covered in the report, where rising oncology drug volumes and hospital capacity constraints are opening selective opportunities for controlled home-delivery frameworks.
Competitive Aligners for Market Players
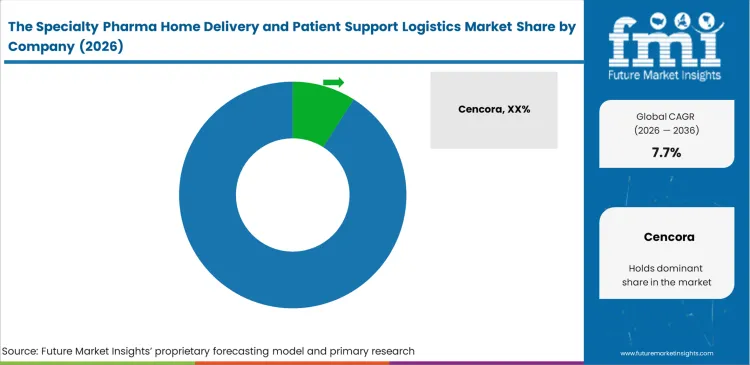
Network scale and the level of clinical integration increasingly determine which vendors remain competitive in the specialty delivery space. Companies such as Cencora and McKesson Corporation build on extensive wholesale networks while layering patient access programs to capture higher-margin specialty distribution. Pharmacy decision-makers assess these players not only on delivery reliability but also on how effectively they manage complex benefit investigations and specialty pharmacy workflows. UnitedHealth Group, through Optum Rx, integrates the full pathway from prescription to home infusion, creating a tightly controlled ecosystem that is difficult for smaller competitors to access. Competition is increasingly centered on securing exclusive distribution agreements for new gene therapies, where control over patient pathways and data is critical.
Renowned players benefit from long-standing integration with payer systems and electronic medical records, creating barriers that are difficult for new entrants to overcome. CVS Health and The Cigna Group use their pharmacy benefit manager platforms to streamline prior authorizations, significantly reducing delays in therapy initiation. Emerging players are investing in patient registry software and adherence tracking tools to demonstrate value to drug sponsors. Many independent specialty logistics providers sustain their position by partnering with niche hub service operators, assembling flexible service models that replicate key capabilities of larger integrated systems without requiring similar capital investment.
Large pharmaceutical sponsors remain cautious about concentrating their rare disease portfolios within a single integrated network, as this can limit negotiating flexibility and reduce visibility into patient data. This creates space for focused providers such as Option Care Health, which offer specialized infusion services across multiple payer networks without exclusivity constraints. Companies that embed predictive analytics and home healthcare software into their delivery and routing models are beginning to differentiate themselves more clearly. Sponsors are evaluating specialty pharmacy logistics partners based on their ability to show that operational execution directly contributes to improved patient outcomes, particularly by reducing avoidable hospitalizations.
Key Players in Specialty Pharma Home Delivery and Patient Support Logistics Market
- Cencora
- McKesson Corporation
- CVS Health
- The Cigna Group
- UnitedHealth Group
- Cardinal Health
- Option Care Health
Scope of the Report
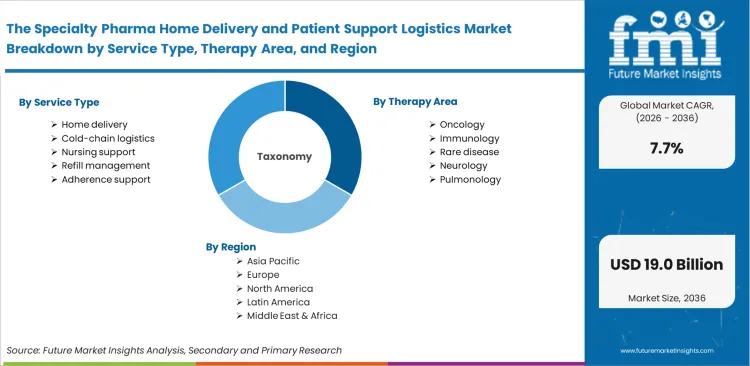
| Metric | Value |
|---|---|
| Quantitative Units | USD 8.4 billion to USD 17.7 billion, at a CAGR of 7.7% |
| Market Definition | Patient support logistics defines the operational architecture linking high-value therapeutics from central pharmacies directly to patient residences. Vendors execute specialized protocols including hub services, benefits verification, and targeted nursing support for complex regimens. |
| Segmentation | By Service Type, Therapy Area, Delivery Mode, Customer Type, Support Function, and Region |
| Regions Covered | North America, Latin America, Western Europe, Eastern Europe, East Asia, South Asia and Pacific, Middle East and Africa |
| Countries Covered | United States, Germany, United Kingdom, China, Japan, South Korea, India |
| Key Companies Profiled | Cencora, McKesson Corporation, CVS Health, The Cigna Group, UnitedHealth Group, Cardinal Health, Option Care Health |
| Forecast Period | 2026 to 2036 |
| Approach | Top-down and bottom-up methodology utilizing CMS reimbursement codes and home infusion claim volumes. |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Specialty Pharma Home Delivery and Patient Support Logistics Market Analysis by Segments
Service Type:
- Home delivery
- Cold-chain logistics
- Nursing support
- Refill management
- Adherence support
Therapy Area:
- Oncology
- Immunology
- Rare disease
- Neurology
- Pulmonology
Delivery Mode:
- Direct-to-patient
- Provider-office
- Home infusion
- White-glove
- Scheduled refill
Customer Type:
- Manufacturers
- Specialty pharmacies
- Health systems
- Payers
- Providers
Support Function:
- Hub services
- Benefits verification
- Prior authorization
- Copay assistance
- Care coordination
Region:
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East and Africa
Bibliography
- Centers for Medicare & Medicaid Services. (2026, February 12). Home Infusion Therapy/Home IVIG Services.
- IQVIA Institute for Human Data Science. (2025, April 30). Understanding the Use of Medicines in the USA 2025.
- National Association of Boards of Pharmacy. (n.d.). Specialty Pharmacy Accreditation | Eligibility.
- The Cigna Group. (2026, February 26). Annual report on Form 10-K for the fiscal year ended December 31, 2025.
- UnitedHealth Group. (2026, March 2). Annual report on Form 10-K for the fiscal year ended December 31, 2025.
- URAC. (2025, October 2). URAC Updates Pharmacy Programs.
- Zuckerman, A. D., et al. (2025). Capturing Value: How Health-System Specialty Pharmacies Define and Document Pharmacist Interventions. Pharmacy, 13(6), 172.
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary source documentation.
This Report Addresses
- Payer reimbursement mandates driving the transition of infusion protocols from clinical facilities to domestic care settings.
- Integration requirements combining cold-chain scheduling with specialized nursing visits to prevent costly dose waste.
- Compliance bottlenecks surrounding prior authorizations and copay assistance programs that dictate therapy initiation times.
- Ergonomic design necessities for direct-to-patient transport boxes to ensure adherence among elderly populations.
- Structural differences in national health policies shaping specialized fulfillment adoption across East Asia and Western Europe.
- Liability distribution strategies pushing manufacturers to fund independent hub services as compliance firewalls.
- Incumbent advantages linked to deep electronic medical record and pharmacy benefit manager database integrations.
- Emerging automation capabilities leveraging clinical notes to accelerate complex benefit verification algorithms.
Frequently Asked Questions
What is specialty pharma home delivery and patient support logistics?
It defines the operational architecture linking high-value therapeutics from central pharmacies directly to patient residences. Vendors combine cold-chain custody, benefit navigation, and nursing coordination to ensure complex therapies reach beneficiaries safely and affordably.
How big is the specialty pharma home delivery sector?
The sector was valued at USD 7.8 billion in 2025 and is projected to reach USD 17.7 billion by 2036. This growth is sustained by a 7.7% CAGR as payers aggressively force high-cost therapeutics out of hospital outpatient departments.
What services are included in patient support logistics?
Operations encompass direct-to-patient fulfillment, cold-chain handling, and white-glove coordination for advanced therapies. Vendors integrate clinical coordination teams to handle prior authorizations, benefit verification, and specialized nursing support for complex regimens.
Why are specialty drugs delivered to patients at home?
Health plan administrators refuse to authorize high-cost infusion therapies administered in hospital outpatient facilities to eliminate facility fees. Delivering directly to patients meets convenience expectations while preventing severe infection risks associated with central pharmacy pickups for immunocompromised populations.
How does specialty pharmacy home delivery work?
Logistics software perfectly aligns courier arrival times with specialized nursing visits to prevent dose spoilage. Access coordinators confirm patient readiness before dispatch, while specialized temperature-controlled packaging maintains tight thermal bands during the entire transit sequence.
What is a specialty pharmacy hub service?
Hub services centralize prior authorization and copay assistance into a single automated workflow. Independent hubs act as compliance firewalls, executing financial assistance programs while preventing regulatory violations between the manufacturer and the patient.
How do hub services improve specialty drug access?
Securing coverage dictates the entire timeline of care. Hub operators use specialized software to format clinical documentation to match specific payer algorithms, rapidly clearing step-therapy requirements and eliminating financial barriers to therapy initiation.
Which companies lead patient support logistics for specialty drugs?
Incumbents like Cencora, McKesson Corporation, CVS Health, and UnitedHealth Group anchor operations on massive wholesale and pharmacy benefit manager infrastructure. Focused players like Option Care Health thrive by offering neutral, specialized infusion capabilities across multiple payer networks.
Compare specialty pharmacy logistics with standard pharma logistics?
Standard logistics centers purely on bulk wholesale movement and general commercial freight handling. Specialty operations require regulatory chain-of-custody protocols, real-time temperature tracking, and patient-facing clinical coordination teams handling prior authorizations.
Is home infusion part of specialty pharma logistics?
Yes, home infusion functions as a core delivery mode within the operational architecture. Network design leaders must integrate clinical nursing support directly with last-mile courier schedules to prevent administration delays for complex rare disease regimens.
What limits adoption speed in this sector?
Complex benefit verification structures act as the primary constraint. Providers lacking deep integration with pharmacy benefit manager databases face extensive initiation delays, forcing patients to abandon high-cost therapeutic regimens regardless of underlying physical courier capabilities.
Why does home delivery command the largest share?
Procurement directors prioritize home delivery at 36.8% because it synchronizes physical transport directly with nursing schedules. Guaranteeing that the drug and the clinician arrive concurrently prevents immediate dose spoilage and secures chronic care contract renewals.
What structural gate triggers self-reinforcing expansion?
Expansion accelerates once a specialty pharmacy links real-time benefit investigation with final-mile tracking. Achieving this integration drops patient onboarding time from weeks to hours, allowing providers to capture exclusive distribution rights from manufacturers.
How does direct-to-patient fulfillment change operational metrics?
Shifting to direct channels forces logistics managers to prioritize unpacking ergonomics over bulk transit efficiency. Beneficiaries must successfully interact with complex packaging, meaning return logistics systems dictate overall program profitability.
Why do manufacturers maintain a heavy presence in this space?
Drug sponsors secure 38.2% share by funding dedicated networks to protect massive investments in rare disease portfolios. Utilizing third-party logistics providers allows manufacturers to clear financial access barriers while complying strictly with anti-kickback statutes.
What distinguishes India's growth trajectory?
India leads at 9.8% due to the rapid scaling of digital pharmacy networks addressing severe rural access gaps. Corporate hospital chains deploy decentralized clinical hubs to retain oncology patients migrating away from unorganized local distribution points.
How does oncology shape specialized fulfillment?
Oncology requires intense clinical oversight, driving a 29.4% share. Specialized nurses must verify patient blood counts before releasing subsequent medication cycles, necessitating sophisticated adherence software linking physical delivery to real-time toxicity monitoring.
What prevents hospital systems from dominating home infusion?
Hospital outpatient departments face immense reimbursement pressure as payers aggressively deny high-cost facility fees. Independent specialty providers outmaneuver hospitals by demonstrating equivalent clinical outcomes at significantly lower total care costs in domestic settings.
Why is benefit investigation critical to delivery success?
Securing coverage for advanced infusion systems dictates the entire care timeline. Logistics firms relying solely on transit tracking fail because securing a payer override on a step-therapy requirement constitutes the actual operational delivery mechanism.
What role do hub services play in patient retention?
Hub services centralize prior authorization and financial assistance workflows. Access coordinators using these platforms accelerate financial clearance, preventing script abandonment during the vulnerable thirty-day period following initial diagnosis.
How do European reimbursement models affect operations?
Western European growth remains steady around 7.1% to 7.3% because national health directives prioritize community-based administration to clear hospital beds. Fulfillment directors win regional tenders by aligning delivery schedules explicitly with district nursing visits.
What capability defines competitive advantage for incumbents?
Incumbents leverage deep, established connections with payer databases to automate prior authorizations. Removing days of friction from the initiation timeline provides an unassailable advantage over regional challengers relying on manual data entry processes.
Why do sponsors avoid single integrated health networks?
Large manufacturers resist locking rare disease portfolios into unified networks to maintain negotiating leverage. Distributing volume across specialized, neutral infusion providers ensures sponsors retain granular visibility over patient data and adherence metrics.
What structural difference explains China's high growth?
China tracks at 9.2% as provincial health authorities actively dismantle restrictive dispensing regulations. Allowing specialized couriers to bypass congested urban hospitals and deliver directly from central repositories fundamentally reshapes regional distribution models.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Service Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Service Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Service Type , 2026 to 2036
- Home delivery
- Cold-chain logistics
- Nursing support
- Refill management
- Adherence support
- Home delivery
- Y to o to Y Growth Trend Analysis By Service Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Service Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Therapy Area
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Therapy Area, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Therapy Area, 2026 to 2036
- Oncology
- Immunology
- Rare disease
- Neurology
- Pulmonology
- Oncology
- Y to o to Y Growth Trend Analysis By Therapy Area, 2021 to 2025
- Absolute $ Opportunity Analysis By Therapy Area, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Delivery Mode
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Delivery Mode, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Delivery Mode, 2026 to 2036
- Direct-to-patient
- Provider-office
- Home infusion
- White-glove
- Scheduled refill
- Direct-to-patient
- Y to o to Y Growth Trend Analysis By Delivery Mode, 2021 to 2025
- Absolute $ Opportunity Analysis By Delivery Mode, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Customer Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Customer Type, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Customer Type, 2026 to 2036
- Manufacturers
- Specialty pharmacies
- Health systems
- Payers
- Providers
- Manufacturers
- Y to o to Y Growth Trend Analysis By Customer Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Customer Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Support Function
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Support Function, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Support Function, 2026 to 2036
- Hub services
- Benefits verification
- Prior authorization
- Copay assistance
- Care coordination
- Hub services
- Y to o to Y Growth Trend Analysis By Support Function, 2021 to 2025
- Absolute $ Opportunity Analysis By Support Function, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Service Type
- By Therapy Area
- By Delivery Mode
- By Customer Type
- By Support Function
- By Country
- Market Attractiveness Analysis
- By Country
- By Service Type
- By Therapy Area
- By Delivery Mode
- By Customer Type
- By Support Function
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Service Type
- By Therapy Area
- By Delivery Mode
- By Customer Type
- By Support Function
- By Country
- Market Attractiveness Analysis
- By Country
- By Service Type
- By Therapy Area
- By Delivery Mode
- By Customer Type
- By Support Function
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Service Type
- By Therapy Area
- By Delivery Mode
- By Customer Type
- By Support Function
- By Country
- Market Attractiveness Analysis
- By Country
- By Service Type
- By Therapy Area
- By Delivery Mode
- By Customer Type
- By Support Function
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Service Type
- By Therapy Area
- By Delivery Mode
- By Customer Type
- By Support Function
- By Country
- Market Attractiveness Analysis
- By Country
- By Service Type
- By Therapy Area
- By Delivery Mode
- By Customer Type
- By Support Function
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Service Type
- By Therapy Area
- By Delivery Mode
- By Customer Type
- By Support Function
- By Country
- Market Attractiveness Analysis
- By Country
- By Service Type
- By Therapy Area
- By Delivery Mode
- By Customer Type
- By Support Function
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Service Type
- By Therapy Area
- By Delivery Mode
- By Customer Type
- By Support Function
- By Country
- Market Attractiveness Analysis
- By Country
- By Service Type
- By Therapy Area
- By Delivery Mode
- By Customer Type
- By Support Function
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Service Type
- By Therapy Area
- By Delivery Mode
- By Customer Type
- By Support Function
- By Country
- Market Attractiveness Analysis
- By Country
- By Service Type
- By Therapy Area
- By Delivery Mode
- By Customer Type
- By Support Function
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Therapy Area
- By Delivery Mode
- By Customer Type
- By Support Function
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Therapy Area
- By Delivery Mode
- By Customer Type
- By Support Function
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Therapy Area
- By Delivery Mode
- By Customer Type
- By Support Function
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Therapy Area
- By Delivery Mode
- By Customer Type
- By Support Function
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Therapy Area
- By Delivery Mode
- By Customer Type
- By Support Function
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Therapy Area
- By Delivery Mode
- By Customer Type
- By Support Function
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Therapy Area
- By Delivery Mode
- By Customer Type
- By Support Function
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Therapy Area
- By Delivery Mode
- By Customer Type
- By Support Function
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Therapy Area
- By Delivery Mode
- By Customer Type
- By Support Function
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Therapy Area
- By Delivery Mode
- By Customer Type
- By Support Function
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Therapy Area
- By Delivery Mode
- By Customer Type
- By Support Function
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Therapy Area
- By Delivery Mode
- By Customer Type
- By Support Function
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Therapy Area
- By Delivery Mode
- By Customer Type
- By Support Function
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Therapy Area
- By Delivery Mode
- By Customer Type
- By Support Function
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Therapy Area
- By Delivery Mode
- By Customer Type
- By Support Function
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Therapy Area
- By Delivery Mode
- By Customer Type
- By Support Function
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Therapy Area
- By Delivery Mode
- By Customer Type
- By Support Function
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Therapy Area
- By Delivery Mode
- By Customer Type
- By Support Function
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Therapy Area
- By Delivery Mode
- By Customer Type
- By Support Function
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Therapy Area
- By Delivery Mode
- By Customer Type
- By Support Function
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Therapy Area
- By Delivery Mode
- By Customer Type
- By Support Function
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Therapy Area
- By Delivery Mode
- By Customer Type
- By Support Function
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Service Type
- By Therapy Area
- By Delivery Mode
- By Customer Type
- By Support Function
- Competition Analysis
- Competition Deep Dive
- Cencora
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- McKesson Corporation
- CVS Health
- The Cigna Group
- Value (USD Million)edHealth Group
- Cardinal Health
- Option Care Health
- Cencora
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Service Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Therapy Area, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Delivery Mode, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by Customer Type, 2021 to 2036
- Table 6: Global Market Value (USD Million) Forecast by Support Function, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Service Type , 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Therapy Area, 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by Delivery Mode, 2021 to 2036
- Table 11: North America Market Value (USD Million) Forecast by Customer Type, 2021 to 2036
- Table 12: North America Market Value (USD Million) Forecast by Support Function, 2021 to 2036
- Table 13: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Latin America Market Value (USD Million) Forecast by Service Type , 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Therapy Area, 2021 to 2036
- Table 16: Latin America Market Value (USD Million) Forecast by Delivery Mode, 2021 to 2036
- Table 17: Latin America Market Value (USD Million) Forecast by Customer Type, 2021 to 2036
- Table 18: Latin America Market Value (USD Million) Forecast by Support Function, 2021 to 2036
- Table 19: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: Western Europe Market Value (USD Million) Forecast by Service Type , 2021 to 2036
- Table 21: Western Europe Market Value (USD Million) Forecast by Therapy Area, 2021 to 2036
- Table 22: Western Europe Market Value (USD Million) Forecast by Delivery Mode, 2021 to 2036
- Table 23: Western Europe Market Value (USD Million) Forecast by Customer Type, 2021 to 2036
- Table 24: Western Europe Market Value (USD Million) Forecast by Support Function, 2021 to 2036
- Table 25: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: Eastern Europe Market Value (USD Million) Forecast by Service Type , 2021 to 2036
- Table 27: Eastern Europe Market Value (USD Million) Forecast by Therapy Area, 2021 to 2036
- Table 28: Eastern Europe Market Value (USD Million) Forecast by Delivery Mode, 2021 to 2036
- Table 29: Eastern Europe Market Value (USD Million) Forecast by Customer Type, 2021 to 2036
- Table 30: Eastern Europe Market Value (USD Million) Forecast by Support Function, 2021 to 2036
- Table 31: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 32: East Asia Market Value (USD Million) Forecast by Service Type , 2021 to 2036
- Table 33: East Asia Market Value (USD Million) Forecast by Therapy Area, 2021 to 2036
- Table 34: East Asia Market Value (USD Million) Forecast by Delivery Mode, 2021 to 2036
- Table 35: East Asia Market Value (USD Million) Forecast by Customer Type, 2021 to 2036
- Table 36: East Asia Market Value (USD Million) Forecast by Support Function, 2021 to 2036
- Table 37: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 38: South Asia and Pacific Market Value (USD Million) Forecast by Service Type , 2021 to 2036
- Table 39: South Asia and Pacific Market Value (USD Million) Forecast by Therapy Area, 2021 to 2036
- Table 40: South Asia and Pacific Market Value (USD Million) Forecast by Delivery Mode, 2021 to 2036
- Table 41: South Asia and Pacific Market Value (USD Million) Forecast by Customer Type, 2021 to 2036
- Table 42: South Asia and Pacific Market Value (USD Million) Forecast by Support Function, 2021 to 2036
- Table 43: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 44: Middle East & Africa Market Value (USD Million) Forecast by Service Type , 2021 to 2036
- Table 45: Middle East & Africa Market Value (USD Million) Forecast by Therapy Area, 2021 to 2036
- Table 46: Middle East & Africa Market Value (USD Million) Forecast by Delivery Mode, 2021 to 2036
- Table 47: Middle East & Africa Market Value (USD Million) Forecast by Customer Type, 2021 to 2036
- Table 48: Middle East & Africa Market Value (USD Million) Forecast by Support Function, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Service Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Service Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Service Type
- Figure 6: Global Market Value Share and BPS Analysis by Therapy Area, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Therapy Area, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Therapy Area
- Figure 9: Global Market Value Share and BPS Analysis by Delivery Mode, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Delivery Mode, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Delivery Mode
- Figure 12: Global Market Value Share and BPS Analysis by Customer Type, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Customer Type, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Customer Type
- Figure 15: Global Market Value Share and BPS Analysis by Support Function, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Support Function, 2026-2036
- Figure 17: Global Market Attractiveness Analysis by Support Function
- Figure 18: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 19: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 20: Global Market Attractiveness Analysis by Region
- Figure 21: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 23: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 24: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 25: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 26: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 27: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 28: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 29: North America Market Value Share and BPS Analysis by Service Type , 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Service Type , 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Service Type
- Figure 32: North America Market Value Share and BPS Analysis by Therapy Area, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Therapy Area, 2026-2036
- Figure 34: North America Market Attractiveness Analysis by Therapy Area
- Figure 35: North America Market Value Share and BPS Analysis by Delivery Mode, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by Delivery Mode, 2026-2036
- Figure 37: North America Market Attractiveness Analysis by Delivery Mode
- Figure 38: North America Market Value Share and BPS Analysis by Customer Type, 2026 and 2036
- Figure 39: North America Market Y-o-Y Growth Comparison by Customer Type, 2026-2036
- Figure 40: North America Market Attractiveness Analysis by Customer Type
- Figure 41: North America Market Value Share and BPS Analysis by Support Function, 2026 and 2036
- Figure 42: North America Market Y-o-Y Growth Comparison by Support Function, 2026-2036
- Figure 43: North America Market Attractiveness Analysis by Support Function
- Figure 44: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 45: Latin America Market Value Share and BPS Analysis by Service Type , 2026 and 2036
- Figure 46: Latin America Market Y-o-Y Growth Comparison by Service Type , 2026-2036
- Figure 47: Latin America Market Attractiveness Analysis by Service Type
- Figure 48: Latin America Market Value Share and BPS Analysis by Therapy Area, 2026 and 2036
- Figure 49: Latin America Market Y-o-Y Growth Comparison by Therapy Area, 2026-2036
- Figure 50: Latin America Market Attractiveness Analysis by Therapy Area
- Figure 51: Latin America Market Value Share and BPS Analysis by Delivery Mode, 2026 and 2036
- Figure 52: Latin America Market Y-o-Y Growth Comparison by Delivery Mode, 2026-2036
- Figure 53: Latin America Market Attractiveness Analysis by Delivery Mode
- Figure 54: Latin America Market Value Share and BPS Analysis by Customer Type, 2026 and 2036
- Figure 55: Latin America Market Y-o-Y Growth Comparison by Customer Type, 2026-2036
- Figure 56: Latin America Market Attractiveness Analysis by Customer Type
- Figure 57: Latin America Market Value Share and BPS Analysis by Support Function, 2026 and 2036
- Figure 58: Latin America Market Y-o-Y Growth Comparison by Support Function, 2026-2036
- Figure 59: Latin America Market Attractiveness Analysis by Support Function
- Figure 60: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 61: Western Europe Market Value Share and BPS Analysis by Service Type , 2026 and 2036
- Figure 62: Western Europe Market Y-o-Y Growth Comparison by Service Type , 2026-2036
- Figure 63: Western Europe Market Attractiveness Analysis by Service Type
- Figure 64: Western Europe Market Value Share and BPS Analysis by Therapy Area, 2026 and 2036
- Figure 65: Western Europe Market Y-o-Y Growth Comparison by Therapy Area, 2026-2036
- Figure 66: Western Europe Market Attractiveness Analysis by Therapy Area
- Figure 67: Western Europe Market Value Share and BPS Analysis by Delivery Mode, 2026 and 2036
- Figure 68: Western Europe Market Y-o-Y Growth Comparison by Delivery Mode, 2026-2036
- Figure 69: Western Europe Market Attractiveness Analysis by Delivery Mode
- Figure 70: Western Europe Market Value Share and BPS Analysis by Customer Type, 2026 and 2036
- Figure 71: Western Europe Market Y-o-Y Growth Comparison by Customer Type, 2026-2036
- Figure 72: Western Europe Market Attractiveness Analysis by Customer Type
- Figure 73: Western Europe Market Value Share and BPS Analysis by Support Function, 2026 and 2036
- Figure 74: Western Europe Market Y-o-Y Growth Comparison by Support Function, 2026-2036
- Figure 75: Western Europe Market Attractiveness Analysis by Support Function
- Figure 76: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 77: Eastern Europe Market Value Share and BPS Analysis by Service Type , 2026 and 2036
- Figure 78: Eastern Europe Market Y-o-Y Growth Comparison by Service Type , 2026-2036
- Figure 79: Eastern Europe Market Attractiveness Analysis by Service Type
- Figure 80: Eastern Europe Market Value Share and BPS Analysis by Therapy Area, 2026 and 2036
- Figure 81: Eastern Europe Market Y-o-Y Growth Comparison by Therapy Area, 2026-2036
- Figure 82: Eastern Europe Market Attractiveness Analysis by Therapy Area
- Figure 83: Eastern Europe Market Value Share and BPS Analysis by Delivery Mode, 2026 and 2036
- Figure 84: Eastern Europe Market Y-o-Y Growth Comparison by Delivery Mode, 2026-2036
- Figure 85: Eastern Europe Market Attractiveness Analysis by Delivery Mode
- Figure 86: Eastern Europe Market Value Share and BPS Analysis by Customer Type, 2026 and 2036
- Figure 87: Eastern Europe Market Y-o-Y Growth Comparison by Customer Type, 2026-2036
- Figure 88: Eastern Europe Market Attractiveness Analysis by Customer Type
- Figure 89: Eastern Europe Market Value Share and BPS Analysis by Support Function, 2026 and 2036
- Figure 90: Eastern Europe Market Y-o-Y Growth Comparison by Support Function, 2026-2036
- Figure 91: Eastern Europe Market Attractiveness Analysis by Support Function
- Figure 92: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 93: East Asia Market Value Share and BPS Analysis by Service Type , 2026 and 2036
- Figure 94: East Asia Market Y-o-Y Growth Comparison by Service Type , 2026-2036
- Figure 95: East Asia Market Attractiveness Analysis by Service Type
- Figure 96: East Asia Market Value Share and BPS Analysis by Therapy Area, 2026 and 2036
- Figure 97: East Asia Market Y-o-Y Growth Comparison by Therapy Area, 2026-2036
- Figure 98: East Asia Market Attractiveness Analysis by Therapy Area
- Figure 99: East Asia Market Value Share and BPS Analysis by Delivery Mode, 2026 and 2036
- Figure 100: East Asia Market Y-o-Y Growth Comparison by Delivery Mode, 2026-2036
- Figure 101: East Asia Market Attractiveness Analysis by Delivery Mode
- Figure 102: East Asia Market Value Share and BPS Analysis by Customer Type, 2026 and 2036
- Figure 103: East Asia Market Y-o-Y Growth Comparison by Customer Type, 2026-2036
- Figure 104: East Asia Market Attractiveness Analysis by Customer Type
- Figure 105: East Asia Market Value Share and BPS Analysis by Support Function, 2026 and 2036
- Figure 106: East Asia Market Y-o-Y Growth Comparison by Support Function, 2026-2036
- Figure 107: East Asia Market Attractiveness Analysis by Support Function
- Figure 108: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 109: South Asia and Pacific Market Value Share and BPS Analysis by Service Type , 2026 and 2036
- Figure 110: South Asia and Pacific Market Y-o-Y Growth Comparison by Service Type , 2026-2036
- Figure 111: South Asia and Pacific Market Attractiveness Analysis by Service Type
- Figure 112: South Asia and Pacific Market Value Share and BPS Analysis by Therapy Area, 2026 and 2036
- Figure 113: South Asia and Pacific Market Y-o-Y Growth Comparison by Therapy Area, 2026-2036
- Figure 114: South Asia and Pacific Market Attractiveness Analysis by Therapy Area
- Figure 115: South Asia and Pacific Market Value Share and BPS Analysis by Delivery Mode, 2026 and 2036
- Figure 116: South Asia and Pacific Market Y-o-Y Growth Comparison by Delivery Mode, 2026-2036
- Figure 117: South Asia and Pacific Market Attractiveness Analysis by Delivery Mode
- Figure 118: South Asia and Pacific Market Value Share and BPS Analysis by Customer Type, 2026 and 2036
- Figure 119: South Asia and Pacific Market Y-o-Y Growth Comparison by Customer Type, 2026-2036
- Figure 120: South Asia and Pacific Market Attractiveness Analysis by Customer Type
- Figure 121: South Asia and Pacific Market Value Share and BPS Analysis by Support Function, 2026 and 2036
- Figure 122: South Asia and Pacific Market Y-o-Y Growth Comparison by Support Function, 2026-2036
- Figure 123: South Asia and Pacific Market Attractiveness Analysis by Support Function
- Figure 124: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 125: Middle East & Africa Market Value Share and BPS Analysis by Service Type , 2026 and 2036
- Figure 126: Middle East & Africa Market Y-o-Y Growth Comparison by Service Type , 2026-2036
- Figure 127: Middle East & Africa Market Attractiveness Analysis by Service Type
- Figure 128: Middle East & Africa Market Value Share and BPS Analysis by Therapy Area, 2026 and 2036
- Figure 129: Middle East & Africa Market Y-o-Y Growth Comparison by Therapy Area, 2026-2036
- Figure 130: Middle East & Africa Market Attractiveness Analysis by Therapy Area
- Figure 131: Middle East & Africa Market Value Share and BPS Analysis by Delivery Mode, 2026 and 2036
- Figure 132: Middle East & Africa Market Y-o-Y Growth Comparison by Delivery Mode, 2026-2036
- Figure 133: Middle East & Africa Market Attractiveness Analysis by Delivery Mode
- Figure 134: Middle East & Africa Market Value Share and BPS Analysis by Customer Type, 2026 and 2036
- Figure 135: Middle East & Africa Market Y-o-Y Growth Comparison by Customer Type, 2026-2036
- Figure 136: Middle East & Africa Market Attractiveness Analysis by Customer Type
- Figure 137: Middle East & Africa Market Value Share and BPS Analysis by Support Function, 2026 and 2036
- Figure 138: Middle East & Africa Market Y-o-Y Growth Comparison by Support Function, 2026-2036
- Figure 139: Middle East & Africa Market Attractiveness Analysis by Support Function
- Figure 140: Global Market - Tier Structure Analysis
- Figure 141: Global Market - Company Share Analysis

