Long-Acting Implant & Ocular Drug-Delivery Polymer Systems Market
Long-Acting Implant & Ocular Drug-Delivery Polymer Systems Market Size and Share Forecast Outlook 2026 to 2036
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Long-Acting Implant & Ocular Drug-Delivery Polymer Systems Market Forecast & Outlook 2026 to 2036
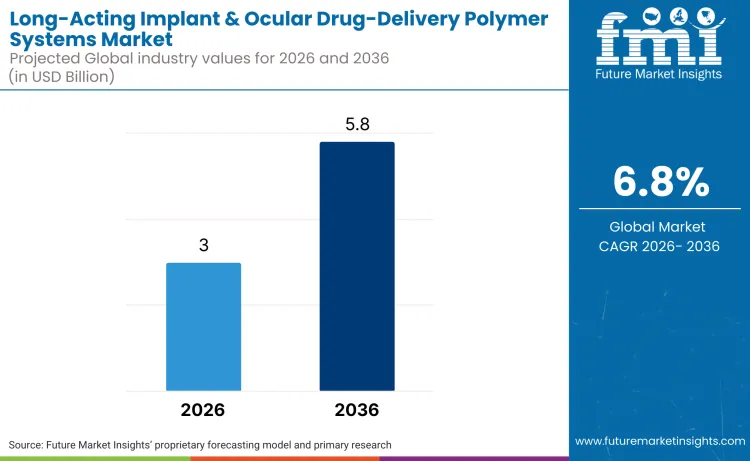
The long-acting implant & ocular drug-delivery polymer systems market is expected to reach USD 3.0 billion in 2026, and ultimately to USD 5.8 billion by 2036. As per FMI’s findings, demand is anticipated to expand at a 6.8% CAGR.
The landscape is currently defined by a decisive transition from episodic intravitreal injections to continuous-elution architectures. As of early 2026, it is likely to be characterized by a durability race, where the strategic objective is to extend the therapeutic window from weeks to a full calendar year, thereby addressing the chronic issue of patient non-compliance.
Current activity is bifurcated between biodegradable and non-biodegradable systems. While non-erodible reservoirs like Roche’s Susvimo® have validated the refillable port delivery model for DME and DR, the momentum is shifting toward bioerodible hydrogels. Ocular Therapeutix is a primary example, utilizing a PEG-based hydrogel platform for AXPAXLI™. This system is designed to biodegrade fully after its 12-month release cycle, eliminating the need for surgical removal and reducing long-term inflammatory risks.
Corporate strategy in 2025-2026 has focused on high-value partnerships targeting the "polymer-free" frontier. Firms such as Ripple Therapeutics are disrupting the space with prodrug-based systems that bypass traditional synthetic matrices entirely, securing licensing deals with AbbVie and Glaukos.
This shift suggests that the next generation of long-acting implant & ocular drug-delivery polymer systems will prioritize zero-order release kinetics and minimal localized toxicity. With major Phase 3 readouts like SOL-1 expected this quarter, the industry is poised to move from proof-of-concept to a new standard of care in retinal health.
Industry leaders emphasize that these technological advancements are not merely incremental updates, but foundational shifts intended to dominate the commercial landscape by fundamentally changing the patient experience.
"Ocular aims to redefine the retina experience by reducing the treatment burden and providing an alternative to the pulsatile nature of available treatments, in hopes of preserving vision for the long-term. Beyond wet AMD, we are thrilled with the FDA feedback supporting our NPDR and DME program, and we look forward to sharing more details at our Investor Day in September, along with the global commercial outlook for AXPAXLI and more. Our refreshed corporate branding, launched in June, reflects the Company we've become: retina-focused, patient-driven, and boldly advancing a potential new standard-of-care in retinal disease." Dr. Pravin U. Dugel, Executive Chairman, President, and CEO, Ocular Therapeutix Source: Ocular Therapeutix™ Reports Second Quarter 2025 Financial Results
Summary of the Long-Acting Implant & Ocular Drug-Delivery Polymer Systems Market
How fast is the Long-Acting Implant & Ocular Drug-Delivery Polymer Systems Market Expanding?
The market is projected to grow from USD 3.0 billion in 2026 to USD 5.8 billion by 2036, registering a CAGR of 6.8%. Growth is supported by rising adoption of sustained-release delivery systems in chronic ophthalmic and systemic therapies.
FMI Research Approach: Proprietary forecasting model based on approved pipeline conversion, delivery-system substitution rates, and chronic disease prevalence.
Which Delivery Routes Account for the Largest Share of Demand?
Subcutaneous and intramuscular implants represent the largest delivery route, accounting for 37% of total demand. These systems dominate due to established safety profiles, ease of administration, and broad use in systemic long-acting therapies.
FMI Research Approach: Route-level adoption mapping across approved and late-stage pipeline products.
Which Therapy Areas Drive Market Volume?
Oncology and chronic pain applications account for 43% of total market demand. High utilization is linked to the need for continuous drug exposure, opioid-sparing pain management, and long-term hormonal and palliative therapies.
FMI Research Approach: Therapy-wise volume modeling using implant usage frequency and patient pool estimates.
What Polymer Systems Dominate Material Selection?
Biodegradable polyesters such as PLGA, PLA, and PCL lead the market with a 45% share. Their dominance reflects regulatory acceptance, predictable degradation kinetics, and elimination of surgical removal requirements.
FMI Research Approach: Material-level analysis of approved products and late-stage clinical platforms.
Which Countries exhibit the Highest Growth Rates?
China is the fastest-growing market with a CAGR of 9.6%, followed by Brazil at 9.2%, the United States at 9.0%, and the UK at 8.0%. Regulatory pathway maturity, aging populations, and expansion of office-based drug delivery drive growth in these countries.
FMI Research Approach: Country-wise forecasting based on regulatory approvals, reimbursement frameworks, and clinical adoption rates.
How is the Market Structurally Positioned Going Forward?
The market is shifting toward long-duration, bioerodible, and refill-free delivery platforms, with competitive differentiation increasingly defined by durability benchmarks of 6-12 months rather than incremental release extensions.
FMI Research Approach: Competitive benchmarking of clinical durability targets and polymer platform strategies.
Long-Acting Implant & Ocular Drug-Delivery Polymer Systems Market Key Takeaways
| Metric | Value |
|---|---|
| Expected Value (2026E) | USD 3.0 billion |
| Projected Value (2036F) | USD 5.8 billion |
| CAGR (2026-2036) | 6.8% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Why is the Long-Acting Implant & Ocular Drug Delivery Polymer Systems Market Growing?
The expansion of the long-acting implant & ocular drug-delivery polymer systems market is driven by a convergence of regulatory efficiency, demographic pressure, and a fundamental shift in clinical site-of-service economics.
The FDA's 505(b)(2) regulatory pathway has emerged as a primary growth catalyst, allowing manufacturers to bypass the "Valley of Death" associated with New Chemical Entities (NCE). By leveraging existing safety data for established APIs and focusing innovation strictly on the polymer delivery matrix, companies can reduce development costs by an estimated 90%. As of 2026, over 60% of all approved NDAs utilize this pathway, incentivizing firms to reformulate legacy molecules into high-margin, sustained-release polymer formats.
The 2026 Medicare Physician Fee Schedule (MPFS) final rule introduced an efficiency adjustment that favors office-based procedures over facility-based care. Specifically, CMS increased the conversion factor for non-facility services by over 3% while implementing substantial cuts for procedures in ASCs. This pivot incentivizes the adoption of injectable polymer-based implants over surgically implanted reservoirs that require facility resources.
Demographic shifts are creating an unprecedented volume of patients requiring chronic therapy. By 2026, global incidence rates for POAG have reached 23.46 cases per 10,000 person-years for those aged 40-79, with rates skyrocketing to 64.36 in the 75-79 age bracket. This rising prevalence, combined with a 2.7% annual growth in practice costs, is forcing a transition toward long-acting systems that reduce the frequency of high-cost clinical visits.
How is the Long-Acting Implant & Ocular Drug-Delivery Polymer Systems Market Segmented?
The long-acting implant and ocular drug-delivery polymer systems market is segmented by administration route, therapeutic application, and material composition, each responding to distinct clinical demands and technological breakthroughs.
What is the Scope for Subcutaneous and Intramuscular Implants?
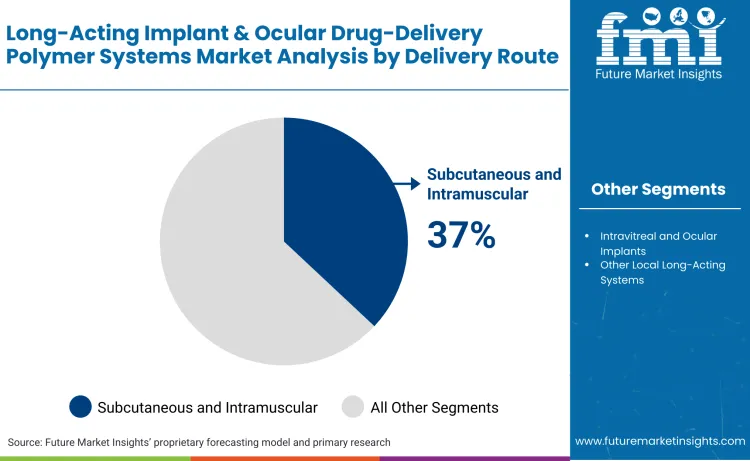
Subcutaneous and intramuscular implants command a 37% market share, serving as the bedrock for systemic long-term therapy. These routes are favored for their established safety profiles and high patient tolerance, particularly in therapies requiring steady-state plasma concentrations over several months.
Recent developments in in situ forming implants (ISFIs) are driving this segment's growth, as seen with Janssen’s Invega Hafyera, a twice-yearly injectable for schizophrenia that utilizes a proprietary depot technology to ensure six-month adherence. Similarly, the rise of large-molecule delivery via subcutaneous "scaffolds" is gaining traction for chronic autoimmune management.
Which Therapy Areas are Dictating Market Volume?
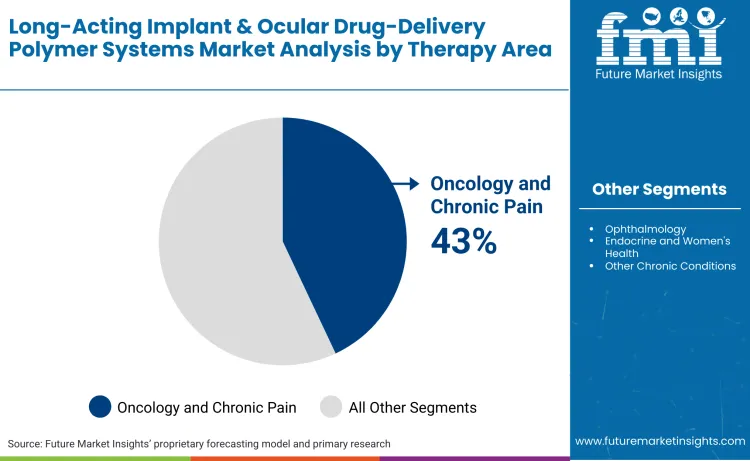
Oncology and chronic pain leads the market with a 43% share, propelled by the urgent need for opioid-sparing pain management and localized chemotherapy. The shift toward palliative care and quality-of-life improvements has made sustained-release (SR) systems a clinical gold standard for terminal and chronic indications.
Growth is highlighted by the adoption of high-frequency neurostimulators and dual-mechanism implants, such as Boston Scientific’s advanced drug-eluting platforms. In oncology, the use of Zoladex (goserelin acetate) as a polymer-based subcutaneous implant continues to set the pace for hormonal therapy in prostate and breast cancers.
What Role do Biodegradable polyesters play in Material Science?
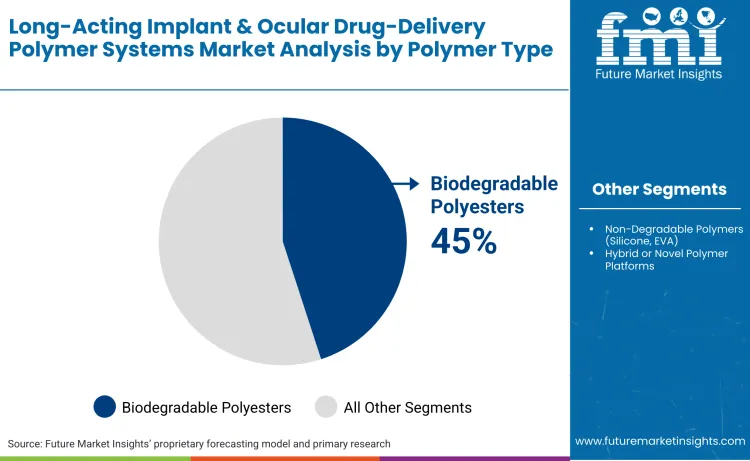
Biodegradable polyesters (PLGA, PLA, PCL) represent the largest material segment with a 45% market share. Their dominance is rooted in their FDA-approved biocompatibility and tuneable degradation rates, which allow manufacturers to program drug release from days to years without surgical retrieval.
Innovation in this segment is exemplified by the Epidel™ platform from Ripple Therapeutics, which uses a polymer-free prodrug approach that mimics biodegradable polyester kinetics to eliminate synthetic residue. PolyActiva’s PA5108 biodegradable latanoprost implant demonstrates the specific utility of these materials in providing 6-month glaucoma therapy while resorbing completely into the ocular tissue.
What Drivers, Restraints, Opportunities and Trends influence the Long-Acting Implant & Ocular Drug-Delivery Polymer Systems Landscape?
The expansion of long-acting implant and ocular drug-delivery polymer systems is increasingly tied to a favorable tilt in outpatient economics. Recent updates to the Medicare Physician Fee Schedule for 2026 have introduced a 3.26% increase in the conversion factor for office-based services.
This adjustment widens the payment gap between in-office drug administration and traditional ambulatory surgery centers, where some procedures face facility-based payment reductions. Such a shift creates a significant financial incentive for clinics to adopt injectable polymer implants that can be delivered during a standard consultation, bypassing the higher overhead of surgical theaters.
International intellectual property standards are also acting as a primary catalyst, particularly APAC. The revised Implementing Regulations of the Drug Administration Law in China, effective May 15, 2026, now grant up to 6 years of data protection for innovative drugs containing novel chemical entities.
This regulatory strengthening, combined with up to 2 years of market exclusivity for new pediatric dosage forms, provides a secured window for global manufacturers to recoup R&D investments in high-tech polymer delivery platforms within the world’s second-largest pharmaceutical market.
Despite these tailwinds, the sector faces a specific restraint related to the rigorous clinical thresholds required to displace the current standard of care. Major players are now moving away from non-inferiority trials toward superiority-labeled registrational programs.
This strategic pivot is evident in the SOL-1 Phase 3 program, where the primary goal is to demonstrate that continuous-release polymer systems can prevent the long-term vision loss associated with the pulsatile nature of traditional monthly injections. While this trend promises higher clinical outcomes, it significantly raises the evidentiary bar and capital requirements for new market entrants.
How is the Long-Acting Implant & Ocular Drug-Delivery Polymer Systems Market performing in Key Countries?
The geographical landscape of long-acting implant and ocular drug-delivery polymer systems is increasingly defined by regional regulatory maturation and the decentralization of high-tech manufacturing.
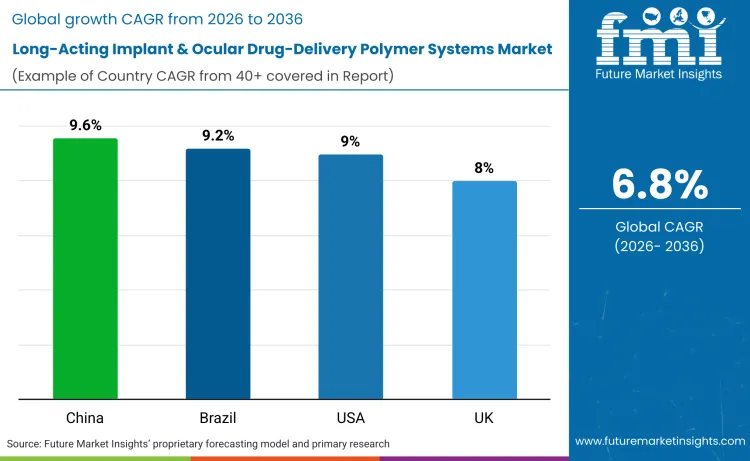
| Country | CAGR (2026-2036) |
|---|---|
| China | 9.6% |
| Brazil | 9.2% |
| USA | 9.0% |
| UK | 8.0% |
Source: FMI analysis based on primary research and proprietary forecasting model
How is China accelerating Domestic Polymer Innovation?
China is projected to lead global expansion with a CAGR of 9.6% through 2036. This growth is underpinned by a surge in "localization" strategies from global leaders. Santen Pharmaceutical officially launched TAPCOM® in the Chinese market in September 2025, marking the first preservative-free, fixed-dose combination for glaucoma in the region. By eliminating preservatives, these formulations reduce long-term ocular surface damage, a critical factor for the 21.8 million glaucoma patients in China.
Furthermore, the domestic pipeline is maturing through strategic collaborations. The recent acceptance of the Biologic License Application (BLA) for RC28-E, a dual-target fusion protein for Diabetic Macular Edema developed by RemeGen and Santen, highlights a shift toward high-durability injectables. As these advanced delivery platforms enter the National Reimbursement Drug List (NRDL), volume-driven growth is expected to sustain high momentum in the tier-2 and tier-3 city hospitals.
Why is Brazil emerging as a Latin American Powerhouse?
Brazil follows closely with an estimated CAGR of 9.2%. The nation’s unique dual-track healthcare system-comprised of the public SUS and a robust private insurance sector-is increasingly favoring bundled contracts that include capital equipment and polymer-based consumables. In late 2025, the Brazilian government announced expanded trade agreements with India to specifically bolster the local pharmaceutical and vaccine supply chains, signaling a long-term intent to localize the production of complex drug-delivery systems.
Private clinics in the Southeast economic corridor are also driving high-end technology adoption. The rapid integration of femtosecond laser platforms for refractive and cataract procedures has created a secondary market for specialized, post-operative polymer inserts. With diabetic retinopathy prevalence adding nearly 1% to the national growth rate annually, the demand for sustained-release implants that reduce the frequency of clinical visits is becoming a public health priority.
What Role does the USA play in Next Generation of Ocular Drug Delivery?
The United States maintains a significant market position with a CAGR of 9.0%. Growth here is primarily focused on dropless therapy and bioerodible scaffolds. AbbVie has signaled a 2026 strategic priority for DURYSTA®, its intracameral implant, by initiating real-world evidence studies to identify long-term predictors of intraocular pressure lowering. This focus on "early therapy adoption" aim to transition patients away from daily eye drops to 3-year durability models.
In addition to established therapies, the USA market is a testing ground for suprachoroidal delivery. The clinical progress of ABBV-RGX-314, which utilizes a subretinal and suprachoroidal delivery approach for wet AMD, represents a shift toward targeting the underlying pathology via a one-time gene therapy application. This "one-and-done" paradigm is expected to redefine the value proposition of polymer-based delivery systems in the North American market.
How is UK positioning itself for Specialized Ophthalmic Care?
UK’s market is expected to grow at a CAGR of 8.0%. The National Health Service (NHS) is increasingly adopting "personalized medicine" frameworks to streamline the management of chronic ocular inflammation. In early 2026, Bausch + Lomb announced a partnership with major UK glaucoma foundations to address irreversible blindness through the deployment of next-generation scleral lenses and bioactive polymer materials that incorporate hyaluronic acid directly into the lens backbone.
Innovation in the UK is also driven by regulatory adaptations that allow for accelerated approvals of preservative-free and long-acting injectables. As the fastest-growing formulation segment in the country, injectables are replacing traditional topical solutions in NHS trusts to reduce the administrative burden on eye clinics. This structural change ensures that even with a more modest growth rate, the UK remains a hub for high-value, clinically superior delivery technologies.
Competitive Landscape
The competitive intensity in the long-acting implant and ocular drug-delivery polymer systems market has reached a critical inflection point, driven by a race for therapeutic durability. Traditional dominance is being dismantled by a cohort of multi-disciplinary giants that no longer compete solely on drug efficacy but on the material science of the delivery matrix itself. This environment has fostered a convergence where pharmaceutical manufacturers are integrating vertically with polymer engineering firms to secure proprietary control over bioerodible scaffolds and non-erodible reservoirs.
Competition is further intensified by a shift in clinical endpoints from simple safety to the reduction of overall treatment burden. Companies are under immense pressure to prove that their polymer systems can maintain consistent intraocular pressure or retinal drying for twelve months or longer without the performance troughs associated with frequent injections. This has led to an era of high-stakes registrational trials and strategic patent layering. The barrier to entry has shifted from biological innovation to the ability to manufacture high-precision, medical-grade polymers at scale while navigating complex outpatient reimbursement frameworks.
Important Developments
- In September 2025, Evonik made its VECOLLAN® collagen available in a grade suitable for clinical trials. This fermentation-based biomaterial provides a highly soluble and sustainable alternative to animal-derived collagen for use in bioresorbable ocular inserts and tissue engineering.
- DSM has positioned its TheraPEA™ PEA technology as a leading vehicle for small-molecule and biologic delivery. In early 2026, the company emphasized its continued progress in ophthalmology, supporting partners in developing next-generation treatments for wet AMD.
- Lubrizol Life Science Health reached announced in June 2025 that its Apisolex™ injectable formulation successfully entered Phase 1 clinical trials, proving the technology's ability to enhance the solubility of hydrophobic active ingredients by up to 50,000-fold.
Key Players Profiled
- Medtronic plc
- Becton, Dickinson and Company
- BASF SE-Pharma Solutions
- Evonik Industries AG (Health Care)
- DSM Biomedical Inc.
- The Lubrizol Corporation (Life Science)
- Ashland Global Holdings Inc. (Specialty Ingredients)
- Corbion N.V. (Biomaterials)
- Alcon Inc.
- Allergan plc
Market Definition
The long-acting implant & ocular drug-delivery polymer systems market comprises polymer-based platforms designed to deliver therapeutic agents over extended durations, typically ranging from several months to one year. These systems are used to maintain controlled, continuous drug release in chronic conditions, particularly where frequent dosing or repeated injections reduce adherence or clinical efficiency. The market spans both biodegradable and non-biodegradable polymer matrices used in systemic and ocular applications .
The report includes polymer systems used in subcutaneous, intramuscular, intravitreal, and other localized long-acting delivery routes across therapy areas such as ophthalmology, oncology, chronic pain, endocrine disorders, and women’s health. It covers key material classes including biodegradable polyesters such as PLGA, PLA, and PCL, non-degradable polymers such as silicone and EVA, and emerging hybrid or novel polymer platforms evaluated in approved products and late-stage clinical pipelines .
The scope excludes conventional short-acting injectables, oral and topical dosage forms, standalone active pharmaceutical ingredients, and non-polymer-based delivery technologies such as pumps or electronic devices. It also excludes surgical instruments, clinical administration services, and drug discovery activities not directly related to polymer-based long-acting or ocular delivery systems .
Scope of Report
| Items | Values |
|---|---|
| Quantitative Units (2026) | USD 3.0 billion |
| Delivery Route | Subcutaneous and Intramuscular Implants, Intravitreal and Ocular Implants, Other Local Long-Acting Systems |
| Therapy Area | Oncology and Chronic Pain, Ophthalmology, Endocrine and Women's Health, Other Chronic Conditions |
| Polymer Type | Biodegradable Polyesters (PLGA, PLA, PCL), Non-Degradable Polymers (Silicone, EVA), Hybrid or Novel Polymer Platforms |
| Regions Covered | North America, Western Europe, Eastern Europe, East Asia, South Asia & Pacific, Latin America, Middle East & Africa |
| Countries Covered | USA, UK, Brazil and 40+ Countries |
| Key Companies Profiled | Medtronic plc, Becton, Dickinson and Company, BASF SE-Pharma Solutions, Evonik Industries AG (Health Care), DSM Biomedical Inc., The Lubrizol Corporation (Life Science), Ashland Global Holdings Inc. (Specialty Ingredients), Corbion N.V. (Biomaterials), Alcon Inc., Allergan plc |
Long-Acting Implant & Ocular Drug-Delivery Polymer Systems Market by Key Segments
Delivery Route
- Subcutaneous and Intramuscular Implants
- Intravitreal and Ocular Implants
- Other Local Long-Acting Systems
Therapy Area
- Oncology and Chronic Pain
- Ophthalmology
- Endocrine and Women's Health
- Other Chronic Conditions
Polymer Type
- Biodegradable Polyesters (PLGA, PLA, PCL)
- Non-Degradable Polymers (Silicone, EVA)
- Hybrid or Novel Polymer Platforms
Region
- North America
- Western Europe
- Eastern Europe
- East Asia
- South Asia & Pacific
- Latin America
- Middle East & Africa
Bibliography
- Bourges, J. L., Bloquel, C., Thomas, A., Froussart, F., Bochot, A., Azan, F., … Behar-Cohen, F. (2024). Intraocular drug delivery systems: Recent advances and future perspectives. Advanced Drug Delivery Reviews, 202, 114149.
- Centers for Medicare & Medicaid Services. (2025). Medicare Physician Fee Schedule Final Rule for Calendar Year 2026. USA Department of Health & Human Services.
- Del Amo, E. M., Rimpelä, A. K., Heikkinen, E., Kari, O. K., Ramsay, E., Lajunen, T., … Urtti, A. (2024). Pharmacokinetic aspects of retinal drug delivery. Progress in Retinal and Eye Research, 89, 101036.
- Evonik Industries AG. (2025). Advanced biomaterials for long-acting parenteral and ocular drug delivery. Evonik Health Care Technical White Paper.
- Patel, A., Cholkar, K., Agrahari, V., & Mitra, A. K. (2024). Ocular drug delivery systems: An overview. World Journal of Pharmacology, 13(1), 1-23.
- Urtti, A., & Del Amo, E. M. (2025). Challenges and opportunities in ocular pharmacokinetics and drug delivery. Pharmacological Reviews, 77(2), 259-307.
Frequently Asked Questions
How opportunistic is the long-acting implant and ocular drug-delivery polymer systems market?
The market is valued at around USD 3.0 billion in 2026, reflecting growing clinical adoption of sustained-release systems for chronic and degenerative conditions.
What unmet clinical need is driving demand for long-acting polymer delivery systems?
Demand is driven by the need to reduce dosing frequency, improve patient adherence, and avoid the therapeutic peaks and troughs associated with repeated injections or daily administration.
Which therapy areas account for the highest utilization of these systems?
Ophthalmology leads adoption, followed by oncology and chronic pain, where continuous drug exposure improves disease control and lowers procedural burden on patients and providers.
How do regulatory and reimbursement pathways shape market growth?
Regulatory routes such as 505(b)(2) enable faster development by reformulating known drugs, while reimbursement policies increasingly favor office-based, minimally invasive delivery over surgical interventions.
What are the main barriers limiting wider clinical adoption?
Key barriers include stringent clinical evidence requirements to demonstrate superiority over existing therapies, long development timelines, and the complexity of manufacturing high-precision, medical-grade polymers.
Table of Content
- Executive Summary
- Market Outlook and Forecast (2026-2036)
- Why is the Long-Acting Implant & Ocular Drug-Delivery Polymer Systems Market Growing?
- Regulatory Pathway Efficiency and Reformulation Economics
- Shift toward Office-based and Minimally Invasive Delivery
- Demographic Pressure and Chronic Disease Burden
- How is the Long-Acting Implant & Ocular Drug-Delivery Polymer Systems Market Segmented?
- By Delivery Route
- By Therapy Area
- By Polymer Type
- What Drivers, Restraints, Opportunities and Trends Influence the Market?
- Reimbursement and Site-of-Care Economics
- Regulatory Exclusivity and Intellectual Property Protection
- Clinical Superiority Requirements
- Emerging Technology Trends
- How is the Long-Acting Implant & Ocular Drug-Delivery Polymer Systems Market Performing in Key Countries?
- China
- Brazil
- United States
- United Kingdom
- Competitive Landscape
- Market Structure and Competitive Intensity
- Strategic Positioning and Polymer Platform Control
- Key Industry Developments
- Key Players Profiled
- Medtronic plc
- Becton, Dickinson and Company
- BASF SE - Pharma Solutions
- Evonik Industries AG (Health Care)
- DSM Biomedic
List of Tables
- Long-Acting Implant & Ocular Drug-Delivery Polymer Systems Market Key Takeaways
- Global Long-Acting Implant & Ocular Drug-Delivery Polymer Systems Market Size (USD Billion), 2026E-2036F
- Long-Acting Implant & Ocular Drug-Delivery Polymer Systems Market Growth Outlook by Country (CAGR %), 2026-2036
- Market Segmentation by Delivery Route
- Market Segmentation by Therapy Area
- Market Segmentation by Polymer Type
- Country-wise CAGR Comparison
- Scope of Report - Market Coverage and Definitions
List of Figures
- Long-Acting Implant & Ocular Drug-Delivery Polymer Systems Market Growth Trajectory, 2026-2036
- Market Share by Delivery Route (%)
- Market Share by Therapy Area (%)
- Market Share by Polymer Type (%)
- Country-wise CAGR Comparison Chart (2026-2036)
- Competitive Landscape Overview of Long-Acting Implant & Ocular Drug-Delivery Polymer Systems Market
- Clinical Durability Benchmarking of Polymer Delivery Platforms
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

