Oral Obesity Therapies Market
Oral Obesity Therapies Market Analysis Size and Share Forecast Outlook 2026 to 2036
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Oral Obesity Therapies Market Forecast and Outlook 2026 to 2036
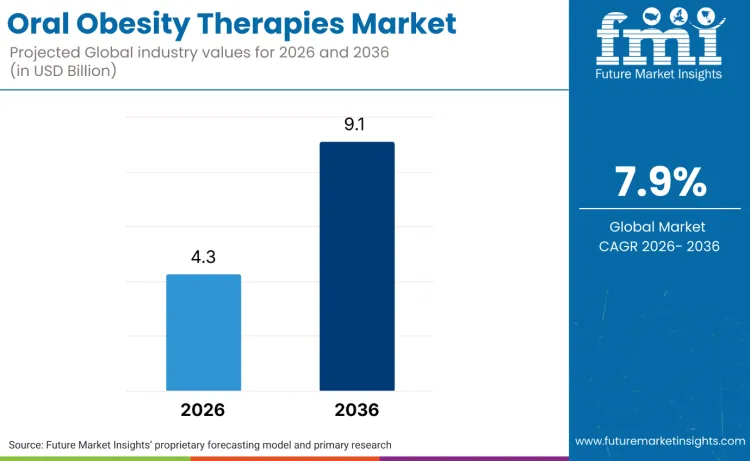
The global oral obesity therapies market is forecasted to reach USD 4.3 billion in 2026 and expand to USD 9.1 billion by 2036, advancing at a CAGR of 7.9%. According to FMI analysis, the trajectory of the oral obesity market reflects a structural shift from episodic weight-loss interventions toward chronic, biology-led metabolic management systems. Patients are increasingly moving away from "quick-fix" solutions in favor of sustained, oral-first protocols that prioritize ease of adherence and the management of metabolic health beyond the scale. Product development is responding to this demand by transitioning from injectable-only formats to high-bioavailability small molecules and "pill-form" GLP-1 agonists designed for long-term daily integration. The rising incidence of obesity-induced chronic diseases, such as cardiovascular diseases, cancer, and others is boosting product innovations in this segment.
“Over the past decade we’ve taken bold new approaches to translating scientific research into effective medicines for people living with cancer, and we have pioneered several breakthroughs in breast cancer, genitourinary cancer, hematological malignancies and precision medicine,” - Chris Boshoff, Chief Development Officer Oncology and Rare Disease, Pfizer.
The focus on consumer-driven accessibility is validated by Novo Nordisk’s 2025 to 2026 strategic pivot following the FDA approval of their high-dose oral obesity pill. By launching the "Wegovy Pill" across USA pharmacies in early 2026, Novo Nordisk is slated to position metabolic treatment from a clinical "procedure" (injections) into a "prescriptive" daily habit. This move confirms that the market’s future value is rooted in the democratization of weight-loss therapy, transitioning patients from specialized obesity clinics to proactive, medicalized maintenance through standard retail pharmacy channels.
The market’s value proposition is increasingly defined by oral regimens that integrate weight loss, cardiovascular protection, and long-term tolerability. Pharmaceutical leaders are expanding patient access while maintaining clinical credibility through "cash-pay" digital platforms and direct-to-consumer delivery. Roche’s 2025 acceleration of its oral GLP-1 pipeline, alongside Sun Pharma’s focus on oral metabolic solutions for liver health, underscores how professional-grade obesity care is moving from a specialist-managed "crisis" setting into a routine-driven, daily health discovery channel.
Summary of the Oral Obesity Therapies Market
Future Market Insights anticipates that the oral obesity therapies market will be reshaped by the reclassification of obesity as a chronic metabolic disease rather than a short-term weight loss condition, driving demand for scalable, oral-first pharmaceutical solutions that support long-term cardiometabolic risk management and daily treatment adherence.
What is the Growth Outlook on the Oral Obesity Therapies Market as per Future Market Insights Projection?
Future Market Insights projects the oral obesity therapies market to grow at a CAGR of 7.9% from 2026 to 2036, reflecting sustained expansion driven by oral GLP-1 innovation, chronic maintenance protocols, and broader insurance integration.
FMI research approach:
- FMI proprietary bottom-up revenue modeling by drug class and patient indication
- Transition analysis from injectable-only regimens to oral-first metabolic protocols
- Regulatory approvals for high-bioavailability oral GLP-1 and small-molecule candidates
- Long-term adoption benchmarking against statins and chronic cardiometabolic drugs
How Do FMI Analysts Perceive the Oral Obesity Therapies Market to Evolve?
FMI analysts perceive the market evolving from episodic weight-loss usage toward routine, long-term metabolic maintenance, where oral therapies function as daily preventive interventions similar to cholesterol and hypertension medications.
FMI research approach:
- Clinical pathway analysis showing step-down treatment models from injectables to pills
- Expansion of obesity drug labels into cardiovascular, liver, and sleep apnea indications
- Manufacturing advances enabling non-peptide, refrigeration-free oral formulations
- Increasing payer alignment with chronic disease management frameworks
Which Country Holds the Largest Share in the Global Oral Obesity Therapies Market?
The United States holds the largest share of the global oral obesity therapies market by value.
FMI research approach:
- FMI country-level revenue modeling across retail, telehealth, and outpatient channels
- Medicare and Medicaid coverage expansion for anti-obesity pharmacotherapy
- FDA approvals for next-generation oral obesity drugs and small-molecule candidates
- Strong direct-to-consumer telehealth and retail pharmacy penetration
How Large Will the Oral Obesity Therapies Market Be by 2036?
The global oral obesity therapies market is estimated to reach USD 9.1 billion by 2036.
FMI research approach:
- FMI long-term forecast derived from drug class, sales channel, and treatment duration mix
- Scaling assumptions for oral GLP-1 adoption and generic entry post-patent expiry
- Inclusion of chronic maintenance therapy duration in revenue modeling
- Cross-validation against global cardiometabolic drug market growth trajectories
What is the Definition of the Oral Obesity Therapies Market?
The oral obesity therapies market comprises pharmaceutical-grade oral medications designed to treat obesity through appetite modulation, metabolic regulation, or hormonal signaling, intended for both short-term weight reduction and long-term metabolic maintenance.
FMI research approach:
- FMI market taxonomy with defined drug class, indication, and delivery criteria
- Alignment with pharmaceutical regulatory standards for obesity indications
- Inclusion of prescription oral GLP-1, combination therapies, and appetite suppressants
- Explicit exclusion of injectables, devices, supplements, and non-pharmaceutical aids
What Are the Globally Unique Trends Shaping the Oral Obesity Therapies Market?
Globally unique trends include oral-first GLP-1 deployment, step-down maintenance models, expansion into comorbidity-led indications, and integration with telehealth-driven care delivery.
FMI research approach:
- Rising adoption of oral GLP-1s to overcome injection aversion and cold-chain barriers
- Step-down protocols transitioning patients from injectables to daily pills
- Regulatory recognition of obesity-linked cardiovascular and metabolic conditions
- Divergence between emerging market access expansion and developed market reimbursement depth
Oral Obesity Therapies Market Key Takeaways
| Metric | Value |
|---|---|
| Market Value (2026) | USD 4.3 billion |
| Market Forecast Value (2036) | USD 9.1 billion |
| Forecast CAGR (2026-2036) | 7.9% |
Source: Future Market Insights (FMI) historical analysis and forecast data.
Which Factors Drive Adoption of Oral Obesity Therapies?
Rising global obesity prevalence is structurally reshaping pharmaceutical demand, translating into sustained uptake of daily, compliance-friendly treatment options that fit long-term weight management routines. Novo Nordisk reinforced this shift with late-stage development of oral GLP-1 formulations during 2024, signaling market intent to move beyond injection-dependent regimens. Convenience, dosing familiarity, and reduced stigma are accelerating patient preference for pills over injectables. FMI analysis indicates that oral delivery formats improve therapy initiation and persistence rates across primary care settings. Post-pandemic health awareness continues to emphasize metabolic risk prevention, sustaining payer and provider alignment around scalable oral obesity therapies.
How is the Oral Obesity Therapies Market Segmented?
The oral obesity therapies market has been segmented based on drug class, patient indication, sales channel, and region. In terms of drug class, the market is divided into lipase inhibitors, sympathomimetic appetite suppressants, combination therapies, oral GLP-1 agonists, and other oral mechanisms. By patient indication, the market is divided into BMI ≥30 primary obesity, overweight with metabolic comorbidities, short-term weight reduction, weight maintenance, and adolescent populations. By sales channel, the market is segmented into retail pharmacies, mail-order pharmacies, telehealth platforms, hospital outpatient pharmacies, and specialty pharmacies. Regionally, the market is divided into North America, Europe, East Asia, South Asia, Latin America, and Middle East & Africa.
Why Do Sympathomimetic Appetite Suppressants Lead the Oral Obesity Therapies Market?
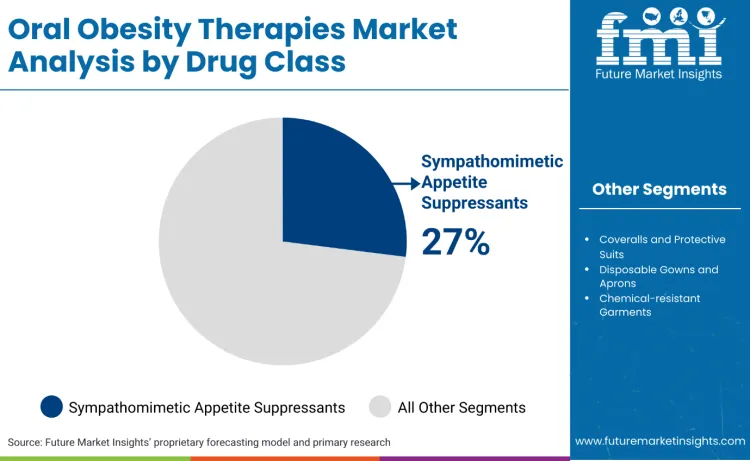
Sympathomimetic appetite suppressants continue to lead the market, accounting for 27.0% of the share. Sympathomimetic appetite suppressants, such as phentermine and diethylpropion, remain widely popular due to their high accessibility and rapid onset of action within a price-sensitive market. Vivus, Inc. markets a sympathomimetic-based oral therapy through Qsymia containing phentermine. These medications are significantly more affordable than newer hormonal therapies, making them the primary choice for patients without comprehensive insurance coverage or those seeking a lower-cost entry point into medical weight management. Their oral tablet format offers a familiar and convenient alternative to the cold-storage requirements and needle-based administration associated with many high-potency injectables currently on the market.
The popularity of these agents is also driven by their unique dual-action mechanism which provides both immediate hunger suppression and a modest boost in energy expenditure. By stimulating the release of norepinephrine in the brain, these drugs trigger a fight-or-flight response that effectively reduces the urge to eat while slightly raising the resting metabolic rate to enhance fat breakdown. This immediate physiological feedback often leads to measurable weight loss within the first few weeks of treatment, providing the rapid results and psychological reinforcement that many consumers prioritize when starting a weight-reduction regimen.
How Do Patient Indication Requirements Influence Oral Obesity Therapy Selection?
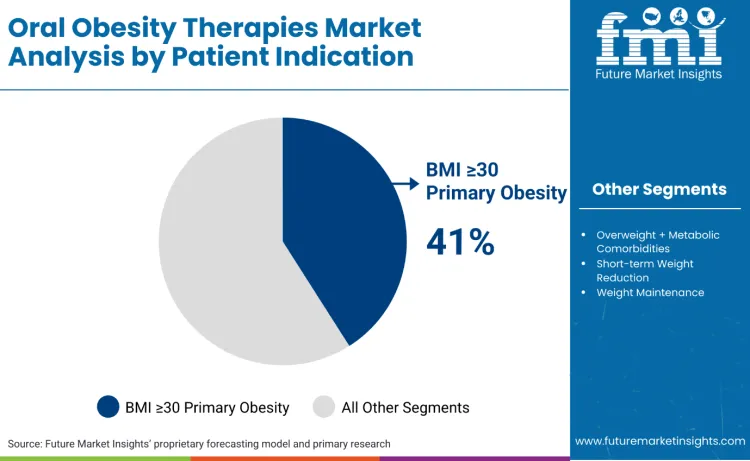
BMI ≥30 primary obesity stands as the dominant segment, commanding 41.0% of the market share. Individuals with a body mass index of 30 or higher typically utilize oral obesity therapies because they qualify for clinical intervention where lifestyle modifications alone have proven insufficient for sustainable weight loss. Novo Nordisk offers Wegovy® (semaglutide) as a prescription medication for chronic weight management in adults. For this population, oral medications serve as a vital medical tool to address the biological resistance to weight reduction by modulating hunger hormones and brain chemistry to reduce caloric intake. These therapies are often preferred over injectables because they eliminate the psychological and physical barriers of self-injection while providing a more discreet and portable treatment option for daily use.
In addition to appetite control, people in this BMI category use oral therapies to proactively manage or prevent the onset of weight-related comorbidities such as type 2 diabetes and hypertension. The convenience of a pill-based regimen supports higher long-term adherence, which is critical for the chronic management of obesity as a lifelong metabolic condition. By integrating these treatments into a daily routine, patients can achieve the clinically significant weight loss necessary to improve joint health, cardiovascular function, and overall quality of life without the complexity of cold-chain storage or specialized medical administration.
How Is the Shift Toward Oral-First Metabolic Care Restructuring Obesity Treatment?
The oral obesity therapies market is being redefined as obesity is increasingly treated as a chronic metabolic disease rather than a short-term weight loss condition. Pharmaceutical development is moving away from episodic intervention models toward daily, adherence-driven oral regimens designed for long-term cardiometabolic risk management. This transition reflects healthcare system pressure to deploy scalable, non-invasive therapies that reduce downstream costs linked to diabetes, cardiovascular disease, and liver disorders. The shift toward high-bioavailability small molecules and oral GLP-1 candidates signals a structural move away from injection-centric care pathways toward routine, primary-care-led metabolic maintenance.
Why Is the Commercialization of Oral GLP-1s a Market Inflection Point?
The market’s trajectory was materially reinforced by Novo Nordisk’s 2025-2026 pivot toward oral obesity therapeutics following regulatory progress on high-dose oral GLP-1 formulations. The launch of the Wegovy pill across USA retail pharmacies in early 2026 reframed obesity treatment from a clinic-based procedure into a standardized prescription habit. This transition lowers initiation barriers, supports long-term persistence, and expands access beyond specialty obesity centers. Parallel pipeline acceleration by Roche and Eli Lilly confirms that future value creation lies in daily oral maintenance models that integrate weight loss, cardiovascular protection, and tolerability within a single therapeutic framework.
How Are Accessibility and Sustainability Shaping Competitive Advantage?
Market leadership is increasingly tied to accessibility, affordability, and manufacturing scalability rather than efficacy alone. Pharmaceutical players are pairing oral therapies with direct-to-consumer platforms, cash-pay models, and telehealth distribution to bypass coverage gaps and accelerate uptake. At the same time, regulators and payers are tightening scrutiny on long-term safety and manufacturing consistency, particularly for mass-volume metabolic drugs. FMI analysis indicates that companies unable to align oral obesity portfolios with chronic-disease reimbursement frameworks, safety expectations, and scalable production economics will struggle to compete as obesity pharmacotherapy becomes embedded into routine healthcare delivery.
How Is the Oral Obesity Therapies Market Evolving Globally?
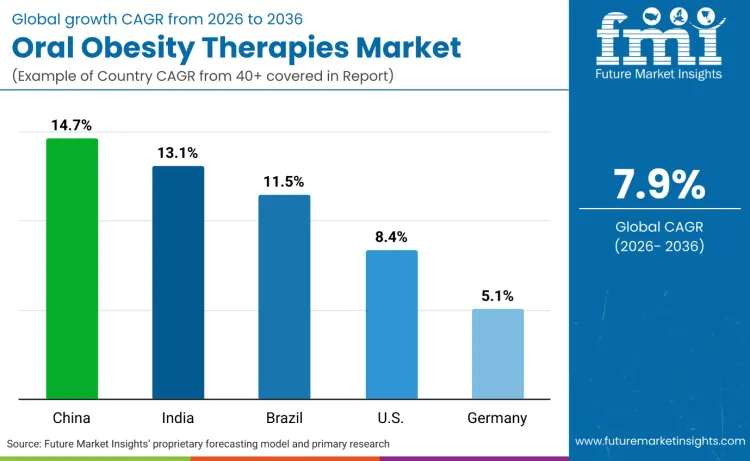
Global demand for oral obesity therapy products is increasing as healthcare providers expand treatment capabilities while addressing diverse patient populations, metabolic comorbidity management, and long-term weight maintenance requirements. Growth demonstrates rising adoption of evidence-based obesity treatment protocols, targeted therapeutic positioning, and efficacy-optimized formulations across endocrinology practices, primary care settings, and specialized weight management centers. Product selection focuses on mechanism of action, safety profile, and clinical outcomes under various patient scenarios. China records 14.7% CAGR, India records 13.1% CAGR, Brazil records 11.5% CAGR, USA records 8.4% CAGR, and Germany records 5.1% CAGR. Adoption remains driven by clinical efficacy requirements and safety considerations rather than cost reduction alone.
| Country | CAGR (2026-2036) |
|---|---|
| China | 14.7% |
| India | 13.1% |
| Brazil | 11.5% |
| USA | 8.4% |
| Germany | 5.1% |
China: How is the Expanding Healthcare Infrastructure Driving Oral Obesity Therapy Adoption?
The primary driver in China is the unprecedented growth of obesity prevalence and healthcare system modernization focusing on chronic disease management. With the rapid urbanization and dietary pattern changes, Chinese metropolitan areas are experiencing significant increases in obesity rates, making traditional lifestyle interventions insufficient for population-level impact. A key development is the Healthy China Initiative, which has promoted pharmaceutical intervention adoption for obesity management to ensure comprehensive healthcare capabilities and treatment standardization. Driven by this national scale of healthcare transformation, the Chinese oral obesity therapies market is set to grow at a 14.7% CAGR during the study period, led by massive demand in urban healthcare systems and specialized metabolic disease centers.
China’s market is expanding rapidly due to an unprecedented surge in obesity rates linked to urbanization and a fast-growing middle class. The government has responded with aggressive health reforms and chronic disease management initiatives that prioritize the adoption of oral therapies to alleviate the burden on the national healthcare system. Additionally, the entry of local biotechs developing domestic oral GLP-1 alternatives is making these treatments more affordable and culturally accessible than imported injectables.
India: Is the Rising Middle Class Healthcare Demand Elevating Treatment Standards?
In India, the market is dominated by the evolution toward comprehensive metabolic disease management and the expansion of private healthcare infrastructure. Indian healthcare providers are implementing evidence-based obesity treatment protocols alongside diabetes and cardiovascular disease management within integrated care frameworks. This comprehensive approach is essential for Indian healthcare systems currently addressing the dual burden of communicable and non-communicable diseases in urban populations. Reflecting this healthcare transformation, the Indian oral obesity therapies market is projected to grow at a 13.1% CAGR during the study period.
Over the next five years, the rise of telemedicine and digital health platforms will necessitate oral therapies that support remote patient monitoring and virtual care delivery. Second, India's growing pharmaceutical manufacturing capabilities create opportunities for cost-effective generic formulations that improve accessibility across diverse socioeconomic populations. Finally, there is expanding demand for combination therapies that address obesity alongside diabetes and hypertension, supporting integrated chronic disease management protocols in both urban and rural healthcare settings.
Brazil: Will Healthcare System Modernization Accelerate Treatment Integration?
Brazil's oral obesity therapies market is uniquely shaped by healthcare system expansion and the integration of obesity treatment into chronic disease management protocols. Brazilian healthcare providers are increasingly adopting evidence-based obesity treatment protocols, maintaining clinical positioning for improved patient outcomes while addressing broader metabolic health challenges. Consequently, the Brazilian oral obesity therapies market is expected to grow at an 11.5% CAGR during the study period, as healthcare organizations invest in comprehensive obesity management capabilities.
Brazil is experiencing strong market growth fueled by the integration of obesity management into both the public health system (SUS) and expanding private employer wellness programs. Local firms such as EMS have invested heavily in peptide production to offer more affordable oral options to a population increasingly affected by sedentary lifestyles. The Brazilian market is particularly driven by a strong consumer preference for non-invasive, pill-based treatments that can be easily distributed across the country's vast geographic regions without the need for complex refrigeration.
Germany: Is the Healthcare Digitalization Initiative Enhancing Treatment Access?
In Germany, the market is being enhanced by the healthcare digitalization strategy, which integrates advanced patient monitoring and evidence-based obesity treatment protocols into comprehensive care delivery systems. A major development is healthcare providers upgrading to digital health platforms that feature integrated obesity management capabilities to compete against traditional weight loss approaches in clinical practice applications. As German healthcare institutions prioritize evidence-based medicine and comprehensive chronic disease management, the German oral obesity therapies market is set to grow at a 5.1% CAGR during the study period, focusing on clinical effectiveness and long-term patient outcomes.
Germany’s market growth is characterized by a "clinical-first" approach where new 2025-2026 European pharmacotherapy frameworks have officially prioritized oral GLP-1s as a first-line defense against obesity-related comorbidities. German healthcare providers are increasingly prescribing oral therapies to address the specific long-term costs of heart disease and type 2 diabetes, supported by a regulatory environment that has expedited the approval of small-molecule alternatives. The growth is further bolstered by Germany’s position as a European leader in metabolic research, with domestic clinical practices shifting toward evidence-based, daily maintenance protocols.
USA: How is the Healthcare Coverage Expansion Driving Market Growth?
The USA market is increasingly focused on expanding insurance coverage for anti-obesity medications and integrating obesity treatment into comprehensive healthcare delivery models. A key driver is the Medicare and Medicaid coverage expansions, which have prioritized obesity pharmacotherapy coverage to improve long-term healthcare outcomes while reducing obesity-related healthcare costs. This transition aims at transforming obesity from an uncovered lifestyle condition to a recognized medical disease requiring pharmaceutical intervention. Supported by these coverage expansion mandates, the USA oral obesity therapies market is set to grow at an 8.4% CAGR during the study period.
Who are the Major Players Active in the Oral Obesity Therapies Market?
The oral obesity therapies market is highly competitive with a mix of established pharmaceutical companies and specialized biotechnology firms. Novo Nordisk A/S leads with a strong pipeline of oral treatments and significant investment in research focused on efficacy and safety. Roche Holding AG leverages its global reach and experience in metabolic disorders to maintain a strong position. Pfizer Inc. brings extensive drug development capabilities and global marketing infrastructure, while Teva Pharmaceutical Industries Ltd. uses its generics and specialty portfolio to expand access and affordability. Sun Pharmaceutical Industries Ltd. focuses on emerging markets with cost effective therapy options. Currax Pharmaceuticals, LLC and Vivus, Inc. operate in niche segments with targeted oral obesity therapies and strategic partnerships to enhance product reach. Arena Pharmaceuticals, Inc. contributes with novel mechanisms of action under development. The competitive landscape emphasizes innovation, regulatory approvals, strategic collaborations, and efforts to differentiate products through improved patient outcomes and convenience, with companies striving to balance clinical benefits with pricing and market access.
Key Industry Development:
- In September 2025, Roche acquired 89bio for the therapy of moderate to severe Metabolic Dysfunction-Associated Steatohepatitis (MASH), one of the common comorbidities of obesity.
- In September 2024, Currax Pharmaceuticals expanded its obesity therapy portfolio through strategic partnerships focused on novel combination therapy development and digital health integration platforms.
Key Players in the Oral Obesity Therapies Market
- Currax Pharmaceuticals, LLC
- Vivus, Inc.
- Roche Holding AG
- Teva Pharmaceutical Industries Ltd.
- Sun Pharmaceutical Industries Ltd.
- Novo Nordisk A/S
- Pfizer Inc.
- Arena Pharmaceuticals, Inc.
- Rhythm Pharmaceuticals, Inc.
- Gelesis, Inc.
Market Definition
The oral obesity therapies market refers to the global pharmaceutical applications of oral medications designed to treat obesity and facilitate weight loss through various mechanisms of action. It includes both prescription and over-the-counter formulations, with segments that cater to various patient populations such as primary obesity, metabolic comorbidities, and weight maintenance. The market size is measured in USD billion and analyzed over the 2026 to 2036 period.
The oral obesity therapies market includes drug classes such as lipase inhibitors, appetite suppressants, combination therapies, and incretin-based oral formulations, utilized through both clinical practice and direct patient applications. The market is expanding due to growing recognition of obesity as a chronic disease requiring pharmaceutical intervention, advances in oral drug delivery technologies, and evolving treatment guidelines toward comprehensive weight management approaches.
Market Inclusion
Included in the oral obesity therapies market scope are pharmaceutical products categorized by drug class (lipase inhibitors, appetite suppressants, combination therapies, incretin agonists), with segmentation based on patient indication (primary obesity, metabolic comorbidities, weight maintenance), healthcare delivery channel (retail pharmacy, specialty pharmacy, telehealth), and treatment duration (short-term, long-term maintenance). The scope includes oral obesity medications with established safety and efficacy profiles, regulatory approval status, and clinical evidence supporting weight loss outcomes, particularly those marketed through healthcare providers, pharmacy networks, and direct-to-consumer channels.
Geographically, the market encompasses key regions such as North America, Europe, East Asia, South Asia, Latin America, and the Middle East & Africa, with country-level analysis for significant markets like the USA, China, India, Brazil, and Germany.
Market Exclusion
Excluded from the oral obesity therapies market scope are non-oral obesity treatments such as injectable medications, surgical interventions, and medical devices designed for weight loss. Additionally, products not specifically indicated for obesity treatment, such as diabetes medications used off-label for weight loss, are not included unless they have specific obesity indications.
The market excludes dietary supplements, herbal products, and over-the-counter weight loss aids that do not meet pharmaceutical regulatory standards. Furthermore, products without established clinical efficacy data or those that have been withdrawn from markets due to safety concerns are outside the market's defined scope.
Scope of the Report
| Items | Values |
|---|---|
| Quantitative Units | USD billion |
| Drug Class | Lipase Inhibitors; Sympathomimetic Appetite Suppressants; Combination Therapies; Oral GLP-1 Agonists; Other Oral Mechanisms |
| Patient Indication | BMI ≥30 Primary Obesity; Overweight + Metabolic Comorbidities; Short-term Weight Reduction; Weight Maintenance; Adolescent Populations |
| Sales Channel | Retail Pharmacies; Mail-order Pharmacies; Telehealth Platforms; Hospital Outpatient Pharmacies; Specialty Pharmacies |
| Regions Covered | North America, Europe, East Asia, South Asia, Latin America, Middle East & Africa |
| Countries Covered | USA, China, India, Brazil, Germany, and 40+ countries |
| Key Companies Profiled | Currax Pharmaceuticals; Vivus; Roche; Teva Pharmaceutical Industries; Sun Pharmaceutical Industries; Others |
| Additional Attributes | Revenue analysis by drug class, patient indication, and sales channel; performance across clinical applications and therapeutic effectiveness in endocrinology practices, primary care settings, and specialized obesity management centers; weight loss optimization, metabolic improvement, and therapy benefits under various patient scenarios; impact on clinical outcomes, treatment adherence, and therapy differentiation during prescribing decisions; compatibility with treatment protocols and physician preferences; prescribing dynamics driven by clinical positioning, evidence generation programs, and long-term patient management partnerships. |
Oral Obesity Therapies Market by Segment
Drug Class:
- Lipase Inhibitors
- Sympathomimetic Appetite Suppressants
- Combination Therapies
- Oral GLP-1 Agonists
- Other Oral Mechanisms
Patient Indication:
- BMI ≥30 Primary Obesity
- Overweight + Metabolic Comorbidities
- Short-term Weight Reduction
- Weight Maintenance
- Adolescent Populations
Sales Channel:
- Retail Pharmacies
- Mail-order Pharmacies
- Telehealth Platforms
- Hospital Outpatient Pharmacies
- Specialty Pharmacies
Region:
- North America
- USA
- Canada
- Mexico
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Nordic
- Netherlands
- Rest of Europe
- East Asia
- China
- Japan
- South Korea
- South Asia
- India
- Bangladesh
- Rest of South Asia
- Latin America
- Brazil
- Argentina
- Chile
- Colombia
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Rest of Middle East & Africa
Bibliography
- International Association for the Study of Obesity. (2024). Clinical Guidelines for Oral Anti-Obesity Medications: Evidence-Based Treatment Protocols. IASO.
- Global Obesity Medicine Council. (2024). Pharmaceutical Standards for Oral Obesity Therapies: Safety and Efficacy Requirements. GOMC.
- Endocrine Society. (2024). Clinical Practice Guidelines for Pharmacological Management of Obesity (updated therapeutic specifications for clinical applications). Endocrine Society.
Frequently Asked Questions
How big is the oral obesity therapies market in 2026?
The global oral obesity therapies market is estimated to be valued at USD 4.3 billion in 2026.
What will be the size of the oral obesity therapies market in 2036?
The market size for the oral obesity therapies market is projected to reach USD 9.1 billion by 2036.
How much will the oral obesity therapies market grow between 2026 and 2036?
The oral obesity therapies market is expected to grow at a 7.9% CAGR between 2026 and 2036.
What are the key drug classes in the oral obesity therapies market?
The key drug classes in the oral obesity therapies market include sympathomimetic appetite suppressants, combination therapies, oral GLP-1 agonists, and lipase inhibitors.
Which patient indication will contribute a significant share in the oral obesity therapies market in 2026?
In terms of patient indication, the BMI ≥30 primary obesity segment is set to command a 41.0% share in the oral obesity therapies market in 2026.
Table of Content
- Executive Summary
- Global Market Overview
- Key Insights
- Market Forecast
- Strategic Recommendations
- Market Overview
- Market Definition
- Market Taxonomy
- Scope and Limitations
- Research Methodology
- Research Approach
- Data Sources
- Analytical Framework
- Market Dynamics
- Drivers
- Restraints
- Opportunities
- Trends
- Market Sizing and Forecast
- Market Value (2026-2036)
- Y-o-Y Growth Analysis
- Market Share by Drug Class
- Market Share by Patient Indication
- Market Share by Sales Channel
- Market Segmentation Analysis
- Drug Class
- Lipase Inhibitors
- Sympathomimetic Appetite Suppressants
- Combination Therapies
- Oral GLP-1 Agonists
- Other Oral Mechanisms
- Patient Indication
- BMI ≥30 Primary Obesity
- Overweight with Metabolic Comorbidities
- Short-term Weight Reduction
- Weight Maintenance
- Adolescent Populations
- Sales Channel
- Retail Pharmacies
- Mail-order Pharmacies
- Telehealth Platforms
- Hospital Outpatient Pharmacies
- Specialty Pharmacies
- Drug Class
- Regional Market Analysis
- North America
- Europe
- East Asia
- South Asia
- Latin America
- Middle East & Africa
- Competitive Landscape
- Market Share of Key Players
- Competitive Benchmarking
- Key Player Strategies
- Key Takeaways
- Market Summary
- Future Trends and Forecast
- Strategic Recommendations
List of Tables
- Global Oral Obesity Therapies Market Size (USD Billion), 2021-2036
- Oral Obesity Therapies Market Y-o-Y Growth (%), 2021-2036
- Global Market Share by Drug Class, 2026
- Global Market Share by Patient Indication, 2026
- Global Market Share by Sales Channel, 2026
- Oral Obesity Therapies Market Share by Region, 2026
- Market Share by Drug Class (USD Billion), 2026-2036
- Market Share by Patient Indication (USD Billion), 2026-2036
- Key Player Market Share in the Oral Obesity Therapies Market
- Oral Obesity Therapies Market Absolute $ Opportunity (USD Billion), 2026-2036
- Oral Obesity Therapies Market CAGR (%) by Region, 2026-2036
List of Figures
- Global Oral Obesity Therapies Market Size (USD Billion), 2021-2036
- Oral Obesity Therapies Market Y-o-Y Growth (%) from 2021-2036
- Oral Obesity Therapies Market Share by Drug Class (%) in 2026
- Oral Obesity Therapies Market Share by Patient Indication (%) in 2026
- Global Oral Obesity Therapies Market by Region (%) 2026-2036
- Global Oral Obesity Therapies Market by Sales Channel
- Competitive Landscape of Key Oral Obesity Therapies Market Players
- Market Trends in Oral Obesity Therapies
- Oral Obesity Therapies Adoption by Region

