Intratumoral Cancer Therapies Market
Intratumoral Cancer Therapies Market Size and Share Forecast Outlook 2025 to 2035
Historical Data Covered: 2015 to 2023 | Base Year: 2024 | Estimated Year: 2025 | Forecast Period: 2026 to 2035
Intratumoral Cancer Therapies Market Snapshot (2023 to 2033)
The Intratumoral Cancer Therapies Market is estimated to be valued at USD 257.7 billion in 2025 and is projected to reach USD 668.4 billion by 2035, registering a compound annual growth rate (CAGR) of 10.0% over the forecast period.
Quick Stats for Intratumoral Cancer Therapies Market
- Intratumoral Cancer Therapies Market Industry Value (2025): USD 257.7 billion
- Intratumoral Cancer Therapies Market Forecast Value (2035): USD 668.4 billion
- Intratumoral Cancer Therapies Market Forecast CAGR: 10.0%
- Leading Segment in Intratumoral Cancer Therapies Market in 2025: Lung Cancer (27.5%)
- Key Growth Region in Intratumoral Cancer Therapies Market: North America, Asia-Pacific, Europe
- Top Key Players in Intratumoral Cancer Therapies Market: Amgen Inc., F. Hoffmann-La Roche Ltd., Novartis AG, Bayer AG, Pfizer Inc., Bausch Health Companies Inc., Regeneron Pharmaceuticals Inc., Biogen, Samsung Bioepis, Ophthotech Corporation
Rationale for Segmental Growth in the Intratumoral Cancer Therapies Market
Market Overview:
The intratumoral cancer therapies market is undergoing significant transformation as advancements in precision medicine, growing oncology burden, and demand for targeted therapies converge. Rising incidence of solid tumors, coupled with limitations of systemic treatments, has accelerated the adoption of localized intratumoral approaches.
These therapies are being viewed as an effective way to enhance immune response, minimize systemic toxicity, and improve patient outcomes. Future growth is expected to be supported by ongoing clinical trials, regulatory support for breakthrough therapies, and the integration of novel delivery platforms with immuno-oncology agents.
Innovations in formulation technologies and strategic collaborations between biopharmaceutical companies and research institutions are paving the path for broader clinical adoption and expansion into additional tumor types.
Segmental Analysis:
The market is segmented by Application, Technology Type, and End User and region. By Application, the market is divided into Lung Cancer, Breast Cancer, Colorectal Cancer, Melanoma, Prostate Cancer, Head & Neck Cancer, and Others. In terms of Technology Type, the market is classified into Monoclonal Antibodies, Vaccines, Checkpoint Inhibitors, Cell Therapies, Immune System Modulators, Adoptive Cell Transfer, Cytokines, and Others. Based on End User, the market is segmented into Hospital, Cancer Research Centres, and Clinical. Regionally, the market is classified into North America, Latin America, Western Europe, Eastern Europe, Balkan & Baltic Countries, Russia & Belarus, Central Asia, East Asia, South Asia & Pacific, and the Middle East & Africa.
The market is segmented by Application, Technology Type, and End User and region. By Application, the market is divided into Lung Cancer, Breast Cancer, Colorectal Cancer, Melanoma, Prostate Cancer, Head & Neck Cancer, and Others. In terms of Technology Type, the market is classified into Monoclonal Antibodies, Vaccines, Checkpoint Inhibitors, Cell Therapies, Immune System Modulators, Adoptive Cell Transfer, Cytokines, and Others. Based on End User, the market is segmented into Hospital, Cancer Research Centres, and Clinical. Regionally, the market is classified into North America, Latin America, Western Europe, Eastern Europe, Balkan & Baltic Countries, Russia & Belarus, Central Asia, East Asia, South Asia & Pacific, and the Middle East & Africa.
Insights into the Lung Cancer Application Segment
When segmented by application, lung cancer is projected to contribute 27.5 of the total market revenue in 2025, positioning it as the leading application segment. This leadership is attributed to the high prevalence and mortality rates of lung cancer worldwide, which have necessitated innovative localized treatment options.
The anatomical accessibility of certain lung tumors has facilitated the clinical feasibility of intratumoral injections, while the urgent unmet need in advanced-stage patients has accelerated their inclusion in clinical practice. The ability of these therapies to induce both local and systemic immune responses has been particularly relevant in lung cancer management, where traditional modalities have shown limited efficacy.
Increased awareness among oncologists, patient willingness to explore novel therapies, and promising trial outcomes have further solidified this segment’s prominence within the market.
Insights into the Monoclonal Antibodies Technology Type Segment
Segmented by technology type, monoclonal antibodies are expected to account for 25.0 of the intratumoral cancer therapies market revenue in 2025, securing their position as the leading technology segment. This dominance has been driven by their proven specificity, ability to target tumor antigens effectively, and established regulatory pathways.
Their integration into intratumoral delivery has enabled enhanced local concentration at the tumor site, reducing systemic exposure and improving efficacy. The adaptability of monoclonal antibodies to conjugation with cytotoxic agents or immune modulators has further expanded their therapeutic potential in this setting.
Continuous innovation in antibody engineering and the development of bispecific and multifunctional formats have reinforced their relevance in the market, allowing them to maintain a competitive advantage in intratumoral applications.
Insights into the Hospital End User Segment
When segmented by end user, hospitals are forecast to hold 33.0 of the market revenue in 2025, establishing themselves as the leading end user segment. This leadership stems from hospitals’ central role in delivering complex oncology care, supported by multidisciplinary teams and advanced procedural infrastructure.
The technical expertise required for intratumoral administration, along with the need for real-time imaging and monitoring, has concentrated these therapies within hospital settings. Institutional focus on offering cutting-edge treatments, combined with the ability to participate in clinical trials and adopt emerging protocols swiftly, has reinforced hospitals as the preferred venue for such interventions.
Additionally, the integration of oncology departments with research initiatives and the availability of specialized personnel have further strengthened hospitals’ dominance in facilitating access to intratumoral cancer therapies.
2020 to 2024 Intratumoral Cancer Therapies Market Demand Analysis vs. Forecast 2025 to 2035
The complication in various people have increased. People suffering from even stage 1 cancer faced complications as the effects of the virus affected the immune system gravely. This paced up the demand for cancer therapies and also made the companies expand the product portfolio to provide enhanced immunity to the patients.
Thus, the market for Intratumoral Cancer Therapies is expected to register a CAGR of 10% in the forecast period 2025 to 2035.
Which are Some Prominent Drivers of Intratumoral Cancer Therapies Market?
Increasing incidence of cancer to push the market growth
According to the Globocan 2024 fact sheet, an estimated 19,292,789 new cancer cases were diagnosed worldwide, with nearly 9,958,133 deaths due to cancers, globally. Additionally, according to estimates from the International Agency for Research on Cancer (IARC), by 2040, the global burden of cancers is expected to grow to 27.5 million new cancer cases and 16.3 million deaths worldwide. The increasing incidence of cancer across the globe has been rapidly adding to the growth in the field of cancer research.
In April 2024, the US FDA approved Trodelvy (sacituzumab govitecan-hziy) for the treatment of adult patients with triple-negative breast cancer. Furthermore, in May 2024, the US FDA approved Tabrecta (capmatinib) for the treatment of adult patients with non-small cell lung cancer (NSCLC), and it is the first FDA-approved targeted therapy to treat NSCLC with specific mutations. Thus, the high research activities related to targeted therapies against cancers are expected to grow over the forecast period.
What are the Challenges Faced by the Intratumoral Cancer Therapies Market?
Strict Regulations and Higher Costs to restrict Market Growth
Inadequate reimbursement regulations are predicted to restrain market expansion. In recent years, there have been several developments in the cancer therapy industry. However, most medical insurance companies do not cover procedures that use computer-aided detection (CAD). As a consequence, individuals choose traditional diagnostic methods such as biopsies, and pathology tests, or conventional imaging methods such as mammography and MRI treatments. Furthermore, the high cost of intratumoral cancer therapy is projected to stifle market expansion.
Region-Wise Insights
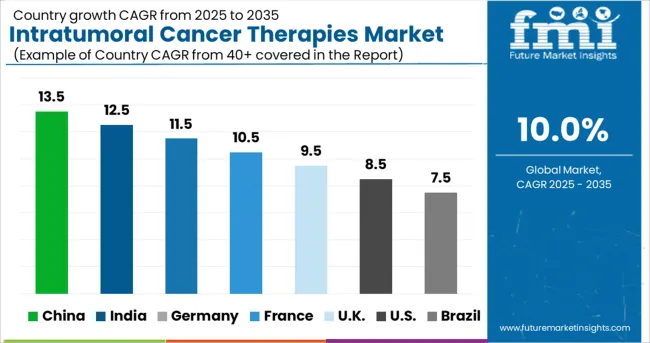
How is the Adoption of the Intratumoral Cancer Therapies Market in Europe Turning Out?
Increased government funding propelling the growth of Intratumoral Cancer Therapies in Europe
The European market is predicted to increase significantly throughout the forecast period, with a CAGR of 5% during the forecast period. Europe is estimated to be the fastest-growing region over the forecast period due to an increase in research funding by government bodies to help combat the growing incidence of cancer cases across the region. In addition, the existence of major players promotes expansion in the United Kingdom. These firmsʜ.49; strategies include new releases and growth initiatives, which will continue to help overall expansion in Europe.
In addition to this, the European Union’s ‘Horizon Europe Mission on Cancer’ was launched in September 2024 to offer funds to a broad spectrum of activities that are intended to lower Europe’s cancer burden by accelerating research and innovation in cancer therapeutics. The mission is anticipated to help over 668.4 million cancer survivors by the year 20668.40.
What are the Factors Boosting the Market for Intratumoral Cancer Therapies in North America?
High Concentration of Key Players shaping the landscape for Intratumoral Cancer Therapies in North America
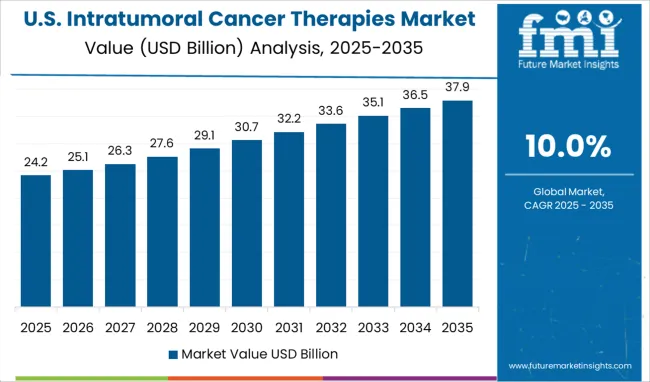
North America is expected to increase its market share in the future, owing to the increased adoption of cancer therapy and the growing burden of cancers in the USA. According to the Globocan 2024 report, an estimated 2,281,658 new cancer cases were diagnosed in the USA in 2024, with nearly 612,668.490 deaths. In 2024, The most common cancers were breast (25668.4,465), lung (227,875), prostate (209,512), and colon (101,809) in the USA.
In terms of market share and revenue, the North American market is predicted to be the most lucrative market during the forecast period with a revenue share of 40%. One of the primary factors propelling the industry in the region is the presence of various biotechnology and medical device firms. North America is likely to maintain its lead during the projection period due to factors such as increased spending for Research and Development initiatives and widespread use of new technology.
The regionʜ.49;s high concentration of important players, growing FDA approvals, strategic partnerships, and substantial expenditure in research & development activities are projected to enhance the market. For instance, in November 2024, Merck & Co. Inc entered into a definitive agreement to acquire Velosbio Inc., which is a privately held clinical-stage biopharmaceutical company committed to developing first-in-class cancer therapies targeting receptor tyrosine kinase-like orphan receptor 1 (ROR1).
Thus, given the aforementioned factors, the cancer therapy market is expected to grow significantly over the forecast period in North America.
Category-Wise Insights
Based on Technology, Which Segment is Pushing the Market Growth?
The superior efficiency of checkpoint inhibitors bolsters the growth
On the basis of technology, the checkpoint inhibitors segment is estimated to witness the highest CAGR. The high growth can be attributed to the benefits offered by this therapeutic drug. Furthermore, the increasing product approvals by the regulatory authorities are expected to contribute to the market's growth. For instance, in August 2024, FDA approved the Glaxosmithkline PD-1 checkpoint inhibitor Jemperli (dostarlimab) for adults with mismatch repair-deficient recurrent or advanced endometrial cancer. Jemperli is an anti-PD-1 antibody that binds to the PD-1 receptor and blocks its interaction with the PD-1 ligands PD-L1 and PD-L2.
By Application, Which Segment Dominates the Market?
Increased incidence of lung cancer to drive market growth
By application, lung cancer is estimated to account for the largest share of the market. Due to the increasing number of patients suffering from lung diseases, the lung cancer segment is expected to hold the largest share in the next ten years as well. Increasing incidences of lung cancer due to the rising smoking population is one of the major factors driving the growth of the lung cancer therapeutics market. The risk of lung cancer is tenfold higher in smokers as compared to non-smokers.
The market is being driven by a growing demand for tailored therapy, the availability of highly effective medications, an increase in the elderly population, and an increase in the prevalence of unhealthy lifestyles.
Start-up in the Intratumoral Cancer Therapies Market
Key start-up players in the Intratumoral Cancer Therapies market are-
- Race Oncology is an ASX-listed precise oncology firm that is developing Zantrene®, a Phase 2/3 cancer medication. Zantrene® is a powerful fatso/fat mass and obesity-linked (FTO) protein inhibitor. Overexpression of FTO has been identified as a genetic driver of a variety of malignancies. Race is investigating the potential of Zantrene® as a novel treatment for melanoma and clear cell renal cell carcinoma, two malignancies that frequently over-express FTO. In groundbreaking preclinical research, Race revealed that Zantrene® protects against anthracycline-induced heart damage while also functioning in combination with anthracyclines to boost their capacity to target breast cancer. Race is assessing this discovery.
- Carer, which was founded in 2020, aims to improve the quality of life of every cancer patient in the convenience of their own home. It attempts to give patients with the best degree of support and therapy both during and after treatment to help recovery. Their therapy has a significant benefit in that it can be tailored to each patient's diagnosis, prior health difficulties, and chosen lifestyle, making it more feasible in the long run. This is accomplished through their three-pronged approach, which includes a dietary protocol, a movement and meditation protocol, and a mental well-being protocol that educates and motivates patients and caregivers to make life-changing adjustments for more successful healing.
Market Competition
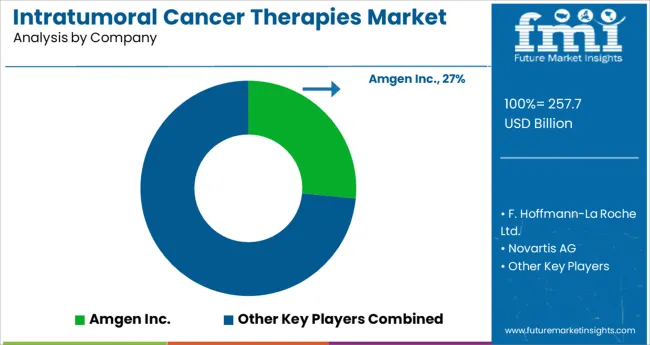
Key players in the Intratumoral Cancer Therapies market are Amgen Inc, AstraZeneca, Bayer AG, Bristol-Myers Squibb Company, Pfizer Inc, Novartis AG, Johnson & Johnson, and Eli Lily Company, F.
- Gland Pharma has launched the cancer treatment medicine Bortezomib for Injection in the USA in 2025. The drug business stated in a statement that it has released the medicine in the US market through a partner who has acquired permission from the US Food and Drug Administration (USFDA). The business claims that its medicine is medically similar to Velcade for Injection from Takeda Pharmaceuticals. Bortezomib for Intravenous is used to treat cancers such as myeloma and mantle cell lymphoma.
- Bristol-Myers Squibb K.K. announced in 2025 that the Japanese Ministry of Health, Labour, and Welfare had approved Abecma (idecabtagene vicleucel), a B-cell maturation antigen (BCMA)-directed chimeric antigen receptor (CAR) T cell immunotherapy, for the treatment of adult patients with refractory or relapsed (R/R) multiple myeloma who had received a minimum of three prior therapies. In Japan, Abecma was the first CAR T cell therapy licensed for treating R/R multiple myeloma in 2024. In Japan, Abecma is the first CAR T cell therapy licensed for treating R/R multiple myeloma in 2024. Bristol Myers Squibb is currently the only firm in Japan with two authorized CAR T cell therapies: the CD19-directed Breyanzi, which was approved in March 2024, and the BCMA-directed Abecma.
Report Scope
| Report Attribute | Details |
|---|---|
| Market Value in 2025 | USD 257.7 billion |
| Market Value in 2035 | USD 668.4 billion |
| Growth Rate | CAGR of 10% from 2025 to 2035 |
| Base Year for Estimation | 2025 |
| Historical Data | 2020 to 2024 |
| Forecast Period | 2025 to 2035 |
| Quantitative Units | Revenue in billion and CAGR from 2025 to 2035 |
| Report Coverage | Revenue Forecast, Volume Forecast, Company Ranking, Competitive Landscape, Growth Factors, Trends, and Pricing Analysis |
| Segments Covered | End-Users, Application, Technology, Region |
| Regions Covered | North America; Latin America; Europe; Asia Pacific; Middle East and Africa (MEA) |
| Key Countries Profiled | USA, Canada, Brazil, Argentina, Germany, UK, France, Spain, Italy, Nordics, BENELUX, Australia & New Zealand, China, India, ASIAN, GCC, South Africa |
| Key Companies Profiled | Amgen Inc; AstraZeneca; Bayer AG; Bristol-Myers Squibb Company; Pfizer Inc; Novartis AG; Johnson & Johnson; and Eli Lily Company; F. |
| Customization | Available Upon Request |
Key Segments Profiled in the Intratumoral Cancer Therapies Industry Survey
By Technology:
- Monoclonal Antibodies
- Vaccines
- Checkpoint Inhibitors
- Cell Therapies
- Immune System Modulators
- Adoptive Cell Transfer
- Cytokines
By Application:
- Lung Cancer
- Breast Cancer
- Melanoma
- Prostate Cancer
- Head & Neck Cancer
- Others
By End-users:
- Hospitals
- Cancer Research Centres
- Clinics
By Region:
- North America
- Latin America
- Europe
- Asia Pacific
- Middle East and Africa (MEA)
Frequently Asked Questions
How big is the intratumoral cancer therapies market in 2025?
The global intratumoral cancer therapies market is estimated to be valued at USD 257.7 billion in 2025.
What will be the market size for the intratumoral cancer therapies market in 2035?
It is projected to reach USD 668.4 billion by 2035.
How much will be the CAGR of the intratumoral cancer therapies market between 2025 and 2035?
The market is expected to grow at a 10.0% CAGR between 2025 and 2035.
What are the key product types in the intratumoral cancer therapies market?
The key product types are lung cancer, breast cancer, colorectal cancer, melanoma, prostate cancer, head & neck cancer and others.
Which monoclonal antibodies segment is expected to dominate the intratumoral cancer therapies market in 2025?
monoclonal antibodies segment is expected to dominate with a 25.0% industry share in 2025.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand-side Trends
- Supply-side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2020 to 2024 and Forecast, 2025 to 2035
- Historical Market Size Value (USD Million) Analysis, 2020 to 2024
- Current and Future Market Size Value (USD Million) Projections, 2025 to 2035
- Y-o-Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2020 to 2024 and Forecast 2025 to 2035
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application , 2020 to 2024
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application , 2025 to 2035
- Lung Cancer
- Breast Cancer
- Colorectal Cancer
- Melanoma
- Prostate Cancer
- Head & Neck Cancer
- Others
- Y-o-Y Growth Trend Analysis By Application , 2020 to 2024
- Absolute $ Opportunity Analysis By Application , 2025 to 2035
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Technology Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Technology Type, 2020 to 2024
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Technology Type, 2025 to 2035
- Monoclonal Antibodies
- Vaccines
- Checkpoint Inhibitors
- Cell Therapies
- Immune System Modulators
- Adoptive Cell Transfer
- Cytokines
- Others
- Y-o-Y Growth Trend Analysis By Technology Type, 2020 to 2024
- Absolute $ Opportunity Analysis By Technology Type, 2025 to 2035
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User, 2020 to 2024
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User, 2025 to 2035
- Hospital
- Cancer Research Centres
- Clinical
- Y-o-Y Growth Trend Analysis By End User, 2020 to 2024
- Absolute $ Opportunity Analysis By End User, 2025 to 2035
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2020 to 2024
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2025 to 2035
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- USA
- Canada
- Mexico
- By Application
- By Technology Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Application
- By Technology Type
- By End User
- Key Takeaways
- Latin America Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Application
- By Technology Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Application
- By Technology Type
- By End User
- Key Takeaways
- Western Europe Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Application
- By Technology Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Application
- By Technology Type
- By End User
- Key Takeaways
- Eastern Europe Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Application
- By Technology Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Application
- By Technology Type
- By End User
- Key Takeaways
- East Asia Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- China
- Japan
- South Korea
- By Application
- By Technology Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Application
- By Technology Type
- By End User
- Key Takeaways
- South Asia and Pacific Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Application
- By Technology Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Application
- By Technology Type
- By End User
- Key Takeaways
- Middle East & Africa Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Application
- By Technology Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Application
- By Technology Type
- By End User
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2024
- By Application
- By Technology Type
- By End User
- Canada
- Pricing Analysis
- Market Share Analysis, 2024
- By Application
- By Technology Type
- By End User
- Mexico
- Pricing Analysis
- Market Share Analysis, 2024
- By Application
- By Technology Type
- By End User
- Brazil
- Pricing Analysis
- Market Share Analysis, 2024
- By Application
- By Technology Type
- By End User
- Chile
- Pricing Analysis
- Market Share Analysis, 2024
- By Application
- By Technology Type
- By End User
- Germany
- Pricing Analysis
- Market Share Analysis, 2024
- By Application
- By Technology Type
- By End User
- UK
- Pricing Analysis
- Market Share Analysis, 2024
- By Application
- By Technology Type
- By End User
- Italy
- Pricing Analysis
- Market Share Analysis, 2024
- By Application
- By Technology Type
- By End User
- Spain
- Pricing Analysis
- Market Share Analysis, 2024
- By Application
- By Technology Type
- By End User
- France
- Pricing Analysis
- Market Share Analysis, 2024
- By Application
- By Technology Type
- By End User
- India
- Pricing Analysis
- Market Share Analysis, 2024
- By Application
- By Technology Type
- By End User
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2024
- By Application
- By Technology Type
- By End User
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2024
- By Application
- By Technology Type
- By End User
- China
- Pricing Analysis
- Market Share Analysis, 2024
- By Application
- By Technology Type
- By End User
- Japan
- Pricing Analysis
- Market Share Analysis, 2024
- By Application
- By Technology Type
- By End User
- South Korea
- Pricing Analysis
- Market Share Analysis, 2024
- By Application
- By Technology Type
- By End User
- Russia
- Pricing Analysis
- Market Share Analysis, 2024
- By Application
- By Technology Type
- By End User
- Poland
- Pricing Analysis
- Market Share Analysis, 2024
- By Application
- By Technology Type
- By End User
- Hungary
- Pricing Analysis
- Market Share Analysis, 2024
- By Application
- By Technology Type
- By End User
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2024
- By Application
- By Technology Type
- By End User
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2024
- By Application
- By Technology Type
- By End User
- South Africa
- Pricing Analysis
- Market Share Analysis, 2024
- By Application
- By Technology Type
- By End User
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Application
- By Technology Type
- By End User
- Competition Analysis
- Competition Deep Dive
- Amgen Inc.
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- F. Hoffmann-La Roche Ltd.
- Novartis AG
- Bayer AG
- Pfizer Inc.
- Bausch Health Companies Inc.
- Regeneron Pharmaceuticals Inc.
- Biogen
- Samsung Bioepis
- Ophthotech Corporation
- Amgen Inc.
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2020-2035
- Table 2: North America Market Value (USD Million) Forecast by Country, 2020-2035
- Table 3: Latin America Market Value (USD Million) Forecast by Country, 2020-2035
- Table 4: Western Europe Market Value (USD Million) Forecast by Country, 2020-2035
- Table 5: Eastern Europe Market Value (USD Million) Forecast by Country, 2020-2035
- Table 6: East Asia Market Value (USD Million) Forecast by Country, 2020-2035
- Table 7: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2020-2035
- Table 8: Middle East & Africa Market Value (USD Million) Forecast by Country, 2020-2035
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2020-2035
- Figure 3: Global Market Value (USD Million) Share and BPS Analysis by Region, 2025 and 2035
- Figure 4: Global Market Y-o-Y Growth Comparison by Region, 2025-2035
- Figure 5: Global Market Attractiveness Analysis by Region
- Figure 6: North America Market Incremental $ Opportunity, 2025-2035
- Figure 7: Latin America Market Incremental $ Opportunity, 2025-2035
- Figure 8: Western Europe Market Incremental $ Opportunity, 2025-2035
- Figure 9: Eastern Europe Market Incremental $ Opportunity, 2025-2035
- Figure 10: East Asia Market Incremental $ Opportunity, 2025-2035
- Figure 11: South Asia and Pacific Market Incremental $ Opportunity, 2025-2035
- Figure 12: Middle East & Africa Market Incremental $ Opportunity, 2025-2035
- Figure 13: North America Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 14: Latin America Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 15: Western Europe Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 16: Eastern Europe Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 17: East Asia Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 18: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 19: Middle East & Africa Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 20: Global Market - Tier Structure Analysis
- Figure 21: Global Market - Company Share Analysis

