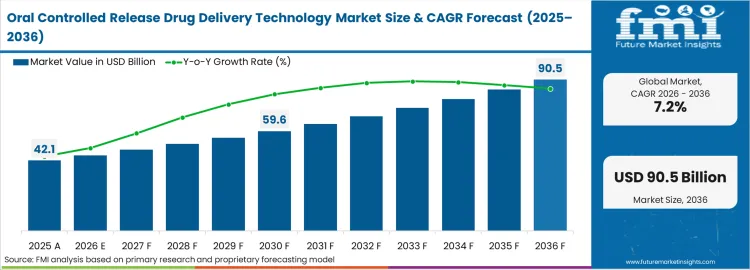
In 2025, the oral controlled release drug delivery technology market was valued at USD 42.10 billion. Based on Future Market Insights' analysis, demand is estimated to grow to USD 45.13 billion in 2026 and USD 90.45 billion by 2036. FMI projects a CAGR of 7.20% during the forecast period.
The market is set to add approximately USD 45.32 billion in absolute terms between 2026 and 2036. Chronic disease prevalence growth requiring long-term pharmacotherapy and pharmaceutical lifecycle management strategies extending patent protection through reformulation are the dual drivers sustaining controlled release technology adoption. Osmotic-controlled and matrix systems dominate new formulation development because they deliver the most predictable zero-order or near-zero-order release kinetics validated by regulatory agencies.
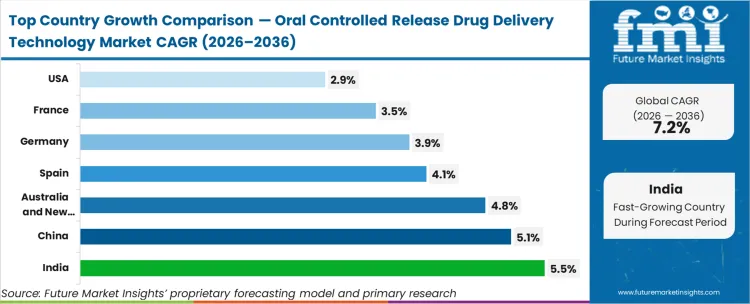
As per FMI, country-level growth rates through 2036 are projected as follows: India at 5.50%, China at 5.10%, South Korea at 5.00%, Australia and New Zealand at 4.80%, Spain at 4.10%, Germany at 3.90%, France at 3.50%, USA at 2.90%. India records the fastest expansion driven by concentrated industry investment.
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 45.13 Billion |
| Industry Value (2036) | USD 90.45 Billion |
| CAGR (2026 to 2036) | 7.20% |
Source: Future Market Insights, 2026
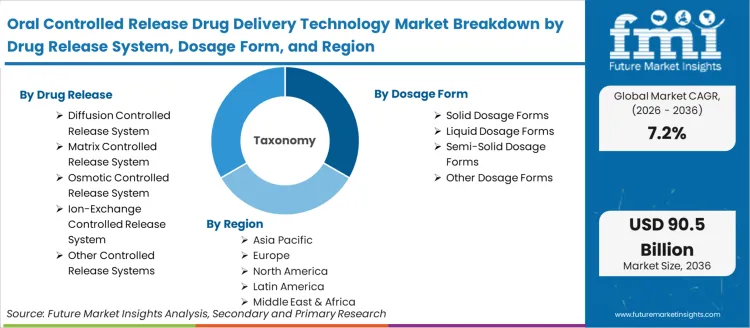
The oral controlled release drug delivery technology market encompasses formulation platforms that modulate oral drug release kinetics to optimize therapeutic outcomes. System categories span diffusion-controlled membrane systems, matrix-controlled hydrophilic and lipophilic polymer systems, osmotic-controlled OROS and push-pull platforms, and ion-exchange resin-based systems. FMI is of the opinion that the market is driven by the dual forces of chronic disease prevalence growth (requiring sustained drug release over extended periods) and pharmaceutical lifecycle management (extending branded product revenue through controlled release reformulation of immediate-release predecessors).
Market scope covers diffusion-controlled, matrix-controlled, osmotic-controlled, and ion-exchange-controlled release drug delivery systems in solid, liquid, and semi-solid oral dosage forms across all therapeutic areas.
Immediate-release oral dosage forms, parenteral (injectable) controlled release systems, transdermal drug delivery patches, and implantable drug delivery devices are excluded.
The Integration of 3D Printing Technology in Pharmaceutical Manufacturing Is a Significant Driver for The Market Growth.
The integration of 3D printing technology is transforming drug formulation since it can develop personalized oral dosage forms with the most tailored release profiles. Differing from conventional manufacturing approaches, 3D printing allows for an accurate control on drug layering and dosage and in the spatial arrangement of APIs and thus the formulation of complex drug delivery systems intended to meet special therapeutic needs.
This technology enables the customization of drug release kinetics, allowing for controlled, delayed, or multi-phase release patterns. By designing formulations that match individual patient needs, such as age, weight, metabolism, or disease progression, 3D printing significantly enhances treatment efficacy and patient compliance. Moreover, the technology supports the development of polypill formulations-combining multiple APIs into a single tablet-while maintaining distinct release profiles for each drug, addressing polypharmacy challenges.
It also produces small, patient-specific batches on demand, thus reducing waste and streamlining supply chains for more cost-effective solutions in specialized treatment regimens. Additionally, the versatility of 3D printing to accommodate a wide range of APIs, especially those with poor solubility or stability, makes it a versatile tool for advancing oral controlled release technologies. With the regulatory frameworks adapting to support such innovations, the adoption of 3D printing in drug delivery continues to propel market growth.
Expansion of Biopharmaceuticals and Complex Molecules Is Propelling the Market Growth for Oral Controlled Release Drug Delivery Technology.
Oral controlled release drug delivery technology market growth is driven by ongoing development and expansion of biopharmaceuticals and development of highly complex drugs. Biopharmaceuticals-MAbs, peptides, and nucleic acid-based therapies-continue to overshadow the pharmaceutical landscape for their targeted actions and superior therapeutic potentials.
However, these complex constituents, unfortunately, witness unfavorably low bioavailability, instability while traversing the gastrointestinal terrain, and hard-to-maintain therapeutic concentrations over a significant period.
Oral controlled release systems are being innovatively derived to redress the issues that they could have. Different advanced delivery systems function in protecting sensitive biopharmaceuticals from enzymatic degradation and from harsh gastric conditions; thus, their stability and efficacy remain enhanced.
Controlled release formulations also facilitate the sustained release of these drugs, optimizing therapeutic outcomes by maintaining consistent plasma drug levels while minimizing the frequency of administration. This becomes more critical in chronic disease conditions requiring long-term treatment where patient compliance becomes an issue of great importance.
Moreover, the customizability of controlled release systems will allow tuning of drug delivery profiles to derive pharmacokinetics and pharmacodynamics of complex compounds. Among others, microencapsulation, nanoformulation, and bioadhesive systems are gaining preeminence in enhancing the delivery of these drugs.
As the pipeline for biopharmaceuticals and other complex drugs continues to grow, the demand for innovative oral controlled release technologies is expected to rise, driving the growth of the market.
Competition from Alternative Drug Delivery Systems Is a Significant Restraint for The Oral Controlled Release Drug Delivery Technology Market.
While oral controlled release systems provide sustained drug release and improved patient compliance, they compete with other delivery methods that may be preferred in specific therapeutic areas.
Injectable drug delivery systems, such as subcutaneous, intravenous, or intramuscular injections, allow for a quicker onset of action and the ability to achieve higher bioavailability.
In some conditions, such as cancer, pain management, or emergencies, injectable systems are preferred because they can deliver drugs directly into the bloodstream, bypassing the digestive system and avoiding the variability in absorption seen with oral formulations. The injectable systems can be dosed more accurately and are, therefore, suitable for drugs that require rapid or high concentration release.
Implantable devices also compete with oral controlled release systems, like drug-eluting implants. The benefit of implantable devices is the localized and long-term drug delivery, thereby less frequent dosing. This kind of therapy has been of value in chronic conditions wherein targeted delivery to a particular site is needed; for example, in ophthalmology, oncology, or hormone therapy.
The preference for these alternative systems in some therapeutic areas limits the adoption of oral controlled release systems because they might offer improved treatment outcomes or have faster and even more precise drug action. Such competition does discourage the growth prospects of some sectors in the pharmaceutical market on oral controlled release technologies.
The Growing Demand for Personalized Medicine Presents a Significant Opportunity for The Market.
As healthcare shifts towards a more individualized treatment plan, the requirement for custom-made drug formulations increases. This trend fits excellently with oral controlled release systems that can be engineered for each individual patient.
Personalized medicine needs drug delivery systems that can be adapted to several factors, including age, weight, comorbidities, and genetic profiles. Furthermore, some pharmacokinetic characteristics, including individual absorption rates and metabolism, must be integrated. An oral controlled release system allows the release rate of drug formulations to be changed so that medication reaches the proper level in the bloodstream at the right time while reducing side effects.
This opportunity also opens up a greater role for drug compounding services. Pharmacists can prepare patient-specific oral controlled release medications for special health needs: IV and weighing adjustments for pediatric or geriatric populations; and combination products that reduce polypharmacy congestions combined into a single dosage form.
As the healthcare system increasingly focuses on individualized treatment, the market of customized oral controlled release systems is expected to grow; more patients will seek individualized treatments that match their specific needs.
The market for oral controlled release drug delivery technology has witnessed tremendous evolution in the past few decades. Early formulations essentially addressed the issue of prolonged drug release, whereas recent innovations are more towards individualized and targeted approaches. The industry has seen the emergence of different technologies over the years, including microencapsulation, polymer-based systems, and advanced biocompatible materials.
In the near future, revolutionary trends will dominate this market with the focus on the advanced drug delivery technologies such as 3D printing, nanotechnology, and smart drug delivery systems.
These emerging technologies are also enhancing the capabilities to produce extremely personalized formulations having intricate release profiles that enable controlling the release kinetics with great precision. Bio-responsive materials will find integration along with sustainable and eco-friendly approaches leading to an even more efficient drug delivery ecosystem which is more patient-centric.
As the healthcare landscape continues to focus on individualized treatment, the oral controlled release market is expected to further evolve with technology advancements that make drug delivery more efficient, targeted, and sustainable. This shift will ensure that patients receive optimized treatments tailored to their unique medical needs.
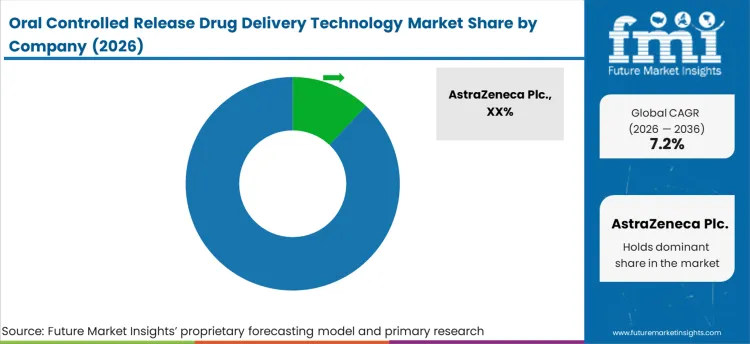
Tier 1 companies comprise market leaders with a significant market share of 52.3% in global market. These companies engage in strategic partnerships and acquisitions to expand their product portfolios and access cutting-edge technologies. Additionally, they emphasize extensive clinical trials to validate the efficacy and safety of their products. Prominent companies in tier 1 include AstraZeneca Plc., Bristol-Myers Squibb Company and Merck & Co. Inc.
Tier 2 companies include mid-size players having presence in specific regions and highly influencing the local market and holds around 23.7% market share. They typically pursue partnerships with multispecialty hospitals and research organizations to leverage emerging technologies and expedite product development.
These companies often emphasize agility and adaptability, allowing them to quickly bring new products to market, additionally targeting specific types medical needs. Additionally, they focus on cost-effective production methods to offer competitive pricing. Prominent companies in tier 2 include Novartis AG, Pfizer Inc., F. Hoffman-La-Roche Ltd. and GlaxoSmithKline Plc.
Finally, Tier 3 companies, such as Sun Pharmaceuticals, BioNTech and Gilead Sciences and others. They specialize in specific products and cater to niche markets, adding diversity to the industry.
Overall, while Tier 1 companies are the primary drivers of the market, Tier 2 and 3 companies also make significant contributions, ensuring the oral controlled release drug delivery technology sales remains dynamic and competitive.
The section below covers the industry analysis for the market for oral controlled release drug delivery technology for different countries. Market demand analysis on key countries in several regions of the globe, including North America, Latin America, East Asia, South Asia & Pacific, Western Europe, Eastern Europe and Middle Ease & Africa, is provided. The United States is anticipated to remain at the forefront in North America, with higher market share through 2036. In South Asia & Pacific, India is projected to witness a CAGR of 5.5% by 2036.
.webp)
| Countries | Value CAGR (2026 to 2036) |
|---|---|
| United States | 2.9% |
| Germany | 3.9% |
| France | 3.5% |
| Spain | 4.1% |
| China | 5.1% |
| India | 5.5% |
| Australia & New Zealand | 4.8% |
| South Korea | 5.0% |
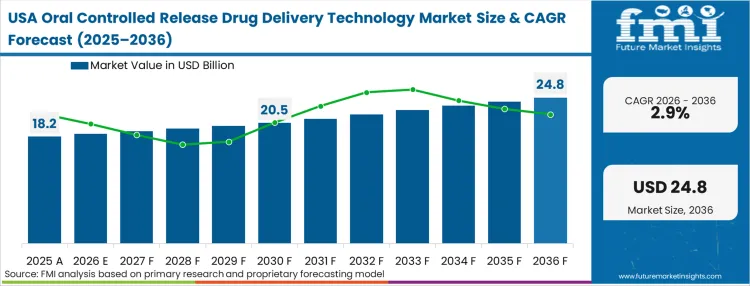
United States oral controlled release drug delivery technology market is poised to exhibit a CAGR of 2.9% between 2026 and 2036. Currently, it holds the highest share in the North American market.
The established pharmaceutical compounding services market in the United States contributes significantly to the growth of oral controlled release drug delivery systems. Compounding pharmacies are able to formulate drugs specifically for individual patient needs, especially when off-the-shelf medicines are not applicable. Tailored oral controlled release formulations from these services offer a customized solution based on specific health conditions, patient demographics, and drug compatibility.
This customization is very important to patients who have their drug dosage needs, release profiles, or active ingredients combined from multiple drugs. It is mainly useful for children and elderly people and those who have special metabolic conditions.
As personalized medicine continues to be on the forefront in the USA, the requirement for patient-specific drug delivery solutions is increasing and, therefore, compounding services are a vital growth driver for the oral controlled release market. This trend will continue to expand because healthcare is putting more emphasis on individualized treatments.
India market is poised to exhibit a CAGR of 5.5% between 2026 and 2036. Currently, it holds the highest share in the South Asia & Pacific market, and the trend is expected to continue during the forecast period.
The oral controlled release drug delivery market is growing in India due to the strong pharma industry, which is seeing continuous investment in R&D. India is home to a thriving pharmaceutical sector with many generic drug manufacturers and the growing presence of multinational pharmaceutical companies. The country's strong R&D capabilities have made it an abode of innovation, primarily of drug delivery technologies.
Pharmaceutical companies are looking forward to investment in developing innovative oral controlled release formulation for addressing the unmet medical needs, enhanced patient compliance, and improved therapeutic effects. The affordable production environment in India is an added benefit, and the advancement of drug delivery systems can be offered at cost-effective prices, targeting both national and international markets.
With government support for pharmaceutical innovation and a growing emphasis on healthcare access, India's pharmaceutical industry is playing a crucial role in advancing oral controlled release technologies, fostering market expansion.
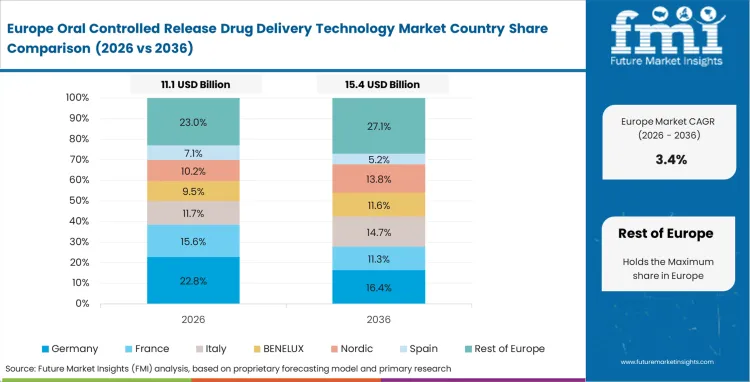
Germany’s market is poised to exhibit a CAGR of 3.9% between 2026 and 2036. Currently, it holds the highest share in the Western Europe market, and the trend is expected to continue during the forecast period.
Germany's dominance in the oral controlled drug delivery technology market can be attributed to its strong export opportunities and access to broader European and global markets. It is the largest economy in Europe where the European pharmaceutical industry is centralized. With well-established pharmaceutical companies inclusive of multinational corporations and innovative biotechnology houses, the German pharmaceutical sector has a strong focus on the production of quality and advanced drug delivery systems, including oral controlled drug delivery systems.
Germany’s strategic location within the European Union (EU) makes it easy for it to act as a doorway to the EU market, granting direct access to a unitary regulatory regime, and a huge customer base spread across the EU. Hence, German pharmaceutical companies have easy access to the EU for a rapid launch of new drug-delivery technologies and continue to meet the demands for effective, patient-friendly treatments.
Apart from a strong domestic market, Germany is among the top exporters of pharmaceuticals. The country becomes important in the global trade of oral controlled release technologies because of a favorable manufacturing environment, advanced research and development capabilities, and fierce competition in pricing in relation to other regions, including North America and Asia.
The section contains information about the leading segments in the industry. By drug release system, the diffusion controlled release system segment holds the highest market share of 25.5% in 2025.
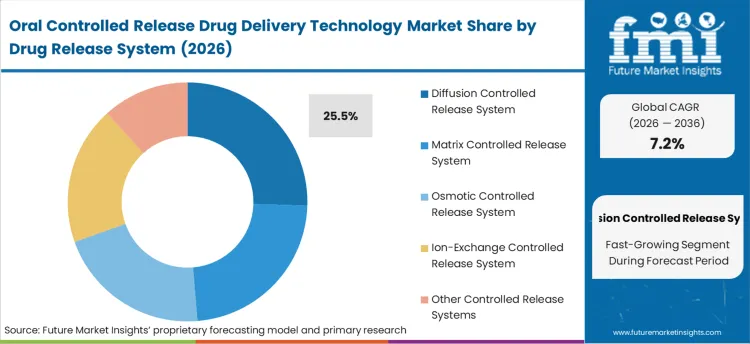
| By Drug Release System | Diffusion Controlled Release System |
|---|---|
| Value Share (2026) | 25.5% |
The diffusion controlled drug delivery system will be a key judgment factor for dominance in the market for oral controlled release drug delivery technology, thanks to its vast application and practicality in achieving steady state drug release profiles. A controlled release system provides a slow and correlative release of drug from the delivery system into the gastrointestinal tract over time through the principle of diffusion. By reducing the frequency of dosing, it helps in better bioavailability and improves patient compliance.
Some drugs release more efficiently by this mechanism over a longer duration of time, thus better suited for treating chronic ailments like diabetes, hypertension, and cardiovascular diseases. Pharmaceutical companies appreciate diffusion-controlled systems for their versatility and rate of release modulation according to the properties of the drug.
The ease of formulation along with cost-effectiveness has contributed to their acceptance all across the realm of therapeutic areas, further consolidating their dominance all over the market.
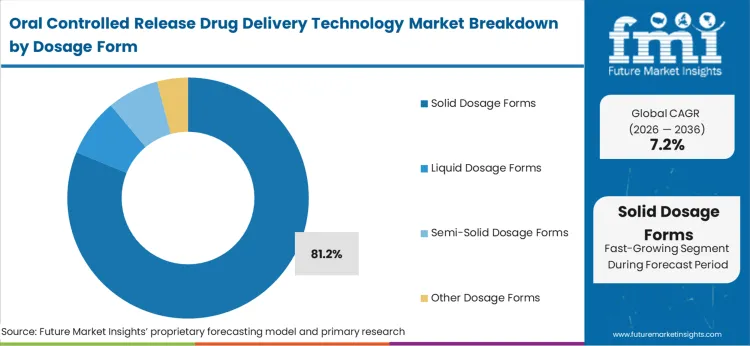
| By Dosage Form | Solid Dosage Forms |
|---|---|
| Value Share (2026) | 81.2% |
Solid dosage forms, such as tablets and capsules, dominate the oral controlled release drug delivery market due to their numerous advantages. Ease of manufacturing, stability, and enhanced patient compliance reduce the number of complications associated with dosage administration. Sustained dosage forms effectively provide a recurrent supply of medicines for an extended period of time and these have a greater place in the management of chronic conditions, lessening side effects of treatment.
Additionally, solid dosage forms are cost-effective in manufacturing and distribution, therefore making them so widely accepted. Ease of swallowing tablets or capsules contributes to increased adherence to medications by patients, helping lessen the burden of complex regimens.
New technology developed for solid dosage forms (for instance, polymeric-based matrices and advanced coating techniques) has also contributed a lot to augmenting their status in controlled release applications. These advantages ensure that solid dosage forms are the favored supply used in the market for oral controlled release.
The market players are using strategies to stay competitive, such as product differentiation through innovative formulations, strategic partnerships with healthcare providers for distribution. Another key strategic focus of these companies is to actively look for strategic partners to bolster their product portfolios and expand their global market presence.
| Metric | Value |
|---|---|
| Quantitative Units | USD 45.13 Billion to USD 90.45 Billion, at a CAGR of 7.20% |
| Market Definition | Oral controlled release drug delivery technology encompasses pharmaceutical formulation platforms (diffusion, matrix, osmotic, and ion-exchange systems) that modulate the rate, duration, and location of drug release from solid, liquid, and semi-solid oral dosage forms to optimize therapeutic efficacy, reduce dosing frequency, and minimize side effects. |
| Drug Release System Segmentation | Diffusion Controlled Release System, Matrix Controlled Release System, Osmotic Controlled Release System, Ion-Exchange Controlled Release System, Other Controlled Release Systems |
| Dosage Form Segmentation | Solid Dosage Forms, Liquid Dosage Forms, Semi-Solid Dosage Forms, Other Dosage Forms |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia, Oceania, Middle East & Africa |
| Countries Covered | India, China, South Korea, Australia and New Zealand, Spain, Germany, France, USA, and 40 plus countries |
| Key Companies Profiled | AstraZeneca Plc., Bristol-Myers Squibb Company, Merck & Co. Inc., Novartis AG, Pfizer Inc., F. Hoffmann-La Roche Ltd., GlaxoSmithKline Plc., Sun Pharmaceuticals, BioNTech, Gilead Sciences |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid bottom-up and top-down modeling validated against primary research inputs and publicly available industry data. |
In terms of drug release system, the industry is divided into- dissolution controlled release system, diffusion controlled release system, osmotically controlled release system, dissolution and diffusion controlled release system, ion exchange resins controlled release system, hydro-dynamically balanced drug delivery system and others.
In terms of dosage form, the industry is segregated into- solid dosage forms and semisolid/liquid/suspensions
Key countries of North America, Latin America, Western Europe, Eastern Europe, South Asia and Pacific, East Asia and Middle East and Africa (MEA) have been covered in the report.
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary research documentation.
How large is the demand for Oral Controlled Release Drug Delivery Technology in the global market in 2026?
Demand for Oral Controlled Release Drug Delivery Technology in the global market is estimated to be valued at USD 45.13 billion in 2026.
What will be the market size of Oral Controlled Release Drug Delivery Technology by 2036?
Market size for Oral Controlled Release Drug Delivery Technology is projected to reach USD 90.45 billion by 2036.
What is the expected demand growth between 2026 and 2036?
Demand is expected to grow at a CAGR of 7.20% between 2026 and 2036.
Which Drug Release System is poised to lead by 2026?
Diffusion Controlled Release System accounts for 25.5% in 2026.
How significant is Solid Dosage Forms in driving adoption?
Solid Dosage Forms represents 81.2% of segment share in 2026.
What country records the fastest growth?
India is projected to grow at a CAGR of 5.50% during 2026 to 2036.
What is the projected growth for China?
China is projected to expand at a CAGR of 5.10% during 2026 to 2036.
What is included in the scope of this report?
Market scope covers diffusion-controlled, matrix-controlled, osmotic-controlled, and ion-exchange-controlled release drug delivery systems in solid, liquid, and semi-solid oral dosage forms across all therapeutic areas.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.