Tibial Neuromodulation Devices Market
Tibial Neuromodulation Devices Market Size and Share Forecast Outlook 2025 to 2035
Historical Data Covered: 2015 to 2023 | Base Year: 2024 | Estimated Year: 2025 | Forecast Period: 2026 to 2035
Tibial Neuromodulation Devices Market Size and Share Forecast Outlook 2025 to 2035
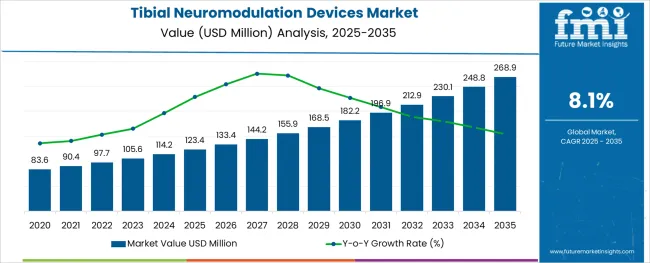
The Tibial Neuromodulation Devices Market is estimated to be valued at USD 123.4 million in 2025 and is projected to reach USD 268.9 million by 2035, registering a compound annual growth rate (CAGR) of 8.1% over the forecast period.
Quick Stats for Tibial Neuromodulation Devices Market
- Industry Value (2025): USD 123.4 million
- Forecast Value (2035): USD 268.9 million
- Forecast CAGR: 8.1%
- Leading Segment: Overactive Bladder (57.5%)
- Key Growth Region: North America, Asia-Pacific, Europe
- Top Key Players: Cogentix Medical, Boston Scientific Corporation, Medtronic plc., Palex Medical SA, Nevro Corporation, Neuronetics, Rainbow Medical Group, NeuroSigma, Synapse Biomedical, Inc.
Rationale for Segmental Growth in the Tibial Neuromodulation Devices Market
Market Overview
The tibial neuromodulation devices market is undergoing strong expansion as demand for non pharmacological treatment of urological disorders continues to rise. Increasing diagnosis of overactive bladder and the shift towards less invasive neuromodulation therapies are central to the market’s current momentum.
Healthcare providers are adopting tibial nerve stimulation due to its favorable safety profile, outpatient applicability, and growing clinical validation in managing chronic bladder dysfunction. The devices are being integrated into outpatient care models and ambulatory surgical workflows, supported by technological improvements that offer portable, patient controlled systems with minimal procedural time.
The growing geriatric population and increasing awareness of neuromodulation alternatives among urologists and patients alike are enhancing treatment uptake. As device miniaturization, wireless connectivity, and personalized stimulation protocols become standard, the market is expected to witness broader penetration across secondary care centers and international markets.
Segmental Analysis
The market is segmented by Application and End User and region. By Application, the market is divided into Overactive Bladder and Faecal Incontinence. In terms of End User, the market is classified into Hospital, Ambulatory Surgical centers, Clinics, Specialty Care Unit, and Others. Regionally, the market is classified into North America, Latin America, Western Europe, Eastern Europe, Balkan & Baltic Countries, Russia & Belarus, Central Asia, East Asia, South Asia & Pacific, and the Middle East & Africa.
Insights into the Overactive Bladder Application Segment
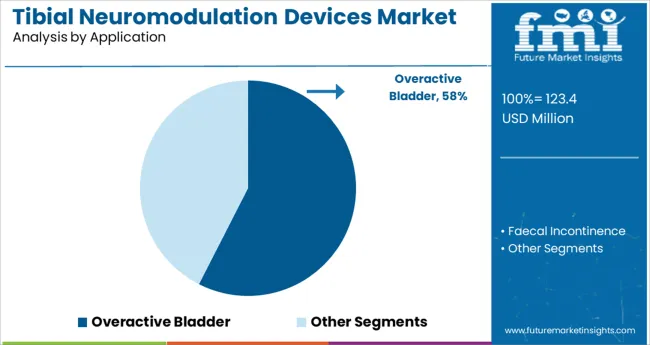
Overactive bladder is projected to account for 57.5% of the total revenue share in 2025, establishing it as the dominant application segment. This leadership is supported by the high prevalence of overactive bladder among aging populations and the limited long term effectiveness of pharmacological treatments.
Tibial neuromodulation has emerged as a preferred therapeutic alternative due to its minimally invasive nature, durable symptom relief, and favorable side effect profile. Clinicians are increasingly recommending the modality in cases where patients experience poor tolerance to antimuscarinics or beta 3 agonists.
Advancements in device usability, such as simplified interfaces and wearable models, have improved adherence and outcomes in overactive bladder treatment pathways. The sustained demand for safe and effective second line therapy is expected to reinforce this segment’s leading share through 2025 and beyond.
Insights into the Hospital End User Segment
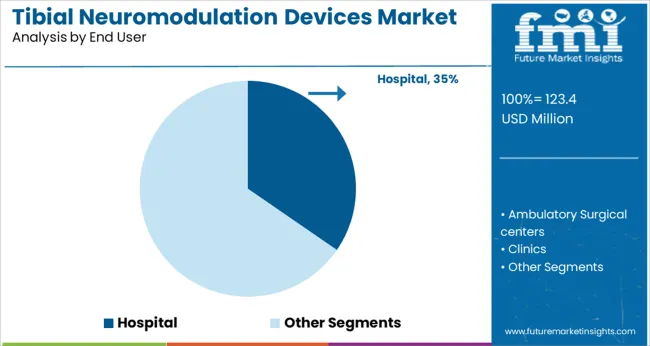
Hospitals are projected to hold 34.6% of the market revenue in 2025, making them the leading end user segment in the tibial neuromodulation devices market. This prominence is attributed to hospitals' capacity to offer advanced urological services, manage complex patient profiles, and provide comprehensive diagnostics prior to initiating neuromodulation therapy.
Multidisciplinary care teams and reimbursement infrastructure within hospitals have enabled greater procedural volumes and streamlined patient onboarding. The availability of trained urologists, access to sterile environments, and integration of supporting technologies such as ultrasound and EMG monitoring have made hospitals the preferred setting for initial device implantation and therapy titration.
Additionally, hospitals play a critical role in clinical research and adoption of next generation devices, further solidifying their contribution to early market leadership and expanded clinical acceptance.
What are the Growth Drivers for the Tibial Neuromodulation Devices Market?
The rising frequency of chronic conditions, combined with the limits of pharmaceutical medications in effectively treating these diseases, has propelled the demand for tibial neuromodulation devices. The functional capabilities of various nerves in the human body, as well as the consequences of electrical stimulation of these nerves for the therapy of various illnesses, have been continuously explored.
As a result, new strategies and gadgets for addressing and treating chronic illnesses have emerged, boosting the tibial neuromodulation devices market growth.
These treatments range from spinal cord stimulation for pain relief to vagus nerve stimulation for epilepsy and other diseases. The tibial neuromodulation devices market is characterized by the introduction of specialized devices and methodologies to treat specific illnesses, as well as ongoing improvements in neurostimulation techniques.
A considerable proportion of patients suffering from chronic diseases such as chronic back pain, depression, epilepsy, and sleep apnea, among others, are resistant to treatment and drugs. Resistance is seen in the treatment's slow progression and delayed or limited consequences. As a result, healthcare professionals are moving their focus to neurostimulation approaches for chronic disease treatment, which is driving the sales of neuromodulation devices during the forecast period.
According to the Centers for Disease Control and Prevention (CDC), an estimated 50 million people in the United States suffer from chronic pain each year, with an additional 19 million suffering from severe chronic pain. Epilepsy, treatment-resistant depression, and other chronic illnesses impose a significant financial burden on countries. As a result, the tibial neuromodulation devices market share is anticipated to rise significantly during the forecast period.
What are the Driving Challenges in the Tibial Neuromodulation Devices Market?
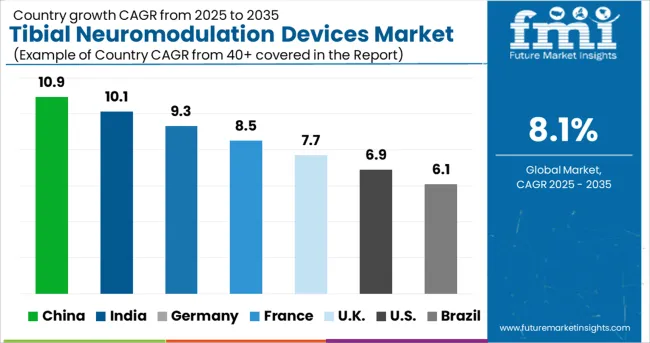
In growing countries such as China, Brazil, and India, the prevalence of neurological diseases such as epilepsy, depression, and others is increasing at a rapid pace. Despite a higher frequency, lower diagnostic rates in developing nations limit the number of individuals who require treatment. As a result of this, the sales of tibial neuromodulation devices are anticipated to decline over the forecast period.
Lower diagnosis rates in these nations are caused by a variety of causes, including a general lack of awareness and limited access to diagnostic facilities due to inadequate healthcare infrastructure. This is exacerbated by the fact that a percentage of the diagnosed population does not receive treatment for a variety of reasons. These are some of the primary factors restricting the number of patients receiving treatment, hence limiting the tibial neuromodulation devices market growth.
Regional Analysis
How is the Performance of the North America Tibial Neuromodulation Devices Market?
North America is projected to hold 34.1% of the tibial neuromodulation devices market share during the forecast period. The primary reasons for the high demand for tibial neuromodulation devices in the region include higher rates of diagnosis and treatment among the patient population, as well as increased adoption of neuromodulation devices for the treatment of chronic illnesses. This is exacerbated by the larger prevalence of chronic illnesses in the United States, as well as suitable reimbursement regulations for these devices in the country.
What is the Growth Outlook for the Europe Tibial Neuromodulation Devices Market?
Europe is projected to dominate the tibial neuromodulation devices market with a share of 40.1% during the forecast period. Major factors propelling the tibial neuromodulation devices market share in Europe include a well-developed healthcare infrastructure, combined with an increase in non-invasive device approvals in the region. As a result of these factors, the sales of tibial neuromodulation devices are projected to grow rapidly from 2025 to 2035.
From 2025 to 2035, new entrants and regional players are projected to get their CE mark approvals for their non-invasive devices which is further expected to boost the tibial neuromodulation devices market growth in Europe.
Start-up Ecosystem
How are New Entrants Revolutionizing the Tibial Neuromodulation Devices Market?
The start-up ecosystem in the tibial neuromodulation devices market is moderate with a few startups conducting clinical trials to determine the therapeutic efficacy of their products. Deep Brain Innovations LLC, Scion NeuroStim, and ALEVA NEUROTHERAPEUTICS SA are some of the startups in the tibial neuromodulation devices market.
Competitive Landscape
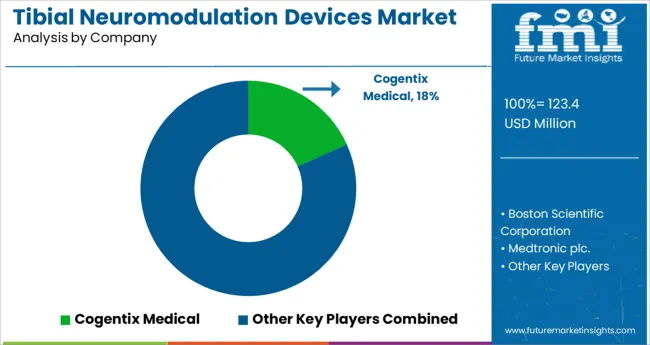
Who are some of the Key Market Players in the Tibial Neuromodulation Devices Market?
The neuromodulation devices market is consolidated, with the top four firms accounting for more than 90% of the market in 2025. Medtronic's extensive portfolio, along with strong brand awareness, widespread regional presence, and a solid distribution network, is responsible for propelling the business to market leadership in 2025.
The key market players profiled in the report include Cogentix Medical, Palex Medical SA, and Medtronic plc. Rainbow Medical Group. The key players in this market adopted product launch, partnership, and expansion as their key strategies to meet the changing consumer demands.
Recent Developments in the Tibial Neuromodulation Devices Market:
- ElectroCore Inc., a commercial-stage medical device startup, acquired US FDA approval for their gammaCore non-invasive vagus nerve stimulation (nVNS) device in November 2020. In the United States, the device was approved as an additional therapy for the treatment of cluster headaches.
- SenTiva, a vagus nerve stimulation device from LivaNova PLC, was approved by the US Food and Drug Administration in October 2020 for the treatment of epilepsy. The gadget is approved for drug-resistant epilepsy patients over the age of four.
Report Scope
| Report Attribute | Details |
|---|---|
| Growth Rate | CAGR of 8.1% from 2025 to 2035 |
| Base Year for Estimation | 2025 |
| Historical Data | 2020 to 2024 |
| Forecast Period | 2025 to 2035 |
| Quantitative Units | Revenue in million and CAGR from 2025 to 2035 |
| Report Coverage | Revenue Forecast, Volume Forecast, Company Ranking, Competitive Landscape, Growth Factors, Trends, and Pricing Analysis |
| Segments Covered | Application, End-User, Region |
| Regions Covered | North America; Latin America; Europe; East Asia; South Asia; Oceania; Middle East and Africa |
| Key Countries Profiled | United States of America; Canada; Brazil; Argentina; Germany; United Kingdom; France; Spain; Italy; Nordics; BENELUX; Australia & New Zealand; China; India; ASIAN; GCC Countries; South Africa |
| Key Companies Profiled | Cogentix Medical, Palex Medical SA, Medtronic plc. Rainbow Medical Group. |
| Customization | Available Upon Request |
Key Market Segments in the Tibial Neuromodulation Devices Market
By Application:
- Faecal Incontinence
- Overactive Bladder
By End User:
- Hospital
- Ambulatory Surgical centers
- Clinics
- Specialty Care Unit
- Others
By Region:
- North America
- Latin America
- Asia Pacific
- Middle East and Africa (MEA)
- Europe
Frequently Asked Questions
How big is the tibial neuromodulation devices market in 2025?
The global tibial neuromodulation devices market is estimated to be valued at USD 123.4 million in 2025.
What will be the market size for the tibial neuromodulation devices market in 2035?
It is projected to reach USD 268.9 million by 2035.
How much will be the CAGR of the tibial neuromodulation devices market between 2025 and 2035?
The market is expected to grow at a 8.1% CAGR between 2025 and 2035.
What are the key product types in the tibial neuromodulation devices market?
The key product types are overactive bladder and faecal incontinence.
Which hospital segment is expected to dominate the tibial neuromodulation devices market in 2025?
hospital segment is expected to dominate with a 34.6% industry share in 2025.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand-side Trends
- Supply-side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage/Taxonomy
- Market Definition/Scope/Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2020-2024 and Forecast, 2025-2035
- Historical Market Size Value (USD Million) Analysis, 2020-2024
- Current and Future Market Size Value (USD Million) Projections, 2025-2035
- Y-o-Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2020-2024 and Forecast 2025-2035
- Global Market Analysis 2020-2024 and Forecast 2025-2035, By Application
- Introduction/Key Findings
- Historical Market Size Value (USD Million) Analysis By Application , 2020-2024
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application , 2025-2035
- Overactive Bladder
- Faecal Incontinence
- Y-o-Y Growth Trend Analysis By Application , 2020-2024
- Absolute $ Opportunity Analysis By Application , 2025-2035
- Global Market Analysis 2020-2024 and Forecast 2025-2035, By End User
- Introduction/Key Findings
- Historical Market Size Value (USD Million) Analysis By End User, 2020-2024
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User, 2025-2035
- Hospital
- Ambulatory Surgical centers
- Clinics
- Specialty Care Value (USD Million)
- Others
- Y-o-Y Growth Trend Analysis By End User, 2020-2024
- Absolute $ Opportunity Analysis By End User, 2025-2035
- Global Market Analysis 2020-2024 and Forecast 2025-2035, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2020-2024
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2025-2035
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2020-2024 and Forecast 2025-2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020-2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025-2035
- By Country
- USA
- Canada
- Mexico
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Application
- By End User
- Key Takeaways
- Latin America Market Analysis 2020-2024 and Forecast 2025-2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020-2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025-2035
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Application
- By End User
- Key Takeaways
- Western Europe Market Analysis 2020-2024 and Forecast 2025-2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020-2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025-2035
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Application
- By End User
- Key Takeaways
- Eastern Europe Market Analysis 2020-2024 and Forecast 2025-2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020-2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025-2035
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Application
- By End User
- Key Takeaways
- East Asia Market Analysis 2020-2024 and Forecast 2025-2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020-2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025-2035
- By Country
- China
- Japan
- South Korea
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Application
- By End User
- Key Takeaways
- South Asia and Pacific Market Analysis 2020-2024 and Forecast 2025-2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020-2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025-2035
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Application
- By End User
- Key Takeaways
- Middle East & Africa Market Analysis 2020-2024 and Forecast 2025-2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020-2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025-2035
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Application
- By End User
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2024
- By Application
- By End User
- Canada
- Pricing Analysis
- Market Share Analysis, 2024
- By Application
- By End User
- Mexico
- Pricing Analysis
- Market Share Analysis, 2024
- By Application
- By End User
- Brazil
- Pricing Analysis
- Market Share Analysis, 2024
- By Application
- By End User
- Chile
- Pricing Analysis
- Market Share Analysis, 2024
- By Application
- By End User
- Germany
- Pricing Analysis
- Market Share Analysis, 2024
- By Application
- By End User
- UK
- Pricing Analysis
- Market Share Analysis, 2024
- By Application
- By End User
- Italy
- Pricing Analysis
- Market Share Analysis, 2024
- By Application
- By End User
- Spain
- Pricing Analysis
- Market Share Analysis, 2024
- By Application
- By End User
- France
- Pricing Analysis
- Market Share Analysis, 2024
- By Application
- By End User
- India
- Pricing Analysis
- Market Share Analysis, 2024
- By Application
- By End User
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2024
- By Application
- By End User
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2024
- By Application
- By End User
- China
- Pricing Analysis
- Market Share Analysis, 2024
- By Application
- By End User
- Japan
- Pricing Analysis
- Market Share Analysis, 2024
- By Application
- By End User
- South Korea
- Pricing Analysis
- Market Share Analysis, 2024
- By Application
- By End User
- Russia
- Pricing Analysis
- Market Share Analysis, 2024
- By Application
- By End User
- Poland
- Pricing Analysis
- Market Share Analysis, 2024
- By Application
- By End User
- Hungary
- Pricing Analysis
- Market Share Analysis, 2024
- By Application
- By End User
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2024
- By Application
- By End User
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2024
- By Application
- By End User
- South Africa
- Pricing Analysis
- Market Share Analysis, 2024
- By Application
- By End User
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Application
- By End User
- Competition Analysis
- Competition Deep Dive
- Cogentix Medical
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Boston Scientific Corporation
- Medtronic plc.
- Palex Medical SA
- Nevro Corporation
- Neuronetics
- Rainbow Medical Group
- NeuroSigma
- Synapse Biomedical, Inc.
- Cogentix Medical
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2020-2035
- Table 2: North America Market Value (USD Million) Forecast by Country, 2020-2035
- Table 3: Latin America Market Value (USD Million) Forecast by Country, 2020-2035
- Table 4: Western Europe Market Value (USD Million) Forecast by Country, 2020-2035
- Table 5: Eastern Europe Market Value (USD Million) Forecast by Country, 2020-2035
- Table 6: East Asia Market Value (USD Million) Forecast by Country, 2020-2035
- Table 7: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2020-2035
- Table 8: Middle East & Africa Market Value (USD Million) Forecast by Country, 2020-2035
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2020-2035
- Figure 3: Global Market Value (USD Million) Share and BPS Analysis by Region, 2025 and 2035
- Figure 4: Global Market Y-o-Y Growth Comparison by Region, 2025-2035
- Figure 5: Global Market Attractiveness Analysis by Region
- Figure 6: North America Market Incremental $ Opportunity, 2025-2035
- Figure 7: Latin America Market Incremental $ Opportunity, 2025-2035
- Figure 8: Western Europe Market Incremental $ Opportunity, 2025-2035
- Figure 9: Eastern Europe Market Incremental $ Opportunity, 2025-2035
- Figure 10: East Asia Market Incremental $ Opportunity, 2025-2035
- Figure 11: South Asia and Pacific Market Incremental $ Opportunity, 2025-2035
- Figure 12: Middle East & Africa Market Incremental $ Opportunity, 2025-2035
- Figure 13: North America Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 14: Latin America Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 15: Western Europe Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 16: Eastern Europe Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 17: East Asia Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 18: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 19: Middle East & Africa Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 20: Global Market - Tier Structure Analysis
- Figure 21: Global Market - Company Share Analysis

