Smart Implants Market
The smart implants market is segmented by Implant type (Cardiovascular implants, Orthopedic implants, Neurostimulation implants, Others), Surgery (Open surgery, Minimally invasive surgery), End use (Hospitals, Cardiac care centers, Ambulatory surgical centers, Other end use) and Region. Forecast for 2026 to 2036.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Smart Implants Market forecast and outlook By FMI
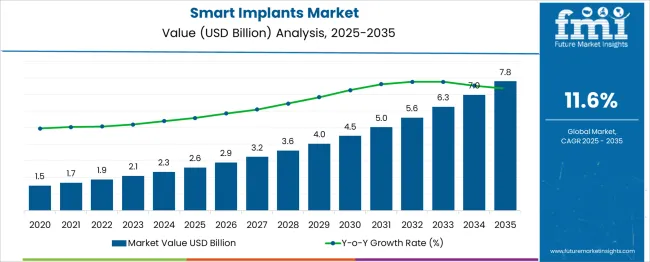
In 2025, the smart implants market was valued at USD 2.6 billion. Based on Future Market Insights’ analysis, demand for smart implants is estimated to grow to USD 2.9 billion in 2026 and USD 8.7 billion by 2036. FMI projects a CAGR of 11.6% during the forecast period.
Absolute dollar growth of USD 5.8 billion over the decade signals a scale-up that depends on clinical workflow readiness and payer acceptance rather than unit volume alone. Growth is held back by regulatory and compliance load (clinical evidence, cybersecurity documentation, post-market surveillance), yet sustained by the economic case for avoiding avoidable admissions through earlier intervention and remote follow-up.
Summary of Smart Implants Market
- Market definition
- Smart implants combine implanted therapy with sensing and connectivity so clinicians can monitor status and intervene earlier across cardiac, neurostimulation, and orthopedic pathways.
- Demand drivers
- Hospitals adopt implant-linked monitoring to reduce avoidable admissions and support earlier intervention in chronic disease pathways.
- Payers expand structured reimbursement for telemonitoring and digital medical devices, improving the business case for connected implants.
- OEM roadmaps push longer battery life, smaller form factors, and algorithm-enabled risk alerts, increasing clinical acceptability.
- Key segments analyzed
- Implant type: Cardiovascular implants lead with 75.4% share in 2025 due to the installed base of CIEDs and routine remote follow-up pathways.
- Surgery: Open surgery leads with 63.5% share in 2025 because complex implant placement still needs direct visualisation and anchoring.
- End use: Hospitals lead with 53.2% share in 2025 because they manage implantation, post-op follow-up, and device clinics.
- Analyst opinion at FMI
- Sabyasachi Ghosh, Principal Analyst at Future Market Insights, opines, "Buyers should treat smart implants as a service-enabled clinical pathway purchase, where reimbursement, cybersecurity readiness, and remote monitoring capacity determine ROI more than device list price."
- Strategic implications
- OEMs that package device, software documentation, and clinic workflow tools shorten adoption cycles in hospital systems.
- Providers that invest in device clinic staffing and triage protocols capture the admission-avoidance value that supports payer discussions.
- Distributors and service partners can win share by supporting claims, coding, and post-market reporting alongside device supply.
- Methodology
- Country forecasts incorporate reimbursement rules, HTA recommendations, and regulator guidance updates that affect adoption speed.
- Company evidence is limited to first-party disclosures such as press releases, investor updates, and regulator notices.
- Segment shares are stress-tested against clinical setting mix and procedure pathway constraints.
| Metric | Value |
|---|---|
| Estimated value in (2026E) | USD 2.9 billion |
| Forecast value in (2036F) | USD 8.7 billion |
| CAGR (2026 to 2036) | 11.6% |
As Geoff Martha, Chairman and Chief Executive Officer of Medtronic, noted regarding product-cycle momentum and AI rollout, "We’re beginning new product cycles in some of MedTech’s most attractive markets, which is further enhanced as we apply AI across our portfolio." [1]
China (15.7% CAGR) and India (14.5% CAGR) lead due to capacity build-out in specialty care and digital health infrastructure. Germany (13.3%) and France (12.2%) grow on reimbursed telemonitoring pathways and hospital digitisation budgets. The UK (11.0%) expands through NICE-backed algorithm-based remote monitoring in cardiac implant populations. The United States (9.9%) remains a large revenue pool driven by replacement cycles and premium device mix, with growth moderated by coverage rules and hospital capital prioritisation.
Smart Implants Market definition
Smart implants are implantable medical devices that combine a therapeutic function (such as pacing, stimulation, or structural support) with embedded sensing and connectivity that can record patient data and share it with clinicians. They are used most often in cardiovascular care (for rhythm and heart failure monitoring), neurostimulation (for chronic pain or epilepsy management), and orthopedics (for recovery tracking after joint replacement). The market measures device revenues and associated platform-enabled implant systems used in hospitals and specialist centers.
Market inclusions
This report covers global and regional market sizing for 2026 to 2036, with 2025 as the base year. It provides segment forecasts by implant type, surgery approach, and end use, along with country-level growth rates. The scope includes demand mapping tied to reimbursement pathways, remote monitoring workflow adoption, and regulatory factors that influence hospital purchasing and clinician uptake.
Market exclusions
The scope excludes non-connected conventional implants that do not generate or transmit patient data. It also excludes external wearables and stand-alone remote monitoring services when not linked to implantable devices. Consumer health apps, generic telemedicine platforms, and non-medical IoT sensors are omitted unless they are part of regulated implant-linked monitoring pathways.
Smart Implants Market Research Methodology
- Primary research: Interviews were conducted with implant OEM product leads, hospital procurement teams, electrophysiology and orthopedic clinicians, and remote monitoring program managers.
- Desk research: Public sources included regulator guidance documents, payer and health-technology assessment publications, and company investor/press disclosures.
- Market-sizing and forecasting: A hybrid model was used, combining bottom-up adoption by care pathway and end-use setting with top-down checks against device franchise revenues and procedure volume trends.
- Data validation and update cycle: Estimates were triangulated across primary inputs, public policy documents, and manufacturer disclosures, then refreshed with new approvals, reimbursement updates, and hospital program rollouts.
Segmental analysis
Smart Implant Market Analysis by Implant Type
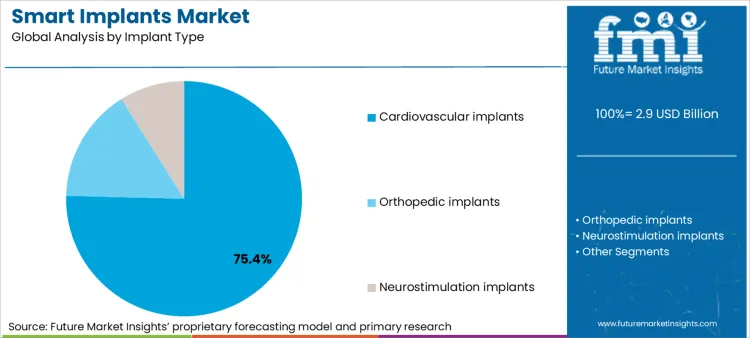
Based on FMI’s smart implants market report, consumption of cardiovascular implants is estimated to hold 75.4% share in 2025. This dominance comes from the installed base of cardiac implantable electronic devices and established follow-up pathways where remote alerts can reduce avoidable admissions and support therapy optimisation.
- Policy pull-through: NICE guidance reviewed algorithm-based remote monitoring for CIED populations, backing structured use of tools linked to implantable devices in heart failure management pathways. [5]
- OEM portfolio expansion: Abbott’s Assert family of insertable cardiac monitors targets long-duration monitoring and workflow efficiency, reinforcing category depth in cardiac monitoring. [6]
- Clinical workflow scaling: Boston Scientific highlighted NICE recommendation for HeartLogic, framing algorithm-enabled monitoring as a pathway that health systems can standardise. [4]
Smart Implants Market analysis by Surgery
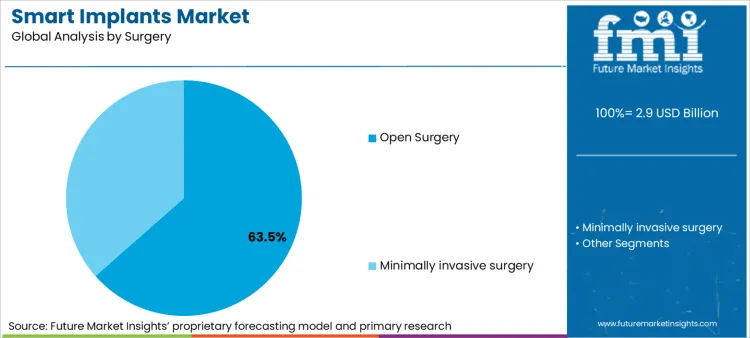
Based on FMI’s smart implants market report, consumption via open surgery is estimated to hold 63.5% share in 2025. Complex implant placement still depends on direct access for anchoring, lead management, and intraoperative testing, especially for cardiac and neurostimulation systems where placement accuracy affects outcomes.
- Hospital operating model: Open implant procedures remain anchored in tertiary centers because they combine implantation, imaging, and post-op monitoring in one controlled pathway. [5]
- Robotics adjacency: Zimmer Biomet’s 2024 distribution agreement around a handheld robotic system for knee arthroplasty shows how surgical platform investments can raise procedure throughput and standardisation, supporting implant utilisation. [8]
- Post-op tracking pull: Sensor-enabled orthopedic implants, such as Zimmer Biomet’s Persona IQ concept, illustrate how implant data collection is being built into recovery pathways, reinforcing surgical adoption. [7]
Smart Implants Market analysis by End use
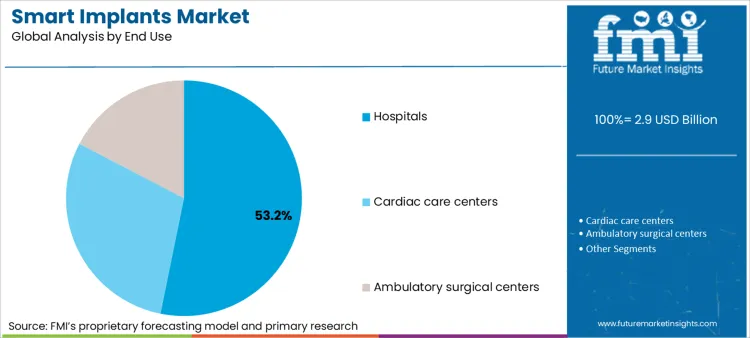
Based on FMI’s smart implants market report, consumption in hospitals is estimated to hold 53.2% share in 2025. Hospitals concentrate implantation volume, device clinics, and complication management, so they capture the clinical and operational value of connected follow-up.
- HTA and pathway adoption: NICE guidance creates a framework for hospitals to justify investment in remote monitoring workflows tied to implanted devices. [5]
- Regulatory proof points: Medtronic’s disclosures around approvals and submissions across cardiovascular and neuromodulation portfolios show how hospital adoption tracks regulator-cleared product cycles. [1]
- Data and claims readiness: USA policy frameworks for physician fee schedule and remote monitoring codes shape how hospitals and clinicians structure billing and service delivery. [13]
Smart Implants Drivers, restraints, opportunities
Future Market Insights analysis links market scale to chronic disease management pathways where clinicians need continuous visibility between visits. Cardiac rhythm management and heart failure monitoring have built the strongest operating model because implant clinics already run scheduled interrogations and can shift part of that workload into remote algorithms under formal guidance. NICE’s assessment of algorithm-based remote monitoring in CIED populations illustrates how public evaluators weigh admission reduction and clinical workload in adoption decisions. [5]
The main restraint is the compliance stack: clinical evidence, cybersecurity documentation, data governance, and post-market surveillance add cost and time before broad rollouts. FDA’s cybersecurity guidance for medical devices raises expectations for design controls, labeling, and premarket documentation for devices with cybersecurity risk, which affects connected implants and their companion ecosystems. [2]
- Reimbursement formalisation: France’s LATM pathway and similar schemes make telemonitoring a billable activity, improving the economics of implant-linked monitoring programs. [9]
- Algorithm-based triage: NICE guidance that covers HeartLogic, TriageHF and other algorithms supports structured adoption in health systems that need standardised pathways. [5]
- Safety-performance compliance: Brazil’s RDC 848/2024 on essential safety and performance requirements increases documentation expectations, pushing suppliers toward stronger regulatory files and post-market processes. [12]
Regional analysis
The market is analysed across North America, Europe, East Asia, South Asia & Pacific, and Latin America, with country-level pathways reflecting reimbursement, regulator requirements, and hospital workflow maturity. The full report provides market attractiveness analysis by segment and region.
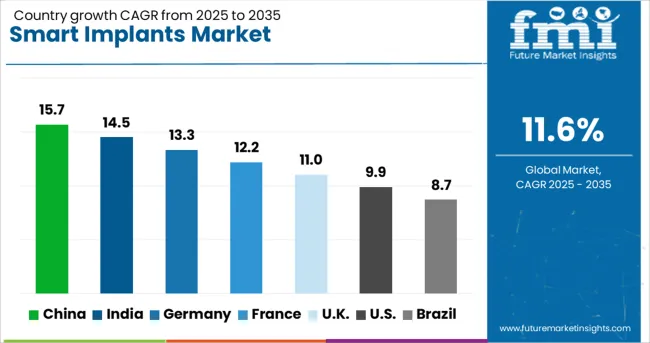
| Country | CAGR (2026-2036) |
|---|---|
| China | 15.7% |
| India | 14.5% |
| Germany | 13.3% |
| France | 12.2% |
| United Kingdom | 11.0% |
| United States | 9.9% |
| Brazil | 8.7% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
North America market analysis
North America is the reimbursement-and-compliance benchmark where coding, coverage rules, and cybersecurity expectations shape supplier behaviour. Medtronic and Abbott anchor large installed bases in cardiac rhythm management and monitoring, while hospital systems influence purchasing through standardised device formularies and clinic capacity planning. [2]
- United States: Demand for smart implants in the United States is projected to rise at 9.9% CAGR through 2036. Adoption is strongly shaped by documentation and billing rules for implant-linked remote monitoring. In the Medicare policy stack, remote monitoring and payment policies under the Physician Fee Schedule are published through Federal Register rulemaking, which providers use to plan service delivery and staffing for monitoring programs. [13] A second adoption lever is cybersecurity compliance, since FDA guidance describes cybersecurity design and documentation expectations for devices with cybersecurity risk, which directly affects connected implants and their software environments. [2] On the supplier side, cardiac device makers publish reimbursement and coding guides that reference CMS rule sets used by providers to align claims and device clinic workflows for implantable cardiovascular monitoring. [14]
FMI’s analysis of smart implants market in North America consists of country-wise assessment that includes the United States. Readers can find device adoption constraints, coding exposure, and how compliance expectations change procurement timelines.
Europe market analysis
Europe is a health-technology assessment-led market where pathway endorsements and reimbursement structures determine scale-up speed. Boston Scientific, Medtronic, and Biotronik compete through algorithm performance, clinic workflow impact, and evidence packages aligned to evaluator requirements. [5]
- Germany: Demand for smart implants in Germany is projected to rise at 13.3% CAGR through 2036. Cardiac telemonitoring is structurally supported because statutory insurance reimbursement for heart failure remote patient monitoring has been in place, enabling providers to build telemedical center models that connect patient follow-up to clinician triage capacity. [10] In parallel, published clinical reviews in 2024 describe Germany’s integration of telemedicine into standard heart failure care, which supports scaling protocols and staffing models inside cardiology practices and hospital networks. [11] This environment favours implant systems that reduce alert fatigue, integrate into clinic workflows, and support evidence-based escalation pathways.
- United Kingdom: Demand for smart implants in the UK is projected to rise at 11.0% CAGR through 2036. NICE issued guidance on algorithm-based remote monitoring for people with cardiac implantable electronic devices, covering solutions such as HeartLogic and TriageHF, giving hospitals an evaluator-backed framework for adoption decisions. [5] This matters because device clinics must manage workload, triage thresholds, and escalation protocols as remote alerts increase. Public-facing reporting on the NICE recommendation highlighted that these tools monitor readings from cardiac implantable devices so clinicians can detect deterioration earlier and intervene before admission is needed, aligning with NHS operational targets. [3]
- France: Demand for smart implants in France is projected to rise at 12.2% CAGR through 2036. The French system is moving toward ordinary-law reimbursement for remote monitoring via the List of Medical Telemonitoring Activities (LATM), creating a more predictable path for providers and digital medical device operators involved in telemonitoring services. [9] This improves commercial viability for implant-linked monitoring where providers need a stable payment model to fund staffing and infrastructure. France’s approach creates demand for suppliers that can support both sides of the pathway: clinical evidence for evaluators and practical deployment support for hospitals and telemonitoring operators.
FMI’s analysis of smart implants market in Europe consists of country-wise assessment that includes Germany, the United Kingdom, and France. Readers can find reimbursement mechanics, evaluator decision drivers, and how clinic workload constraints affect vendor selection.
East Asia market analysis
East Asia is the hospital-capacity build-out region where high procedure volumes and domestic manufacturing ecosystems can accelerate adoption once regulatory pathways and procurement cycles align. Multinationals compete with regional suppliers through evidence packages, pricing, and platform integration.
- China: Demand for smart implants in China is projected to rise at 15.7% CAGR through 2036. A major driver is the regulator’s expanding experience with software-driven and AI-enabled medical devices, which supports broader comfort with connected device ecosystems. A 2024 comparative regulatory review documented the count of AI medical devices approved in China (as of mid-2023) using governmental database searches, illustrating the scale and speed of software-enabled medical device review activity. [15] In parallel, hospital investment and specialty care expansion in large urban systems increase the installed base for implant follow-up programs where remote monitoring can reduce outpatient load and support earlier intervention.
FMI’s analysis of smart implants market in East Asia consists of country-wise assessment that includes China. Readers can find regulatory context for software-enabled devices, adoption constraints inside hospital systems, and supplier go-to-market strategies.
South Asia & Pacific market analysis
South Asia & Pacific growth is driven by specialty care expansion and digitisation of health records, which lowers friction for follow-up workflows and remote care models when implant programs scale. Hospital groups and cardiac centers are the main adoption engines.
- India: Demand for smart implants in India is projected to rise at 14.5% CAGR through 2036. Adoption is supported by the build-out of national digital health rails that can make patient identification and record linkage easier across providers, which matters for chronic disease follow-up after implantation. A 2024 peer-reviewed analysis of the Ayushman Bharat Digital Mission describes its role in building a national digital health ecosystem and integrating health data records, supporting continuity of care where implant patients often move between providers. [16] The Ministry of Health and Family Welfare also sets out national telemedicine and digital health initiatives that frame how public systems approach remote services and citizen-centric access. [17]
FMI’s analysis of smart implants market in South Asia & Pacific consists of country-wise assessment that includes India. Readers can find how digital health infrastructure affects care continuity, follow-up capacity, and adoption pacing.
Latin America market analysis
Latin America is a regulatory documentation-and-access market where safety-performance requirements and tendering practices shape procurement. Hospitals and specialist centers drive adoption, with variation by payer coverage and public system capacity.
- Brazil: Demand for smart implants in Brazil is projected to rise at 8.7% CAGR through 2036. Regulatory requirements and procurement readiness shape adoption more than consumer demand signals. Brazil’s RDC No. 848 (March 2024) sets essential safety and performance principles that must be considered in device design and manufacturing, raising compliance expectations across medical devices and influencing supplier documentation and readiness for high-risk implant categories. [12] For connected implant ecosystems, this intersects with Brazil’s growing attention to software-regulated medical products, since ANVISA has a defined framework for software as a medical device, which sets expectations for how digital components are classified and authorised. [18]
FMI’s analysis of smart implants market in Latin America consists of country-wise assessment that includes Brazil. Readers can find how regulatory requirements and procurement models affect adoption pacing and supplier strategy.
Competitive aligners for market players
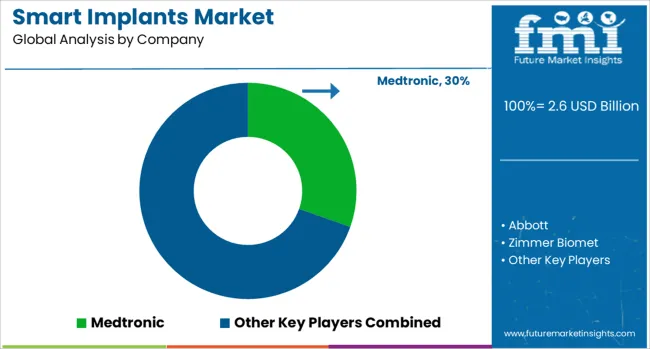
Competition is moderately concentrated in cardiovascular implants and more fragmented in orthopedic and neurostimulation segments due to differing clinical pathways and incumbent positions. Large OEMs with deep cardiac franchises benefit from installed base economics: device clinics, programmer ecosystems, and long-standing clinician relationships keep switching costs high. In neurostimulation and orthopedics, the field is more open to challengers when they can prove measurable improvements in recovery tracking, therapy optimisation, or reduced follow-up burden.
Structural advantage is strongest where companies control both implant hardware and the monitoring or algorithm layer. Payers and hospitals increasingly assess "total pathway cost," including clinic time, alert handling, and readmission rates. That pushes suppliers to bundle monitoring software, onboarding, clinician dashboards, and training support into the commercial offer, since hospitals struggle to staff remote monitoring without predictable workload tools.
Buyer behaviour is shifting toward standardisation and vendor rationalisation. Large hospital groups reduce vendor count to improve training consistency and supply reliability, then negotiate multi-year pricing with service add-ons. In cardiac monitoring, buyers also assess reimbursement exposure and coding support, since coverage rules and billing requirements affect program sustainability. Suppliers that provide coding guidance, integration support, and evidence packs are more likely to hold formulary placement.
Recent Developments
- May 2024: Medtronic reported multiple portfolio approvals and submissions across cardiovascular and neuromodulation areas, pointing to active product-cycle refresh that supports hospital adoption planning. [1]
- June 2024: Zimmer Biomet announced a distribution agreement with THINK Surgical for a handheld robotic system for total knee arthroplasty, supporting throughput and standardisation in orthopedic procedures linked to implant volumes. [8]
- October 2024: NICE reviewed and maintained guidance on algorithm-based remote monitoring for people with cardiac implantable electronic devices, supporting structured adoption of implant-linked monitoring tools. [5]
Key players in Smart implants Market
- Medtronic plc
- Abbott Laboratories
- Zimmer Biomet Holdings, Inc.
- BIOTRONIK SE & Co. KG
- Boston Scientific Corporation
- DirectSync Surgical
- Intelligent Implants
- NeuroPace, Inc.
Scope of the Report
| Items | Value |
|---|---|
| Quantitative units | USD 2.9 billion (2026) to USD 8.7 billion (2036), at a CAGR of 11.6% |
| Market definition | Smart implants are implantable medical devices with sensing and connectivity that transmit patient data to support monitoring, early intervention, and therapy optimisation across cardiac, neurostimulation, and orthopedic pathways. |
| Implant type segmentation | Cardiovascular implants; Orthopedic implants; Neurostimulation implants; Others |
| Surgery segmentation | Open surgery; Minimally invasive surgery |
| End use segmentation | Hospitals; Cardiac care centers; Ambulatory surgical centers; Other end use |
| Regions covered | North America; Europe; East Asia; South Asia & Pacific; Latin America; Middle East & Africa |
| Countries covered | United States; United Kingdom; Germany; China; India |
| Industry players | Medtronic plc; Abbott Laboratories; Zimmer Biomet Holdings, Inc.; BIOTRONIK SE & Co. KG; Boston Scientific Corporation; NeuroPace, Inc. |
| Forecast period | 2026 to 2036 |
| Market evolution coverage | 2020 to 2024 historical trend review; 2025 base year; 2026 to 2036 forecast |
| Additional attributes | Revenue by implant type, surgery, and end use; country growth rates; reimbursement and HTA pathway tracking; cybersecurity and regulatory documentation impacts; hospital workflow adoption patterns |
Smart implants Market analysis by segments
By Implant type:
- Cardiovascular implants
- Orthopedic implants
- Neurostimulation implants
- Others
By Surgery:
- Open surgery
- Minimally invasive surgery
By End use:
- Hospitals
- Cardiac care centers
- Ambulatory surgical centers
- Other end use
By Region:
- North America
- Europe
- East Asia
- South Asia & Pacific
- Latin America
- Middle East & Africa
Bibliography
- [1] Medtronic. (2024, May). Medtronic reports full year and fourth quarter fiscal 2024 financial results; announces dividend increase.
- [2] USA Food and Drug Administration. (2023, September). Cybersecurity in medical devices: Quality management system considerations and content of premarket submissions (Web page updated February 2026).
- [3] Digital Health. (2024, October). NICE approves remote monitoring tools for heart failure patients.
- [4] Boston Scientific. (2024, October). NICE recommends Boston Scientific HeartLogic heart failure diagnostic.
- [5] National Institute for Health and Care Excellence. (2024, October). Heart failure algorithms for remote monitoring in people with cardiac implantable electronic devices (HTG730).
- [6] Abbott. (2026, February). About Assert-IQ insertable cardiac monitor (ICM) (Accessed February 2026).
- [7] Zimmer Biomet. (2026, February). Persona IQ: The smart knee implant (Accessed February 2026).
- [8] Zimmer Biomet. (2024, June). Zimmer Biomet enters distribution agreement with THINK Surgical.
- [9] G_NIUS (French eHealth). (2026, February). Remote monitoring reimbursement (LATM) (Accessed February 2026).
- [10] Koehler, F., et al. (2022). Telemonitoring of heart failure patients is reimbursed in Germany: What’s next? European Heart Journal - Digital Health.
- [11] Spethmann, S., et al. (2024). Telemonitoring for chronic heart failure: Narrative review. Journal of Medical Internet Research, 26, e63391.
- [12] Agência Nacional de Vigilância Sanitária. (2024, March). Resolution RDC No. 848, of March 6, 2024.
- [13] Federal Register. (2023, November). Medicare and Medicaid programs; CY 2024 payment policies under the Physician Fee Schedule and other changes.
- [14] Abbott. (2026, February). National Medicare reimbursement guide: Implantable cardiovascular physiologic monitoring - Remote (Accessed February 2026).
- [15] Liu, Y., et al. (2024). Regulatory responses and approval status of artificial intelligence-enabled medical devices. npj Digital Medicine, 7, Article 1254.
- [16] Mishra, USA, et al. (2024). The Ayushman Bharat Digital Mission of India. Journal of Global Health Reports.
- [17] Ministry of Health and Family Welfare, Government of India. (2025, July). e-Health / Telemedicine (Updated July 2025).
- [18] Agência Nacional de Vigilância Sanitária. (2024). RDC No. 657 of 24 March 2022 (Software as a Medical Device - SaMD), English version (2024 file posting).
This bibliography is provided for reader reference and is not exhaustive. The full report contains the complete reference list and detailed citations.
This report addresses
- Market sizing for 2026 and 2036 with CAGR projection for 2026 to 2036
- Segment forecasts by implant type, surgery approach, and end use setting
- Country growth mapping across major adoption markets and procedure pathways
- Reimbursement and HTA pathway tracking that affects hospital adoption speed
- Regulatory and cybersecurity documentation factors shaping connected implant rollouts
- Competitive assessment of OEM positioning, installed base advantages, and bundling strategy
- Deployment constraints analysis including device clinic workload, alert handling, and integration needs
- Deliverables in analyst report format with segment taxonomy, tables, and evidence-based bibliography
Frequently Asked Questions
How large is the smart implants market in 2025?
The global smart implants market was valued at USD 2.6 billion in 2025.
What is the projected size of the smart implants market in 2026?
The market is estimated to reach USD 2.9 billion in 2026.
What will be the size of the smart implants market by 2036?
The market is projected to reach USD 8.7 billion by 2036.
What is the expected CAGR for smart implants from 2026 to 2036?
FMI projects the market to expand at a CAGR of 11.6% from 2026 to 2036.
How much absolute dollar growth is expected from 2026 to 2036?
The market is expected to add about USD 5.8 billion from 2026 to 2036.
Which implant type holds the largest share in 2025?
Cardiovascular implants lead, estimated at 75.4% share in 2025.
Which surgery approach leads smart implant procedures in 2025?
Open surgery leads, estimated at 63.5% share in 2025.
Which end use setting accounts for the highest demand in 2025?
Hospitals lead, estimated at 53.2% share in 2025.
Which country is expected to grow fastest through 2036?
China is projected to grow fastest at 15.7% CAGR through 2036.
What is the growth outlook for India through 2036?
India is projected to expand at 14.5% CAGR through 2036.
How fast is Germany expected to grow through 2036?
Germany is projected to expand at 13.3% CAGR through 2036.
What is the growth outlook for France through 2036?
France is projected to expand at 12.2% CAGR through 2036.
What is the growth outlook for the UK through 2036?
The UK is projected to expand at 11.0% CAGR through 2036.
How fast is the United States market expected to grow through 2036?
The United States is projected to expand at 9.9% CAGR through 2036.
What is the growth outlook for Brazil through 2036?
Brazil is projected to expand at 8.7% CAGR through 2036.
What are the primary adoption settings for smart implants?
Adoption concentrates in hospitals and specialist centers because implantation and device clinic follow-up sit in those settings.
What is included in the market’s quantitative unit definition in this report?
This report quantifies market value from USD 2.9 billion (2026) to USD 8.7 billion (2036), at a CAGR of 11.6%.
Which segmentations are used for forecasting in this report?
Forecasts are provided by implant type, surgery, end use, and region.
What is the forecast period covered in this report?
The forecast period is 2026 to 2036, with 2025 as the base year.
What is the single growth metric FMI uses for the forecast period?
The forecast uses a CAGR of 11.6% from 2026 to 2036.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Implant Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Implant Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Implant Type , 2026 to 2036
- Cardiovascular implants
- Orthopedic implants
- Neurostimulation implants
- Cardiovascular implants
- Y to o to Y Growth Trend Analysis By Implant Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Implant Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Surgery
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Surgery, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Surgery, 2026 to 2036
- Open Surgery
- Minimally invasive surgery
- Open Surgery
- Y to o to Y Growth Trend Analysis By Surgery, 2021 to 2025
- Absolute $ Opportunity Analysis By Surgery, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End Use
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End Use, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End Use, 2026 to 2036
- Hospitals
- Cardiac care centers
- Ambulatory surgical centers
- Hospitals
- Y to o to Y Growth Trend Analysis By End Use, 2021 to 2025
- Absolute $ Opportunity Analysis By End Use, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Implant Type
- By Surgery
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Implant Type
- By Surgery
- By End Use
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Implant Type
- By Surgery
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Implant Type
- By Surgery
- By End Use
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Implant Type
- By Surgery
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Implant Type
- By Surgery
- By End Use
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Implant Type
- By Surgery
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Implant Type
- By Surgery
- By End Use
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Implant Type
- By Surgery
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Implant Type
- By Surgery
- By End Use
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Implant Type
- By Surgery
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Implant Type
- By Surgery
- By End Use
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Implant Type
- By Surgery
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Implant Type
- By Surgery
- By End Use
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Implant Type
- By Surgery
- By End Use
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Implant Type
- By Surgery
- By End Use
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Implant Type
- By Surgery
- By End Use
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Implant Type
- By Surgery
- By End Use
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Implant Type
- By Surgery
- By End Use
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Implant Type
- By Surgery
- By End Use
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Implant Type
- By Surgery
- By End Use
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Implant Type
- By Surgery
- By End Use
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Implant Type
- By Surgery
- By End Use
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Implant Type
- By Surgery
- By End Use
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Implant Type
- By Surgery
- By End Use
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Implant Type
- By Surgery
- By End Use
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Implant Type
- By Surgery
- By End Use
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Implant Type
- By Surgery
- By End Use
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Implant Type
- By Surgery
- By End Use
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Implant Type
- By Surgery
- By End Use
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Implant Type
- By Surgery
- By End Use
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Implant Type
- By Surgery
- By End Use
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Implant Type
- By Surgery
- By End Use
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Implant Type
- By Surgery
- By End Use
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Implant Type
- By Surgery
- By End Use
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Implant Type
- By Surgery
- By End Use
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Implant Type
- By Surgery
- By End Use
- Competition Analysis
- Competition Deep Dive
- Medtronic plc
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Abbott Laboratories
- Zimmer Biomet Holdings, Inc.
- BIOTRONIK SE & Co. KG
- Boston Scientific Corporation
- DirectSync Surgical
- Intelligent Implants
- Medtronic plc
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Implant Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Surgery, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Implant Type , 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Surgery, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 10: Latin America Market Value (USD Million) Forecast by Implant Type , 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by Surgery, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 13: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Western Europe Market Value (USD Million) Forecast by Implant Type , 2021 to 2036
- Table 15: Western Europe Market Value (USD Million) Forecast by Surgery, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 17: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 18: Eastern Europe Market Value (USD Million) Forecast by Implant Type , 2021 to 2036
- Table 19: Eastern Europe Market Value (USD Million) Forecast by Surgery, 2021 to 2036
- Table 20: Eastern Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 21: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: East Asia Market Value (USD Million) Forecast by Implant Type , 2021 to 2036
- Table 23: East Asia Market Value (USD Million) Forecast by Surgery, 2021 to 2036
- Table 24: East Asia Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 25: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: South Asia and Pacific Market Value (USD Million) Forecast by Implant Type , 2021 to 2036
- Table 27: South Asia and Pacific Market Value (USD Million) Forecast by Surgery, 2021 to 2036
- Table 28: South Asia and Pacific Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 29: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 30: Middle East & Africa Market Value (USD Million) Forecast by Implant Type , 2021 to 2036
- Table 31: Middle East & Africa Market Value (USD Million) Forecast by Surgery, 2021 to 2036
- Table 32: Middle East & Africa Market Value (USD Million) Forecast by End Use, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Implant Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Implant Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Implant Type
- Figure 6: Global Market Value Share and BPS Analysis by Surgery, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Surgery, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Surgery
- Figure 9: Global Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by End Use
- Figure 12: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Region
- Figure 15: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 20: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 21: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 23: North America Market Value Share and BPS Analysis by Implant Type , 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Implant Type , 2026-2036
- Figure 25: North America Market Attractiveness Analysis by Implant Type
- Figure 26: North America Market Value Share and BPS Analysis by Surgery, 2026 and 2036
- Figure 27: North America Market Y-o-Y Growth Comparison by Surgery, 2026-2036
- Figure 28: North America Market Attractiveness Analysis by Surgery
- Figure 29: North America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 31: North America Market Attractiveness Analysis by End Use
- Figure 32: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 33: Latin America Market Value Share and BPS Analysis by Implant Type , 2026 and 2036
- Figure 34: Latin America Market Y-o-Y Growth Comparison by Implant Type , 2026-2036
- Figure 35: Latin America Market Attractiveness Analysis by Implant Type
- Figure 36: Latin America Market Value Share and BPS Analysis by Surgery, 2026 and 2036
- Figure 37: Latin America Market Y-o-Y Growth Comparison by Surgery, 2026-2036
- Figure 38: Latin America Market Attractiveness Analysis by Surgery
- Figure 39: Latin America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 40: Latin America Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 41: Latin America Market Attractiveness Analysis by End Use
- Figure 42: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 43: Western Europe Market Value Share and BPS Analysis by Implant Type , 2026 and 2036
- Figure 44: Western Europe Market Y-o-Y Growth Comparison by Implant Type , 2026-2036
- Figure 45: Western Europe Market Attractiveness Analysis by Implant Type
- Figure 46: Western Europe Market Value Share and BPS Analysis by Surgery, 2026 and 2036
- Figure 47: Western Europe Market Y-o-Y Growth Comparison by Surgery, 2026-2036
- Figure 48: Western Europe Market Attractiveness Analysis by Surgery
- Figure 49: Western Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 50: Western Europe Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 51: Western Europe Market Attractiveness Analysis by End Use
- Figure 52: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 53: Eastern Europe Market Value Share and BPS Analysis by Implant Type , 2026 and 2036
- Figure 54: Eastern Europe Market Y-o-Y Growth Comparison by Implant Type , 2026-2036
- Figure 55: Eastern Europe Market Attractiveness Analysis by Implant Type
- Figure 56: Eastern Europe Market Value Share and BPS Analysis by Surgery, 2026 and 2036
- Figure 57: Eastern Europe Market Y-o-Y Growth Comparison by Surgery, 2026-2036
- Figure 58: Eastern Europe Market Attractiveness Analysis by Surgery
- Figure 59: Eastern Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 60: Eastern Europe Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 61: Eastern Europe Market Attractiveness Analysis by End Use
- Figure 62: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 63: East Asia Market Value Share and BPS Analysis by Implant Type , 2026 and 2036
- Figure 64: East Asia Market Y-o-Y Growth Comparison by Implant Type , 2026-2036
- Figure 65: East Asia Market Attractiveness Analysis by Implant Type
- Figure 66: East Asia Market Value Share and BPS Analysis by Surgery, 2026 and 2036
- Figure 67: East Asia Market Y-o-Y Growth Comparison by Surgery, 2026-2036
- Figure 68: East Asia Market Attractiveness Analysis by Surgery
- Figure 69: East Asia Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 70: East Asia Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 71: East Asia Market Attractiveness Analysis by End Use
- Figure 72: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 73: South Asia and Pacific Market Value Share and BPS Analysis by Implant Type , 2026 and 2036
- Figure 74: South Asia and Pacific Market Y-o-Y Growth Comparison by Implant Type , 2026-2036
- Figure 75: South Asia and Pacific Market Attractiveness Analysis by Implant Type
- Figure 76: South Asia and Pacific Market Value Share and BPS Analysis by Surgery, 2026 and 2036
- Figure 77: South Asia and Pacific Market Y-o-Y Growth Comparison by Surgery, 2026-2036
- Figure 78: South Asia and Pacific Market Attractiveness Analysis by Surgery
- Figure 79: South Asia and Pacific Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 80: South Asia and Pacific Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 81: South Asia and Pacific Market Attractiveness Analysis by End Use
- Figure 82: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 83: Middle East & Africa Market Value Share and BPS Analysis by Implant Type , 2026 and 2036
- Figure 84: Middle East & Africa Market Y-o-Y Growth Comparison by Implant Type , 2026-2036
- Figure 85: Middle East & Africa Market Attractiveness Analysis by Implant Type
- Figure 86: Middle East & Africa Market Value Share and BPS Analysis by Surgery, 2026 and 2036
- Figure 87: Middle East & Africa Market Y-o-Y Growth Comparison by Surgery, 2026-2036
- Figure 88: Middle East & Africa Market Attractiveness Analysis by Surgery
- Figure 89: Middle East & Africa Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 90: Middle East & Africa Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 91: Middle East & Africa Market Attractiveness Analysis by End Use
- Figure 92: Global Market - Tier Structure Analysis
- Figure 93: Global Market - Company Share Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

