Smart Implant Telemetry and Reliability Test Equipment Market
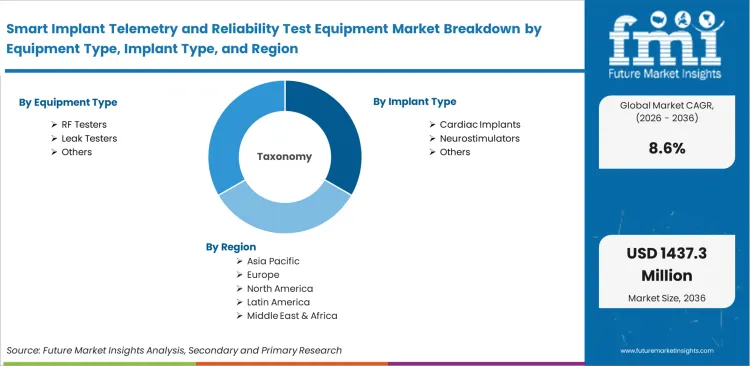
The smart implant telemetry and reliability test equipment market Is Segmented By Equipment Type (RF Testers, EMC Analyzers, Battery Cyclers, Aging Chambers, Leak Testers), Implant Type (Cardiac Implants, Neurostimulators, Cochlear Implants, Biosensors, Other Implants), Test Function (Telemetry Validation, Reliability Cycling, MRI Safety, Battery Endurance, Cybersecurity), Deployment (R&D Labs, Pilot Lines, Production QA, Third-Party Labs), End User (Implant OEMs, CDMOs, Test Labs, Research Institutes), And Region. Forecast for 2026 To 2036.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Smart Implant Telemetry and Reliability Test Equipment Market Size, Market Forecast and Outlook By FMI
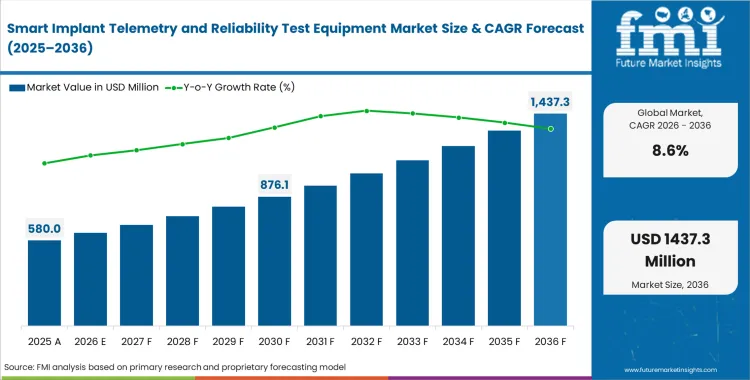
The smart implant telemetry and reliability test equipment market crossed a valuation of USD 530 million in 2025. Revenue is estimated at USD 580 million in 2026 and is projected to reach USD 1,320 million by 2036, reflecting a CAGR of 8.6% from 2026 to 2036. Growth is being supported by tighter validation requirements for active implants and rising demand for advanced telemetry, battery endurance, MRI safety, and cybersecurity testing systems.
Summary of Smart Implant Telemetry and Reliability Test Equipment Market
- The market is forecast to reach USD 1,320 million by 2036.
- The market is expected to grow at a CAGR of 8.6% from 2026 to 2036.
- The market was estimated at USD 530 million in 2025.
- The market represents an incremental opportunity of USD 740 million over the forecast period.
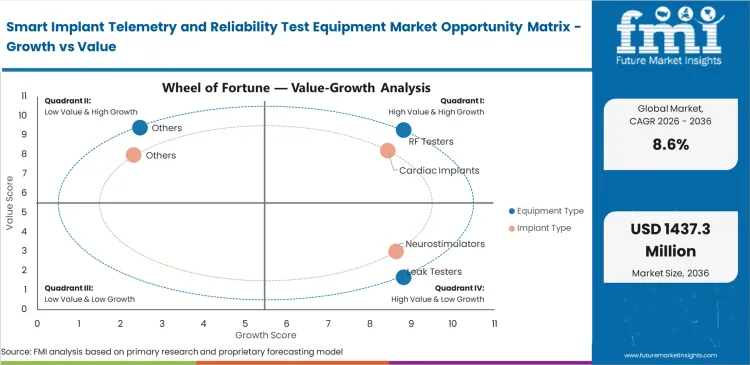
- RF testers lead the equipment segment with a 34.0% share in 2026.
- Cardiac implants dominate by application with a 38.0% share in 2026.
- Telemetry validation leads by function with a 31.0% share in 2026.
- R&D labs lead deployment usage with a 46.0% share in 2026.
- Implant OEMs dominate end users with a 52.0% share in 2026.
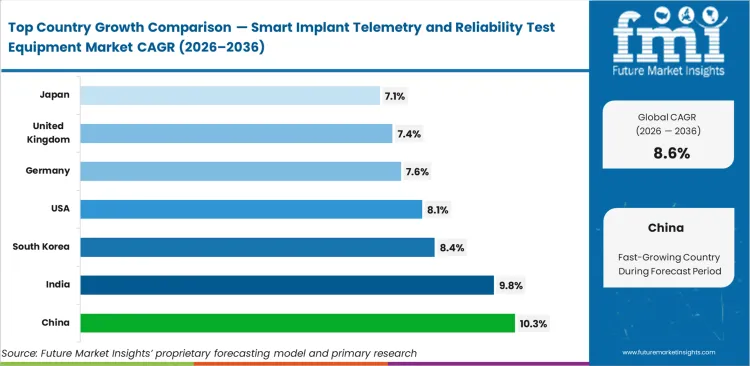
- China is the fastest-growing market at 10.3%, followed by India at 9.8% and South Korea at 8.4%.
- The market is driven by increasing complexity of smart implants, rising demand for wireless reliability testing, and stricter regulatory validation requirements.
- The competitive landscape includes RF testing equipment providers, medical validation system developers, and integrated compliance solution suppliers.
Manufacturers face rising pressure to validate smart implants thoroughly before commercialization. Wireless reliability concerns are reshaping investment priorities in active implantable device test equipment. Basic functional checks are no longer sufficient to support approval readiness and lifecycle reliability. A flawed software update can disrupt implant performance after deployment, increasing regulatory and commercial risk for manufacturers. Advanced validation routines help identify intermittent faults that conventional test cycles may overlook. FMI analysts point out that shifting from manual checks to automated wireless verification prevents severe safety issues after the product hits the market.
Building implants that transmit and receive data changes how companies invest in medical device tester hardware. Wireless health tracking becomes a standard product feature, leading manufacturers to stop buying separate, standalone instruments. Demand shifts toward fully integrated implantable medical device test systems capable of checking multiple functions simultaneously. Catching signal delays early in the laboratory saves manufacturers from spending millions to redesign a nearly finished product.
Divergent regulatory timelines across Asia and Europe create distinct investment waves for specialized validation hardware. China expects to scale at a CAGR of 10.3% as local device manufacturers upgrade quality control infrastructure. India estimates growth at a CAGR of 9.8% through 2036, supported by expansion of third-party diagnostic ecosystems. South Korea projects advancement at a CAGR of 8.4%, backed by continued investment in local biomedical hubs. United States demand leads Western markets with an estimated CAGR of 8.1%, while Germany likely expands at 7.6% through 2036. United Kingdom and Japan reflect mature replacement cycles, projected at 7.4% and 7.1% respectively.
Segmental Analysis
Smart Implant Telemetry and Reliability Test Equipment Market Analysis by Equipment Type
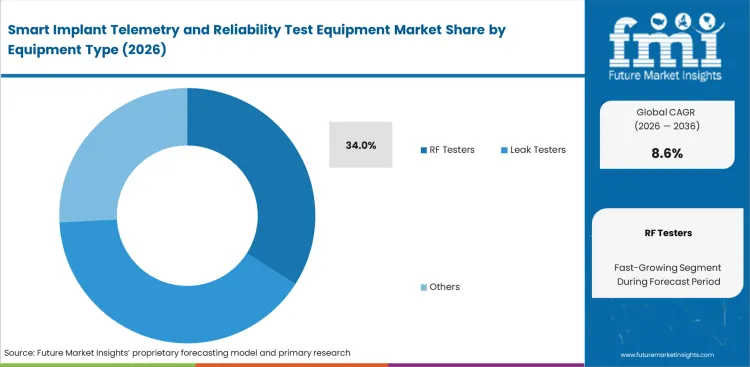
Validating wireless signals correctly decides whether a new biomedical device reaches patients. RF testers category represents 34% of the market in 2026, as testing groups realize machines cannot easily swap out later in the process. Once a lab establishes a baseline telemetry profile using specific implant RF test equipment, switching to a different machine restarts the entire regulatory approval clock. Manufacturers accept this technical lock-in as a necessary cost to guarantee reliable two-way communication. Avoiding a complete recertification cycle creates strong loyalty toward established equipment brands capable of precise medical device RF validation.
- Communication Dropout Prevention: Specialized RF instruments detect micro-second latency issues during burst data transmission. Compliance groups avoid critical real-world signal failures.
- Interference Vulnerability: Complex hospital environments generate significant electromagnetic noise that corrupts basic telemetry signals. Validators must execute rigorous spectrum analysis using specialized implant EMC test equipment to secure approvals.
- Protocol Adherence: Standard validation routines struggle with proprietary encryption handshakes used in modern implants. Manufacturing supervisors need dedicated hardware to confirm data packet security.
Smart Implant Telemetry and Reliability Test Equipment Market Analysis by Implant Type
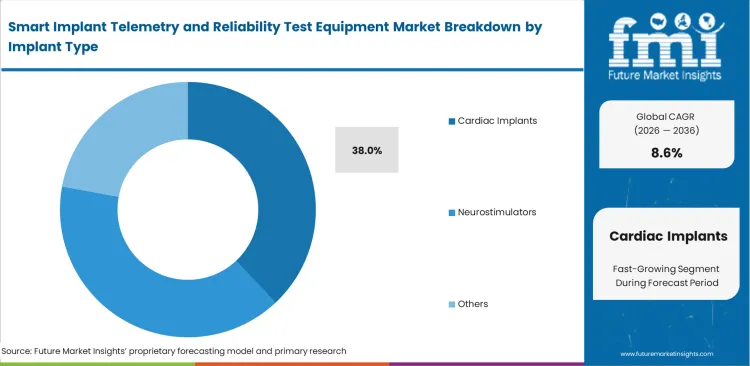
Cardiac implants segment anticipates capturing 38% of the market in 2026, driven by an ongoing clash between shrinking device sizes and expanding wireless data needs. Laboratories at cardiovascular medical devices companies face intense pressure to prove hardware handles continuous monitoring without draining the battery. Older pacemakers barely interacted with outside equipment, but modern defibrillators send significant amounts of physiological data every week. Checking heavy data traffic without draining tiny power cells forces manufacturers to buy highly specialized cardiac implant validation systems.
- Baseline Energy Evaluation: Initial component selection relies heavily on specialized battery cyclers to project 10-year lifespan viability. R&D functions eliminate power-hungry communication chips early.
- Stress Testing Qualification: Regulatory bodies demand proof that active cardiovascular units survive extreme physiological strain. Quality functions deploy advanced aging chambers simulating decade-long fluid exposure.
- Continuous Telemetry Renewal: Firmware updates alter power consumption profiles dramatically post-launch. Test laboratories require persistent validation hardware to certify every software patch.
Smart Implant Telemetry and Reliability Test Equipment Market Analysis by Test Function
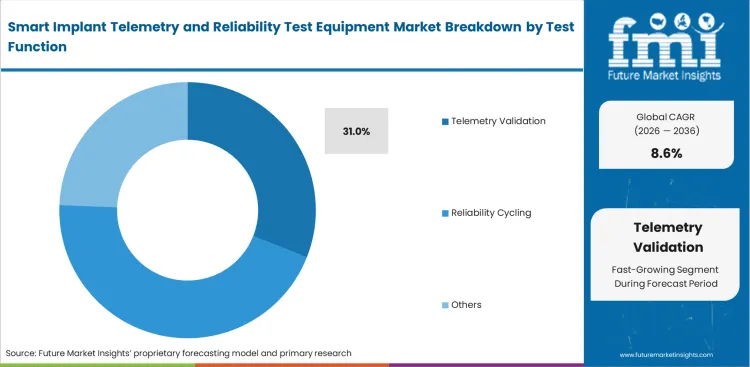
Getting federal clearance requires clear proof that an active implant communicates securely without putting the patient in danger. Checking bidirectional device technologies forces compliance staff to choose between slow manual testing and faster automated platforms. Relying on manual checks introduces high risks of human error during complex encryption handovers. To eliminate mistakes, labs invest heavily in software-driven instruments because telemetry validation holds a share of 31% in 2026. Automated approach allows manufacturing staff to run thousands of active implant telemetry validation scenarios at the exact same time.
- Basic Link Verification: Standard instruments confirm simple pairing between internal units and external controllers. Quality technicians capture baseline connection metrics quickly.
- Complex Physiological Simulation: Advanced chambers introduce tissue attenuation models that degrade signal strength realistically. Manufacturing supervisors identify true operational limits under dense physical blockages.
- Absolute Cybersecurity Qualification: Elite hardware executes continuous penetration testing alongside dedicated implant MRI safety test systems to ensure holistic device integrity. Security functions validate encryption strength before regulatory submission.
Smart Implant Telemetry and Reliability Test Equipment Market Analysis by Deployment
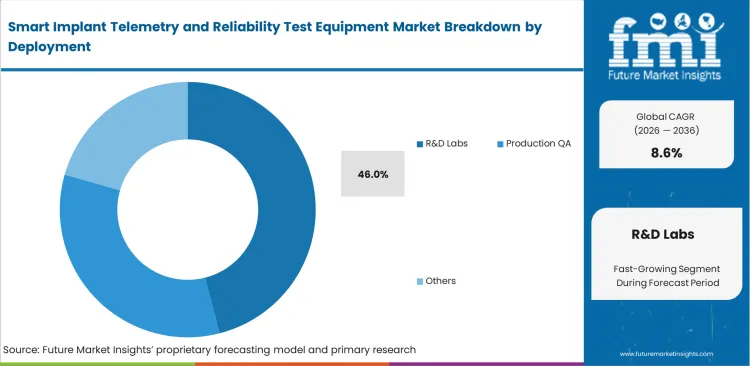
Testing new biomedical platforms early in the design cycle heavily influences whether product survives long-term commercial use. Research groups understand this risk, which is why R&D labs contribute 46% of total market share in 2026. Preclinical functions run early prototypes through sophisticated patient monitoring devices testing arrays to finalize component choices well before expensive human trials start. Centralizing evaluation work prevents major design flaws from showing up late in development. Pushing comprehensive hardware checks off until the pilot production phase virtually guarantees costly recalls unless the company uses a unified implantable electronics test platform.
- Early Conceptual Validation: Innovation labs deploy flexible benchtop RF analyzers to test raw component viability. Lead researchers secure proof-of-concept data for initial funding.
- Integrated Prototype Testing: Moving toward functional models requires specialized environmental chambers simulating core body temperatures. Compliance functions confirm basic physical resilience.
- Final Production Transition: Scaling manufacturing necessitates high-throughput implant manufacturing test automation setups. Operations functions standardize testing protocols across global facilities.
Smart Implant Telemetry and Reliability Test Equipment Market Analysis by End User
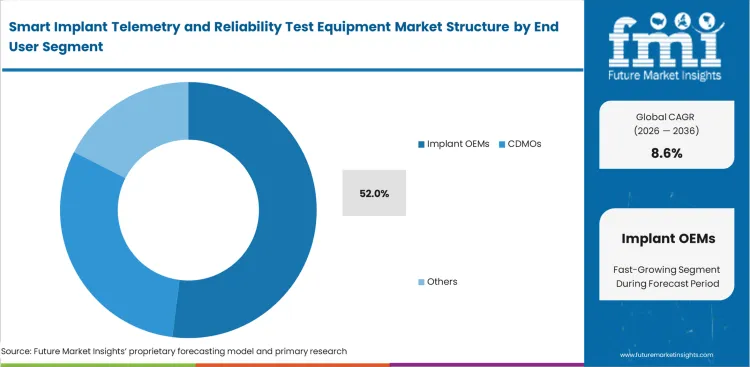
In 2026, the implant OEMs segment accounts for 52% of market share as major medical companies fight to keep proprietary wireless protocols secret. Sending complex ambulatory monitoring hardware to outside labs exposes valuable trade secrets to third-party vulnerabilities. Large biomedical firms prefer building own dedicated testing facilities inside headquarters, despite heavy upfront costs. Owning the AIMD compliance testing equipment internally lets quality assurance functions speed up design cycles without waiting in line at external testing vendors.
- Internal Capitalization Benefits: Purchasing dedicated aging chambers reduces long-term reliance on expensive hourly contract laboratory fees. Operations functions achieve predictable testing budgets.
- Hidden Maintenance Burdens: Highly calibrated RF equipment requires frequent expensive recertification to satisfy medical regulations. Facility functions must allocate continuous funding for hardware upkeep.
- Total Lifecycle Advantages: Complete internal ownership allows immediate testing of emergency firmware patches. Compliance functions avoid costly regulatory delays during critical post-market software updates.
Smart Implant Telemetry and Reliability Test Equipment Market Drivers, Restraints, and Opportunities
Health authorities now require active heart devices to monitor patients continuously. This strict rule forces laboratories to upgrade their validation equipment rather than rely on outdated manual checks. Missing complete testing data blocks product launches and causes severe financial losses. Modern mobile cardiac telemetry features need clear proof that two-way communication stays active inside the human body under heavy physical stress. Testing basic signals on a dry workbench fails to meet these new standards because reviewers expect complete environmental simulation. Buying advanced automated hardware clears these regulatory hurdles faster.
Getting new testing hardware approved inside a medical company creates unexpected delays. Proving that a new testing instrument works accurately requires almost as much paperwork as certifying the actual cardiovascular devices. Engineering leads face major technical hurdles when they try to connect modern software-driven testers with older, hardcoded compliance databases. This heavy administrative workload slows down equipment upgrades, even when labs have the budget to buy them. Setting up new automatic calibration tools takes time away from actual product development.
- Predictive Fault Tracking: Using software to read historical testing data helps spot tiny component weaknesses before they cause actual failures. Lab staff speed up the development of neurostimulation products without taking safety risks.
- Compact Simulation Chambers: Building smaller testing boxes lets labs evaluate thousands of tiny implants at the exact same time. Facility planners save valuable space on the testing floor and increase their daily testing volume.
- Connected Compliance Networks: Linking testing sites together online organizes regulatory paperwork automatically. Compliance staff submit their international filings faster by gathering data from multiple global sites instantly.
Regional Analysis
Regional demand for smart implant testing hardware shifts based on local medical device manufacturing goals and specific regulatory deadlines. While Western hubs focus on strict compliance updates to keep products on the market, Asian countries are rapidly building out their own testing infrastructure to capture a larger share of global exports. Based on regional analysis, the market is segmented into Asia Pacific, North America, and Europe across 40 plus countries.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| China | 10.3% |
| India | 9.8% |
| South Korea | 8.4% |
| United States | 8.1% |
| Germany | 7.6% |
| United Kingdom | 7.4% |
| Japan | 7.1% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Asia Pacific Smart Implant Telemetry and Reliability Test Equipment Market Analysis
Asian tech hubs are buying more validation equipment as they build their own advanced biomedical manufacturing centers. Local device makers are expanding their internal testing labs so they can compete directly with Western cardiovascular companies. Government-funded science parks now include specialized environmental testing centers to help regional start-ups validate their designs. Upgrading basic MRI safe neurostimulation testing capabilities helps these Asian manufacturers meet the strict export rules required to sell their devices in Europe.
- China: State-backed funding for domestic medical technology development is accelerating the uptake of advanced RF testing hardware. Demand for smart implant telemetry and reliability test equipment in China is projected to grow at a CAGR of 10.3% through 2036 as laboratories expand compliance testing capacity. That build-out is giving export-oriented biomedical manufacturers more room to complete validation work within the country instead of relying on overseas testing labs.
- India: Cost-effective validation platforms remain important as India’s third-party diagnostic testing base continues to expand. The market for smart implant telemetry and reliability test equipment in India is expected to increase at a CAGR of 9.8% through 2036, supported by a growing volume of outsourced compliance work from global medical device companies. Regional testing centers are using this installed base to strengthen their position in international validation services.
- South Korea: Micro-electronics expertise continues to support demand for specialized battery endurance cyclers in South Korea. Smart implant telemetry and reliability test equipment demand in the country is likely to rise at a CAGR of 8.4% during the study period as developers work to extend the operating life of continuous monitoring devices used inside the body. That testing depth is helping local neurostimulator and implant developers stay aligned with global performance benchmarks.
- Japan: Long-term reliability remains a central requirement in Japan’s implant testing environment as aging demographics keep focus on durable geriatric monitoring devices. The market in Japan is forecast to expand at a CAGR of 7.1% through 2036 because validation programs place strong weight on device endurance over extended operating life. This continues to support established biomedical manufacturers known for dependable implant hardware.
FMI's report includes Australia and Southeast Asian nations. Advanced regulatory alignment across ASEAN borders streamlines regional validation equipment deployment.
North America Smart Implant Telemetry and Reliability Test Equipment Market Analysis
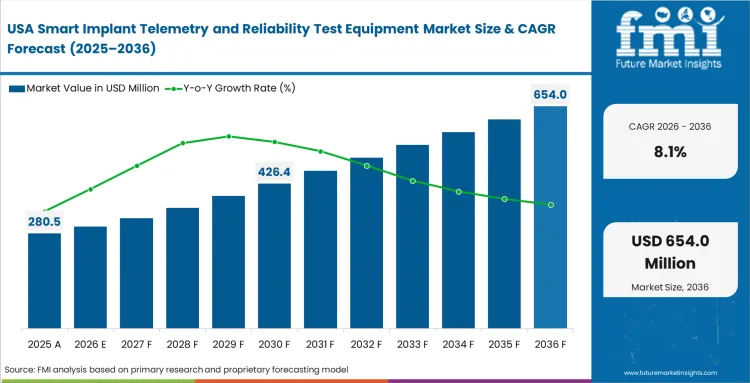
Strict FDA documentation rules force American biomedical hubs to spend heavily on automated testing platforms. Leading medical innovators are buying integrated cybersecurity testing hardware now that wireless two-way communication is required for new device approvals. Increased regulatory focus on implantable infusion pumps running out of battery power drives clear demand for advanced environmental cycling chambers. Engineers simply cannot afford manual testing errors during the final pre-market checks.
- United States: Local laboratories continue investing in advanced telemetry validation instruments to support their lead in emerging neurostimulation research. The United States market is projected to grow at a CAGR of 8.1% over the forecast period, with added spending directed toward proprietary encryption testing setups. Domestic bionic implant developers rely on this in-house validation capacity to maintain tighter control over sensitive intellectual property.
FMI's report includes Canada. Cross-border biomedical research collaboration accelerates unified North American testing standards.
Europe Smart Implant Telemetry and Reliability Test Equipment Market Analysis
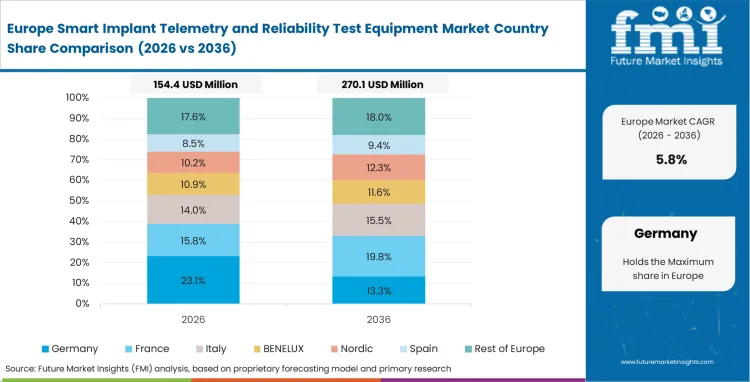
The shift to strict Medical Device Regulation (MDR) rules requires established European manufacturers to test their older products again using modern hardware. Rules for continuous active monitoring mean companies must perform thorough RF signal integrity testing that accounts for crowded urban hospital environments. Centralized testing labs are upgrading their older equipment to handle complex two-way telemetry protocols. Testers use this new hardware to gather the certification data needed to keep their products on the market.
- Germany: Strong engineering capabilities continue to support investment in precision EMC analyzers across Germany’s biomedical manufacturing hubs. Preventing electromagnetic interference in complex clinical environments remains a core testing priority. The validation equipment market in Germany is projected to grow at a CAGR of 7.6% during the assessment period, which is reinforcing the country’s role in cross-border compliance testing work for precision device manufacturers.
- United Kingdom: A well-established academic research base is sustaining demand for flexible benchtop evaluation instruments. The market in the United Kingdom is expected to expand at a CAGR of 7.4% through 2036 as research teams validate experimental 3D medical implants using advanced tissue simulation chambers. This testing base supports a more reliable transition from prototype-stage development into human clinical evaluation.
FMI's report includes France, Italy, and Spain. Expanding continental manufacturing footprints require standardized validation protocols across all facilities.
Competitive Aligners for Market Players
Active implant validation leaves very little tolerance for error, which keeps the supplier base limited to a relatively small group of specialized test equipment providers. Keysight Technologies and Rohde & Schwarz remain strong in radio frequency testing because their systems combine measurement hardware with deep integrated software. Two-way wireless communication testing depends on signal measurement functions that standard medical device testers usually cannot provide. Platform selection often comes down to how efficiently the software can generate regulatory documentation, not just the underlying hardware speed.
Established testing companies also benefit from existing libraries of medical compliance protocols. Averna and Emerson Test & Measurement use prebuilt software validation checklists to secure long-term programs with large medical device manufacturers early in the design cycle. Modeling how human tissue interferes with telemetry signals depends on years of accumulated test data, which new entrants cannot replicate quickly. FMI notes that implant telemetry test system buyers tend to avoid unfamiliar equipment brands because a weak compliance record can slow FDA review and create added administrative risk.
Large medical device manufacturers still try to avoid dependence on a single equipment supplier. Flexible test configurations remain important during design and qualification because they preserve room to compare RF and EMC test equipment for implants before a major commitment is made. Common interface standards support that flexibility across laboratory setups. As wireless requirements continue to evolve, established vendors will need to rely more on software-adaptable systems rather than depending only on fixed-function hardware.
Key Players in Smart Implant Telemetry and Reliability Test Equipment Market
- Keysight Technologies
- Rohde & Schwarz
- Anritsu
- Emerson Test & Measurement (formerly NI)
- Averna
Scope of the Report
| Metric | Value |
|---|---|
| Quantitative Units | USD 580 million to USD 1,320 million, at a CAGR of 8.6% |
| Market Definition | Hardware and software instruments that validate wireless communication, electronic durability, and operational safety for active medical implants prior to human deployment. |
| Segmentation | Equipment type, Implant type, Test function, Deployment, End user, Region |
| Regions Covered | North America, Latin America, Europe, Asia Pacific, Middle East and Africa |
| Countries Covered | United States, Canada, Brazil, Mexico, Germany, United Kingdom, France, Italy, Spain, China, India, Japan, South Korea, Australia, GCC, South Africa |
| Key Companies Profiled | Keysight Technologies, Rohde & Schwarz, Anritsu, Emerson Test & Measurement (formerly NI), Averna |
| Forecast Period | 2026 to 2036 |
| Approach | Annual capital expenditure budgets for validation laboratories establish baseline hardware demand. |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Key Segments
Equipment Type
- RF testers
- EMC analyzers
- Battery cyclers
- Aging chambers
- Leak testers
Implant Type
- Cardiac implants
- Neurostimulators
- Cochlear implants
- Biosensors
- Other implants
Test Function
- Telemetry validation
- Reliability cycling
- MRI safety
- Battery endurance
- Cybersecurity
Deployment
- R&D labs
- Pilot lines
- Production QA
- Third-party labs
End User
- Implant OEMs
- CDMOs
- Test labs
- Research institutes
Regions:
- Asia Pacific
- India
- China
- Japan
- South Korea
- Indonesia
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- Italy
- France
- United Kingdom
- Spain
- Benelux
- Nordics
- Central & Eastern Europe
- Rest of Europe
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Argentina
- Chile
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- United Arab Emirates
- South Africa
- Turkey
- Rest of Middle East & Africa
Bibliography
- Medicines and Healthcare products Regulatory Agency. (2026). Implementation of the future regulations. GOV.UK.
- Harwood, M., Fahrenholtz, S. J., Wellnitz, C. V., Kawashima, A., & Panda, A. (2024). MRI in adult patients with active and inactive implanted MR-conditional, MR-nonconditional, and other devices. RadioGraphics, 44(3), e230102.
- Vivarelli, C., Mattei, E., Ricci, F., D’Eramo, S., & Calcagnini, G. (2025). A tool for the assessment of electromagnetic compatibility in active implantable devices: The pacemaker physical twin. Bioengineering, 12(7), 689.
- Ries, M., Kelava, I., Ajduk, J., Košec, A., Žaja, R., & Trotić, R. (2024). Correlation between neural response telemetry measurements and fitting levels. International Journal of Pediatric Otorhinolaryngology, 184, 112001.
- Mariello, M. (2025). Reliability and stability of Bioelectronic Medicine: A critical and pedagogical perspective. Bioelectronic Medicine, 11(1), 16.
- Zhai, L., Duan, J., Lin, T., & Shao, H. (2024). Recent advances in implantable batteries: Development and challenge. Journal of Alloys and Compounds, 979, 173551.
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary source documentation.
This Report Addresses
- Automated validation capabilities required for continuous active cardiovascular monitoring.
- RF signal integrity degradation testing within dense electromagnetic clinical environments.
- Tissue simulation parameters utilized in accelerated battery lifecycle evaluation.
- Cybersecurity encryption testing protocols mandatory for bidirectional medical communication.
- Preclinical laboratory deployment models minimizing expensive late-stage design flaws.
- In-house capital expenditure patterns among major biomedical original equipment manufacturers.
- Structural differences in regulatory documentation requirements across Asian test hubs.
- Proprietary software protocol lock-in dynamics governing competitive hardware acquisition.
Frequently Asked Questions
What is smart implant telemetry testing?
It is the process of using specialized instruments to validate the wireless communication integrity and electronic durability of active medical implants prior to human deployment.
Why do connected implants need reliability test equipment?
Continuous active monitoring mandates require absolute proof that bidirectional communication modules will not fail or exhaust batteries under severe physiological stress.
What standards apply to active implant testing?
Compliance frameworks like EU MDR and FDA clearance guidelines enforce rigorous verification of RF signal integrity and electromagnetic compatibility for medical devices.
How are implantable devices tested for telemetry?
Engineers execute continuous penetration testing and automated verification cycles inside specialized aging chambers that simulate complex human tissue attenuation.
Compare RF and EMC test equipment for implants.
RF testers detect micro-second latency issues during data transmission, whereas EMC analyzers measure the device's vulnerability to external electromagnetic noise in hospital settings.
Which countries are growing fastest in this market?
China leads regional expansion with a projected 10.3% CAGR, driven by massive state funding aimed at localizing advanced biomedical device manufacturing infrastructure.
What drives demand for implant reliability testing systems?
Regulatory agencies enforcing strict continuous monitoring capabilities compel compliance officers to replace basic manual checks with automated telemetry verification platforms.
Why do RF testers lead equipment types?
Complex bidirectional wireless protocols require dedicated signal integrity validation instruments to prevent severe commercial penalties linked to real-world communication dropouts.
Why do cardiac implants command high hardware demand?
Continuous ambulatory monitoring forces extreme battery cycling and massive data transmission testing to ensure decades-long survival in dynamic cardiovascular environments.
Why do R&D labs dominate deployment?
Finalizing component selection early through centralized testing arrays prevents severe late-stage design failures during expensive clinical trials.
Why do implant OEMs prefer internal testing?
Protecting vital intellectual property prevents biomedical companies from outsourcing proprietary encryption verification to third-party testing facilities.
How does India differ from South Korea?
India expands third-party diagnostic ecosystems while South Korea targets advanced micro-electronic endurance through concentrated government investments.
What limits rapid testing integration?
Administrative friction forces engineers to struggle matching modern software-defined testers with legacy medical compliance databases.
How does cybersecurity influence equipment selection?
Vulnerability testing requires elite hardware to execute continuous penetration protocols securely and validate encryption strength before regulatory submission.
What role do aging chambers play?
Advanced simulation modules replicate decade-long fluid exposure to prove physiological durability and stress resistance in active cardiovascular units.
Why are automated platforms replacing manual tests?
Manual verification introduces massive human error risks during complex encryption handshakes, forcing labs to invest in software-defined instrumentation.
How do engineers prevent communication dropouts?
Specialized instruments detect micro-second latency issues during burst data transmission testing to guarantee bidirectional reliability.
What hidden costs impact internal labs?
Highly calibrated RF equipment demands frequent expensive recertification to maintain medical compliance and satisfy strict regulatory audits.
How do AI models improve validation?
Machine learning algorithms analyze massive reliability datasets to identify subtle component degradation early and accelerate development cycles safely.
Why are miniaturized test fixtures gaining popularity?
Compact simulation chambers allow parallel testing of thousands of micro-implants simultaneously, maximizing laboratory floor space utilization for operations officers.
What capability defines competitive hardware leadership?
Deep proprietary software integration simplifies automated regulatory reporting for compliance groups navigating stringent FDA review circles.
How do buyers resist vendor lock-in?
Quality assurance heads mandate modular hardware architectures using open-standard communication interfaces to efficiently evaluate different instruments before integration.
Why do legacy providers hold an advantage?
Established vendors possess massive libraries of pre-configured medical compliance protocols built on decades of empirical physiological data collection.
What happens if companies delay validation upgrades?
Failing to provide comprehensive lifecycle testing data stalls a multi-million dollar product launches indefinitely and guarantees massive recall risks.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Equipment Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Equipment Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Equipment Type , 2026 to 2036
- RF Testers
- Leak Testers
- Others
- RF Testers
- Y to o to Y Growth Trend Analysis By Equipment Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Equipment Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Implant Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Implant Type, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Implant Type, 2026 to 2036
- Cardiac Implants
- Neurostimulators
- Others
- Cardiac Implants
- Y to o to Y Growth Trend Analysis By Implant Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Implant Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Test Function
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Test Function, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Test Function, 2026 to 2036
- Telemetry Validation
- Reliability Cycling
- Others
- Telemetry Validation
- Y to o to Y Growth Trend Analysis By Test Function, 2021 to 2025
- Absolute $ Opportunity Analysis By Test Function, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Deployment
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Deployment, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Deployment, 2026 to 2036
- R&D Labs
- Production QA
- Others
- R&D Labs
- Y to o to Y Growth Trend Analysis By Deployment, 2021 to 2025
- Absolute $ Opportunity Analysis By Deployment, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User, 2026 to 2036
- Implant OEMs
- CDMOs
- Others
- Implant OEMs
- Y to o to Y Growth Trend Analysis By End User, 2021 to 2025
- Absolute $ Opportunity Analysis By End User, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Equipment Type
- By Implant Type
- By Test Function
- By Deployment
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Equipment Type
- By Implant Type
- By Test Function
- By Deployment
- By End User
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Equipment Type
- By Implant Type
- By Test Function
- By Deployment
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Equipment Type
- By Implant Type
- By Test Function
- By Deployment
- By End User
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Equipment Type
- By Implant Type
- By Test Function
- By Deployment
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Equipment Type
- By Implant Type
- By Test Function
- By Deployment
- By End User
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Equipment Type
- By Implant Type
- By Test Function
- By Deployment
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Equipment Type
- By Implant Type
- By Test Function
- By Deployment
- By End User
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Equipment Type
- By Implant Type
- By Test Function
- By Deployment
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Equipment Type
- By Implant Type
- By Test Function
- By Deployment
- By End User
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Equipment Type
- By Implant Type
- By Test Function
- By Deployment
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Equipment Type
- By Implant Type
- By Test Function
- By Deployment
- By End User
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Equipment Type
- By Implant Type
- By Test Function
- By Deployment
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Equipment Type
- By Implant Type
- By Test Function
- By Deployment
- By End User
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Equipment Type
- By Implant Type
- By Test Function
- By Deployment
- By End User
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Equipment Type
- By Implant Type
- By Test Function
- By Deployment
- By End User
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Equipment Type
- By Implant Type
- By Test Function
- By Deployment
- By End User
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Equipment Type
- By Implant Type
- By Test Function
- By Deployment
- By End User
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Equipment Type
- By Implant Type
- By Test Function
- By Deployment
- By End User
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Equipment Type
- By Implant Type
- By Test Function
- By Deployment
- By End User
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Equipment Type
- By Implant Type
- By Test Function
- By Deployment
- By End User
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Equipment Type
- By Implant Type
- By Test Function
- By Deployment
- By End User
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Equipment Type
- By Implant Type
- By Test Function
- By Deployment
- By End User
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Equipment Type
- By Implant Type
- By Test Function
- By Deployment
- By End User
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Equipment Type
- By Implant Type
- By Test Function
- By Deployment
- By End User
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Equipment Type
- By Implant Type
- By Test Function
- By Deployment
- By End User
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Equipment Type
- By Implant Type
- By Test Function
- By Deployment
- By End User
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Equipment Type
- By Implant Type
- By Test Function
- By Deployment
- By End User
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Equipment Type
- By Implant Type
- By Test Function
- By Deployment
- By End User
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Equipment Type
- By Implant Type
- By Test Function
- By Deployment
- By End User
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Equipment Type
- By Implant Type
- By Test Function
- By Deployment
- By End User
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Equipment Type
- By Implant Type
- By Test Function
- By Deployment
- By End User
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Equipment Type
- By Implant Type
- By Test Function
- By Deployment
- By End User
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Equipment Type
- By Implant Type
- By Test Function
- By Deployment
- By End User
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Equipment Type
- By Implant Type
- By Test Function
- By Deployment
- By End User
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Equipment Type
- By Implant Type
- By Test Function
- By Deployment
- By End User
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Equipment Type
- By Implant Type
- By Test Function
- By Deployment
- By End User
- Competition Analysis
- Competition Deep Dive
- Keysight Technologies
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Rohde & Schwarz
- Anritsu
- Emerson Test & Measurement (formerly NI)
- Averna
- Keysight Technologies
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Equipment Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Implant Type, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Test Function, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by Deployment, 2021 to 2036
- Table 6: Global Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Equipment Type , 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Implant Type, 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by Test Function, 2021 to 2036
- Table 11: North America Market Value (USD Million) Forecast by Deployment, 2021 to 2036
- Table 12: North America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 13: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Latin America Market Value (USD Million) Forecast by Equipment Type , 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Implant Type, 2021 to 2036
- Table 16: Latin America Market Value (USD Million) Forecast by Test Function, 2021 to 2036
- Table 17: Latin America Market Value (USD Million) Forecast by Deployment, 2021 to 2036
- Table 18: Latin America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 19: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: Western Europe Market Value (USD Million) Forecast by Equipment Type , 2021 to 2036
- Table 21: Western Europe Market Value (USD Million) Forecast by Implant Type, 2021 to 2036
- Table 22: Western Europe Market Value (USD Million) Forecast by Test Function, 2021 to 2036
- Table 23: Western Europe Market Value (USD Million) Forecast by Deployment, 2021 to 2036
- Table 24: Western Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 25: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: Eastern Europe Market Value (USD Million) Forecast by Equipment Type , 2021 to 2036
- Table 27: Eastern Europe Market Value (USD Million) Forecast by Implant Type, 2021 to 2036
- Table 28: Eastern Europe Market Value (USD Million) Forecast by Test Function, 2021 to 2036
- Table 29: Eastern Europe Market Value (USD Million) Forecast by Deployment, 2021 to 2036
- Table 30: Eastern Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 31: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 32: East Asia Market Value (USD Million) Forecast by Equipment Type , 2021 to 2036
- Table 33: East Asia Market Value (USD Million) Forecast by Implant Type, 2021 to 2036
- Table 34: East Asia Market Value (USD Million) Forecast by Test Function, 2021 to 2036
- Table 35: East Asia Market Value (USD Million) Forecast by Deployment, 2021 to 2036
- Table 36: East Asia Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 37: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 38: South Asia and Pacific Market Value (USD Million) Forecast by Equipment Type , 2021 to 2036
- Table 39: South Asia and Pacific Market Value (USD Million) Forecast by Implant Type, 2021 to 2036
- Table 40: South Asia and Pacific Market Value (USD Million) Forecast by Test Function, 2021 to 2036
- Table 41: South Asia and Pacific Market Value (USD Million) Forecast by Deployment, 2021 to 2036
- Table 42: South Asia and Pacific Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 43: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 44: Middle East & Africa Market Value (USD Million) Forecast by Equipment Type , 2021 to 2036
- Table 45: Middle East & Africa Market Value (USD Million) Forecast by Implant Type, 2021 to 2036
- Table 46: Middle East & Africa Market Value (USD Million) Forecast by Test Function, 2021 to 2036
- Table 47: Middle East & Africa Market Value (USD Million) Forecast by Deployment, 2021 to 2036
- Table 48: Middle East & Africa Market Value (USD Million) Forecast by End User, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Equipment Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Equipment Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Equipment Type
- Figure 6: Global Market Value Share and BPS Analysis by Implant Type, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Implant Type, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Implant Type
- Figure 9: Global Market Value Share and BPS Analysis by Test Function, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Test Function, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Test Function
- Figure 12: Global Market Value Share and BPS Analysis by Deployment, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Deployment, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Deployment
- Figure 15: Global Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 17: Global Market Attractiveness Analysis by End User
- Figure 18: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 19: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 20: Global Market Attractiveness Analysis by Region
- Figure 21: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 23: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 24: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 25: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 26: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 27: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 28: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 29: North America Market Value Share and BPS Analysis by Equipment Type , 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Equipment Type , 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Equipment Type
- Figure 32: North America Market Value Share and BPS Analysis by Implant Type, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Implant Type, 2026-2036
- Figure 34: North America Market Attractiveness Analysis by Implant Type
- Figure 35: North America Market Value Share and BPS Analysis by Test Function, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by Test Function, 2026-2036
- Figure 37: North America Market Attractiveness Analysis by Test Function
- Figure 38: North America Market Value Share and BPS Analysis by Deployment, 2026 and 2036
- Figure 39: North America Market Y-o-Y Growth Comparison by Deployment, 2026-2036
- Figure 40: North America Market Attractiveness Analysis by Deployment
- Figure 41: North America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 42: North America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 43: North America Market Attractiveness Analysis by End User
- Figure 44: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 45: Latin America Market Value Share and BPS Analysis by Equipment Type , 2026 and 2036
- Figure 46: Latin America Market Y-o-Y Growth Comparison by Equipment Type , 2026-2036
- Figure 47: Latin America Market Attractiveness Analysis by Equipment Type
- Figure 48: Latin America Market Value Share and BPS Analysis by Implant Type, 2026 and 2036
- Figure 49: Latin America Market Y-o-Y Growth Comparison by Implant Type, 2026-2036
- Figure 50: Latin America Market Attractiveness Analysis by Implant Type
- Figure 51: Latin America Market Value Share and BPS Analysis by Test Function, 2026 and 2036
- Figure 52: Latin America Market Y-o-Y Growth Comparison by Test Function, 2026-2036
- Figure 53: Latin America Market Attractiveness Analysis by Test Function
- Figure 54: Latin America Market Value Share and BPS Analysis by Deployment, 2026 and 2036
- Figure 55: Latin America Market Y-o-Y Growth Comparison by Deployment, 2026-2036
- Figure 56: Latin America Market Attractiveness Analysis by Deployment
- Figure 57: Latin America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 58: Latin America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 59: Latin America Market Attractiveness Analysis by End User
- Figure 60: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 61: Western Europe Market Value Share and BPS Analysis by Equipment Type , 2026 and 2036
- Figure 62: Western Europe Market Y-o-Y Growth Comparison by Equipment Type , 2026-2036
- Figure 63: Western Europe Market Attractiveness Analysis by Equipment Type
- Figure 64: Western Europe Market Value Share and BPS Analysis by Implant Type, 2026 and 2036
- Figure 65: Western Europe Market Y-o-Y Growth Comparison by Implant Type, 2026-2036
- Figure 66: Western Europe Market Attractiveness Analysis by Implant Type
- Figure 67: Western Europe Market Value Share and BPS Analysis by Test Function, 2026 and 2036
- Figure 68: Western Europe Market Y-o-Y Growth Comparison by Test Function, 2026-2036
- Figure 69: Western Europe Market Attractiveness Analysis by Test Function
- Figure 70: Western Europe Market Value Share and BPS Analysis by Deployment, 2026 and 2036
- Figure 71: Western Europe Market Y-o-Y Growth Comparison by Deployment, 2026-2036
- Figure 72: Western Europe Market Attractiveness Analysis by Deployment
- Figure 73: Western Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 74: Western Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 75: Western Europe Market Attractiveness Analysis by End User
- Figure 76: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 77: Eastern Europe Market Value Share and BPS Analysis by Equipment Type , 2026 and 2036
- Figure 78: Eastern Europe Market Y-o-Y Growth Comparison by Equipment Type , 2026-2036
- Figure 79: Eastern Europe Market Attractiveness Analysis by Equipment Type
- Figure 80: Eastern Europe Market Value Share and BPS Analysis by Implant Type, 2026 and 2036
- Figure 81: Eastern Europe Market Y-o-Y Growth Comparison by Implant Type, 2026-2036
- Figure 82: Eastern Europe Market Attractiveness Analysis by Implant Type
- Figure 83: Eastern Europe Market Value Share and BPS Analysis by Test Function, 2026 and 2036
- Figure 84: Eastern Europe Market Y-o-Y Growth Comparison by Test Function, 2026-2036
- Figure 85: Eastern Europe Market Attractiveness Analysis by Test Function
- Figure 86: Eastern Europe Market Value Share and BPS Analysis by Deployment, 2026 and 2036
- Figure 87: Eastern Europe Market Y-o-Y Growth Comparison by Deployment, 2026-2036
- Figure 88: Eastern Europe Market Attractiveness Analysis by Deployment
- Figure 89: Eastern Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 90: Eastern Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 91: Eastern Europe Market Attractiveness Analysis by End User
- Figure 92: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 93: East Asia Market Value Share and BPS Analysis by Equipment Type , 2026 and 2036
- Figure 94: East Asia Market Y-o-Y Growth Comparison by Equipment Type , 2026-2036
- Figure 95: East Asia Market Attractiveness Analysis by Equipment Type
- Figure 96: East Asia Market Value Share and BPS Analysis by Implant Type, 2026 and 2036
- Figure 97: East Asia Market Y-o-Y Growth Comparison by Implant Type, 2026-2036
- Figure 98: East Asia Market Attractiveness Analysis by Implant Type
- Figure 99: East Asia Market Value Share and BPS Analysis by Test Function, 2026 and 2036
- Figure 100: East Asia Market Y-o-Y Growth Comparison by Test Function, 2026-2036
- Figure 101: East Asia Market Attractiveness Analysis by Test Function
- Figure 102: East Asia Market Value Share and BPS Analysis by Deployment, 2026 and 2036
- Figure 103: East Asia Market Y-o-Y Growth Comparison by Deployment, 2026-2036
- Figure 104: East Asia Market Attractiveness Analysis by Deployment
- Figure 105: East Asia Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 106: East Asia Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 107: East Asia Market Attractiveness Analysis by End User
- Figure 108: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 109: South Asia and Pacific Market Value Share and BPS Analysis by Equipment Type , 2026 and 2036
- Figure 110: South Asia and Pacific Market Y-o-Y Growth Comparison by Equipment Type , 2026-2036
- Figure 111: South Asia and Pacific Market Attractiveness Analysis by Equipment Type
- Figure 112: South Asia and Pacific Market Value Share and BPS Analysis by Implant Type, 2026 and 2036
- Figure 113: South Asia and Pacific Market Y-o-Y Growth Comparison by Implant Type, 2026-2036
- Figure 114: South Asia and Pacific Market Attractiveness Analysis by Implant Type
- Figure 115: South Asia and Pacific Market Value Share and BPS Analysis by Test Function, 2026 and 2036
- Figure 116: South Asia and Pacific Market Y-o-Y Growth Comparison by Test Function, 2026-2036
- Figure 117: South Asia and Pacific Market Attractiveness Analysis by Test Function
- Figure 118: South Asia and Pacific Market Value Share and BPS Analysis by Deployment, 2026 and 2036
- Figure 119: South Asia and Pacific Market Y-o-Y Growth Comparison by Deployment, 2026-2036
- Figure 120: South Asia and Pacific Market Attractiveness Analysis by Deployment
- Figure 121: South Asia and Pacific Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 122: South Asia and Pacific Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 123: South Asia and Pacific Market Attractiveness Analysis by End User
- Figure 124: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 125: Middle East & Africa Market Value Share and BPS Analysis by Equipment Type , 2026 and 2036
- Figure 126: Middle East & Africa Market Y-o-Y Growth Comparison by Equipment Type , 2026-2036
- Figure 127: Middle East & Africa Market Attractiveness Analysis by Equipment Type
- Figure 128: Middle East & Africa Market Value Share and BPS Analysis by Implant Type, 2026 and 2036
- Figure 129: Middle East & Africa Market Y-o-Y Growth Comparison by Implant Type, 2026-2036
- Figure 130: Middle East & Africa Market Attractiveness Analysis by Implant Type
- Figure 131: Middle East & Africa Market Value Share and BPS Analysis by Test Function, 2026 and 2036
- Figure 132: Middle East & Africa Market Y-o-Y Growth Comparison by Test Function, 2026-2036
- Figure 133: Middle East & Africa Market Attractiveness Analysis by Test Function
- Figure 134: Middle East & Africa Market Value Share and BPS Analysis by Deployment, 2026 and 2036
- Figure 135: Middle East & Africa Market Y-o-Y Growth Comparison by Deployment, 2026-2036
- Figure 136: Middle East & Africa Market Attractiveness Analysis by Deployment
- Figure 137: Middle East & Africa Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 138: Middle East & Africa Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 139: Middle East & Africa Market Attractiveness Analysis by End User
- Figure 140: Global Market - Tier Structure Analysis
- Figure 141: Global Market - Company Share Analysis

