For product managers, biophotonics is best understood as a pipeline rather than a buzzword. At the front end you choose a light source and wavelength regime, for example near infrared fluorescence, broadband low coherence light for optical coherence tomography, or lasers for Raman spectroscopy. That choice encodes depth, resolution, penetration and safety constraints.
The light then interacts with tissue through absorption, scattering, fluorescence or nonlinear effects. That interaction determines what contrast mechanism you are actually selling: structure, function, molecular composition or blood flow. Downstream of that, optics and detectors define how much of that contrast you capture and at what speed. The final layers are computational: image reconstruction, registration, analytics and decision support
From a product lens, risk and cost cluster in three places. First, in the light source and safety regime, because this drives hardware cost and regulatory classification. Second, in the reconstruction and analytics stack, because this is where false positives, false negatives and latency live. Third, in the user interface and workflow integration layer, which decides whether clinicians adopt or bypass the system. When you map your roadmap, you want clear choices about which layer is your differentiator and which layers you deliberately commoditise or buy.
In diagnostics and non invasive imaging, there are already workhorse biophotonics platforms. Optical coherence tomography is embedded in ophthalmology worldwide for cross sectional imaging of the retina and choroid, allowing early detection and monitoring of macular disease and glaucoma. It has expanded into cardiology, dermatology and dentistry, with ongoing development of handheld and intraoperative probes.
For a product manager, OCT is less an experimental technology and more a mature platform with questions like which clinical micro indication you pursue, whether you differentiate through probe design, software or connectivity, and how you handle data standards and integration with existing imaging archives. In practice, your competitive edge often comes from ergonomics, automation and analytics rather than the core interferometer
On the molecular and functional side, fluorescence imaging, Raman spectroscopy and related modalities are used for cancer detection, metabolic imaging and margin assessment in research and early translational settings. Biophotonics for cancer diagnostics leverages the fact that light at specific wavelengths is sensitive to biochemical and structural changes long before gross anatomical changes appear, so you can position products as tools for earlier, less invasive diagnosis or treatment monitoring. The trade off is that these systems often require contrast agents, complex interpretation or high quality motion control, which complicates both regulation and workflow integration.
The field guide implication is simple. Whenever you hear a biophotonics proposal for diagnostics, force the team to write down three things: the exact clinical decision that should change, the existing gold standard they are competing with or complementing, and the minimal data package a hospital or lab director would need to trust that decision. Without that, you may ship an impressive device that remains stuck in pilot mode.
In surgery, biophotonics mostly shows up as an extra layer of vision. Fluorescence guided surgery using agents such as indocyanine green or targeted probes is used to visualise perfusion, lymphatics or tumour margins in real time and can be deployed in open, laparoscopic and robotic procedures. Reviews highlight improved detection of residual disease and more precise resections in several oncology indications, but also flag variability in image interpretation and signal to noise.
From a product standpoint, your constraints are brutal. Surgeons need wide fields of view, stable images, minimal extra hardware in already crowded operating rooms and extremely low latency. Training time must be short and images have to map clearly onto actionable steps such as where to cut, where not to cut, and when to stop. Systems that demand constant manual tuning, complex interpretation or separate displays tend to be used only by champions, not by the wider surgical team.
Emerging biophotonics systems for surgery also combine multiple modalities and artificial intelligence, for example fluorescence with preoperative CT or MRI, or Raman imaging with automated tissue classification. That creates a powerful value story but compounds integration complexity, cybersecurity surface area and clinical validation burden. As a product manager you need to decide whether you are building a stand alone surgical adjunct, an integrated module for existing endoscopy or robotics platforms, or a full stack guidance ecosystem with long sales cycles and deep partnership needs.
Sources

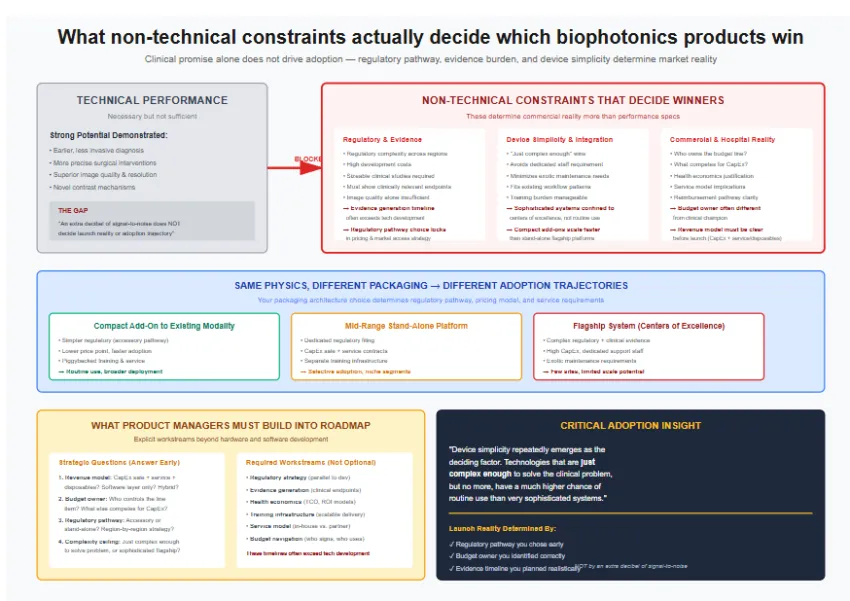
Any system that uses light to generate structural, functional, or molecular contrast: OCT, fluorescence imaging, hyperspectral systems, Raman, photoacoustic platforms, and fluorescence-guided surgery. These extract biological signatures that conventional cameras cannot.
Workflow disruption and evidence gaps dominate. Physicians worry about OR time and training; administrators need clear clinical and economic proof. Integration with navigation, PACS, and analytics is often harder than the optics themselves.
Go where optical contrast is strong and clinical need is obvious: oncologic margin assessment, dermatology triage, ophthalmology, and targeted neuro/breast imaging. These areas already show strong evidence momentum.
Expect Class II/III pathways, multi-center performance studies, human-factors validation, and health-economic evidence. You’re not selling hardware; you’re selling an intervention that alters diagnostic or surgical decision-making.
The Recycling of Automotive Glass and Laminated Windscreens in EU Industry is segmented by Source Stream (ELV Glass, Replacement Glass, OEM Scrap, Dismantled Windscreens, Side and Rear Glass), Glass Type (Laminated Glass, Tempered Glass, Mixed Glass, Windscreen Glass, Side and Rear Glass), Recycling Process (Mechanical Separation, Thermal Delamination, Chemical Recovery, Crushing and Cleaning, PVB Separation), Recovered Output (Glass Cullet, Recycled PVB, Mixed Fines, Glass Powder, Composite Residue), End Application (Flat Glass, Insulation Wool, Container Glass, Polymer Compounds, Construction Fill), Vehicle Type (Passenger Cars, SUVs, Light Commercial Vehicles, Heavy Commercial Vehicles, Buses), End User (Flat Glass Manufacturers, Insulation Producers, Polymer Processors, Aggregates Processors, Specialty Recyclers), Collection Channel (Dismantlers, Replacement Glass Networks, Authorized Treatment Facilities, Scrap Yards, OEM Plants), and Region. Forecast for 2026 to 2036.
The Recycled Technical Textiles for EU Automotive Acoustic Insulation Industry is segmented by Material Type (rPET, Blended Fibers, PP Fibers, Cotton Shoddy, Bio Blends), Feedstock Source (Post-consumer, Pre-consumer, ELV Textiles, Mixed Waste, Bottle Flake), Product Form (Felts, Mats, Pads, Molded Parts, Composites), Application (Floor Insulators, Wheel Liners, Headliners, Trunk Liners, Dash Insulators, Door Panels, Hood Liners), Vehicle Type (Passenger Cars, SUVs, LCVs, BEVs, Hybrids), Manufacturing Process (Needlepunch, Thermobonding, Airlaid, Compression Molding, Lamination), and Region. Forecast for 2026 to 2036.
The High-Brix Fruit Quality Enhancement Fertilizer Market is segmented by Nutrient Focus (Potassium-Rich, Calcium-Rich, Magnesium-Rich, Micronutrient Blends, Carbon-Based Inputs, Amino and Seaweed Inputs), Form (Water-Soluble Powders, Liquid Concentrates, Suspension Blends, Soluble Granules), Delivery Method (Fertigation, Foliar Spray, Soil Drench), Fruit Type (Grapes, Citrus, Berries, Pome Fruits, Stone Fruits, Tropical Fruits), Farm Type (Open-Field Orchards, Protected Cultivation, Export Orchards), and Region. Forecast for 2026 to 2036.
The Urban Rooftop Farm-Safe Pest Management Chemistry Market is segmented by Chemistry Type (Microbials, Botanicals, Minerals), Application Method (Foliar Sprays, Root Drenches, Spot Treatment), Crop Type (Leafy Greens, Fruiting Crops, Herbs), and Region. Forecast for 2026 to 2036.
The Multi-Residue Detoxifying Field Wash Aid Market is segmented by Chemistry Type (Surfactant Blends, Peracetic Acid, Enzyme Blends, Organic Acids, Oxidizing Blends, Mixed Detergents), Function (Residue Removal, Soil Lift, Wax Removal, Surface Sanitizing, Water Conditioning, Biofilm Control), Application Stage (Packhouse Wash, Drench, Brush Wash, Flume Wash, Final Rinse, Dip Tank), Crop Type (Citrus, Apples, Grapes, Berries, Leafy Greens, Tomatoes, Tropical Fruit), Form (Liquid Concentrate, Foam, Powder, Ready-to-Use), End User (Export Packhouses, Grower-Packers, Cooperatives, Fresh-Cut Plants, Contract Packers, Retail Prep Units), Positioning (Conventional, Organic-Compliant, Low-Foam, Low-Residue, Chlorine-Free, No-Rinse), and Region. Forecast for 2026 to 2036.