Telepresence robots are moving beyond showcase deployments. Hospitals are now evaluating them as part of routine specialist access, remote rounding, and coverage planning.
That change depends on more than hardware. It reflects a mix of reimbursement clarity, clinician shortages, and the practical value of mobile bedside access in facilities that cannot place every specialist on site.
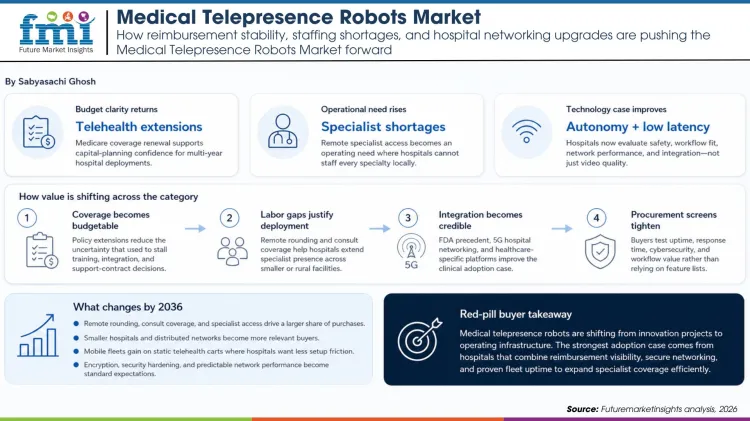
Telehealth reimbursement disruptions in 2025 showed exactly how sensitive the market can be to coverage policy. The later extension and two-year renewal restored enough certainty for health systems to think in capital-planning terms again, which is essential for robots that need network integration, training, and support contracts.
Staffing pressure is the other anchor. AAMC shortage projections make it clear that many hospitals will struggle to staff every specialty locally, especially in smaller or rural settings. Telepresence robots help close that gap by putting specialist presence where the labor market cannot.
FDA precedent for autonomous telepresence in hospitals and newer 5G medical networking trials both improve the technology case. Buyers are no longer evaluating only whether a robot can move and stream video. They are evaluating whether it can do so safely, with acceptable latency, inside real clinical workflows.
The supplier landscape is also becoming more purposeful. Healthcare-focused platforms, product repositioning, and virtual-care stack integration have made the category easier to buy as part of a service model. That reduces the friction hospitals used to face when they had to stitch together software, hardware, and support from multiple partners.
| Market dimension | 2015-2025 | 2026-2036 |
|---|---|---|
| Primary role | Pilot programs and novelty deployments were common. | Remote rounding, consult coverage, and specialist access drive more purchases. |
| Policy backdrop | Reimbursement uncertainty slowed larger rollouts. | More stable coverage improves capital planning confidence. |
| Technology baseline | Mobility and video quality were the main selling points. | Autonomy, latency, security, and uptime become core buying criteria. |
| Buyer type | Large systems led most deployments. | Smaller hospitals and distributed networks become more relevant buyers. |
| Vendor expectation | Hardware differentiation carried more weight. | Hospitals prefer integrated support, workflow fit, and security assurance. |
Even after recent renewals, coverage remains something buyers must watch carefully. A time-bound reimbursement framework changes the way CFOs evaluate long-lived equipment, especially if a business case assumes broad virtual visit monetization.
Operational evidence matters just as much. Hospitals should ask for documented consult response times, fleet uptime, cybersecurity controls, and examples of transfer avoidance or specialist coverage improvement. In this market, unreliable deployment data can undo a strong hardware pitch very quickly.
The category is likely to tilt further toward self-navigating or easier-to-deploy mobile systems because hospitals are trying to save clinician time, not create more setup work. That makes bedside availability and movement efficiency more important than flashy hardware design.
Security and network quality will also become table stakes. Hospitals that invest in local wireless upgrades will expect vendors to show predictable QoS behavior, strong encryption, and evidence of continuous security maintenance. For the full market outlook through 2036, see the Future Market Insights report: Medical Telepresence Robots Market (2026 - 2036) - https://www.futuremarketinsights.com/reports/medical-telepresence-robots-market
Growth in the Medical Telepresence Robots Market is being supported by stronger end-user demand, operational efficiency needs, regulatory pressure, and wider adoption across relevant commercial and industrial applications.
High upfront costs, validation requirements, supply chain constraints, pricing pressure, and slower adoption among cost-sensitive buyers can restrict expansion in the Medical Telepresence Robots Market.
Demand typically comes from manufacturers, service providers, healthcare or industrial operators, distributors, and specialized buyers that need reliable performance, compliance, and cost efficiency.
Regulations are pushing suppliers toward safer materials, better documentation, stronger quality controls, and products that help customers meet environmental, safety, or performance standards.
Companies should track raw material costs, technology upgrades, customer purchasing cycles, regional policy changes, and competitive moves that can alter pricing and adoption rates.
This report covers the coronary heart disease diagnostic imaging market through analysis of market size, revenue forecast, competitive landscape, demand outlook, growth drivers, restraints, imaging pathway shifts, supply-side developments, and strategic growth opportunities.
This report covers the mRNA vaccine formulation stability and stress test systems market through analysis of market size, market share, revenue forecast, system pricing benchmarks, company positioning, competitive landscape, demand outlook, growth drivers, restraints, technology trends, formulation testing requirements, assay integration, analytical method continuity, end-user adoption, supply chain developments, regulatory and quality expectations, investment opportunities, profitability outlook, and strategic growth opportunities during 2026 to 2036.
This report covers the cell therapy cryopreservation and thaw performance test platforms market through analysis of market size, market share, revenue forecast, platform segmentation, assay readout trends, cell type demand, preservation format usage, automation level adoption, end-user demand, workflow stage requirements, regional outlook, technology developments, handling standardization needs, method transfer practices, comparability testing, supply chain dynamics, regulatory and quality expectations, investment opportunities, profitability outlook, and strategic growth opportunities during 2026 to 2036.
The Gene Editing Off-Target Effect Detection Test Systems Market is segmented by Detection Approach (Cell-based Assays, Biochemical Assays, In Silico Tools, Orthogonal Validation), Product Format (Services, Assay Kits, Software, Instruments, Reagents), Workflow Stage (Site Nomination, Site Confirmation, Structural Follow-up, Release Support), Editing Modality (CRISPR-Cas9, Base Editing, Prime Editing, Cas12 Systems, Other Editors), Readout Technology (Amplicon NGS, Short-read WGS, Single-cell DNA, ChIP-seq Workflows, Long-read Sequencing), End User (Biopharma Companies, Academic Laboratories, CROs, CDMOs, Hospitals), Application (IND-enabling Studies, Discovery Screening, Cell Therapy Quality Assessment, In vivo Profiling, Comparability Studies), Deployment (Outsourced Testing, In-house Testing, Hybrid Models), and Region. Forecast for 2026 to 2036.
This report covers the at-home blood collection kits and devices market through analysis of market size, revenue forecast, competitive landscape, demand outlook, growth drivers, restraints, self-collection workflow trends, regulatory developments, and strategic growth opportunities.