Western blotting remains a foundational technique for protein detection and validation across academic research, biopharmaceutical development, and clinical laboratories. Despite the emergence of alternative protein analysis platforms, including capillary electrophoresis systems, mass spectrometry workflows, and high-throughput immunoassays, Western blotting continues to serve as a confirmatory method valued for its specificity and visual validation of protein size and expression. However, the technique has long been criticised for operator-dependent variability, time-intensive manual steps, and challenges in achieving reproducible results across laboratories and analysts. Automated Western blotting processors address these limitations not by fundamentally altering the underlying immunoblotting chemistry, but by standardising execution, improving documentation, and reducing the risk of human error in regulated and high-volume environments.
Manual Western blotting is highly susceptible to operator-dependent variability at nearly every stage of the workflow. Inconsistent handling during membrane blocking, antibody incubation, and washing steps introduces significant variability in signal intensity and background noise. Timing discrepancies between replicates or across different operators can result in under- or over-developed blots, making quantitative comparisons unreliable. Reagent preparation and dilution errors further compound variability, particularly when multiple antibodies or experimental conditions are tested in parallel.
Temperature fluctuations during incubation, uneven agitation during washing, and inconsistent buffer exchange all contribute to assay drift. Even experienced laboratory personnel struggle to maintain identical conditions across multiple blots or over extended time periods. This variability becomes particularly problematic in regulated research settings where reproducibility is not only a scientific goal but a compliance requirement. When Western blots are used to support regulatory submissions, demonstrate batch-to-batch consistency of biologics, or validate manufacturing processes, manual inconsistencies translate directly into regulatory risk and delayed approvals.
Automated Western blotting processors address reproducibility concerns by enforcing standardised protocols across all critical workflow steps. These systems control incubation times, temperatures, and agitation speeds with precision that manual execution cannot match. Automated liquid handling ensures consistent delivery of blocking buffers, primary and secondary antibodies, and wash solutions, eliminating pipetting errors and reagent carryover between steps.
Processors typically include integrated shaking or rocking platforms that maintain uniform membrane exposure to reagents, reducing edge effects and uneven antibody binding. Programmable washing cycles deliver precise buffer volumes and exchange rates, ensuring thorough removal of unbound antibodies and minimising background signal. Many systems incorporate temperature-controlled chambers that maintain optimal conditions for antibody-antigen interactions, further reducing assay-to-assay variability.
Critically, these processors enforce protocol adherence by removing opportunities for operator deviation. Once a validated method is programmed, every run follows identical parameters regardless of who initiates the process or when the assay is performed. This standardisation is particularly valuable in laboratories with high staff turnover, multiple shifts, or distributed sites where maintaining consistent manual technique is logistically challenging.
Biopharmaceutical development and core facility operations increasingly demand higher throughput and consistent batch processing capabilities. During biologics development, Western blotting is used to confirm protein expression, assess post-translational modifications, and validate product quality across multiple manufacturing runs. Manual processing becomes a bottleneck when dozens or hundreds of samples require analysis within compressed timelines.
Automated processors enable parallel processing of multiple blots under identical conditions, significantly increasing laboratory capacity without proportional increases in staffing. This parallel capability is essential in core facilities serving multiple research groups, where demand fluctuates and turnaround expectations are high. Processors allow facilities to batch similar samples, optimise instrument utilisation, and maintain consistent quality across diverse projects.
Throughput gains extend beyond simple speed improvements. By reducing the hands-on time required per blot, processors free skilled personnel to focus on experimental design, data interpretation, and method development rather than repetitive manual tasks. This shift in labour allocation improves overall laboratory productivity and enables more sophisticated experimental approaches that would be impractical with manual execution.
Regulatory environments governed by Good Laboratory Practice and Good Manufacturing Practice principles impose strict documentation and traceability requirements on all analytical methods. Manual Western blotting presents significant challenges in meeting these standards, as critical process parameters such as incubation times, temperatures, and reagent lot numbers must be meticulously recorded and verified.
Automated processors generate comprehensive electronic records of every run, including timestamps, reagent tracking, temperature logs, and protocol parameters. This digital documentation eliminates transcription errors, provides audit-ready records, and supports data integrity requirements mandated by regulatory agencies. Many systems integrate with laboratory information management systems, enabling seamless data transfer and centralised record keeping.
Traceability extends to reagent management, where processors can track antibody lot numbers, expiration dates, and usage history, ensuring that every Western blot can be traced to specific reagent batches if questions arise during regulatory review. This level of documentation is nearly impossible to maintain consistently with manual processes, particularly in high-volume laboratories where dozens of blots are processed weekly.
Audit readiness becomes a driver of automation adoption in regulated laboratories, where inspection findings related to inadequate documentation or uncontrolled processes can result in costly delays, repeat analyses, or regulatory holds on product development programs.
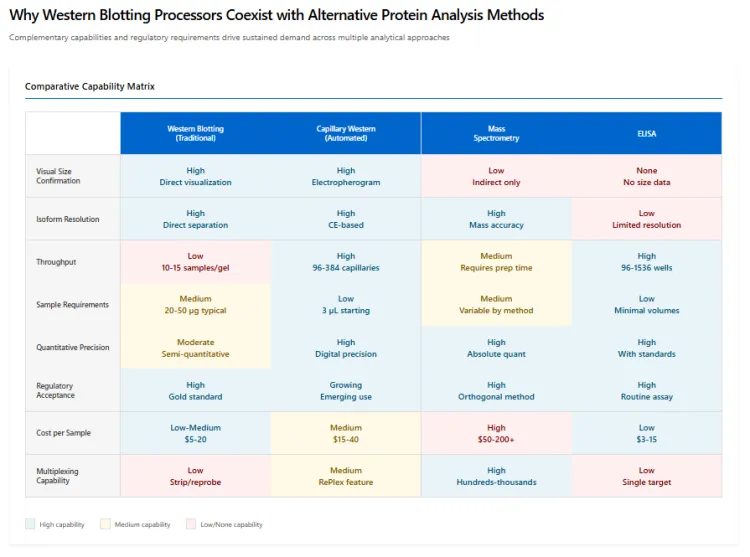
Despite advances in alternative protein analysis technologies, Western blotting processors continue to find adoption because the underlying technique serves a distinct confirmatory role that complementary methods cannot entirely replace. Mass spectrometry provides superior sensitivity and multiplexing capability but lacks the visual confirmation of protein size that Western blotting delivers. Enzyme-linked immunosorbent assays offer higher throughput but do not resolve protein isoforms or confirm antibody specificity as directly.
Capillary-based automated Western blotting systems, while offering improved reproducibility and reduced sample requirements, are often deployed alongside traditional blotting processors rather than replacing them entirely. Laboratories frequently require both approaches: capillary systems for high-throughput screening and quantitative analysis, and traditional processors for confirmatory work, large-format gels, and applications requiring specific membrane types or detection chemistries.
Regulatory agencies often expect Western blotting data as confirmatory evidence even when alternative methods are used for routine analysis. This expectation reinforces continued investment in Western blotting infrastructure and drives demand for processors that can deliver the reproducibility and documentation quality required in regulated submissions.

Bibliography
Automated processors control workflow execution but do not eliminate biological sample variability, antibody quality issues, or pre-analytical factors such as protein extraction efficiency and sample degradation. Processors improve technical reproducibility but cannot compensate for poor experimental design, inappropriate antibody selection, or samples that fall outside optimal assay ranges.
Washing steps, incubation timing, and reagent delivery show the greatest improvement from automation. These steps are highly sensitive to operator variability and directly impact signal-to-noise ratios. Temperature control during incubation and consistent agitation also provide significant reproducibility gains that are difficult to achieve manually.
Processors often reduce antibody consumption through precise liquid handling and optimised incubation chamber designs that minimise required reagent volumes. However, initial capital costs and maintenance expenses must be weighed against reagent savings. Cost-effectiveness is typically achieved in laboratories processing sufficient volumes to justify the investment.
Small laboratories must carefully evaluate whether throughput and reproducibility demands justify processor investment. Benchtop models with lower capital costs exist, but laboratories with infrequent Western blotting needs may find manual methods more cost-effective. Shared core facility access to processors often provides better economics for low-volume users.
Processors provide electronic batch records, audit trails, and documentation of critical process parameters that inspectors expect in regulated environments. Systems with 21 CFR Part 11 compliant software features support data integrity requirements and demonstrate process control. This documentation significantly reduces inspection findings related to analytical method validation and execution.
Western Blotting Market is segmented by product, application and end user from 2025 to 2035
Western Europe Automotive Performance Tuning & Engine Remapping Market Size and Share Forecast Outlook 2025 to 2035
Western Europe Valve Seat Insert Market Size and Share Forecast Outlook 2025 to 2035
Western Europe Automated People Mover Industry Size and Share Forecast Outlook 2025 to 2035
Western Europe Bicycle Component Aftermarket Analysis Size and Share Forecast Outlook 2025 to 2035