Addison Disease Testing Market
The Addison Disease Testing Market Is Segmented by Test Type, and End User from 2025 To 2035
Historical Data Covered: 2015 to 2023 | Base Year: 2024 | Estimated Year: 2025 | Forecast Period: 2026 to 2035
Addison Disease Testing Market Outlook 2025 to 2035
The Addison disease testing market is expected to reach approximately USD 267.6 million in 2025 and expand to around USD 374.6 million by 2035, reflecting a compound annual growth rate (CAGR) of 3.5% over the forecast period.
The Addison disease testing market is ready to grow significantly from 2025 to 2035, with increased awareness of endocrine disorders, improved diagnostics, and awareness on the part of physicians of rare adrenal insufficiencies being major drivers.
Addison's disease or primary adrenal insufficiency is a rare condition, clinically life-threatening, that needs to be carefully diagnosed to avoid even more life-threatening complications.
Depending on the clinical setting, the main diagnosis includes ACTH stimulation tests, cortisol levels, the detection of autoantibodies, and imaging of the adrenal glands.
With the rising incidence of autoimmune conditions, improved laboratory access, point-of-care capabilities in developed regions, improved clinical understanding, and integration in electronic health records which improved trends in referrals and screenings, detection rates have been improving.
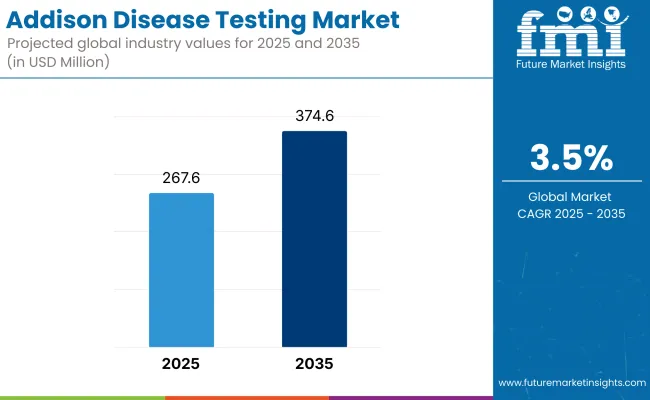
Key Market Metrics
| Metric | Value (USD Million) |
|---|---|
| Industry Size (2025E) | USD 267.6 Million |
| Industry Value (2035F) | USD 374.6 Million |
| CAGR (2025 to 2035) | 3.5% |
Over the past decades, the ever-evolving Addison disease testing market has been buoyed through novel potentials in diagnostics, increasing knowledge on adrenal insufficiency, and improvement of healthcare infrastructure. Traditionally, diagnosis was based on symptomatology, thus resulting in underdiagnosis and often delayed treatment.
The picture changed when hormone assays-such as serum cortisol and ACTH stimulation tests-were introduced in clinical practice for earliest and accurate detection. Development over years of diagnostic accuracy through autoantibody-based tests and adrenal hormone panels has further enhanced diagnostic precision for autoimmune Addison disease.
Moreover, awareness and screening programs have also enhanced growth, particularly in developed regions. On the contrary, significant impediments include the high costs of diagnosis, limited access in emerging economies, and variations in testing protocols hindering market growth.
Regional Market Trends
North America
North America is leading the Addison disease testing market as a result of its strong diagnostic infrastructure, the awareness among endocrinologists, and wide insurance coverage for endocrine testing. The USA dominates these regional growth avenues, underpinned by a solid research network and well-defined guidelines by the Endocrine Society.
ACTH stimulation tests and cortisol assays are performed in an outpatient and hospital settings, while newer advances in immunoassays improve the sensitivity of tests. Canada employs tele-endocrinology programs and regional centers for referral to improve diagnostic capacity in rural and underserved areas. Further, this region has early adoption of digital instruments for endocrine symptom tracking and patient engagement, thereby attaining end-user advantage.
Europe
Europe maintains a strong presence in the Addison disease testing market, bolstered by universal healthcare access, standardized endocrine care pathways, and centralized laboratory services. Countries such as Germany, the UK, France, and the Nordics have well-developed endocrinology networks and are early adopters of autoantibody and ACTH diagnostics.
EU-funded rare disease initiatives and registries support increased awareness and test accessibility. Collaboration between hospitals and research institutions is promoting the development of next-generation cortisol and adrenal antibody assays. The region’s emphasis on early diagnosis and chronic disease management aligns well with market expansion.
Asia-Pacific
Asia-Pacific is anticipated to be the fastest-growing region, driven by expanding healthcare infrastructure, improved laboratory capabilities, and rising detection of autoimmune and endocrine disorders. Japan and South Korea are advanced markets with strong specialty care systems and research in adrenal pathology.
China and India are rapidly scaling up diagnostic capacity, with improved clinician training and increasing public health focus on underdiagnosed rare diseases. Government initiatives promoting rural diagnostics, electronic health systems, and health screening programs are supporting market penetration. Despite current gaps in awareness and test availability, Asia-Pacific offers strong long-term potential as access and endocrinology care improve.
Challenges and Opportunities
Differences in Diagnostic Practices and Protocols Across Regions Lead to Inconsistent Testing Accuracy, Impacting Market Adoption
Access barriers faced by people in rural or underserved regions are some of the leading concerns regarding Addison disease testing market growth, aside from delayed diagnosis owing to nonspecific symptoms and lack of disease awareness in primary care settings. Dynamic testing (such as ACTH stimulation) may also be logistically challenging, can be time-sensitive, and requires trained personnel and lab support.
Reimbursement constraints for autoimmune antibody testing and the lack of standardization between assays used in different areas are contributing factors to the slower adoption. Moreover, Addison’s disease is so rare that there is little incentive for commercial players to invest in large-scale diagnostic production.
The Introduction of Portable and Home-Based Diagnostic Kits Can Improve Patient Convenience and Drive Market Growth
There are opportunities to develop rapid and minimally invasive cortisol testing solutions such as salivary or point-of-care assays that facilitate early detection and remote monitoring. Early suspicion and referral rates can be improved by education campaigns aimed at general practitioners and emergency physicians.
Testing for Addison’s disease within autoimmune disease panels or through routine endocrine testing may increase case-finding. Joint ventures between academic medical centers and diagnostic companies are pushing research to expand biomarker testing. Digital health platforms and symptom checkers are emerging as tools to channel patient awareness and to self-refer for endocrine testing.
Industry-Specific Highlights
ACTH Stimulation Test Innovation
The synthetic ACTH formulations and simplified testing protocols have made the ACTH stimulation test more efficient and widely available. Advancements in synthetic ACTH that render them more stable and less expensive have enabled hundreds of such procedures to be performed more easily in outpatient and urgent care settings.
Moreover, simplified testing protocols lower the winding of the process, leading to reduced time for diagnosis. These innovations allow for faster results, increasing the patient experience and allowing the test to be more widely available to all types of patients. As a result, patients with Addison's disease not only receive advanced treatments but also gain greater access to appropriate monitoring in non-hospital settings.
Emerging Trends
Autoantibody-Based Diagnostics
Autoantibody-based diagnostics, specifically adrenal cortex antibody (ACA) and 21-hydroxylase antibody testing are increasingly valuable for the diagnosis of autoimmuneAddison’sdisease, particularly in patients with polyendocrine syndromes. These will demonstrate the presence of the specific antibodies linked to the autoimmune destruction of the adrenal cortex, which is the hallmark of autoimmune Addison’s disease.
These laboratory tests provide a greater diagnostic opportunity for PE in ambiguous clinical presentations or overlap with other endocrine disorders. Increased awareness and diagnostic capabilities of these tests are providing a useful tool to help identify patients who may be at risk for autoimmune adrenal insufficiency.
Brief Market Overview
From 2020 to 2024, the Addison's disease testing market was gradual owing to lingering awareness of adrenal insufficiency and advancement in test technologies. The Addison's disease testing market showed significant improvement led by increasing incidences of autoimmune disease, new testing technologies, including ACTH stimulation and imaging studies, and a growing healthcare sector. However, continuing challenges related to high costs in testing, low insurance availability, and variation in testing between areas limited the expansion of the market.
Between 2025 and 2035, the Addison's disease testing market will likely accelerate as new technologies come onboard, investment grows in rare disease testing, and focus on early and accurate testing develops. New testing systems will come to market in several forms, including home testing kits, next-gen technologies, and rapid tests incorporating AI diagnostic algorithms. The Addison's disease testing market will grow, as more access in healthcare for patients supports testing, and as healthcare provider and patient education continues to improve around Addison's disease testing.
Market Shifts: A Comparative Analysis 2020 to 2024 vs. 2025 to 2035
| Market Aspect | 2020 to 2024 |
|---|---|
| Regulatory Landscape | The guidelines were implemented to ensure the safety and efficacy of diagnostic tests and thereby facilitate standard protocols and increased surveillance. |
| Technological Advancements | Advanced diagnostic tests ACTH stimulation test, cortisol and aldosterone blood tests, and image studies find better diagnostic accuracy and are also time-efficient. |
| Consumer Demand | Healthcare providers and patients are increasingly embracing diagnostic tests, primarily because Addison's disease needs an accurate and timely diagnosis for treatment and management. |
| Market Growth Drivers | Increase in the number of autoimmune diseases, modernization in the field of diagnostic technology, and an emphasis on greater accuracy to improve patient outcomes. |
| Sustainability | Initial steps have been made to promote sustainable practices in the manufacture of diagnostic devices with a greater focus on environmentally aware materials and energy-efficient manufacturing techniques. |
| Supply Chain Dynamics | The suppliers have to specialize to guarantee the supply of high-reliability reagents and a set of diagnostic equipment that, however, under consideration localizing of production to avoid disruptions to supply chains demonstrated by the globe in recent times. |
| Market Aspect | 2025 to 2035 |
|---|---|
| Regulatory Landscape | A constant monitoring and evolving of regulations across the various countries to achieve the right balance between patient safety and technology innovation, alongside expedited approvals for new diagnostic technology filling the unmet medical need. |
| Technological Advancements | The development of home diagnostic kits and integration of artificial intelligence into diagnostic algorithms increases availability and minimizes the time for diagnosis while enabling a personalized system of diagnostics. |
| Consumer Demand | Due to technologic advancements across various patient populations embracing non-invasive and rapid diagnostic avenues with patient-centered care in mind are the driving forces behind their adoption. |
| Market Growth Drivers | Expanding healthcare across emerging markets, increased investment into rare disease R&D, continuous technology innovation that enhances efficacy of diagnostics, and a worldwide focus on early detection and personalized approaches to medicine. |
| Sustainability | Following sustainable manufacturing processes and creating biodegradable parts will go hand in hand with global sustainability initiatives for a reduction in the environmental impact footprint of diagnostic devices. |
| Supply Chain Dynamics | Increases in local manufacturing capabilities through technology and partnership would cut dependence on imports, improve supply chain resilience and meet the rapidly emerging needs in the diagnosis of Addison's disease-testing. |
Country-wise Outlook
United States
Market Outlook
The Addison’s disease testing market in the United States is expected to expand due to well-established healthcare system and well-versed knowledge about autoimmune diseases. The increasing incidence of autoimmune diseases and endocrine disorders, in addition to technological advancements in diagnostic procedures, supports market growth.
The presence of several diagnostic methods, including ACTH stimulation tests and autoantibody-based diagnostics, also aids in the growth of the market. But barriers such as expensive medical care and restrictions on insurance coverage for specialized tests may block wider uptake. Upcoming growth in the market is estimated to be driven by government initiatives to encourage rare disease research and enhance diagnostic accuracy.
Market Growth Factors
- High Awareness and Clinical Suspicion of Adrenal Disorders: Especially in autoimmune populations.
- Availability of Cortisol and ACTH Testing in Primary and Tertiary Settings: Improves early detection.
- Advanced Laboratory Infrastructure and Diagnostic Reimbursement: Drives high testing volumes.
- Increased Screening in Autoimmune Polyglandular Syndromes (APS): Boosts Addison disease testing uptake.
- Growing Demand for ACTH Stimulation and Salivary Cortisol Tests: Preferred for accurate adrenal evaluation.
Market Forecast
| Country | CAGR (2025 to 2035) |
|---|---|
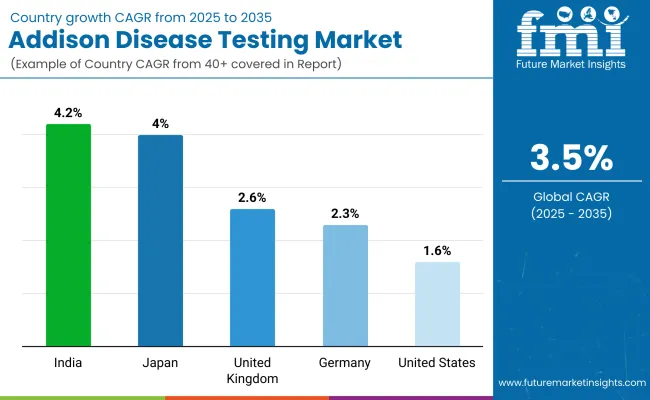
| United States | 1.6% |
Germany
Market Outlook
Germany’s Addison’s disease testing market is backed by a strong healthcare system providing widespread access to advanced diagnostic methods. Rising awareness regarding the autoimmune diseases along with increasing focus on early detection is boosting the market growth.
Germany is uniquely poised to take pits and create new diagnostic tools for Addison’s disease with well-established protocols (ACTH stimulation test) and for autoantibody-based diagnostics (Cohen et al. 2023). In addition, AI-powered diagnostics and next-gen testing solutions are likely to make testing more efficient. However, it is likely that regional differences in healthcare access, coupled with regulatory barriers and the high cost associated with testing, will restrict market growth in some areas.
Market Growth Factors
- Advanced Clinical Protocols for Suspected Adrenal Insufficiency: Standardized across university and community hospitals.
- High Adoption of ELISA and Chemiluminescence Assays: Ensures accuracy in hormone quantification.
- Integration of Addison Disease Screening into Autoimmune Diagnostic Panels: Improves early detection.
- Prevalence of Congenital Adrenal Hyperplasia and APS Cases: Drives pediatric and adult testing.
- Government Coverage for Endocrine Testing Under Public Health Insurance: Ensures accessibility.
Market Forecast
| Country | CAGR (2025 to 2035) |
|---|---|
| Germany | 2.3% |
United Kingdom
Market Outlook
The market for Addison’s disease testing in the UK can be attributed to advanced healthcare infrastructure and growing emphasis on patient care. Wide access to diagnostic services in the UK through the National Health Service (NHS) has also fueled demand for testing. Increased awareness of autoimmune adrenal insufficiency, particularly within the healthcare community, has led to more patients being diagnosed with the disorder.
The increased availability of testing methods (21-hydroxylase antibody tests and synthetic ACTH) allows clinicians to make a more accurate diagnosis. Though the outlook remains promising, economic factors, like limits to the healthcare budget and hold-up of insurance coverage for specialised tests, may hinder adoption.
Market Growth Factors
- National Guidelines for Adrenal Insufficiency Testing (NICE): Standardizes diagnostic practices for consistent and accurate adrenal insufficiency diagnosis.
- Broad Use of Short Synacthen Tests (SST): Recognized as the gold standard for diagnosing Addison’s disease accurately.
- Growth in Autoimmune Disease Diagnosis: Increased co-detection of adrenal insufficiency due to rising autoimmune disease prevalence.
- Integration of Cortisol Tests in A&E and ICU Settings: Enables rapid, life-saving diagnostics for critically ill patients.
- Academic Research in Endocrinology: Advances biomarker-based diagnostics, improving accuracy and early detection of adrenal disorders.
Market Forecast
| Country | CAGR (2025 to 2035) |
|---|---|
| United Kingdom | 2.6% |
Japan
Market Outlook
Japan's Addison's disease testing market is redrawn by the country's elderly population and a rise in autoimmune diseases. Japan possesses an advanced healthcare system with broad access to diagnostic capabilities such as ACTH stimulation tests [9] and autoantibody-based diagnostics .
Nevertheless,more awareness about autoimmune adrenal insufficiency should be raised, both in the healthcare sector and among patients. Technological advancements like home-based testing kits and AI powered diagnostic algorithms, etc., would increase the accessibility ratio of testing which would contribute towards this market segment. However, the market is challenged with the high cost of novel diagnostic technology and heterogeneity in access to healthcare.
Market Growth Factors
- Strong Diagnostic Infrastructure in Endocrinology: High-quality laboratories and specialized centers ensure precise adrenal disorder diagnostics.
- Aging Population Prone to Adrenal Dysfunction: Expanding screening programs target at-risk elderly individuals for early detection.
- Widespread Use of Autoimmune Panels and Cortisol Rhythm Tests: Advanced diagnostics improve accuracy in identifying adrenal disorders.
- Government Reimbursement for Endocrine Workups: Financial support encourages comprehensive hospital-based screening for adrenal dysfunction.
- Collaborative Clinical Trials in Adrenal Disease Management: Partnerships drive innovative diagnostics and expand market reach in adrenal care.
Market Forecast
| Country | CAGR (2025 to 2035) |
|---|---|
| Japan | 4.0% |
India
Market Outlook
The Addison’s disease testing market in India is evolving with rising cognizance about autoimmune disorders and improved diagnostic tools. In both urban and rural areas, we are seeing advancements in access, however the healthcare system must still overcome challenges as it relates to infrastructure and affordability.
This increase in the volume of cases of autoimmune diseases along with the increasing emphasis on early diagnosis is driving the market. Assessing these tests for efficacy also had a high cost and insurance only covered very few specialized tests. In the long term, increasing healthcare access and the rise of relatively affordable and quality diagnostics options are anticipated to boost market growth in this country.
Market Growth Factors
- High Prevalence of TB-Associated Adrenal Insufficiency: Drives demand for ACTH and cortisol tests.
- Growth of Endocrinology Departments in Tertiary Care Hospitals: Increases diagnostic availability.
- Rising Public Awareness of Hormonal Disorders: Accelerates early testing and referrals.
- Increased Adoption of Chemiluminescence and Immunoassay Platforms: Boosting diagnostic capacity.
- Government and Private Investments in Lab Infrastructure: Driving market accessibility and accuracy.
Market Forecast
| Country | CAGR (2025 to 2035) |
|---|---|
| India | 4.2% |
Market Outlook: Addison Disease Testing Market
By Test Type
The ACTH Stimulation Test Dominates the Market Due to Its Established Reliability and Widely Accepted Clinical Use
The ACTH stimulation test is the gold standard in diagnosing Addison's disease (primary adrenal insufficiency). It evaluates adrenal response to synthetic ACTH by measuring cortisol levels pre- and post-injection. The market for ACTH testing is expected to experience growth due to rising awareness of autoimmune adrenal disorders, increasing incidence of adrenal cortex dysfunction caused by autoimmune polyendocrine syndromes, tuberculosis or cancer, and growing availability of point-of-care endocrine diagnostic services.
North America and Europe are major markets with a solid footing fueled by dedicated endocrinology centers and robust reimbursement structures. In the Asia-Pacific region, rising adoption of diagnoses with the advent of better endocrinology infrastructure and increased screening for an acute adrenal crisis in critical care environments are considering to contribute to market strength. Trends for the future are microfluidic ACTH test platforms, integration of testing with electronic health records for tracking endocrine problems, and lab-on-chip cortisol response devices.
Serum Cortisol Testing Dominates the Market Due to Its High Accuracy in Assessing Adrenal Function
An adrenal insufficiency workup usually starts with a serum cortisol test which assesses baseline cortisol levels, most commonly serum, on the 8th hour in the morning. It is less specific than ACTH stimulation, but is widely used in primary and secondary care due to its ease of use, low cost and availability. It is prevalent as the mechanism for triaging patients prior to confirmatory dynamic testing.
The growing incidence of fatigue related disorders, corticosteroid usage and adrenal suppression monitoring in patients with chronic inflammatory diseases is driving the growth. Serum cortisol testing is widely used by hospitals, diagnostic laboratories, and outpatient clinics in the USA, UK, and India. These may come in the forms of saliva based cortisol assays for non-invasive screening, wearable cortisol biosensors and AI-assisted result interpretation to assess individual diurnal cortisol patterns.
By End User
Hospitals and Specialty Endocrine Clinics Dominate the Market Due to Their Access to Advanced Diagnostic Tools
The main diagnostic facilities for Addison’s disease, in particular the testing of patients with hypotension, fatigue, electrolyte imbalance, or assumed adrenal crisis, are hospitals and endocrinology clinics. These include dynamic testing protocols (ACTH stimulation), baseline hormone testing (cortisol, aldosterone, renin), and imaging follow-ups (CT/MRI of adrenal glands).
Integrated diagnostic workflows with endocrinologist oversight, and the ability to provide requisite emergency care, make hospitals the optimal setting for testing. The volume for hospital-based testing leaders is concentrated in North America, Europe, and Gulf countries, with urban centers in the Asia-Pacific and Latin America regions building out capabilities.
Diagnostic Laboratories Dominate the Market Due to Their Extensive Testing Capabilities and Specialized Diagnostic Expertise
High-throughput Addison disease testing is available in standalone and networked diagnostic laboratories, with serum cortisol and plasma ACTH levels being particularly relevant. These labs are also collaborating with tele-endocrinologies, health screening programs, and outpatient clinics to bring hormone testing at your door step.
Everywhere from the USA to the UK to urban Asia, a push towards direct-to-consumer hormone health panels is already beginning to decentralize endocrine diagnostics. Diagnostic capabilities are being augmented by labs with chemiluminescence immunoassay (CLIA), radioimmunoassay (RIA), and LC-MS/MS platforms. Future advances involve home collection kits for hormone testing, mobile delivery of results linked to health status, and cloud-based dashboards interpreting endocrine tests.
Competitive Outlook
Awareness on adrenal insufficiency, advanced diagnostic techniques, and rise in screening of endocrine disorders are shifting the trends in Addison's disease testing market. As it often misdiagnosed and rarer disorders, quick and accurate diagnosis remains a challenge.
However, advances have been made by growing adoption of such diagnostic technologies which include especially autoantibody testing, ACTH stimulation tests and extensive adrenal hormone panels have become pivotal in improving diagnostic accuracy.
Market comprises specialized diagnostic providers, clinical laboratories and medical device companies, which offer varied diagnostic services such as hormone assay kits and immunoassay systems. Innovations lead to more precise detection and monitoring of the disease Addison's, enhancing early diagnosis and treatment outcomes.
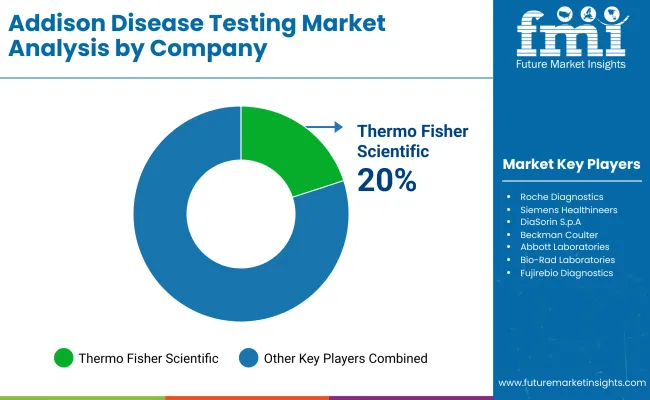
Market Share Analysis by Company
| Company Name | Estimated Market Share (%) |
|---|---|
| Thermo Fisher Scientific | 20-24% |
| Roche Diagnostics | 16-20% |
| Siemens Healthineers | 14-18% |
| DiaSorin S.p.A. | 8-12% |
| Beckman Coulter (Danaher) | 5-9% |
| Other Companies (combined) | 20-30% |
Key Company Offering Indias and Activities
| Company Name | Key Offerings/Activities |
|---|---|
| Thermo Fisher Scientific | Offers immunoassay systems and endocrine panels for ACTH, cortisol, and adrenal antibodies. |
| Roche Diagnostics | Provides automated hormone testing platforms and diagnostic reagents for adrenal insufficiency detection. |
| Siemens Healthineers | Supplies high-throughput immunoassay analyzers and ACTH hormone assays used in endocrine diagnostics. |
| DiaSorin S.p.A. | Specializes in immunodiagnostic solutions including cortisol and 21-hydroxylase antibody tests for Addison disease. |
| Beckman Coulter | Delivers clinical chemistry and immunoassay analyzers with endocrine panels for hormone testing. |
Key Company Insights
Thermo Fisher Scientific (20-24%)
Thermo Fisher offers a broad range of hormone assays and automated analyzers, providing precise diagnostics for adrenal disorders, positioning it as a leader in Addison’s disease testing.
Roche Diagnostics (16-20%)
Roche Diagnostics enhances Addison's disease testing with cutting-edge immunoassay platforms and reliable cortisol testing reagents, ensuring accurate diagnostics and supporting both clinical and research applications.
Siemens Healthineers (14-18%)
Siemens integrates Addison’s disease diagnostics into high-throughput systems, catering to large-scale testing needs in hospitals and reference laboratories, improving efficiency and accessibility for adrenal insufficiency detection.
DiaSorin S.p.A. (8-12%)
DiaSorin specializes in autoimmune-related Addison’s disease diagnostics, offering highly specific tests that support early-stage detection, enabling timely intervention and better patient outcomes.
Beckman Coulter (5-9%)
Beckman Coulter provides reliable endocrine function testing kits and lab automation solutions, facilitating the detection of adrenal insufficiencies, and improving workflow efficiency in clinical laboratories.
Other Key Players (20-30% Combined)
Additional contributors to the Addison disease testing market include:
- Abbott Laboratories
- Bio-Rad Laboratories
- Fujirebio Diagnostics
- Randox Laboratories
- Nova Biomedical
These companies enhance testing accuracy, accessibility, and automation through point-of-care tools, hormone assay innovations, and integrated lab platforms for adrenal health diagnostics.
Key Segmentation
By Test Type:
Laboratory, Comprehensive Metabolic Panel, Thyroid-Stimulating Hormone (TSH) Levels, ACTH Stimulation, Insulin-induced Hypoglycaemia, Autoantibody, Prolactin, Imaging Studies, Chest Radiograph, Computed tomography (CT) Scan and Serum Cortisol Testing.
By End User:
Hospitals, Clinics and Diagnostics Laboratories.
By Region:
North America, Latin America, Western Europe, Eastern Europe, East Asia, South Asia & Pacific, Middle East & Africa.
Frequently Asked Questions
What is the future of Addison disease testing industry?
The Addison disease testing industry is projected to witness CAGR of 3.5% between 2025 and 2035.
What was the worth of the Addison disease testing industry in 2024?
The Addison disease testing industry stood at USD 258.5 million in 2024.
What will the worth of Addison disease testing industry by 2035 end?
The Addison disease testing industry is anticipated to reach USD 374.6 million by 2035 end.
What is the expected CAGR for China during forecast period?
China is expected to show a CAGR of 4.6% in the assessment period.
Who are the key manufacturer of Addison disease testing industry?
The key players operating in the Addison disease testing industry are Thermo Fisher Scientific, Roche Diagnostics, Siemens Healthineers, DiaSorin S.p.A., Beckman Coulter, Abbott Laboratories, Bio-Rad Laboratories, Fujirebio Diagnostics, Randox Laboratories, Nova Biomedical and Others.
Table of Content
- Executive Summary
- Industry Introduction, including Taxonomy and Market Definition
- Market Trends and Success Factors, including Macro-economic Factors, Market Dynamics, and Recent Industry Developments
- Global Market Demand Analysis 2020 to 2024 and Forecast 2025 to 2035, including Historical Analysis and Future Projections
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035
- By Test Type
- By End User
- By Region
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, by Test Type
- Laboratory Testing
- Comprehensive Metabolic Panel
- Thyroid-Stimulating Hormone (TSH) Levels
- ACTH Stimulation
- Insulin-induced Hypoglycaemia
- Autoantibody
- Prolactin
- Imaging Studies
- Chest Radiograph
- Computed tomography (CT) Scan
- Serum Cortisol Testing
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, by End User
- Hospitals
- Clinics
- Diagnostics Laboratories
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, by Region
- North America
- Latin America
- East Asia
- South Asia and Pacific
- Western Europe
- Eastern Europe
- Middle East and Africa
- North America Sales Analysis 2020 to 2024 and Forecast 2025 to 2035, by Key Segments and Countries
- Latin America Sales Analysis 2020 to 2024 and Forecast 2025 to 2035, by Key Segments and Countries
- East Asia Sales Analysis 2020 to 2024 and Forecast 2025 to 2035, by Key Segments and Countries
- South Asia & Pacific Sales Analysis 2020 to 2024 and Forecast 2025 to 2035, by Key Segments and Countries
- Western Europe Sales Analysis 2020 to 2024 and Forecast 2025 to 2035, by Key Segments and Countries
- Eastern Europe Sales Analysis 2020 to 2024 and Forecast 2025 to 2035, by Key Segments and Countries
- Middle East and Africa Sales Analysis 2020 to 2024 and Forecast 2025 to 2035, by Key Segments and Countries
- Sales Forecast 2025 to 2035 by Test Type, and End User for 30 Countries
- Competition Outlook, including Market Structure Analysis, Company Share Analysis by Key Players, and Competition Dashboard
- Company Profile
- Thermo Fisher Scientific
- Roche Diagnostics
- Siemens Healthineers
- DiaSorin S.p.A.
- Beckman Coulter
- Abbott Laboratories
- Bio-Rad Laboratories
- Fuji Rebio Diagnostics
- Randox Laboratories
- Nova Biomedical
- Others
List of Tables
Table 1: Global Market Value (US$ Million) Forecast by Region, 2018 to 2033
Table 2: Global Market Value (US$ Million) Forecast by Testing Type, 2018 to 2033
Table 3: Global Market Value (US$ Million) Forecast by End User, 2018 to 2033
Table 4: North America Market Value (US$ Million) Forecast by Country, 2018 to 2033
Table 5: North America Market Value (US$ Million) Forecast by Testing Type, 2018 to 2033
Table 6: North America Market Value (US$ Million) Forecast by End User, 2018 to 2033
Table 7: Latin America Market Value (US$ Million) Forecast by Country, 2018 to 2033
Table 8: Latin America Market Value (US$ Million) Forecast by Testing Type, 2018 to 2033
Table 9: Latin America Market Value (US$ Million) Forecast by End User, 2018 to 2033
Table 10: Western Europe Market Value (US$ Million) Forecast by Country, 2018 to 2033
Table 11: Western Europe Market Value (US$ Million) Forecast by Testing Type, 2018 to 2033
Table 12: Western Europe Market Value (US$ Million) Forecast by End User, 2018 to 2033
Table 13: Eastern Europe Market Value (US$ Million) Forecast by Country, 2018 to 2033
Table 14: Eastern Europe Market Value (US$ Million) Forecast by Testing Type, 2018 to 2033
Table 15: Eastern Europe Market Value (US$ Million) Forecast by End User, 2018 to 2033
Table 16: South Asia and Pacific Market Value (US$ Million) Forecast by Country, 2018 to 2033
Table 17: South Asia and Pacific Market Value (US$ Million) Forecast by Testing Type, 2018 to 2033
Table 18: South Asia and Pacific Market Value (US$ Million) Forecast by End User, 2018 to 2033
Table 19: East Asia Market Value (US$ Million) Forecast by Country, 2018 to 2033
Table 20: East Asia Market Value (US$ Million) Forecast by Testing Type, 2018 to 2033
Table 21: East Asia Market Value (US$ Million) Forecast by End User, 2018 to 2033
Table 22: Middle East and Africa Market Value (US$ Million) Forecast by Country, 2018 to 2033
Table 23: Middle East and Africa Market Value (US$ Million) Forecast by Testing Type, 2018 to 2033
Table 24: Middle East and Africa Market Value (US$ Million) Forecast by End User, 2018 to 2033
List of Figures
Figure 1: Global Market Value (US$ Million) by Testing Type, 2023 to 2033
Figure 2: Global Market Value (US$ Million) by End User, 2023 to 2033
Figure 3: Global Market Value (US$ Million) by Region, 2023 to 2033
Figure 4: Global Market Value (US$ Million) Analysis by Region, 2018 to 2033
Figure 5: Global Market Value Share (%) and BPS Analysis by Region, 2023 to 2033
Figure 6: Global Market Y-o-Y Growth (%) Projections by Region, 2023 to 2033
Figure 7: Global Market Value (US$ Million) Analysis by Testing Type, 2018 to 2033
Figure 8: Global Market Value Share (%) and BPS Analysis by Testing Type, 2023 to 2033
Figure 9: Global Market Y-o-Y Growth (%) Projections by Testing Type, 2023 to 2033
Figure 10: Global Market Value (US$ Million) Analysis by End User, 2018 to 2033
Figure 11: Global Market Value Share (%) and BPS Analysis by End User, 2023 to 2033
Figure 12: Global Market Y-o-Y Growth (%) Projections by End User, 2023 to 2033
Figure 13: Global Market Attractiveness by Testing Type, 2023 to 2033
Figure 14: Global Market Attractiveness by End User, 2023 to 2033
Figure 15: Global Market Attractiveness by Region, 2023 to 2033
Figure 16: North America Market Value (US$ Million) by Testing Type, 2023 to 2033
Figure 17: North America Market Value (US$ Million) by End User, 2023 to 2033
Figure 18: North America Market Value (US$ Million) by Country, 2023 to 2033
Figure 19: North America Market Value (US$ Million) Analysis by Country, 2018 to 2033
Figure 20: North America Market Value Share (%) and BPS Analysis by Country, 2023 to 2033
Figure 21: North America Market Y-o-Y Growth (%) Projections by Country, 2023 to 2033
Figure 22: North America Market Value (US$ Million) Analysis by Testing Type, 2018 to 2033
Figure 23: North America Market Value Share (%) and BPS Analysis by Testing Type, 2023 to 2033
Figure 24: North America Market Y-o-Y Growth (%) Projections by Testing Type, 2023 to 2033
Figure 25: North America Market Value (US$ Million) Analysis by End User, 2018 to 2033
Figure 26: North America Market Value Share (%) and BPS Analysis by End User, 2023 to 2033
Figure 27: North America Market Y-o-Y Growth (%) Projections by End User, 2023 to 2033
Figure 28: North America Market Attractiveness by Testing Type, 2023 to 2033
Figure 29: North America Market Attractiveness by End User, 2023 to 2033
Figure 30: North America Market Attractiveness by Country, 2023 to 2033
Figure 31: Latin America Market Value (US$ Million) by Testing Type, 2023 to 2033
Figure 32: Latin America Market Value (US$ Million) by End User, 2023 to 2033
Figure 33: Latin America Market Value (US$ Million) by Country, 2023 to 2033
Figure 34: Latin America Market Value (US$ Million) Analysis by Country, 2018 to 2033
Figure 35: Latin America Market Value Share (%) and BPS Analysis by Country, 2023 to 2033
Figure 36: Latin America Market Y-o-Y Growth (%) Projections by Country, 2023 to 2033
Figure 37: Latin America Market Value (US$ Million) Analysis by Testing Type, 2018 to 2033
Figure 38: Latin America Market Value Share (%) and BPS Analysis by Testing Type, 2023 to 2033
Figure 39: Latin America Market Y-o-Y Growth (%) Projections by Testing Type, 2023 to 2033
Figure 40: Latin America Market Value (US$ Million) Analysis by End User, 2018 to 2033
Figure 41: Latin America Market Value Share (%) and BPS Analysis by End User, 2023 to 2033
Figure 42: Latin America Market Y-o-Y Growth (%) Projections by End User, 2023 to 2033
Figure 43: Latin America Market Attractiveness by Testing Type, 2023 to 2033
Figure 44: Latin America Market Attractiveness by End User, 2023 to 2033
Figure 45: Latin America Market Attractiveness by Country, 2023 to 2033
Figure 46: Western Europe Market Value (US$ Million) by Testing Type, 2023 to 2033
Figure 47: Western Europe Market Value (US$ Million) by End User, 2023 to 2033
Figure 48: Western Europe Market Value (US$ Million) by Country, 2023 to 2033
Figure 49: Western Europe Market Value (US$ Million) Analysis by Country, 2018 to 2033
Figure 50: Western Europe Market Value Share (%) and BPS Analysis by Country, 2023 to 2033
Figure 51: Western Europe Market Y-o-Y Growth (%) Projections by Country, 2023 to 2033
Figure 52: Western Europe Market Value (US$ Million) Analysis by Testing Type, 2018 to 2033
Figure 53: Western Europe Market Value Share (%) and BPS Analysis by Testing Type, 2023 to 2033
Figure 54: Western Europe Market Y-o-Y Growth (%) Projections by Testing Type, 2023 to 2033
Figure 55: Western Europe Market Value (US$ Million) Analysis by End User, 2018 to 2033
Figure 56: Western Europe Market Value Share (%) and BPS Analysis by End User, 2023 to 2033
Figure 57: Western Europe Market Y-o-Y Growth (%) Projections by End User, 2023 to 2033
Figure 58: Western Europe Market Attractiveness by Testing Type, 2023 to 2033
Figure 59: Western Europe Market Attractiveness by End User, 2023 to 2033
Figure 60: Western Europe Market Attractiveness by Country, 2023 to 2033
Figure 61: Eastern Europe Market Value (US$ Million) by Testing Type, 2023 to 2033
Figure 62: Eastern Europe Market Value (US$ Million) by End User, 2023 to 2033
Figure 63: Eastern Europe Market Value (US$ Million) by Country, 2023 to 2033
Figure 64: Eastern Europe Market Value (US$ Million) Analysis by Country, 2018 to 2033
Figure 65: Eastern Europe Market Value Share (%) and BPS Analysis by Country, 2023 to 2033
Figure 66: Eastern Europe Market Y-o-Y Growth (%) Projections by Country, 2023 to 2033
Figure 67: Eastern Europe Market Value (US$ Million) Analysis by Testing Type, 2018 to 2033
Figure 68: Eastern Europe Market Value Share (%) and BPS Analysis by Testing Type, 2023 to 2033
Figure 69: Eastern Europe Market Y-o-Y Growth (%) Projections by Testing Type, 2023 to 2033
Figure 70: Eastern Europe Market Value (US$ Million) Analysis by End User, 2018 to 2033
Figure 71: Eastern Europe Market Value Share (%) and BPS Analysis by End User, 2023 to 2033
Figure 72: Eastern Europe Market Y-o-Y Growth (%) Projections by End User, 2023 to 2033
Figure 73: Eastern Europe Market Attractiveness by Testing Type, 2023 to 2033
Figure 74: Eastern Europe Market Attractiveness by End User, 2023 to 2033
Figure 75: Eastern Europe Market Attractiveness by Country, 2023 to 2033
Figure 76: South Asia and Pacific Market Value (US$ Million) by Testing Type, 2023 to 2033
Figure 77: South Asia and Pacific Market Value (US$ Million) by End User, 2023 to 2033
Figure 78: South Asia and Pacific Market Value (US$ Million) by Country, 2023 to 2033
Figure 79: South Asia and Pacific Market Value (US$ Million) Analysis by Country, 2018 to 2033
Figure 80: South Asia and Pacific Market Value Share (%) and BPS Analysis by Country, 2023 to 2033
Figure 81: South Asia and Pacific Market Y-o-Y Growth (%) Projections by Country, 2023 to 2033
Figure 82: South Asia and Pacific Market Value (US$ Million) Analysis by Testing Type, 2018 to 2033
Figure 83: South Asia and Pacific Market Value Share (%) and BPS Analysis by Testing Type, 2023 to 2033
Figure 84: South Asia and Pacific Market Y-o-Y Growth (%) Projections by Testing Type, 2023 to 2033
Figure 85: South Asia and Pacific Market Value (US$ Million) Analysis by End User, 2018 to 2033
Figure 86: South Asia and Pacific Market Value Share (%) and BPS Analysis by End User, 2023 to 2033
Figure 87: South Asia and Pacific Market Y-o-Y Growth (%) Projections by End User, 2023 to 2033
Figure 88: South Asia and Pacific Market Attractiveness by Testing Type, 2023 to 2033
Figure 89: South Asia and Pacific Market Attractiveness by End User, 2023 to 2033
Figure 90: South Asia and Pacific Market Attractiveness by Country, 2023 to 2033
Figure 91: East Asia Market Value (US$ Million) by Testing Type, 2023 to 2033
Figure 92: East Asia Market Value (US$ Million) by End User, 2023 to 2033
Figure 93: East Asia Market Value (US$ Million) by Country, 2023 to 2033
Figure 94: East Asia Market Value (US$ Million) Analysis by Country, 2018 to 2033
Figure 95: East Asia Market Value Share (%) and BPS Analysis by Country, 2023 to 2033
Figure 96: East Asia Market Y-o-Y Growth (%) Projections by Country, 2023 to 2033
Figure 97: East Asia Market Value (US$ Million) Analysis by Testing Type, 2018 to 2033
Figure 98: East Asia Market Value Share (%) and BPS Analysis by Testing Type, 2023 to 2033
Figure 99: East Asia Market Y-o-Y Growth (%) Projections by Testing Type, 2023 to 2033
Figure 100: East Asia Market Value (US$ Million) Analysis by End User, 2018 to 2033
Figure 101: East Asia Market Value Share (%) and BPS Analysis by End User, 2023 to 2033
Figure 102: East Asia Market Y-o-Y Growth (%) Projections by End User, 2023 to 2033
Figure 103: East Asia Market Attractiveness by Testing Type, 2023 to 2033
Figure 104: East Asia Market Attractiveness by End User, 2023 to 2033
Figure 105: East Asia Market Attractiveness by Country, 2023 to 2033
Figure 106: Middle East and Africa Market Value (US$ Million) by Testing Type, 2023 to 2033
Figure 107: Middle East and Africa Market Value (US$ Million) by End User, 2023 to 2033
Figure 108: Middle East and Africa Market Value (US$ Million) by Country, 2023 to 2033
Figure 109: Middle East and Africa Market Value (US$ Million) Analysis by Country, 2018 to 2033
Figure 110: Middle East and Africa Market Value Share (%) and BPS Analysis by Country, 2023 to 2033
Figure 111: Middle East and Africa Market Y-o-Y Growth (%) Projections by Country, 2023 to 2033
Figure 112: Middle East and Africa Market Value (US$ Million) Analysis by Testing Type, 2018 to 2033
Figure 113: Middle East and Africa Market Value Share (%) and BPS Analysis by Testing Type, 2023 to 2033
Figure 114: Middle East and Africa Market Y-o-Y Growth (%) Projections by Testing Type, 2023 to 2033
Figure 115: Middle East and Africa Market Value (US$ Million) Analysis by End User, 2018 to 2033
Figure 116: Middle East and Africa Market Value Share (%) and BPS Analysis by End User, 2023 to 2033
Figure 117: Middle East and Africa Market Y-o-Y Growth (%) Projections by End User, 2023 to 2033
Figure 118: Middle East and Africa Market Attractiveness by Testing Type, 2023 to 2033
Figure 119: Middle East and Africa Market Attractiveness by End User, 2023 to 2033
Figure 120: Middle East and Africa Market Attractiveness by Country, 2023 to 2033

