Balloon Catheters for Bile Stone Removal Market
This report provides an extensive study of the balloon catheters for bile stone removal market, addressing market size estimation, revenue trajectory, competitive analysis, product type trends, demand forecasting, growth opportunities, market restraints, supply chain assessment, and strategic recommendations.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Balloon Catheters for Bile Stone Removal Market Size, Market Forecast and Outlook By FMI
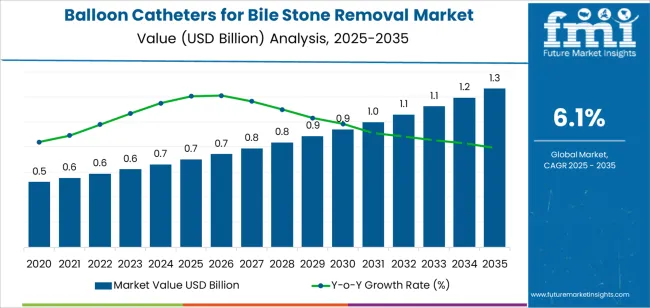
The balloon catheters for bile stone removal market was valued at USD 700.00 million in 2025, projected to reach USD 742.70 million in 2026, and is forecast to expand to USD 1,342.66 million by 2036 at a 6.10% CAGR. Rising global prevalence of choledocholithiasis driven by aging populations and dietary shifts toward high-fat consumption patterns is increasing ERCP procedure volumes that generate per-procedure balloon catheter consumption. Endoscopic retrograde cholangiopancreatography has become the standard-of-care interventional approach for common bile duct stone extraction, creating a procedure-volume-linked demand structure for balloon extraction catheters that expands proportionally with gastroenterology service capacity buildout.
The absolute dollar opportunity of approximately USD 599 million over the forecast period reflects the expansion of ERCP procedure access in emerging markets combined with technology-driven catheter premium pricing in established markets. Multi-lumen catheter designs enabling simultaneous contrast injection and stone extraction within a single catheter insertion are commanding per-unit price premiums over standard single-lumen models by reducing procedure time and radiation exposure. Single-use catheter mandates driven by infection control standards are converting reusable catheter procedure volumes into disposable device procurement, expanding the per-procedure device cost and total addressable market.
Summary of Balloon Catheters for Bile Stone Removal Market
- Balloon Catheters for Bile Stone Removal Market Definition
- Balloon catheters for bile stone removal are specialized ERCP devices with inflatable tips in single, double, and triple-lumen configurations for common bile duct stone extraction, contrast injection, and guidewire passage during minimally invasive biliary interventions.
- Demand Drivers in the Market
- Rising choledocholithiasis prevalence driven by aging populations and dietary shifts is increasing ERCP procedure volumes that generate per-procedure disposable balloon catheter consumption proportional to gastroenterology service capacity expansion.
- Multi-lumen catheter designs enabling simultaneous contrast injection and stone extraction within a single insertion command per-unit price premiums by reducing procedure time and radiation exposure for both patient and endoscopist.
- Single-use catheter mandates driven by infection control standards are converting reusable catheter procedure volumes into disposable device procurement, expanding per-procedure device cost and total addressable market value.
- Key Segments Analyzed in the FMI Report
- Single-lumen Balloon Catheter by Lumen Type: 41.0% share in 2026, reflecting the cost-effectiveness and procedural simplicity of single-lumen designs for standard bile duct stone extraction in high-volume ERCP centres.
- Public Hospital by Application: 55.0% share in 2026, as public hospital gastroenterology departments serve the majority of ERCP referral volumes and generate the largest institutional balloon catheter procurement orders.
- China: 8.2% compound growth driven by digestive endoscopy centre expansion across county-level hospitals and hepatobiliary disease screening programme referral volumes.
- Analyst Opinion at FMI
- Sabyasachi Ghosh Principal Consultant for healthcare opinis,that "The balloon catheters for bile stone removal market is procedure-volume driven, where demand correlates directly with ERCP capacity expansion and hepatobiliary disease prevalence rather than technology adoption cycles. Catheter manufacturers who cannot demonstrate infection control compliance through validated single-use packaging and traceability systems face procurement exclusion as hospital infection prevention committees mandate disposable device policies. The critical market access risk centers on tender pricing pressure in public hospital systems, where balloon catheter procurement is consolidated into competitive bidding processes that compress per-unit margins for suppliers unable to differentiate on procedural outcome data".
- Strategic Implications / Executive Takeaways
- Balloon catheter manufacturers must invest in clinical outcome data generation demonstrating procedure time reduction and stone clearance rates to differentiate multi-lumen products against lower-priced single-lumen alternatives in competitive hospital tenders.
- Hospital gastroenterology department procurement teams must evaluate total procedure cost models that account for catheter-related time savings and reduced fluoroscopy exposure when comparing single-lumen and multi-lumen balloon catheter options.
- Medical device distributors must expand cold chain and single-use packaging logistics capabilities to serve the growing disposable balloon catheter procurement requirements in emerging market ERCP centres.
China sets the pace at an 8.2% CAGR, driven by digestive endoscopy centre expansion across county-level hospitals and national hepatobiliary disease screening programme referral volumes. India follows at 7.6% as gastroenterology department capacity expansion in district hospitals and growing cholelithiasis treatment access generate ERCP procedure volume growth. Germany registers 7.0% growth supported by established gastroenterology centre density and high per-capita ERCP procedure rates. Brazil advances at 6.4% with SUS gastroenterology service expansion and hepatobiliary disease burden-driven procedure demand. USA tracks at 5.8% driven by mature ERCP procedure volumes and technology-driven catheter premium adoption. UK maintains 5.2% growth anchored by NHS gastroenterology service modernization. Japan registers 4.6% supported by established endoscopy infrastructure and aging population biliary disease prevalence.
Balloon Catheters for Bile Stone Removal Market Key Takeaways
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 742.70 Million |
| Industry Value (2036) | USD 1,342.66 Million |
| CAGR (2026-2036) | 6.10% |
Source: Future Market Insights, 2026
Balloon Catheters for Bile Stone Removal Market Definition
Balloon catheters for bile stone removal are specialized endoscopic devices designed for extraction of calculi from the common bile duct during ERCP procedures. These catheters feature inflatable balloon tips in single-lumen, double-lumen, and triple-lumen configurations that enable bile duct stone sweeping, contrast medium injection, and guidewire passage through a single catheter platform for minimally invasive biliary stone management.
Balloon Catheters for Bile Stone Removal Market Inclusions
Market scope includes single-lumen extraction balloon catheters, double-lumen balloon catheters with contrast injection capability, triple-lumen balloon catheters with guidewire channels, and compliant and non-compliant balloon catheter designs. Coverage spans public hospital and private hospital procurement across disposable single-use and limited-reuse configurations.
Balloon Catheters for Bile Stone Removal Market Exclusions
Lithotripsy catheters, biliary stents, sphincterotomy devices, and diagnostic ERCP catheters without balloon extraction capability are excluded. Endoscope capital equipment and accessories not specific to balloon stone extraction fall outside analytical parameters.
Balloon Catheters for Bile Stone Removal Market Research Methodology
- Primary Research: Analysts engaged with gastroenterology department procurement managers, ERCP procedure volume data coordinators, and endoscopic device manufacturer clinical affairs directors to map the procedure volume and device selection triggers for balloon catheter procurement.
- Desk Research: Data collection phases aggregated national ERCP procedure volume registries, hepatobiliary disease prevalence data, and hospital endoscopy centre capacity expansion statistics.
- Market-Sizing and Forecasting: Baseline values derive from a bottom-up aggregation of ERCP procedure volumes multiplied by per-procedure balloon catheter utilization rates and average per-unit prices, applying region-specific gastroenterology capacity growth curves.
- Data Validation and Update Cycle: Projections are tested against publicly reported endoscopic device segment revenues from leading medical device manufacturers and gastroenterology equipment distributors.
Why is the Balloon Catheters for Bile Stone Removal Market Growing?
Market expansion is being supported by the increasing global incidence of gallstone disease and biliary disorders driven by aging populations and dietary lifestyle factors, alongside the corresponding need for minimally invasive intervention technologies that can improve procedural outcomes, reduce patient recovery times, and maintain clinical effectiveness across various endoscopic bile duct procedures and therapeutic interventions. Modern gastroenterology departments and endoscopy centers are increasingly focused on implementing balloon catheter solutions that can facilitate stone extraction, enhance bile duct clearance, and provide consistent performance in complex biliary anatomies.
The growing focus on outpatient procedures and reduced hospital stays is driving demand for balloon catheters that can enable efficient endoscopic interventions, support same-day discharge protocols, and ensure comprehensive patient safety. Clinical practitioners' preference for single-use medical devices that combine procedural reliability with infection prevention and quality assurance is creating opportunities for advanced balloon catheter implementations. The rising influence of minimally invasive surgery adoption and therapeutic endoscopy advancement is also contributing to increased utilization of balloon catheters that can provide superior stone extraction capabilities without compromising procedural safety or clinical outcomes.
What factors drive the leadership of the Single-lumen Balloon Catheter and Public Hospital segments in their respective categories?
The market is segmented by lumen type, application, and region. By lumen type, the market is divided into single-lumen balloon catheter, double-lumen balloon catheter, and triple-lumen balloon catheter. Based on application, the market is categorized into public hospital and private hospital. Regionally, the market is divided into East Asia, Europe, North America, South Asia, Latin America, Middle East & Africa, and Eastern Europe.
By Lumen Type, the Single-lumen Balloon Catheter Segment Leads the Market
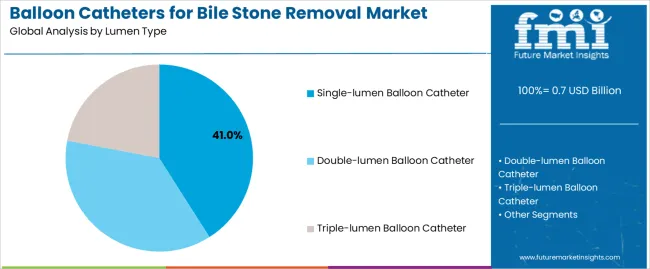
The single-lumen balloon catheter segment is projected to maintain its leading position in the market with 41.0% market share in 2026, reaffirming its role as the preferred device configuration for standard endoscopic bile stone extraction procedures. Gastroenterologists and endoscopy specialists utilize single-lumen balloon catheters for their procedural simplicity, cost-effectiveness, and proven effectiveness in removing bile duct stones while maintaining adequate visualization and working channel access. Single-lumen catheter technology's proven clinical performance and ease of use directly address the procedural requirements for routine bile stone extraction and bile duct clearance across diverse patient populations and stone characteristics.
This device segment forms the foundation of standard endoscopic stone removal procedures, as it represents the catheter configuration with the greatest adoption rate and established safety record across multiple endoscopy applications and clinical settings. Healthcare facility investments in endoscopic intervention capabilities continue to strengthen adoption among gastroenterology departments and ambulatory endoscopy centers. With clinical protocols focusing procedural efficiency and patient safety, single-lumen balloon catheters align with both treatment objectives and economic considerations, making them the central component of comprehensive bile stone management strategies.
By Application, the Public Hospital Segment Dominates Market Demand
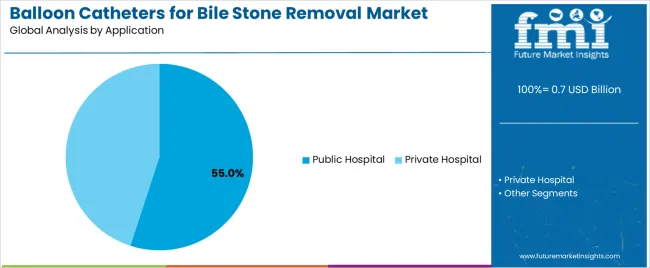
The public hospital application segment is projected to represent the 55.0% market share of balloon catheters for bile stone removal demand in 2026, highlighting its role as the primary setting for endoscopic biliary interventions across emergency presentations, scheduled procedures, and complex case management. Public hospitals utilize balloon catheters for bile stone removal due to their comprehensive gastroenterology departments, high patient volumes, and ability to manage diverse biliary complications while supporting emergency care capabilities and teaching hospital functions. Positioned as essential medical devices for hospital endoscopy units, balloon catheters offer both clinical effectiveness and procedural versatility.
The segment is supported by continuous expansion in public healthcare infrastructure and the growing availability of trained endoscopists that enable wider access to therapeutic endoscopy procedures with improved clinical outcomes and reduced surgical intervention rates. Public hospitals are investing in comprehensive endoscopy programs to support increasingly complex biliary disease management and population health requirements. As healthcare access expands and endoscopic intervention adoption increases, the public hospital application will continue to dominate the market while supporting advanced procedural capabilities and clinical training initiatives.
What are the Drivers, Restraints, and Key Trends of the Balloon Catheters for Bile Stone Removal Market?
The balloon catheters for bile stone removal market is advancing due to increasing prevalence of gallstone disease driven by aging demographics and growing adoption of minimally invasive endoscopic procedures that require specialized medical device technologies providing enhanced clinical outcomes and patient safety benefits across diverse therapeutic endoscopy and biliary intervention applications. The market faces challenges, including procedural complexity and specialist training requirements, competition from alternative stone extraction devices and surgical interventions, and reimbursement constraints related to procedure coding and healthcare payment limitations. Innovation in catheter design technologies and advanced balloon materials continues to influence product development and market expansion patterns.
Expansion of Endoscopic Retrograde Cholangiopancreatography Procedures
The growing adoption of endoscopic retrograde cholangiopancreatography as the preferred intervention for bile duct stone management is driving demand for specialized balloon catheters that address procedural requirements including stone extraction efficiency, bile duct clearance verification, and minimal tissue trauma during stone removal. Hospital endoscopy units require reliable catheter systems that deliver consistent performance across varied stone sizes while maintaining procedural safety and reducing complication rates. Gastroenterology specialists are increasingly recognizing the clinical advantages of balloon catheter integration for biliary stone management and treatment optimization, creating opportunities for advanced catheter designs specifically developed for complex biliary interventions.
Integration of Advanced Balloon Materials and Design Features
Modern medical device manufacturers are incorporating advanced polymer materials and innovative catheter designs to enhance procedural performance, improve stone capture efficiency, and support comprehensive clinical objectives through optimized balloon compliance and controlled inflation characteristics. Leading companies are developing radiopaque marker systems, implementing tapered balloon configurations, and advancing catheter shaft technologies that improve device trackability and procedural control. These technologies improve clinical outcomes while enabling procedural flexibility, including varied stone extraction techniques, balloon dilation capabilities, and enhanced endoscopic visualization. Advanced design integration also allows clinicians to support comprehensive treatment objectives and procedural optimization beyond traditional catheter limitations.
Development of Single-Use Sterile Catheter Systems
The expansion of infection prevention protocols and patient safety initiatives is driving adoption of single-use balloon catheters with sterile packaging and quality assurance systems that eliminate reprocessing concerns and ensure consistent device performance. These disposable medical devices address healthcare facility requirements for infection control compliance, reduce cross-contamination risks, and provide standardized product specifications that support predictable procedural outcomes. Manufacturers are investing in cost-effective production capabilities and quality management systems to serve growing demand for single-use endoscopy devices while supporting innovation in catheter functionality and clinical performance.
Analysis of the Balloon Catheters for Bile Stone Removal Market by Key Countries
.webp)
| Country | CAGR (2026-2036) |
|---|---|
| China | 8.2% |
| India | 7.6% |
| Germany | 7.0% |
| Brazil | 6.4% |
| United States | 5.8% |
| United Kingdom | 5.2% |
| Japan | 4.6% |
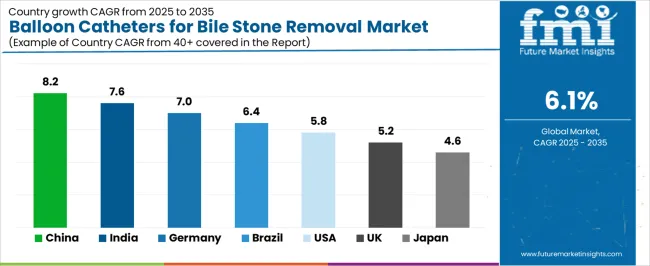
The market is experiencing solid growth globally, with China leading at an 8.2% CAGR through 2036, driven by expanding endoscopy infrastructure, growing prevalence of biliary disorders, and increasing adoption of minimally invasive therapeutic procedures. India follows at 7.6%, supported by rising gallstone disease incidence, expanding gastroenterology departments, and growing healthcare access in tier-2 and tier-3 cities. Germany shows growth at 7.0%, focusing advanced endoscopy techniques, clinical excellence in gastroenterology, and established therapeutic endoscopy training programs. Brazil demonstrates 6.4% growth, supported by expanding healthcare infrastructure, increasing endoscopy procedure volumes, and growing medical device market development. The United States records 5.8%, focusing on outpatient endoscopy expansion, ambulatory surgical center growth, and advanced biliary intervention techniques. The United Kingdom exhibits 5.2% growth, focusing National Health Service endoscopy programs and gastroenterology service expansion. Japan shows 4.6% growth, focusing clinical quality standards and advanced endoscopic device adoption.
The report covers an in-depth analysis of 40 countries, top-performing countries are highlighted below.
China Leads Global Market Growth with Healthcare Infrastructure Expansion
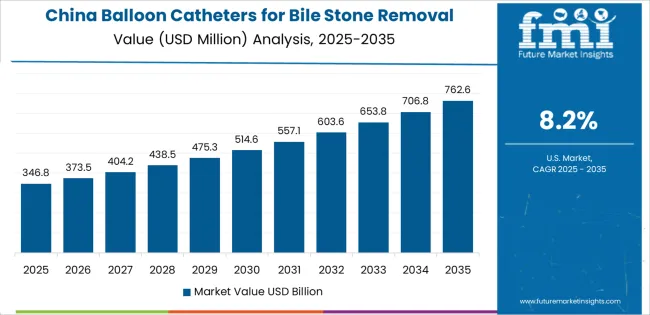
Revenue from balloon catheters for bile stone removal in China is projected to exhibit exceptional growth with a CAGR of 8.2% through 2036, driven by expanding endoscopy infrastructure and rapidly growing gastroenterology departments supported by government healthcare initiatives and hospital modernization programs. The country's massive healthcare sector expansion and increasing investment in minimally invasive procedure capabilities are creating substantial demand for therapeutic endoscopy devices. Major medical device manufacturers and international suppliers are establishing comprehensive distribution and clinical support capabilities to serve both urban tertiary hospitals and expanding regional medical centers.
- Government support for healthcare infrastructure development and digestive disease management is driving demand for balloon catheters throughout major metropolitan regions and provincial medical centers across gastroenterology departments, endoscopy units, and general surgery facilities.
- Strong growth in endoscopy procedure volumes and an expanding network of trained endoscopists are supporting the rapid adoption of single-use catheter technologies among hospitals seeking improved infection control and standardized procedural outcomes.
India Demonstrates Strong Market Potential with Disease Burden and Healthcare Access Growth
Revenue from balloon catheters for bile stone removal in India is expanding at a CAGR of 7.6%, supported by increasing prevalence of gallstone disease, expanding gastroenterology services, and growing adoption of therapeutic endoscopy procedures driven by healthcare infrastructure investments and medical education advancement. The country's substantial patient population and expanding hospital networks are driving demand for bile stone removal technologies throughout diverse clinical settings. Leading medical device companies and local distributors are establishing comprehensive supply chains and clinical training programs to address growing procedural demand.
- Rising incidence of biliary disorders and expanding network of endoscopy facilities are creating opportunities for balloon catheter adoption across corporate hospitals, government medical colleges, and specialty gastroenterology centers in major urban areas and emerging healthcare markets.
- Growing awareness of minimally invasive treatment options and increasing endoscopy training programs are driving adoption of advanced catheter technologies among gastroenterologists seeking improved procedural efficiency and patient outcomes.
Germany Demonstrates Clinical Excellence with Advanced Endoscopy Applications
Revenue from balloon catheters for bile stone removal in Germany is expanding at a CAGR of 7.0%, supported by the country's leadership in gastroenterology, advanced endoscopy infrastructure, and focus on clinical quality standards for therapeutic endoscopy procedures. Germany's medical excellence and established training programs are driving demand for high-quality medical devices throughout gastroenterology departments. Leading university hospitals and endoscopy centers are investing in advanced therapeutic techniques and device technologies.
- Advanced clinical protocols and established endoscopy expertise are creating opportunities for specialized balloon catheter configurations among gastroenterology departments seeking superior procedural outcomes and complex case management capabilities.
- Strong focus on clinical training and procedural standardization is supporting the adoption of quality-assured single-use catheters across hospital endoscopy units throughout major medical centers and regional healthcare facilities.
Brazil Focuses on Healthcare Infrastructure and Procedure Volume Growth
Revenue from balloon catheters for bile stone removal in Brazil is growing at a CAGR of 6.4%, driven by expanding healthcare infrastructure, growing endoscopy procedure volumes, and increasing adoption of minimally invasive biliary interventions across public and private hospital networks. Brazil's developing healthcare sector and population health needs are supporting investment in therapeutic endoscopy capabilities. Medical device distributors and healthcare facilities are establishing endoscopy service capabilities for biliary disease management.
- Healthcare infrastructure expansion and growing prevalence of metabolic disorders are creating demand for bile stone removal procedures throughout major metropolitan hospitals, specialty gastroenterology clinics, and regional medical centers.
- Increasing availability of trained endoscopists and growing awareness of endoscopic treatment options are driving adoption of balloon catheter technologies among healthcare facilities seeking improved patient care and reduced surgical intervention rates.
United States Emphasizes Outpatient Procedures and Ambulatory Care Growth
Revenue from balloon catheters for bile stone removal in the United States is expanding at a CAGR of 5.8%, supported by expansion of ambulatory surgical centers, growing outpatient endoscopy procedures, and established gastroenterology specialty networks providing comprehensive biliary intervention services. The nation's mature healthcare infrastructure and procedure volume migration to outpatient settings are driving demand for efficient single-use medical devices. Ambulatory care facilities and hospital endoscopy departments are investing in quality devices and procedural efficiency optimization.
- Outpatient procedure growth and ambulatory surgical center expansion are creating demand for reliable balloon catheters among gastroenterology practices seeking streamlined procedural workflows and same-day discharge capabilities.
- Strong reimbursement structures and established clinical pathways are driving adoption of quality-assured catheter systems across diverse endoscopy settings throughout major healthcare markets and regional gastroenterology networks.
United Kingdom Shows Healthcare System Integration
Revenue from balloon catheters for bile stone removal in the United Kingdom is expanding at a CAGR of 5.2%, supported by National Health Service endoscopy programs, gastroenterology service expansion, and ongoing efforts to reduce endoscopy procedure waiting times through capacity enhancement initiatives. The country's integrated healthcare system and clinical guideline adherence are driving demand for standardized medical devices. Hospital trusts and endoscopy units are implementing quality improvement programs for biliary intervention procedures.
- National Health Service initiatives to expand endoscopy capacity and reduce procedure backlogs are creating opportunities for balloon catheter adoption among hospital gastroenterology departments seeking improved throughput and procedural efficiency.
- Clinical quality standards and evidence-based treatment protocols are driving demand for reliable single-use catheter systems throughout hospital endoscopy units across regional healthcare networks and teaching hospitals.
Japan Shows Advanced Technology Focus with Clinical Quality Standards
Revenue from balloon catheters for bile stone removal in Japan is expanding at a CAGR of 4.6%, supported by the country's focus on clinical quality, advanced endoscopy technology adoption, and comprehensive gastroenterology training programs ensuring high procedural standards. Japan's medical sophistication and attention to device quality are driving demand for premium balloon catheter products. Leading academic medical centers and community hospitals are investing in advanced therapeutic endoscopy capabilities.
- Advanced clinical standards and established endoscopy expertise are creating opportunities for high-quality balloon catheters throughout university hospitals, specialty gastroenterology centers, and community hospital endoscopy departments.
- Strong focus on procedural safety and device reliability is driving adoption of quality-assured catheter systems meeting stringent performance and regulatory standards across gastroenterology practices and hospital endoscopy services.
What is the market split by country in Europe?
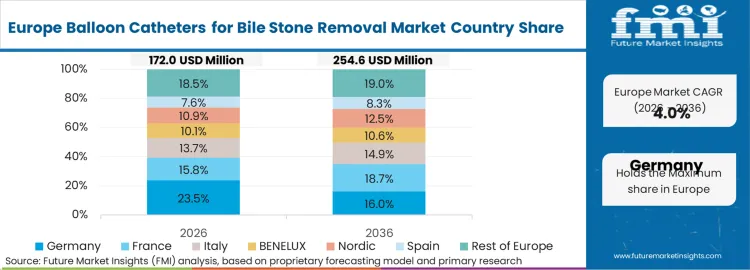
The balloon catheters for bile stone removal market in Europe is projected to grow from USD 245.7 million in 2026 to USD 434.6 million by 2036, registering a CAGR of 5.9% over the forecast period. Germany is expected to maintain leadership with a 26.5% market share in 2026, moderating to 26.2% by 2036, supported by advanced gastroenterology infrastructure, clinical excellence in therapeutic endoscopy, and comprehensive training programs.
France follows with 19.8% in 2026, projected at 20.1% by 2036, driven by established endoscopy networks, academic medical centers, and national digestive health programs. The United Kingdom holds 17.6% in 2026, declining slightly to 17.3% by 2036 due to healthcare budget constraints and procedure volume management. Italy commands 13.4% in 2026, rising to 13.6% by 2036, while Spain accounts for 10.2% in 2026, reaching 10.5% by 2036 aided by gastroenterology service expansion and endoscopy infrastructure development. The Netherlands maintains 6.8% in 2026, up to 7.0% by 2036 due to advanced endoscopy facilities and clinical quality initiatives. The Rest of Europe region, including Nordic countries, Central and Eastern Europe, and other markets, is anticipated to hold 5.7% in 2026 and 5.3% by 2036, reflecting gradual adoption in emerging gastroenterology markets and endoscopy service development programs.
What factors drive the leadership of the Unit/Component Testing segment and the dominance of Large Enterprises in market demand?
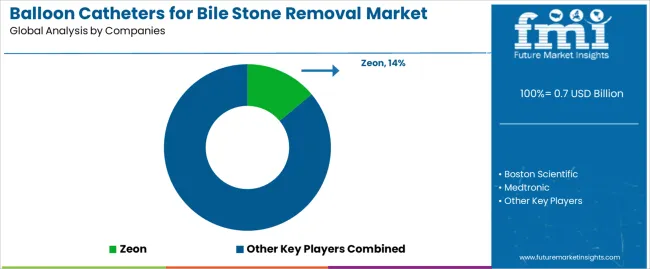
The market is characterized by competition among established medical device manufacturers, specialized endoscopy equipment companies, and regional catheter suppliers. Companies are investing in advanced balloon material development, catheter design innovation, clinical evidence generation, and application-specific product portfolios to deliver safe, effective, and user-friendly balloon catheter solutions. Innovation in radiopaque marker systems, balloon compliance characteristics, and catheter shaft flexibility is central to strengthening market position and competitive advantage.
Zeon leads the market with comprehensive balloon catheter solutions with a focus on clinical performance, device reliability, and established presence in therapeutic endoscopy applications across diverse gastroenterology settings. Boston Scientific provides innovative endoscopy devices with focus on procedural efficiency and advanced catheter technologies for complex biliary interventions. Medtronic offers versatile medical device portfolios with focus on gastroenterology and therapeutic endoscopy solutions. Cook Medical delivers specialized endoscopy products with comprehensive catheter offerings for biliary procedures. CONMED provides endoscopic intervention devices with focus on procedural performance and clinical support. Micro-Tech Endoscopy specializes in therapeutic endoscopy technologies with focus on emerging markets and cost-effective device solutions.
Medorah Medical focuses on specialized catheter technologies for challenging biliary anatomy. Penlon offers medical device solutions with established product lines for endoscopy applications. Edwards Lifesciences provides advanced medical technologies with expertise in catheter-based interventions. B. Braun delivers comprehensive medical device portfolios including endoscopy and intervention products for healthcare facilities worldwide.
Balloon Catheters for Bile Stone Removal Market - Stakeholder Contribution Framework
Balloon catheters for bile stone removal represent a specialized medical device segment within therapeutic endoscopy and gastroenterology applications, projected to grow from USD 0.7 billion in 2025 to USD 1.2 billion by 2035 at a 6.1% CAGR. These single-use intervention devices serve as essential tools in endoscopic procedures for bile duct stone extraction, bile duct clearance verification, and biliary obstruction management where procedural safety, stone removal efficiency, and patient comfort are required. Market expansion is driven by increasing gallstone disease prevalence, growing minimally invasive procedure adoption, advancing endoscopy infrastructure, and rising focus on outpatient therapeutic interventions across diverse hospital and ambulatory care settings.
How Healthcare Regulators Could Strengthen Device Standards and Patient Safety?
- Medical Device Performance Standards: Establish comprehensive technical specifications for balloon catheters, including balloon compliance characteristics, catheter shaft flexibility, radiopaque marker visibility, and dimensional tolerances that ensure consistent quality across therapeutic endoscopy and biliary intervention applications.
- Single-Use Device Guidelines: Develop regulatory frameworks that mandate single-use designation for balloon catheters, requiring manufacturers to provide clear labeling, prevent device reprocessing, and ensure sterility assurance throughout product shelf life and clinical use.
- Clinical Evidence Requirements: Implement mandatory clinical evaluation standards for balloon catheter devices, including safety data collection, efficacy outcome assessment, and adverse event reporting that ensure product performance and patient safety across diverse clinical applications.
- Quality Management Certification: Create specialized guidelines for manufacturing quality systems used in balloon catheter production, addressing cleanroom requirements, sterilization validation, biocompatibility testing, and traceability protocols specific to single-use endoscopy devices.
- Post-Market Surveillance Programs: Provide regulatory oversight for device performance monitoring and reporting systems that enable adverse event tracking, product quality assessment, and continuous safety evaluation throughout commercial distribution and clinical utilization.
How Healthcare Associations Could Advance Clinical Standards and Training Development?
- Clinical Practice Guidelines: Develop comprehensive procedural protocols for bile stone removal techniques, including balloon catheter selection criteria, stone extraction methodologies, and complication management strategies that optimize clinical outcomes, ensure procedural safety, and maintain treatment effectiveness across diverse patient populations.
- Quality Benchmarking Programs: Establish clinical performance metrics for endoscopic bile stone removal procedures, including stone clearance rates, procedure times, complication frequencies, and patient safety indicators that enable comparative analysis and drive continuous improvement toward procedural excellence.
- Endoscopy Training Programs: Create specialized education initiatives for gastroenterologists, endoscopy nurses, and technical personnel covering balloon catheter techniques, device handling procedures, troubleshooting approaches, and safety protocols across therapeutic endoscopy training centers.
- Device Performance Evaluation: Develop standardized assessment methodologies for balloon catheter performance, including stone extraction efficiency, balloon inflation characteristics, catheter trackability, and clinical usability metrics that facilitate objective device comparison and selection decisions.
- Multidisciplinary Collaboration: Facilitate partnerships between gastroenterologists, device manufacturers, endoscopy training centers, and healthcare facilities to advance procedural techniques, address clinical challenges, and accelerate innovation in therapeutic endoscopy technologies.
How Medical Device Manufacturers Could Drive Innovation and Clinical Excellence?
- Advanced Material Development: Invest in medical-grade polymer research, balloon compliance optimization, catheter shaft engineering, and biocompatible coating technologies that improve device performance while enhancing procedural safety and clinical outcomes in biliary interventions.
- Clinical Product Development: Develop next-generation balloon catheters with enhanced features, including optimized balloon shapes, improved radiopaque markers, varied catheter lengths, and application-specific designs for complex biliary anatomy and challenging stone characteristics.
- Manufacturing Quality Excellence: Implement advanced production technologies with automated quality inspection, statistical process control, sterility assurance validation, and comprehensive documentation systems that ensure consistent product quality and regulatory compliance across global manufacturing operations.
- Clinical Support Services: Establish comprehensive healthcare provider support capabilities providing device training programs, procedural technique assistance, clinical evidence resources, and technical expertise that strengthen physician relationships and enable successful device adoption.
- Global Distribution Networks: Develop regional distribution partnerships, strategic inventory positioning, cold chain logistics capabilities, and responsive customer service that ensure product availability, minimize delivery delays, and support clinical procedure scheduling requirements worldwide.
How Healthcare Facilities Could Optimize Clinical Outcomes and Procedural Efficiency?
- Evidence-Based Device Selection: Conduct systematic assessments of clinical requirements, procedural complexity, patient characteristics, and cost considerations to optimize balloon catheter selection and achieve desired clinical outcomes while managing endoscopy department budgets.
- Procedural Protocol Standardization: Implement standardized clinical pathways utilizing evidence-based techniques, quality checklists, and systematic approaches to maximize balloon catheter effectiveness, ensure procedural consistency, and maintain patient safety throughout endoscopy operations.
- Staff Training Investment: Develop rigorous education programs utilizing hands-on training, simulation exercises, and ongoing competency assessment to maximize clinical proficiency, improve device utilization, and maintain procedural quality throughout gastroenterology departments.
- Quality Monitoring Programs: Incorporate outcome tracking, complication surveillance, and performance analysis methodologies into clinical quality programs that support continuous improvement, identify training needs, and optimize procedural techniques.
- Manufacturer Partnerships: Establish collaborative relationships with device manufacturers for clinical education, product evaluation, technical support, and joint quality improvement initiatives that address specific procedural challenges and enhance therapeutic endoscopy capabilities.
How Research Institutions Could Enable Technology Advancement?
- Device Technology Research: Conduct scientific investigations into balloon catheter mechanics, stone extraction dynamics, tissue interaction phenomena, and procedural optimization principles that advance fundamental understanding and enable breakthrough innovations in therapeutic endoscopy devices.
- Material Science Development: Develop novel catheter materials, advanced balloon polymers, biocompatible coatings, and functional surface modifications that improve device performance while enhancing clinical safety and procedural outcomes in biliary interventions.
- Clinical Outcomes Studies: Investigate comparative effectiveness of balloon catheter designs, procedural technique variations, and patient outcome predictors that create evidence foundations for clinical practice guidelines and inform device development priorities.
- Advanced Characterization Capabilities: Provide specialized testing services utilizing mechanical property analysis, imaging modalities, biocompatibility assessment, and clinical simulation methodologies that enable detailed understanding of balloon catheter performance and clinical behavior.
- Technology Commercialization Support: Facilitate innovation translation through licensing agreements, startup incubation programs, regulatory guidance, and industry collaborations that accelerate novel device development and strengthen medical device industry advancement.
How Investors and Financial Enablers Could Support Market Growth and Innovation?
- Manufacturing Capacity Investment: Provide capital for medical device production facility construction, cleanroom infrastructure development, and manufacturing technology implementation that address growing market demand while ensuring regulatory compliance and product quality standards.
- Innovation Financing: Fund research and development of advanced balloon catheter technologies, including novel materials, innovative designs, and enhanced functionality that address current clinical limitations and create competitive advantages in therapeutic endoscopy markets.
- Clinical Evidence Development: Support clinical trial programs, registry studies, and real-world evidence generation that demonstrate device safety, validate clinical effectiveness, and support regulatory approvals across global markets and diverse patient populations.
- Market Expansion Enablement: Finance strategic acquisitions, geographic expansion initiatives, distribution network development, and clinical support capabilities that accelerate market penetration and strengthen competitive positions in high-growth regions and emerging markets.
- Emerging Market Development: Provide financing and technical assistance for medical device distribution infrastructure in developing economies, creating local supply capabilities, supporting healthcare access improvement, and expanding global availability of therapeutic endoscopy technologies.
Key Players in the Balloon Catheters for Bile Stone Removal Market
- Zeon
- Boston Scientific
- Medtronic
- Cook Medical
- CONMED
- Micro-Tech Endoscopy
- Medorah Medical
- Penlon
- Edwards Lifesciences
- B. Braun
Scope of the Report

| Metric | Value |
|---|---|
| Quantitative Units | USD 742.70 Million to USD 1,342.66 Million, at a CAGR of 6.10% |
| Market Definition | Balloon catheters for bile stone removal are specialized ERCP devices with inflatable tips in single, double, and triple-lumen configurations for common bile duct stone extraction, contrast injection, and guidewire passage during minimally invasive biliary interventions. |
| Segmentation | Lumen Type: Single-lumen Balloon Catheter, Double-lumen Balloon Catheter, and Triple-lumen Balloon Catheter; Application: Public Hospital and Private Hospital |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia, Oceania, Middle East & Africa |
| Countries Covered | China, India, Germany, Brazil, USA, UK, Japan, and 40 plus countries |
| Key Companies Profiled | Zeon, Boston Scientific, Medtronic, Cook Medical, CONMED, Micro-Tech Endoscopy, Medorah Medical, Penlon, Edwards Lifesciences, B. Braun |
| Forecast Period | 2026 to 2036 |
| Approach | Forecasting models apply a bottom-up methodology starting with installed base metrics and projecting conversion rates to technology adoption. |
Balloon Catheters for Bile Stone Removal Market by Segments
Lumen Type:
- Single-lumen Balloon Catheter
- Double-lumen Balloon Catheter
- Triple-lumen Balloon Catheter
Application:
- Public Hospital
- Private Hospital
Region:
East Asia
- China
- Japan
- South Korea
- Taiwan
- Rest of East Asia
Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Netherlands
- Nordic
- BENELUX
- Rest of Europe
North America
- United States
- Canada
- Mexico
South Asia
- India
- Pakistan
- Bangladesh
- Rest of South Asia
Latin America
- Brazil
- Argentina
- Chile
- Rest of Latin America
Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Other African Union
- Rest of Middle East & Africa
Eastern Europe
- Russia
- Poland
- Czech Republic
- Rest of Eastern Europe
Bibliography
- 1. World Health Organization. (2024). Global Status Report on Digestive Diseases: Cholelithiasis Prevalence and Treatment Access. WHO.
- 2. European Society of Gastrointestinal Endoscopy. (2024). ERCP Procedure Guidelines: Device Selection and Infection Control Standards. ESGE.
- 3. American Society for Gastrointestinal Endoscopy. (2024). ERCP Quality Indicators and Device Reprocessing Standards. ASGE.
- 4. National Health Commission of China. (2024). County Hospital Capacity Building: Digestive Endoscopy Centre Expansion Targets. NHC.
- 5. Indian Council of Medical Research. (2024). Hepatobiliary Disease Burden: Epidemiological Survey and Treatment Access Report. ICMR.
- 6. National Institute for Health and Care Excellence. (2024). Interventional Procedure Guidance: ERCP for Bile Duct Stones. NICE.
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary research documentation.
Frequently Asked Questions
How large is the demand for Balloon Catheters for Bile Stone Removal in the global market in 2026?
Demand for balloon catheters for bile stone removal in the global market is estimated to be valued at USD 742.70 million in 2026.
What will be the market size of Balloon Catheters for Bile Stone Removal by 2036?
Market size for balloon catheters for bile stone removal is projected to reach USD 1,342.66 million by 2036.
What is the expected CAGR between 2026 and 2036?
Demand for balloon catheters for bile stone removal is expected to grow at a CAGR of 6.10% between 2026 and 2036.
Which Lumen Type is poised to lead global sales by 2026?
Single-lumen Balloon Catheter accounts for 41.0% share in 2026 reflecting cost-effectiveness and procedural simplicity for standard bile duct stone extraction.
How is Public Hospital positioned as an Application in 2026?
Public Hospital represents 55.0% share in 2026, serving the majority of ERCP referral volumes and generating the largest institutional procurement orders.
What is driving demand in China?
Digestive endoscopy centre expansion across county-level hospitals and hepatobiliary screening referral volumes fuel the fastest growth at 8.2% CAGR through 2036.
What is included in the scope of this report?
The market covers single, double, and triple-lumen balloon extraction catheters in disposable and limited-reuse configurations across public and private hospital ERCP applications.
What is excluded from the scope of this report?
Lithotripsy catheters, biliary stents, sphincterotomy devices, diagnostic ERCP catheters, and endoscope capital equipment are excluded.
Table of Content
- Executive Summary
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Lumen Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Lumen Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Lumen Type , 2026 to 2036
- Single-lumen Balloon Catheter
- Double-lumen Balloon Catheter
- Triple-lumen Balloon Catheter
- Single-lumen Balloon Catheter
- Y to o to Y Growth Trend Analysis By Lumen Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Lumen Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application, 2026 to 2036
- Public Hospital
- Private Hospital
- Public Hospital
- Y to o to Y Growth Trend Analysis By Application, 2021 to 2025
- Absolute $ Opportunity Analysis By Application, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Lumen Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Lumen Type
- By Application
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Lumen Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Lumen Type
- By Application
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Lumen Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Lumen Type
- By Application
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Lumen Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Lumen Type
- By Application
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Lumen Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Lumen Type
- By Application
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Lumen Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Lumen Type
- By Application
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Lumen Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Lumen Type
- By Application
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Lumen Type
- By Application
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Lumen Type
- By Application
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Lumen Type
- By Application
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Lumen Type
- By Application
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Lumen Type
- By Application
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Lumen Type
- By Application
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Lumen Type
- By Application
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Lumen Type
- By Application
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Lumen Type
- By Application
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Lumen Type
- By Application
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Lumen Type
- By Application
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Lumen Type
- By Application
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Lumen Type
- By Application
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Lumen Type
- By Application
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Lumen Type
- By Application
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Lumen Type
- By Application
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Lumen Type
- By Application
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Lumen Type
- By Application
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Lumen Type
- By Application
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Lumen Type
- By Application
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Lumen Type
- By Application
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Lumen Type
- By Application
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Lumen Type
- By Application
- Competition Analysis
- Competition Deep Dive
- Zeon
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Boston Scientific
- Medtronic
- Cook Medical
- CONMED
- Micro-Tech Endoscopy
- Medorah Medical
- Penlon
- Edwards Lifesciences
- B. Braun
- Zeon
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Lumen Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Lumen Type , 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by Lumen Type , 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by Lumen Type , 2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Lumen Type , 2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by Lumen Type , 2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Lumen Type , 2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Lumen Type , 2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by Application, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Lumen Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Lumen Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Lumen Type
- Figure 6: Global Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Application
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by Lumen Type , 2026 and 2036
- Figure 21: North America Market Y-o-Y Growth Comparison by Lumen Type , 2026-2036
- Figure 22: North America Market Attractiveness Analysis by Lumen Type
- Figure 23: North America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 25: North America Market Attractiveness Analysis by Application
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by Lumen Type , 2026 and 2036
- Figure 28: Latin America Market Y-o-Y Growth Comparison by Lumen Type , 2026-2036
- Figure 29: Latin America Market Attractiveness Analysis by Lumen Type
- Figure 30: Latin America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 31: Latin America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 32: Latin America Market Attractiveness Analysis by Application
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by Lumen Type , 2026 and 2036
- Figure 35: Western Europe Market Y-o-Y Growth Comparison by Lumen Type , 2026-2036
- Figure 36: Western Europe Market Attractiveness Analysis by Lumen Type
- Figure 37: Western Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 38: Western Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 39: Western Europe Market Attractiveness Analysis by Application
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Lumen Type , 2026 and 2036
- Figure 42: Eastern Europe Market Y-o-Y Growth Comparison by Lumen Type , 2026-2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by Lumen Type
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 45: Eastern Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by Application
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by Lumen Type , 2026 and 2036
- Figure 49: East Asia Market Y-o-Y Growth Comparison by Lumen Type , 2026-2036
- Figure 50: East Asia Market Attractiveness Analysis by Lumen Type
- Figure 51: East Asia Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 52: East Asia Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 53: East Asia Market Attractiveness Analysis by Application
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Lumen Type , 2026 and 2036
- Figure 56: South Asia and Pacific Market Y-o-Y Growth Comparison by Lumen Type , 2026-2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Lumen Type
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 59: South Asia and Pacific Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by Application
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Lumen Type , 2026 and 2036
- Figure 63: Middle East & Africa Market Y-o-Y Growth Comparison by Lumen Type , 2026-2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Lumen Type
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 66: Middle East & Africa Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

