Blow-Fill-Seal Technology Market Size, Market Forecast and Outlook By FMI
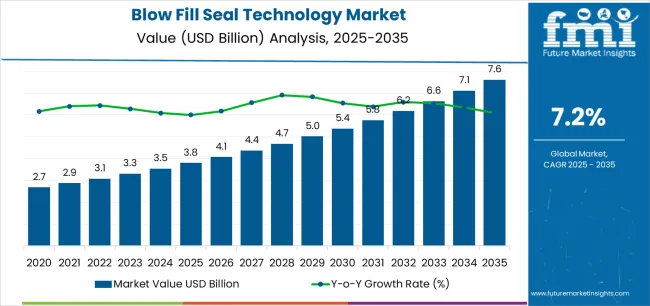
In 2025, the blow-fill-seal technology market was valued at USD 5,300 million. Based on Future Market Insights’ analysis, demand for blow-fill-seal technology is estimated to grow to USD 6,200 million in 2026 and USD 10,400 million by 2036. FMI projects a CAGR of 5.3% during the forecast period.
Summary of Blow-Fill-Seal Technology Market
- Market Definition
- Blow-fill-seal technology enables inline extrusion-fill-seal sterile packaging platforms supporting high-volume production of preservative-free ophthalmic therapies, respiratory nebulizer solutions, saline flush products, and irrigation liquids across pharmaceutical manufacturing environments.
- Demand Drivers in the Market
- Increasing production of preservative-free ophthalmic therapies continues accelerating adoption of BFS ampoule packaging formats globally.
- Localization of respiratory therapy manufacturing across Asia Pacific is strengthening deployment of single-dose BFS packaging infrastructure supporting nebulizer solution production.
- Hospital transition toward ready-to-use saline flush packaging formats is reinforcing demand for BFS container manufacturing platforms across inpatient treatment environments.
- Key Segments Analyzed in the FMI Report
- By Container Size: Containers below 0.2 L dominate demand due to widespread adoption across preservative-free ophthalmic therapies and respiratory nebulizer solutions.
- By Geography: Asia Pacific represents maximum growth opportunity driven by ophthalmic export manufacturing expansion and respiratory therapy localization across India and China.
- Analyst Opinion at FMI
- , Principal Consultant for Packaging Industry at Future Market Insights, opines, “Stakeholders will find in this report strategic clarity on how ophthalmic export manufacturing expansion and hospital unit-dose packaging adoption are redefining sterile liquid packaging infrastructure investment priorities globally.”
- Strategic Implications / Executive Takeaways
- Prioritize BFS deployment across ophthalmic and respiratory therapy packaging portfolios supporting single-dose sterile delivery formats.
- Align saline flush packaging infrastructure with ready-to-use injectable hospital procurement strategies.
- Strengthen polymer compatibility validation programs supporting injectable support liquid packaging transitions from glass to polymer containers.
- Methodology
- Market estimates were validated through interviews with sterile packaging equipment suppliers and contract manufacturing organizations.
- Secondary research included pharmaceutical manufacturing infrastructure expansion disclosures and respiratory therapy packaging demand indicators.
- Forecasts were developed using therapy-class packaging consumption modeling aligned with ophthalmic export manufacturing expansion trends.
The market is expected to generate an absolute dollar growth of USD 4.2 billion between 2026 and 2036. This represents steady structural expansion supported by increasing production of preservative-free ophthalmic therapies, localization of respiratory nebulizer solution manufacturing, and adoption of ready-to-use saline flush packaging across hospital supply chains. Growth is further reinforced by expansion of sterile contract manufacturing infrastructure across Asia Pacific supporting export-oriented ophthalmic formulation production. However, extractables-leachables qualification requirements and oxygen permeability sensitivity continue limiting BFS deployment across biologics packaging applications traditionally served by glass vial filling platforms. Blow-fill-seal technology continues to support reliable single-dose sterile packaging solutions across ophthalmic and respiratory therapies where contamination control and high-throughput production consistency remain critical manufacturing priorities.
Dr. Uwe Hanenberg, Head of Product Development at Recipharm, commented: “With the addition of these new product development and pilot scale capabilities, alongside our ReciPredict Platform, we can now offer an even faster, cost-efficient and fully integrated solution for oral solids from clinical development to commercialisation.”
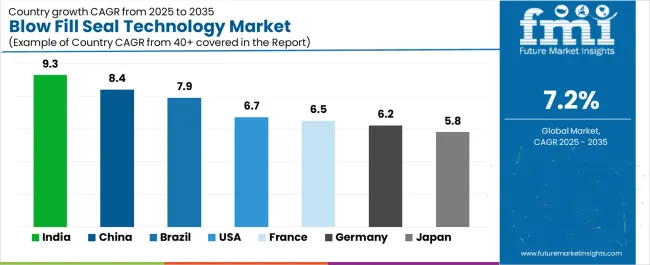
India (8.2%) is projected to register the fastest growth, driven by expansion of ophthalmic export manufacturing and increasing localization of respiratory therapy production across domestic sterile formulation facilities. China (7.4%) follows, supported by large-scale sterile generic manufacturing capacity and strengthening aseptic compliance alignment across export-oriented injectable production clusters. Brazil (6.3%) benefits from modernization of hospital procurement systems and increasing adoption of ready-to-use saline flush and irrigation packaging formats. The United States (4.9%) and Germany (4.4%) represent mature markets where growth is largely replacement-driven, supported by steady respiratory therapy demand and established ophthalmic manufacturing infrastructure rather than rapid expansion of new sterile packaging installations.
Blow-Fill-Seal Technology Market Definition
Blow-fill-seal technology refers to an automated aseptic packaging process in which polymer containers are formed, filled, and sealed within a continuous sterile enclosure without intermediate container handling steps. The technology eliminates container washing, depyrogenation tunnels, and stopper sterilization workflows required in conventional glass vial filling environments. BFS technology is widely deployed across preservative-free ophthalmic therapies, respiratory nebulizer solutions, sterile irrigation liquids, saline flush products, and vaccine diluent packaging where contamination-resilient single-dose delivery formats are required across pharmaceutical manufacturing environments.
Blow-Fill-Seal Technology Market Inclusions
Our report provides global and regional market size estimates for 2025 and 2026 with forecasts through 2036 and CAGR analysis. It includes segmentation by product type, machine configuration, container size, material type, end-use application, and region. The study covers sterile manufacturing infrastructure expansion trends, ophthalmic export manufacturing growth, respiratory therapy localization initiatives, and hospital injectable packaging transitions influencing BFS deployment across pharmaceutical supply chains.
Blow-Fill-Seal Technology Market Exclusions
The scope excludes conventional glass vial filling systems, prefilled syringe assembly platforms, blister packaging technologies, and oral solid-dose packaging formats. Upstream polymer resin production is excluded unless directly associated with BFS extrusion container manufacturing. Finished pharmaceutical formulation markets are excluded except where sterile packaging format selection influences manufacturing infrastructure investment decisions.
Blow-Fill-Seal Technology Market Research Methodology
- Primary Research: Interviews were conducted with sterile packaging equipment suppliers, pharmaceutical manufacturers, ophthalmic formulation producers, respiratory therapy packaging specialists, and contract development and manufacturing organizations operating BFS platforms.
- Desk Research: Regulatory publications, pharmaceutical infrastructure investment announcements, respiratory therapy packaging adoption indicators, ophthalmic export manufacturing disclosures, and sterile fill-finish expansion updates were reviewed.
- Market-Sizing and Forecasting: A hybrid top-down and bottom-up approach reconstructed demand using ophthalmic therapy output volumes, respiratory nebulizer packaging demand indicators, saline flush consumption estimates, and irrigation solution manufacturing benchmarks.
- Data Validation and Update Cycle: Market estimates were cross-verified across equipment installation benchmarks, therapy-class packaging demand indicators, and pharmaceutical sterile manufacturing expansion disclosures prior to structured expert validation.
Key Segments
Blow-Fill-Seal Technology Market Analysis by Product Type
Based on FMI's Blow-Fill-Seal Technology market report, consumption of Ampoules is estimated to hold the leading share in 2026. Ampoule formats dominate due to their compatibility with preservative-free ophthalmic therapies and respiratory unit-dose nebulizer solutions manufactured primarily using low-density polyethylene (LDPE) extrusion resins. These single-dose container architectures support contamination-resilient sterile delivery workflows while enabling continuous polymer consumption efficiency across high-volume pharmaceutical packaging environments. Ampoule-based BFS packaging is widely deployed across export-oriented ophthalmic formulation production and inhalation therapy manufacturing programs requiring precise dose control and break-free container handling compared with glass alternatives.
- Preservative-Free Ophthalmic Compatibility: Single-dose LDPE ampoules are widely used for preservative-free eye-drop formulations requiring sterile packaging architectures aligned with export-grade pharmaceutical manufacturing standards.
- Respiratory Therapy Packaging Alignment: Unit-dose nebulizer solution containers rely on BFS ampoule formats supporting controlled dosing accuracy and contamination-resilient administration workflows.
- Material Consumption Efficiency: Continuous LDPE extrusion across multi-cavity BFS tooling supports stable polymer utilization efficiency across large-volume sterile liquid packaging production environments.
Blow-Fill-Seal Technology Market Analysis by Material Type
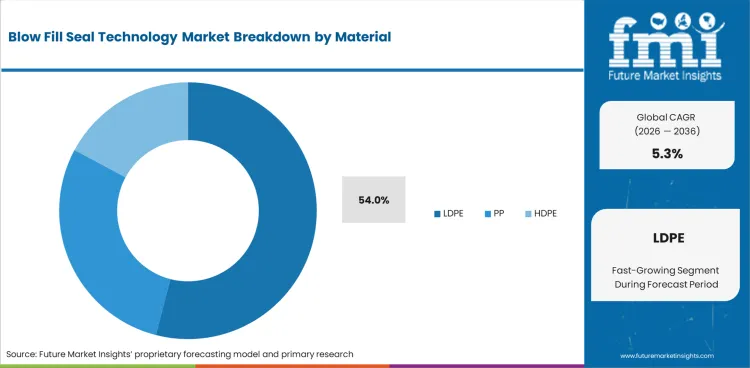
Based on FMI's Blow-Fill-Seal Technology market report, consumption of Polyethylene materials is estimated to hold the leading share in 2026. Polyethylene-based extrusion resins, particularly low-density polyethylene (LDPE) accounts for more than 54% share due to their compatibility with preservative-free ophthalmic ampoules, respiratory unit-dose containers, and saline flush packaging formats requiring breakage-resistant sterile container architectures. These materials support continuous extrusion during inline container formation while enabling lightweight logistics handling across export-oriented pharmaceutical manufacturing environments.
- Preservative-Free Ophthalmic Compatibility: LDPE containers are widely used across single-dose ophthalmic therapies requiring contamination-resilient sterile packaging aligned with global pharmaceutical export manufacturing standards.
- Respiratory Therapy Packaging Alignment: Unit-dose nebulizer solution containers are primarily produced using polyethylene extrusion formats supporting stable polymer consumption across high-volume inhalation therapy production environments.
- Hospital Injectable Support Liquid Applications: HDPE-based saline flush containers between 10 mL and 100 mL are increasingly deployed across hospital supply chains supporting ready-to-use injectable packaging transitions away from glass alternatives.
Blow-Fill-Seal Technology Market Analysis by Container Size
Based on FMI's Blow-Fill-Seal Technology market report, consumption of containers below 0.2 L is estimated to hold the leading share in 2026. Sub-0.2 L container formats dominate due to widespread adoption across preservative-free ophthalmic therapies and respiratory unit-dose nebulizer solutions manufactured primarily using LDPE extrusion containers supporting single-dose sterile delivery workflows across pharmaceutical manufacturing environments.
- Ophthalmic Therapy Packaging Dominance: Single-dose ophthalmic ampoules typically ranging between 0.3 mL and 10 mL represent the largest global BFS container category supporting preservative-free ocular drug administration.
- Respiratory Nebulizer Solution Alignment: Unit-dose inhalation therapy containers between 2 mL and 5 mL continue supporting expansion of BFS packaging infrastructure across respiratory treatment supply chains.
- Export Manufacturing Deployment: Asia Pacific pharmaceutical formulation clusters increasingly deploy sub-0.2 L BFS packaging platforms supporting global sterile eye-drop and respiratory therapy export programs
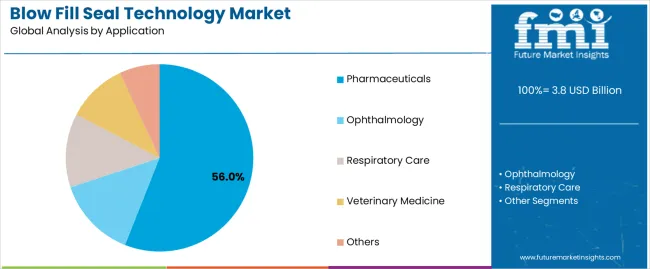
Blow-Fill-Seal Technology Market Analysis by End-Use Application
Based on FMI's Blow-Fill-Seal Technology market report, consumption of Ophthalmic applications is estimated to hold 37% share in 2026. The ophthalmic segment leads due to strong demand for preservative-free single-dose packaging formats supporting sterile ocular therapy administration across global pharmaceutical supply chains.
- Respiratory Therapy Localization: Increasing nebulizer therapy adoption supports expansion of BFS packaging infrastructure across respiratory solution manufacturing environments.
- Saline Flush Adoption: Hospital procurement systems continue transitioning toward ready-to-use injectable packaging formats supporting BFS container demand.
- Single-Dose Delivery Compliance: Preservative-free ophthalmic therapies increasingly rely on BFS ampoule formats supporting contamination-resilient administration workflows.
Blow-Fill-Seal Technology Market: Drivers, Restraints, and Opportunities
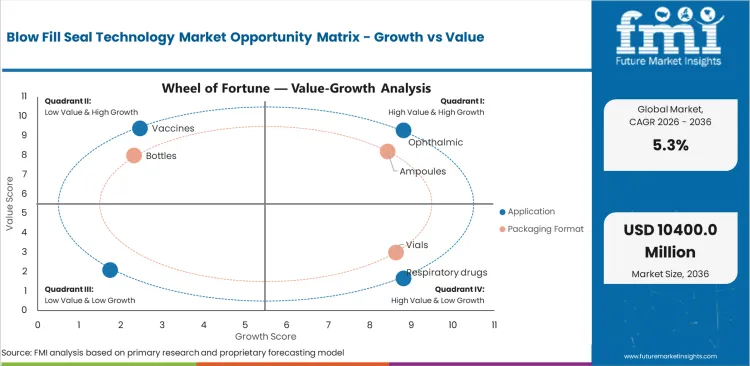
Future Market Insights analysis indicates the blow-fill-seal technology market is structurally expanding as pharmaceutical manufacturers transition toward contamination-resilient single-dose packaging architectures supporting ophthalmic therapies, respiratory nebulizer solutions, and hospital injectable support liquids. The market exists at its current scale because BFS technology enables inline container formation, filling, and sealing within closed sterile environments supporting high-volume production efficiency across liquid pharmaceutical supply chains.
FMI analysts observe a transition from preservative-stabilized multi-dose packaging toward single-dose sterile delivery formats across ophthalmic and respiratory therapy portfolios. While glass vial platforms remain dominant across biologics packaging applications, BFS containers continue expanding across injectable support liquid packaging environments requiring breakage resistance and simplified logistics handling. Advanced BFS container architectures carry higher installation qualification requirements but support long-term cost efficiency across high-volume sterile packaging production environments.
- Ophthalmic Export Manufacturing Expansion: Asia Pacific pharmaceutical manufacturers continue strengthening preservative-free ophthalmic production infrastructure supporting global BFS container demand growth.
- Respiratory Therapy Localization: Expansion of nebulizer therapy manufacturing across India and China continues supporting installation of BFS platforms aligned with single-dose respiratory packaging requirements.
- Hospital Injectable Standardization: Transition toward ready-to-use saline flush packaging across hospital supply chains continues supporting deployment of BFS container manufacturing platforms.
Regional Analysis
Region Introduction
The global blow-fill-seal technology market is segmented across Asia Pacific, North America, Europe, and Latin America. Regional growth varies based on ophthalmic export manufacturing expansion, respiratory therapy localization initiatives, hospital injectable packaging transitions, and sterile pharmaceutical infrastructure modernization trends. The full report provides comprehensive regional market attractiveness analysis.
.webp)
| Country |
CAGR |
| USA |
4.9% |
| Germany |
4.4% |
| China |
7.4% |
| India |
8.2% |
| Brazil |
6.3% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Asia Pacific - Ophthalmic Export Manufacturing Growth Engine
Asia Pacific serves as the primary growth engine for BFS deployment supported by expansion of ophthalmic export manufacturing infrastructure and respiratory therapy localization across India and China.
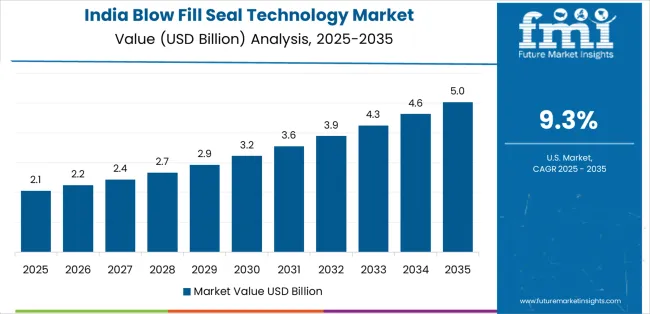
- India: Demand for blow-fill-seal technology in India is projected to rise at 8.2% CAGR through 2036. Growth is driven by expansion of ophthalmic export manufacturing aligned with regulatory-compliant sterile formulation production infrastructure and increasing localization of respiratory therapy packaging capacity supporting domestic inhalation solution supply chains.
- China: Demand for blow-fill-seal technology in China is projected to rise at 7.4% CAGR through 2036. The market benefits from large-scale sterile generic manufacturing capacity and strengthening aseptic production compliance alignment supporting export-oriented injectable production clusters.
FMI's analysis of blow-fill-seal technology market in Asia Pacific consists of country-wise assessment that includes India and China. Readers can find insights on ophthalmic export manufacturing expansion and respiratory therapy localization trends.
North America - Hospital Unit-Dose Packaging Adoption Hub
North America represents a mature but innovation-driven BFS deployment region supported by respiratory therapy packaging demand and hospital injectable supply chain modernization initiatives.
- United States: Demand for blow-fill-seal technology in the United States is projected to rise at 4.9% CAGR through 2036. Growth is supported by increasing adoption of ready-to-use saline flush packaging across hospital supply chains and expansion of respiratory therapy unit-dose delivery infrastructure across outpatient treatment environments. FMI's analysis of blow-fill-seal technology market in North America consists of country-wise assessment that includes the United States. Readers can find insights on hospital injectable packaging trends and respiratory therapy infrastructure expansion.
Europe - Sterile Packaging Equipment Innovation Center
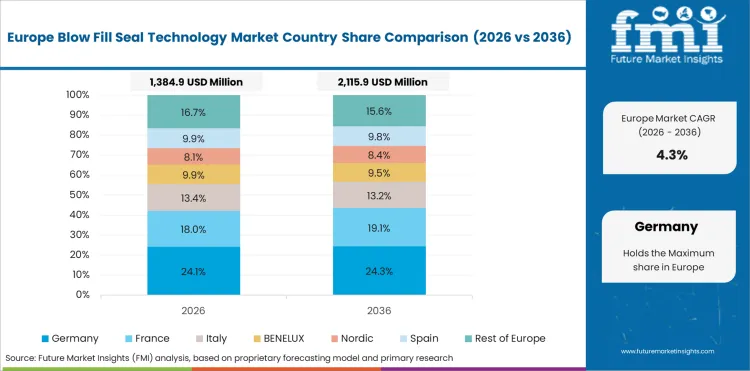
Europe functions as a leading BFS deployment region supported by strong ophthalmic formulation manufacturing clusters and the presence of specialized aseptic packaging equipment suppliers.
- Germany: Demand for blow-fill-seal technology in Germany is projected to rise at 4.4% CAGR through 2036. Growth is influenced by continued investments in sterile fill-finish infrastructure modernization supporting ophthalmic formulation packaging capacity across European pharmaceutical manufacturing networks. The report consists of country-wise assessment that includes Germany along with other EU-5 countries. Readers can find insights on ophthalmic therapy packaging capacity expansion and sterile manufacturing infrastructure modernization.
Latin America - Expanding Hospital Injectable Packaging Infrastructure Region
Latin America represents an emerging BFS deployment region supported by modernization of hospital injectable procurement systems and expansion of regional pharmaceutical manufacturing capacity.
- Brazil: Demand for blow-fill-seal technology in Brazil is projected to rise at 6.3% CAGR through 2036. Growth is supported by expansion of sterile pharmaceutical manufacturing infrastructure serving public healthcare procurement programs and increasing adoption of ready-to-use saline flush packaging formats.
Competitive Aligners for Market Players
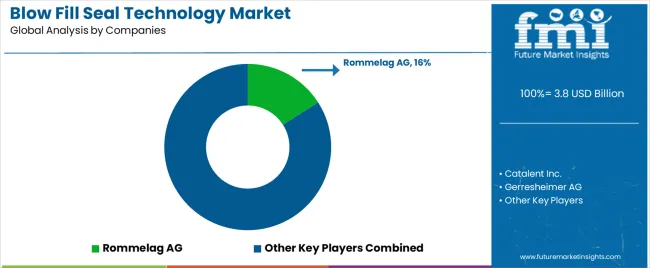
The blow-fill-seal technology market is moderately consolidated and led by specialized aseptic packaging equipment suppliers with established installation validation track records across pharmaceutical sterile manufacturing environments. Companies operating BFS platforms benefit from high switching costs associated with tooling qualification, enclosure validation requirements, and therapy-specific sterile production infrastructure alignment.
Equipment suppliers maintain structural advantages through multi-cavity tooling capability, container geometry flexibility, and compatibility with ophthalmic and respiratory therapy packaging workflows. Contract development and manufacturing organizations strengthen competitive positioning through ophthalmic specialization, respiratory therapy packaging infrastructure scale, and export-market regulatory inspection readiness.
Buyer leverage varies across therapy classes. Large ophthalmic manufacturers typically deploy captive BFS infrastructure supporting preservative-free formulation production environments, while emerging respiratory therapy developers increasingly rely on CDMO outsourcing strategies supporting flexible sterile packaging deployment. As hospital injectable supply chains transition toward ready-to-use saline flush packaging architectures, pharmaceutical manufacturers increasingly prioritize suppliers capable of supporting high-throughput BFS container production aligned with contamination-resilient packaging requirements.
Recent Developments
- In March 2026, Ritedose advanced BFS through key innovation steps including investment in advanced BFS capabilities, expansion of manufacturing capacity, enhancement of automation, development of capabilities for complex formulations, and strengthening of quality systems and infrastructure. These innovations enable reliable large-scale production, support advanced respiratory and ophthalmic therapies, and position Ritedose as a high-quality, scalable, and regulatory-compliant CDMO partner.
- In June, 2025, Recipharm, a leading global contract development and manufacturing organisation (CDMO), has secured major product development contracts, further solidifying its leadership in Blow-Fill-Seal (BFS) technology. Recipharm, also announced development of its new Pilot Scale Development Centre in Germany. Complements existing product development and manufacturing capabilities in oral solid, sterile fill & finish and biologics
Key Players
- Rommelag Group
- Weiler Engineering Inc.
- Unither Pharmaceuticals
- Recipharm AB
- Curida AS
- The Ritedose Corporation
- Catalent Inc.
- Takeda Pharmaceutical Company Limited
Report Scope

| Metric |
Value |
| Quantitative Units |
USD 5.3 billion (2025); USD 6.2 billion (2026) to USD 10.4 billion (2036), at a CAGR of 5.3% |
| Market Definition |
The blow-fill-seal technology market comprises automated sterile extrusion-fill-seal packaging platforms supporting production of ophthalmic therapies, respiratory nebulizer solutions, saline flush products, irrigation liquids, and vaccine diluent containers across pharmaceutical manufacturing environments. |
| Form Segmentation |
Ampoules; Bottles; Vials and Others |
| End Use Segmentation |
Ophthalmics; Respiratory Therapies; Injectable Support Fluids; Vaccines & Biologics Support Liquids; Irrigation & Saline Solutions |
| Application Coverage |
Preservative-free ophthalmic therapies; nebulizer respiratory solutions; saline flush packaging; irrigation liquids; injectable support liquids |
| Regions Covered |
North America; Latin America; East Asia; South Asia & Pacific; Eastern Europe; Western Europe; MEA |
| Countries Covered |
United States; Germany; China; India; Brazil; Japan; France; and 40+ countries |
| Key Companies Profiled |
Rommelag Group; Weiler Engineering Inc.; Unither Pharmaceuticals; Recipharm AB; Curida AS; The Ritedose Corporation; Catalent Inc.; Others |
| Forecast Period |
2026 to 2036 |
| Approach |
Hybrid top-down and bottom-up market modeling validated through interviews with sterile packaging equipment suppliers, pharmaceutical manufacturers, ophthalmic formulation producers, respiratory therapy packaging specialists, and contract development organizations supported by therapy-class packaging consumption benchmarking. |
Blow Fill Seal Technology Market by Segments
By Product Type:
- Vials
- Ampoules
- Bottles
- Others
By Application:
- Pharmaceuticals
- Ophthalmology
- Respiratory Care
- Veterinary Medicine
- Others
By Container Capacity:
- Small Volume (Less than 10ml)
- Medium Volume (10-100ml)
- Large Volume (Above 100ml)
By Material Type:
- Polyethylene (PE)
- Polypropylene (PP)
- Others
By End-Use Industry:
- Pharmaceutical Manufacturing
- Contract Packaging
- Biotechnology
- Others
By Region:
- North America
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Nordic
- BENELUX
- Rest of Europe
- East Asia
- China
- Japan
- South Korea
- Taiwan
- South Asia & Pacific
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia & Pacific
- Latin America
- Brazil
- Chile
- Argentina
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Other African Union
- Rest of Middle East & Africa
Bibilography
- European Medicines Agency (EMA), Sterile Manufacturing and Aseptic Processing Guidance for Medicinal Products
- USA Food and Drug Administration (FDA), Guidance for Industry - Sterile Drug Products Produced by Aseptic Processing
- World Health Organization (WHO), Technical Report Series on Sterile Pharmaceutical Production and Aseptic Processing
- European Commission, EU Guidelines for Good Manufacturing Practice - Annex 1 Manufacture of Sterile Medicinal Products (2022 Revision)
- International Council for Harmonisation (ICH), Q9 Quality Risk Management and Q10 Pharmaceutical Quality System Guidelines
- International Organization for Standardization (ISO), ISO 13408 Aseptic Processing of Healthcare Products - General Requirement
- ASTM International, ASTM F1980 Standard Guide for Accelerated Aging of Sterile Barrier System
- Parenteral Drug Association (PDA), Technical Reports on Aseptic Processing and Sterile Fill-Finish Manufacturing
- International Society for Pharmaceutical Engineering (ISPE), Baseline Guide Volume 3 - Sterile Product Manufacturing Facilities
- Healthcare Packaging Association (HPA), Sterile Packaging Industry Resources and Compliance Guidanc
- European Federation for Pharmaceutical Sciences (EUFEPS), Sterile Drug Manufacturing Best Practice References
- Organisation for Economic Co-operation and Development (OECD), Health Statistics - Pharmaceutical Manufacturing Indicators
- United Nations Comtrade Database, Trade Statistics for Pharmaceutical Preparations and Sterile Liquid Packaging Input
- World Bank, Pharmaceutical Manufacturing Infrastructure Development Indicator
- Centers for Disease Control and Prevention (CDC), Respiratory Therapy Utilization and Healthcare Infrastructure Statistics
This Report Addresses
- Strategic market intelligence enabling long-term sterile packaging infrastructure planning across pharmaceutical manufacturing ecosystems
- Global market size estimation with 2026-2036 revenue forecasts validated through therapy-class container consumption benchmarking
- Growth opportunity assessment across ophthalmic, respiratory therapy, saline flush, and irrigation solution packaging applications
- Container-format demand mapping across ampoules, bottles, and vials used in BFS sterile extrusion-fill-seal environments
- Material consumption analysis across LDPE and HDPE extrusion container manufacturing supporting polymer-based sterile packaging transitions
- Regional outlook assessment covering Asia Pacific, North America, Europe, and Latin America with country-level sterile manufacturing expansion drivers
- Competitive benchmarking of BFS equipment suppliers and contract development manufacturing organizations based on sterile fill-finish capability positioning
- Application-level adoption insights supporting transition from glass vial packaging toward single-dose polymer container architectures across selected sterile liquid therapy classes
Frequently Asked Questions
How large is the Blow-Fill-Seal Technology Market in 2026?
The blow-fill-seal technology market is valued at USD 6.2 billion in 2026.
What was the market size in 2025?
The market was valued at USD 5.3 billion in 2025.
What will the market size be by 2036?
The market is projected to reach USD 10.4 billion by 2036.
What is the expected CAGR during the forecast period?
The market is expected to grow at a CAGR of 5.3% between 2026 and 2036.
Which container format plays a major role in the Blow-Fill-Seal Technology Market?
Single-dose ampoules play a major role due to widespread adoption across preservative-free ophthalmic therapies and respiratory nebulizer solution packaging.
Which material type dominates BFS container production?
Polyethylene materials, particularly LDPE and HDPE, dominate BFS container manufacturing due to compatibility with sterile extrusion-fill-seal packaging workflows.
Which application segment drives the largest demand for BFS packaging?
Ophthalmic applications represent the leading demand segment supported by preservative-free single-dose sterile eye-drop packaging requirements.




.webp)





