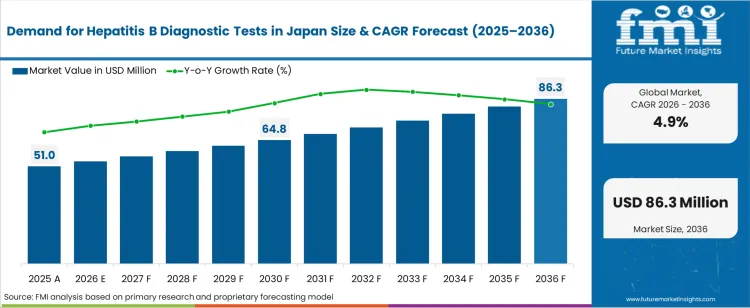
The demand for hepatitis b diagnostic tests in japan was valued at USD 51.00 million in 2025, projected to reach USD 53.50 million in 2026, and is forecast to expand to USD 86.32 million by 2036 at a 4.9% CAGR. Japan's national hepatitis elimination strategy and the mandatory screening requirements for blood donors, pregnant women, and surgical patients are sustaining consistent testing volumes across hospital and diagnostic laboratory networks. The Ministry of Health, Labour and Welfare's subsidised hepatitis screening programme continues to expand its reach into workplace health examinations and community screening events, increasing the addressable testing population beyond traditional clinical settings.
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 53.50 million |
| Industry Value (2036) | USD 86.32 million |
| CAGR (2026 to 2036) | 4.9% |
Source: Future Market Insights, 2026
The incremental opportunity between 2026 and 2036 amounts to USD 32.82 million. As per FMI, the growth trajectory reflects the combination of expanded screening mandates and the higher per-test cost of quantitative HBV DNA assays replacing qualitative serology in treatment monitoring. Japan's universal health insurance reimbursement for hepatitis B surface antigen testing and HBV viral load quantification provides a stable institutional procurement floor. The transition from manual ELISA workflows to automated immunoassay platforms is increasing throughput while concentrating reagent procurement among fewer high-volume analyser manufacturers.
Sub-national growth patterns reflect regional screening programme intensity and hospital laboratory infrastructure. Kyushu & Okinawa at 6.1%, Kanto at 5.6%, Kansai at 4.9%, Chubu at 4.3%, Tohoku at 3.8%, Rest of Japan at 3.6%. Kyushu and Okinawa lead due to higher hepatitis B prevalence rates relative to the national average and active prefectural health department screening subsidies. Kanto follows, anchored by the Tokyo metropolitan area's concentration of high-throughput reference laboratories processing specimens from employer-mandated health examinations. Kansai benefits from Osaka's diagnostic reagent manufacturing cluster supporting rapid test kit distribution, while Chubu and Tohoku reflect incremental gains from rural community screening programme expansion.
Hepatitis B diagnostic tests are in vitro diagnostic products used to detect hepatitis B virus infection markers in blood and serum specimens. The Japan market covers enzyme immunoassay kits and point-of-care testing kits used for hepatitis B surface antigen detection, antibody screening, and viral load quantification. These tests are performed across hospital laboratories, clinics, diagnostic centres, and home care settings for infection screening, treatment monitoring, and blood safety purposes.
Market scope includes enzyme immunoassay kits (ELISA, CLIA, ECLIA), rapid point-of-care testing kits, and confirmatory molecular diagnostic reagents for hepatitis B virus detection. Calibrators, quality controls, and sample preparation consumables specific to HBV testing workflows are included.
General immunoassay analyser hardware, hepatitis C and HIV diagnostic test kits, liver function biochemistry panels, and liver biopsy equipment are excluded. Hepatitis B therapeutic agents, vaccines, and treatment monitoring services are outside the analytical scope.
The demand for hepatitis B diagnostic tests in Japan is growing due to the increasing focus on early detection, prevention, and management of viral hepatitis. Hepatitis B is a serious liver infection caused by the hepatitis B virus (HBV) that can lead to chronic liver disease, cirrhosis, and liver cancer. As Japan continues to prioritize healthcare and disease prevention, the need for reliable diagnostic tests to detect and monitor hepatitis B is expanding.
A key driver of this growth is Japan's aging population, which is at higher risk for chronic diseases, including liver conditions related to hepatitis B. As the elderly population grows, the demand for hepatitis B screening and diagnostic testing increases, particularly for high-risk individuals such as those with a history of drug use, frequent blood transfusions, or healthcare workers. Early detection through diagnostic tests is crucial for managing the progression of the disease and preventing complications.
The growing focus on public health initiatives and preventative care is contributing to the rise in hepatitis B diagnostic tests. With the increasing availability of affordable and accurate testing methods, such as serological and molecular assays, more individuals are being screened for HBV. Japan’s healthcare system continues to emphasize disease prevention, and as awareness about the importance of early diagnosis increases, the demand for hepatitis B diagnostic tests is expected to grow steadily through 2035.
Demand for hepatitis B diagnostic tests in Japan is segmented by product type and end user. By product type, demand is divided into enzyme immunoassay kits (EIA), point-of-care testing kits, and other diagnostic methods, with enzyme immunoassay kits (EIA) leading at 35%. The demand is also segmented by end user, including hospitals, clinics, diagnostic centers, and home care, with hospitals accounting for 40% of the demand. Regionally, demand is divided into Kanto, Kinki, Chubu, Kyushu & Okinawa, Tohoku, and the Rest of Japan.
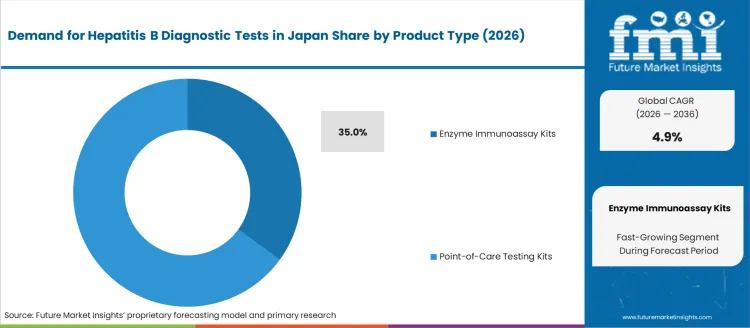
Enzyme immunoassay kits (EIA) account for 35% of the demand for hepatitis B diagnostic tests in Japan due to their high sensitivity, reliability, and widespread use in clinical laboratories. EIA kits are commonly used to detect hepatitis B surface antigens and antibodies, making them an essential tool for both initial screening and confirmation of infection. Their ability to process multiple samples simultaneously and provide accurate, reproducible results makes them the preferred choice for healthcare providers in diagnostic settings. The robust and well-established nature of EIA technology, combined with Japan’s focus on advanced diagnostic capabilities, has solidified its position as the dominant method for hepatitis B testing. As awareness of hepatitis B and the need for early detection grow, EIA kits will continue to lead the industry for hepatitis B diagnostics in Japan.
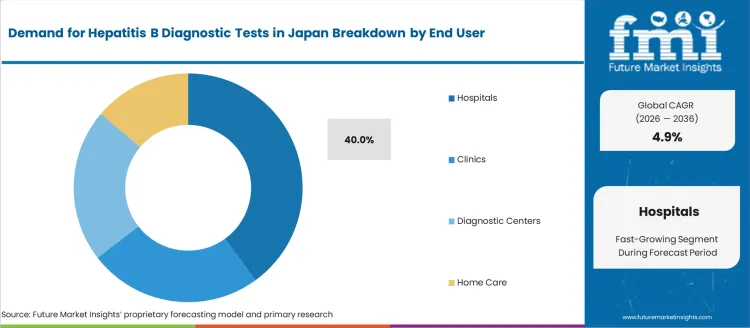
Hospitals account for 40% of the demand for hepatitis B diagnostic tests in Japan, driven by their role as the primary healthcare setting for diagnosing and managing infectious diseases like hepatitis B. Hospitals are equipped with advanced diagnostic tools, including EIA kits, which are necessary for accurately diagnosing hepatitis B in patients. The high volume of patients, particularly those undergoing routine screenings or presenting with liver-related symptoms, contributes to the strong demand for hepatitis B tests in hospital settings. Hospitals also play a critical role in the treatment and ongoing management of hepatitis B patients, necessitating frequent and reliable testing. As the healthcare system in Japan continues to emphasize early detection, prevention, and comprehensive care, hospitals will remain the dominant end user for hepatitis B diagnostic tests.
Demand for Hepatitis B diagnostic tests in Japan is rising driven by the significant number of people living with chronic or undiagnosed HBV infection, and public‑health efforts to detect and treat carriers before they develop serious liver disease. As chronic liver disease, cirrhosis, and liver cancer remain important health burdens linked to HBV, early detection via testing helps enable timely intervention, monitoring, and treatment. Increasing awareness among the general public and medical community about the importance of screening for blood donors, patients prior to surgery or immunosuppressive therapy, pregnant women, and high-risk populations supports demand. On the other hand, stigma, lack of universal screening coverage, and limited routine testing in some workplaces or clinics can restrain widespread uptake.
Why is Demand for Hepatitis B Diagnostic Tests Growing in Japan?
Demand grows because many individuals require testing to know their infection status, especially those with risk factors e.g. older age cohorts (due to historical prevalence), people undergoing surgery or immunosuppressive therapy, pregnant women, or those with elevated liver‑enzyme readings. As treatment and management options for chronic HBV improve, knowing one’s HBV status becomes more relevant for long-term health. National efforts and public‑health policies encourage screening to reduce hepatitis‑related liver disease burden, which encourages more people to seek testing. The fact that some people are asymptomatic yet infected further underscores the importance of diagnostic tests to identify silent carriers before complications arise.
How are Technological & Industry Innovations Driving Hepatitis B Testing Demand in Japan?
Advances in diagnostic technologies such as sensitive antigen/antibody assays, rapid point‑of-care (POC) tests, and improved lab-based immunoassays have made HBV testing more accessible, faster, and reliable. These improvements reduce cost, turnaround time, and logistical barriers, encouraging both patients and healthcare providers to adopt testing. Routine health check-up packages (e.g. for workers, pre‑surgical screening, prenatal care) increasingly include HBV diagnostics, increasing coverage. As awareness grows and guidelines recommend regular screening in certain populations, demand for modern, high‑quality diagnostic tests continues to rise.
What are the Key Challenges and Risks That Could Limit Hepatitis B Testing Demand in Japan?
Despite clear benefits, several factors may limit testing uptake. First, HBV screening is not yet uniformly mandatory in all health checkups or workplaces; coverage depends on policies of individual employers or clinics. This leads to missed opportunities for early detection. Second, social stigma or concerns about privacy/discrimination remain a barrier some individuals avoid testing for fear of social repercussions. Third, some people may underestimate their risk or believe they are healthy, so may not seek testing proactively, especially if asymptomatic. Finally, limited awareness about the long-term consequences of untreated HBV and the benefits of early detection may reduce motivation to test, particularly among younger or low-risk groups.
.webp)
| Region | CAGR (%) |
|---|---|
| Kyushu & Okinawa | 6.1% |
| Kanto | 5.6% |
| Kansai | 4.9% |
| Chubu | 4.3% |
| Tohoku | 3.8% |
| Rest of Japan | 3.6% |
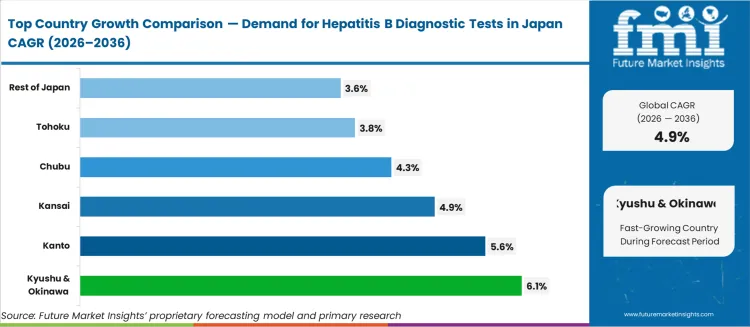
Demand for hepatitis B diagnostic tests in Japan is steadily growing, with Kyushu & Okinawa leading at a 6.1% CAGR, driven by the increasing focus on preventive healthcare and public health awareness. Kanto follows with a 5.6% CAGR, supported by the region’s large urban population and advanced healthcare infrastructure. Kansai shows a 4.9% CAGR, driven by its growing interest in medical dermatology and public health. Chubu experiences a 4.3% CAGR, fueled by improvements in healthcare services and rising awareness of liver diseases. Tohoku and the Rest of Japan show moderate growth at 3.8% and 3.6%, respectively, reflecting increased adoption of testing services in rural areas. As awareness and healthcare access continue to rise, demand for hepatitis B diagnostic tests is expected to grow across Japan.
Kyushu & Okinawa shows the highest demand for hepatitis B diagnostic tests, growing at a 6.1% CAGR. The region’s healthcare infrastructure, along with increasing awareness of liver disease prevention, contributes to the rise in demand for hepatitis B testing. As the population ages, there is a greater focus on early detection and preventive care for chronic conditions like hepatitis. The region’s growing tourism industry, particularly in Okinawa, drives demand as tourists and residents seek hepatitis B diagnostic tests for health assessments. The emphasis on wellness, self-care, and improved lifestyle also influences individuals to opt for regular screenings. As people recognize the importance of managing liver health to prevent complications like liver failure, hepatitis B testing is becoming more integral to health maintenance. With both public health campaigns and increased availability of testing options, the demand for hepatitis B diagnostic tests is expected to remain strong in Kyushu & Okinawa.
Kanto is experiencing steady demand for hepatitis B diagnostic tests, with a 5.6% CAGR. The region’s dense population, particularly in urban areas like Tokyo, significantly influences the growing need for diagnostic solutions to address liver diseases like hepatitis B. As awareness about the long-term effects of untreated hepatitis B, including liver cirrhosis and cancer, increases, more individuals are opting for testing, even if they show no symptoms. Kanto’s advanced healthcare infrastructure and high access to specialized diagnostic centers further facilitate the adoption of hepatitis B testing. The region’s growing focus on proactive healthcare and self-care practices, driven by both the medical community and public health campaigns, is pushing more residents to undergo regular screenings. With healthcare services continuously advancing and preventive care becoming a priority, Kanto's demand for hepatitis B diagnostic tests is expected to remain strong and continue to grow steadily in the coming years.
Kansai is seeing steady demand for hepatitis B diagnostic tests, with a 4.9% CAGR. The region’s rising healthcare awareness and strong focus on both medical and cosmetic dermatology are contributing to the demand for hepatitis B diagnostic tests. As the population ages, there is an increasing need for preventive care, with more people opting for tests that help detect liver disease at an early stage. Kansai's key cities like Osaka and Kyoto, which are healthcare and research hubs, also facilitate the availability of advanced diagnostic technologies. The region’s commitment to health improvement, coupled with a rising interest in managing chronic conditions, drives the growth in hepatitis B testing. Public health initiatives and healthcare campaigns have played an important role in educating people about the risks of untreated hepatitis B. As more residents in Kansai take a proactive approach to their health, demand for hepatitis B diagnostic tests is expected to continue growing steadily.
Chubu is experiencing steady demand for hepatitis B diagnostic tests, growing at a 4.3% CAGR. The region's increasing focus on wellness and preventive healthcare is contributing to the growing adoption of hepatitis B testing. With a higher awareness of the risks associated with liver diseases, especially among older adults, more people in Chubu are seeking diagnostic tests to assess their liver health. The expansion of healthcare services, particularly in urban centers like Nagoya, further facilitates access to hepatitis B testing. As individuals become more health-conscious, the demand for diagnostic solutions that can detect viral infections like hepatitis B has risen. Public health campaigns and the availability of advanced diagnostic options, including non-invasive testing, are also helping drive this growth. As Chubu continues to prioritize healthcare innovation, demand for hepatitis B diagnostic testing is expected to rise steadily, with increasing uptake among both urban and rural populations.
Tohoku is seeing moderate demand for hepatitis B diagnostic tests, with a 3.8% CAGR. While demand in this region is smaller compared to urbanized areas, healthcare improvements and growing awareness are contributing to steady growth. The region’s aging population is a key driver; as older adults are at higher risk for liver diseases such as hepatitis B. As more healthcare facilities in Tohoku offer diagnostic services and raise awareness about the importance of early detection, the demand for hepatitis B testing is rising. The increasing focus on preventive healthcare and the expansion of medical outreach programs are helping boost adoption. While growth in Tohoku may be slower than in more urban regions, the rise in healthcare access and public awareness ensures steady growth in the demand for hepatitis B diagnostic tests. As the healthcare system continues to improve, more individuals in Tohoku are expected to opt for regular screenings.
The Rest of Japan is experiencing steady demand for hepatitis B diagnostic tests, growing at a 3.6% CAGR. While the industry is smaller in rural and less urbanized areas, the growing awareness of hepatitis B risks and increasing access to healthcare services are contributing to steady growth. As more individuals in these areas become aware of the importance of early hepatitis detection and treatment, demand for testing continues to rise. Public health campaigns aimed at rural populations and the expansion of diagnostic testing facilities play a significant role in this growth. As people in rural areas prioritize health maintenance, preventive care, and early intervention, hepatitis B diagnostic testing becomes an essential part of regular health checkups. As healthcare services improve in rural regions, the demand for hepatitis B diagnostic tests in the Rest of Japan is expected to grow steadily, despite lower growth compared to urban areas.
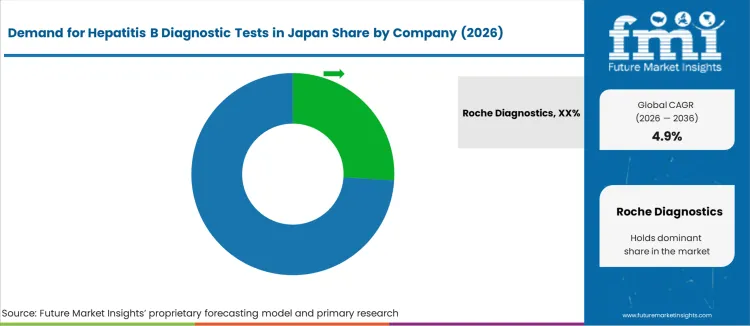
Demand for Hepatitis B diagnostic tests in Japan remains significant, driven by ongoing public-health efforts, screening initiatives, and the need to detect and monitor chronic hepatitis B virus (HBV) infections for treatment and prevention of liver diseases. Even though the overall prevalence among the general population is relatively low (less than 1%), a substantial segment especially older adults and higher-risk groups requires periodic testing, driving steady demand for reliable, accurate HBV detection and monitoring solutions.
Key companies in the Japanese Hepatitis B diagnostics industry include Roche Diagnostics, Abbott Laboratories, Siemens Healthineers, Bio-Rad Laboratories, and Danaher Corporation (Beckman Coulter). Among them, Roche Diagnostics is estimated to have an industry share of around 26.0%, offering a wide spectrum of molecular and immunoassay-based HBV tests for antigen detection, viral load quantification, and disease monitoring. Abbott Laboratories, Siemens Healthineers, Bio-Rad Laboratories, and Danaher/Beckman Coulter also play major roles supplying automated analyzers, ELISA and chemiluminescence kits, and laboratory workflows that cater to hospitals, clinics, and diagnostic labs.
Growth and competitive dynamics are driven by several factors. First, an ongoing need for early detection of HBV carriers including asymptomatic individuals and regular monitoring of chronic cases fuels demand for both screening and follow-up testing. Second, consolidation and modernization of clinical laboratories push adoption of automated, high-throughput diagnostic platforms that improve accuracy and efficiency and reduce turnaround times. Third, growing public-health awareness and screening policies encourage wider use of rapid, sensitive testing methods. Consequently, providers compete on assay sensitivity, automation, reliability, cost-efficiency, and ease of integration into hospital/laboratory workflows.
| Metric | Value |
|---|---|
| Quantitative Units | USD 53.50 to USD 86.32 million, at a CAGR of 4.9% |
| Market Definition | Hepatitis B diagnostic tests are in vitro diagnostic products used to detect hepatitis B virus infection markers in blood and serum specimens. The Japan market covers enzyme immunoassay kits and point-of-care testing kits used for hepatitis B surface antigen detection, antibody screening, and viral load quantification. |
| Segmentation | Product Type (Enzyme Immunoassay Kits, Point-of-Care Testing Kits), End User (Hospitals, Clinics, Diagnostic Centers, Home Care) |
| Regions Covered | Kyushu & Okinawa, Kanto, Kansai, Chubu, Tohoku, Rest of Japan |
| Key Companies Profiled | Roche Diagnostics, Abbott Laboratories, Siemens Healthineers, Bio-Rad Laboratories, Danaher Corporation (Beckman Coulter) |
| Forecast Period | 2026 to 2036 |
| Approach | Forecasting models apply a bottom-up methodology starting with unit shipment and average selling price data, cross-validated against manufacturer financial disclosures and institutional procurement records. |
This bibliography is provided for reader reference. The full Future Market Insights report contains the complete reference list with primary research documentation.
How large is the demand for hepatitis b diagnostic tests in japan in 2026?
The demand for hepatitis b diagnostic tests in japan is estimated to be valued at USD 53.50 million in 2026.
What will be the market size of demand for hepatitis b diagnostic tests in japan by 2036?
Market size for demand for hepatitis b diagnostic tests in japan is projected to reach USD 86.32 million by 2036.
What is the expected growth rate for demand for hepatitis b diagnostic tests in japan between 2026 and 2036?
The demand for hepatitis b diagnostic tests in japan is expected to grow at a CAGR of 4.9% between 2026 and 2036.
Which product type is poised to lead by 2026?
Enzyme Immunoassay Kits accounts for 35.0% share in 2026, driven by established procurement patterns and institutional adoption.
How is hospitals positioned in driving adoption in 2026?
Hospitals holds 40.0% share of the end user segment in 2026, reflecting concentrated institutional demand.
What is the growth outlook for Kyushu & Okinawa?
Kyushu & Okinawa is projected to grow at a CAGR of 6.1% during 2026 to 2036.
What is included in the scope of this report?
Market scope includes enzyme immunoassay kits (ELISA, CLIA, ECLIA), rapid point-of-care testing kits, and confirmatory molecular diagnostic reagents for hepatitis B virus detection. Calibrators, quality controls, and sample preparation consumables specific to HBV testing workflows are included.
How does FMI build and validate the demand for hepatitis b diagnostic tests in japan forecast?
Forecasting models apply a bottom-up methodology starting with unit shipment and average selling price data, cross-validated against manufacturer financial disclosures and institutional procurement records.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.