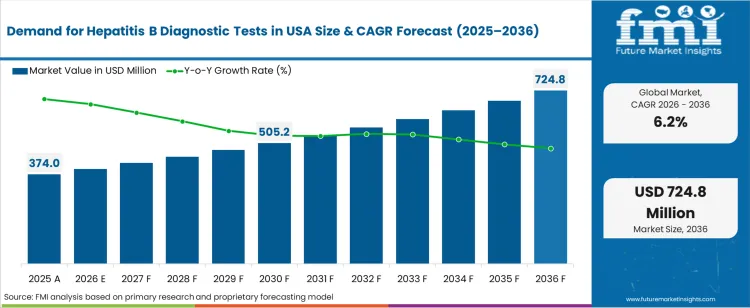
The demand for hepatitis b diagnostic tests in usa was valued at USD 374.00 million in 2025, projected to reach USD 397.19 million in 2026, and is forecast to expand to USD 724.84 million by 2036 at a 6.2% CAGR. CDC-recommended universal hepatitis B screening guidelines for all adults aged 18 and older, implemented since March 2023, are expanding the addressable testing population and driving sustained volume growth across hospital laboratories, community health centres, and commercial reference laboratories. The addition of triple-panel hepatitis B testing (HBsAg, anti-HBs, anti-HBc) to routine adult screening protocols is increasing the number of assays performed per patient encounter compared to prior single-marker screening approaches.
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 397.19 million |
| Industry Value (2036) | USD 724.84 million |
| CAGR (2026 to 2036) | 6.2% |
Source: Future Market Insights, 2026
The incremental opportunity between 2026 and 2036 amounts to USD 327.65 million. FMI is of the opinion that the universal screening mandate is the primary volume catalyst, with an estimated 67 million previously unscreened adults now eligible for routine testing through primary care encounters. The shift from risk-based to universal screening has increased test ordering rates at federally qualified health centres and integrated delivery networks. Quantitative HBV DNA viral load testing for treatment monitoring adds higher-value molecular assay consumption on top of the serology screening base.
Sub-national growth trajectories reflect screening programme adoption rates and immigrant population density. West at 7.1%, South at 6.4%, Northeast at 5.7%, Midwest at 4.9%. The West leads, driven by California's high hepatitis B prevalence among Asian American and Pacific Islander populations and state-funded screening programme infrastructure. The South follows, where expanding Medicaid coverage and community health centre capacity are broadening access to routine screening. The Northeast benefits from dense laboratory networks serving urban immigrant communities with elevated HBV prevalence, while the Midwest reflects steady growth from integrated health system screening protocol adoption.
Hepatitis B diagnostic tests are in vitro diagnostic products used to detect hepatitis B virus infection markers, immune status, and viral load in blood specimens. The USA market covers enzyme immunoassay kits and point-of-care testing kits used for HBsAg screening, anti-HBs immunity assessment, anti-HBc exposure detection, and HBV DNA viral load quantification. These tests are deployed across hospital laboratories, clinics, diagnostic centres, community health facilities, and home care settings.
Market scope includes enzyme immunoassay kits (ELISA, CLIA, ECLIA), rapid point-of-care testing kits, and molecular diagnostic reagents for HBV detection and viral load monitoring. Triple-panel screening kits, confirmatory assays, and associated calibrators and quality controls are included.
General immunoassay analyser hardware, hepatitis C and HIV diagnostic test kits, liver function biochemistry panels, and FibroScan elastography devices are excluded. Hepatitis B vaccines, antiviral therapeutics, and treatment management services are outside the analytical scope.
Demand for hepatitis B diagnostic tests in the USA is increasing because healthcare systems prioritize early identification and long term monitoring of chronic viral infections. Routine screening expands in primary care, prenatal visits and community health programs to prevent mother to child transmission and detect infection before liver complications develop. Large groups of individuals with risk factors such as shared needle exposure, close household contact with infected persons and older adults require ongoing surveillance and confirmatory testing.
Specialty clinics and hospitals rely on viral load assays and serology panels to guide treatment decisions and monitor response to antiviral therapy. Blood banks and organ donation programs maintain strict screening protocols to protect transfusion and transplant safety. Public health initiatives encourage testing among populations with low awareness of infection status, including foreign born residents from regions where hepatitis B prevalence is higher. Constraints include inconsistent follow up after initial testing and insurance variability for certain diagnostic panels. Some individuals avoid screening due to limited access to primary care or concern about stigma. Laboratory capacity planning may fluctuate with changes in vaccination coverage and public funding priorities.
Demand for hepatitis B diagnostic tests in the United States is supported by nationwide screening programs, increased focus on early detection, and continued incidence among high-risk groups such as individuals with chronic liver disease and immunocompromised patients. Hospitals and clinical laboratories rely on advanced immunoassays for accurate viral antigen and antibody assessment. Growth is reinforced by USA public health strategies emphasizing vaccination monitoring, donor screening, and improved access to diagnostic solutions through diverse care settings.
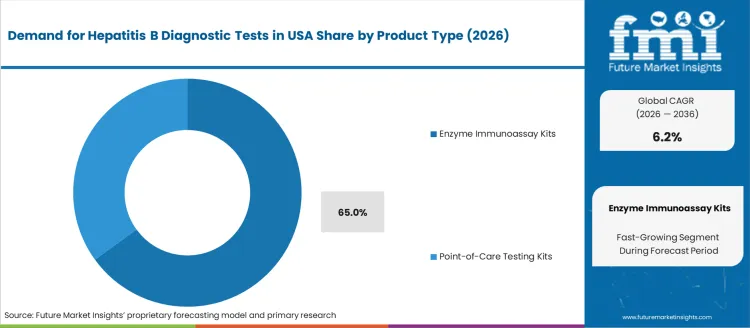
Enzyme Immunoassay (EIA) kits represent 65.0%, positioning them as the leading diagnostic modality due to high sensitivity levels required for hepatitis B surface antigen (HBsAg) and antibody detection. These kits remain essential for screening asymptomatic individuals and confirming viral activity during disease management. Their dominance reflects strong adoption among hospitals and accredited laboratories where automated platforms enable consistent processing of high sample volumes. Point-of-care kits explain the remaining demand share, supporting urgent screening in decentralized settings including emergency departments and outreach programs. These portable formats serve populations that benefit from rapid results despite smaller throughput capabilities. Product demand across the USA aligns with stringent testing standards, reimbursement support, and clinical preference for validated assay performance.
Key Points:
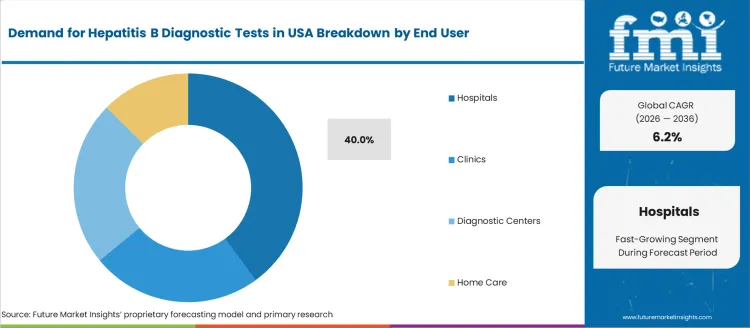
Hospitals hold 40.0%, driven by their central role in acute care, pre-operative screening, emergency diagnostics, and infectious disease management. Clinics and diagnostic centers share the remaining segment, serving routine patient testing and community-based screening programs. Home care represents a smaller presence due to limited use of self-administered tests in hepatitis B detection. End-user demand in the USA reflects clinical complexity, patient flow, and reimbursement coverage, with hospital laboratories continuing to perform confirmatory and highly sensitive viral load assessment.
Key Points:
Growth of immigration screening from endemic regions, increased monitoring in high risk populations and expanding prenatal testing programs are driving demand.
In the United States, hepatitis B diagnostic testing gains adoption as public health agencies conduct routine screening among individuals born in regions with higher infection prevalence, including parts of Asia and Africa. Primary care and infectious disease clinics monitor patients with chronic liver disease and those receiving immunosuppressive therapies to detect viral reactivation early. Prenatal care guidelines recommend hepatitis B screening for all pregnant women to prevent maternal to infant transmission, creating stable test volume within obstetric networks. Substance use treatment facilities and community health centers expand testing to identify undiagnosed infections among at risk adults. These coordinated health efforts support continuous procurement of serology and molecular tests.
Underdiagnosis in uninsured populations, testing hesitancy due to stigma and reimbursement variation for screening restrain demand.
Patients without consistent medical coverage may postpone routine checkups and miss screening opportunities. Stigma related to infection risk factors can discourage individuals from requesting tests or discussing concerns with clinicians. Reimbursement for screening outside guideline directed risk groups may be limited, leading some providers to order tests selectively. Lack of follow through after initial screening can reduce repeat molecular monitoring for chronic cases if patients disconnect from continued care. These gaps slow broader uptake of diagnostic pathways.
Shift toward point of care rapid tests, increased linkage to digital record systems and rising co testing with hepatitis C and HIV define key trends.
Community clinics and emergency departments adopt rapid assays that allow immediate counseling and referral for vaccination or treatment. Integration with electronic medical records streamlines reporting and improves follow up scheduling for chronic disease management. Screening programs increasingly combine hepatitis B tests with hepatitis C and HIV panels to improve detection efficiency among shared risk groups. Pharmaceutical and public health initiatives emphasize early diagnosis to support antiviral therapy access and reduce long term liver complications. These developments indicate sustained, prevention driven demand for hepatitis B diagnostic testing across the United States healthcare system.
Demand for hepatitis B diagnostic tests in the United States is driven by chronic-liver-disease surveillance, immigration-screening protocols, blood-safety requirements, and prenatal testing programs. Laboratories and hospitals use serological panels and molecular assays to identify active infection, prior exposure, and immunity status. Public health initiatives encourage screening in higher-risk populations, including individuals with comorbid infections or family history. Centralized reference labs and hospital networks evaluate analyzers based on throughput, automation compatibility, and linkage to electronic health records. West USA leads at 7.1% CAGR, South USA follows with 6.4%, Northeast USA grows at 5.7%, and Midwest USA records 4.9%, reflecting variations in demographics and healthcare-access infrastructure.
.webp)
| Region | CAGR (2026-2036) |
|---|---|
| West USA | 7.1% |
| South USA | 6.4% |
| Northeast USA | 5.7% |
| Midwest USA | 4.9% |
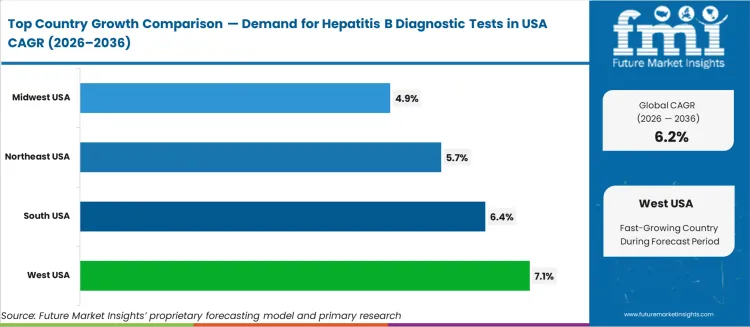
West USA shows the highest regional momentum, growing at 7.1% CAGR, supported by large coastal metropolitan centers in California and Washington. Screenings are integrated into primary-care workflows, community clinics, and hospital admission panels for at-risk patient groups. Immigration-support services encourage serology testing as part of comprehensive infectious-disease evaluation for new arrivals. Blood banks rely on high-sensitivity assays to secure transfusion safety across densely populated catchment areas. Diagnostic laboratories invest in automated platforms that consolidate hepatitis B, hepatitis C, and HIV testing to manage high daily specimen volumes. Public health departments promote testing during outreach events in urban neighborhoods, increasing specimen submissions through community clinics. Procurement teams review analyzer performance data, reagent shelf-life, and interface compatibility with laboratory information systems.
South USA advances at approximately 6.4% CAGR, influenced by demographic structure, regional liver-disease incidence, and strong hospital networks in Texas, Florida, and Georgia. Screening is incorporated into emergency-department panels where patients present with non-specific liver-function abnormalities. Obstetrics practices conduct routine tests in prenatal care, checking infection risk and vaccination history. Community-health centers serving uninsured and underinsured populations rely on compact analyzers or send-out services to reference laboratories. Public-health programs coordinate testing drives that target higher-risk groups, guiding individuals toward follow-up consultation. Procurement emphasizes reagent pricing stability, instrument uptime, and scalable capacity to handle episodic surges during outreach campaigns. Rural facilities favor platforms with straightforward operation and remote-service support.
Northeast USA posts about 5.7% CAGR, reflecting strong adoption within academic medical centers and integrated health systems in New York, New Jersey, and Massachusetts. Hospitals standardize hepatitis B panels within pre-surgical and chronic-liver-disease workups. Infectious-disease clinics manage follow-up testing to monitor treatment decisions and immunity status, especially in patients with additional viral infections. Reference laboratories use high-throughput analyzers connected to regional courier networks to serve community practices. University-affiliated institutions participate in surveillance studies that rely on consistent assay performance across multiple sites. Purchasers examine test-metric precision, lot-to-lot consistency, and data-integration capabilities that support regional health-reporting systems.
Midwest USA grows at around 4.9% CAGR, representing steady adoption across hospital systems and independent laboratories in Illinois, Ohio, Michigan, and neighboring states. Testing demand is tied to routine pre-employment and occupational-health screenings in healthcare workers and other at-risk professions. Primary-care physicians order hepatitis B panels for patients with elevated liver enzymes or risk factors documented during visits. Community hospitals deploy mid-range analyzers that balance cost with reliability, avoiding excessive overcapacity. Rural laboratories often rely on regional reference centers for confirmatory or molecular assays, maintaining essential serology capacity in-house. Procurement strategies prioritize robust technical support and reagent logistics suited to winter-weather disruptions.
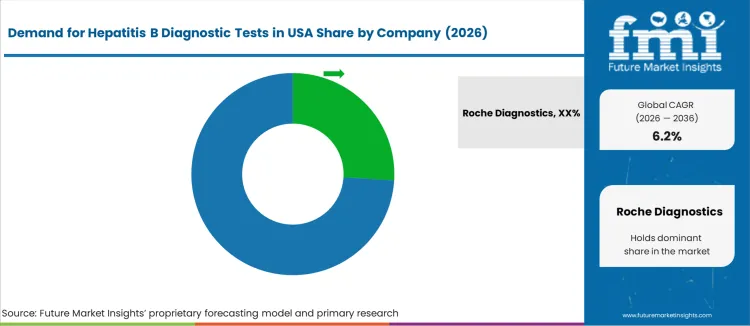
Hepatitis B diagnostic testing in the United States supports screening in hospitals, clinical laboratories, community health programs, and blood-safety networks. Demand is sustained by CDC-recommended screening protocols for high-risk groups, prenatal testing requirements, and monitoring of chronic infection. Laboratories prioritize high-sensitivity assays, automation compatibility, and consistent reagent supply. Interoperability with digital reporting systems strengthens adoption in centralized testing environments that handle high daily sample volumes. Roche Diagnostics holds an estimated 26.2% share. Its immunoassay and molecular testing platforms are broadly installed in national reference laboratories and large hospital systems.
Wide test menu availability increases operational consistency across viral hepatitis workflows. Abbott Laboratories participates with automated assay systems used in routine screening and confirmatory viral marker detection for both acute and chronic cases. Siemens Healthineers supports diagnostic networks with high-throughput immunoassay instruments aligned to diverse laboratory sizes. Bio-Rad Laboratories contributes through specialized hepatitis B test kits used in public-health settings and follow-up research applications. Danaher Corporation (Beckman Coulter) focuses on integrated chemistry and immunoassay analyzers within community hospital networks where dependable uptime and guided workflows are valued. Competitive positioning is influenced by assay accuracy, regulatory compliance, ease of workflow standardization, and the ability to scale from targeted screening programs to continuous monitoring in chronic-care populations across the United States.
| Metric | Value |
|---|---|
| Quantitative Units | USD 397.19 to USD 724.84 million, at a CAGR of 6.2% |
| Market Definition | Hepatitis B diagnostic tests are in vitro diagnostic products used to detect hepatitis B virus infection markers, immune status, and viral load in blood specimens. The USA market covers enzyme immunoassay kits and point-of-care testing kits used for HBsAg screening, anti-HBs immunity assessment, anti-HBc exposure detection, and HBV DNA viral load quantification. |
| Segmentation | Product Type (Enzyme Immunoassay Kits, Point-of-Care Testing Kits), End User (Hospitals, Clinics, Diagnostic Centers, Home Care) |
| Regions Covered | West, South, Northeast, Midwest |
| Key Companies Profiled | Roche Diagnostics, Abbott Laboratories, Siemens Healthineers, Bio-Rad Laboratories, Danaher Corporation (Beckman Coulter) |
| Forecast Period | 2026 to 2036 |
| Approach | Forecasting models apply a bottom-up methodology starting with unit shipment and average selling price data, cross-validated against manufacturer financial disclosures and institutional procurement records. |
This bibliography is provided for reader reference. The full Future Market Insights report contains the complete reference list with primary research documentation.
How large is the demand for hepatitis b diagnostic tests in usa in 2026?
The demand for hepatitis b diagnostic tests in usa is estimated to be valued at USD 397.19 million in 2026.
What will be the market size of demand for hepatitis b diagnostic tests in usa by 2036?
Market size for demand for hepatitis b diagnostic tests in usa is projected to reach USD 724.84 million by 2036.
What is the expected growth rate for demand for hepatitis b diagnostic tests in usa between 2026 and 2036?
The demand for hepatitis b diagnostic tests in usa is expected to grow at a CAGR of 6.2% between 2026 and 2036.
Which product type is poised to lead by 2026?
Enzyme Immunoassay Kits accounts for 65.0% share in 2026, driven by established procurement patterns and institutional adoption.
How is hospitals positioned in driving adoption in 2026?
Hospitals holds 40.0% share of the end user segment in 2026, reflecting concentrated institutional demand.
What is the growth outlook for West?
West is projected to grow at a CAGR of 7.1% during 2026 to 2036.
What is included in the scope of this report?
Market scope includes enzyme immunoassay kits (ELISA, CLIA, ECLIA), rapid point-of-care testing kits, and molecular diagnostic reagents for HBV detection and viral load monitoring. Triple-panel screening kits, confirmatory assays, and associated calibrators and quality controls are included.
How does FMI build and validate the demand for hepatitis b diagnostic tests in usa forecast?
Forecasting models apply a bottom-up methodology starting with unit shipment and average selling price data, cross-validated against manufacturer financial disclosures and institutional procurement records.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.