Jet Needle-free Injectors Market
Jet Needle-free Injectors Market Size and Share Forecast Outlook 2025 to 2035
Historical Data Covered: 2015 to 2023 | Base Year: 2024 | Estimated Year: 2025 | Forecast Period: 2026 to 2035
Jet Needle-free Injectors Market Forecast and Outlook (2025-2035)
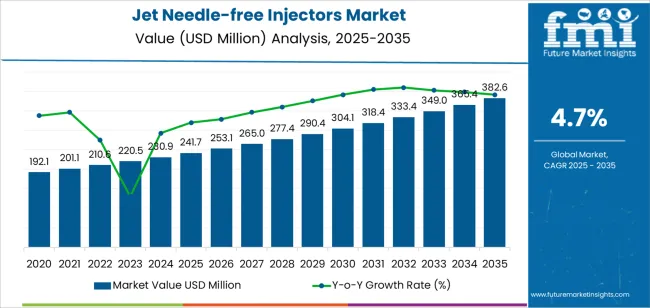
The jet needle-free injectors market is expected to grow from USD 241.7 million in 2025 to USD 382.6 million by 2035, with market momentum shaped by shifting priorities in patient comfort, infection control, and home-based treatment models. A primary trend shaping adoption is the transition from needle-based injections to pain-reduced, pressure-driven liquid delivery systems, particularly in diabetes and chronic care routines where daily or frequent dosing increases patient fatigue and needle anxiety. The growing emphasis on needle-stick injury prevention in hospitals and clinical settings is also accelerating institutional acceptance, as healthcare worker safety protocols increasingly discourage sharps usage unless necessary.
Another key trend is the emergence of self-administration and home-care treatment platforms, where compact, user-friendly needle-free injectors support decentralization of treatment from hospital environments to personal care routines. Pharmaceutical companies are also beginning to co-develop drug–device integrated delivery models, particularly for biologics, peptides, and vaccines requiring controlled intradermal or subcutaneous dispersion. At the same time, improvements in nozzle geometry, pressure modulation, and dose precision calibration are increasing clinical confidence in the consistency of jet injection performance. Beyond clinical healthcare, rising demand in mass vaccination and public health immunization programs is positioning needle-free systems as viable tools for high-volume, rapid-deployment injection campaigns.
Quick Stats for Jet Needle-free Injectors Market
- Jet Needle-free Injectors Market Value (2025): USD 241.7 million
- Jet Needle-free Injectors Market Forecast Value (2035): USD 382.6 million
- Jet Needle-free Injectors Market Forecast CAGR: 4.7%
- Leading Injection Technology in Jet Needle-free Injectors Market: Liquid Injections
- Key Growth Regions in Jet Needle-free Injectors Market: East Asia, South Asia Pacific, and North America
- Top Key Players in Jet Needle-free Injectors Market: Halozyme (Antares Pharma), Inovio Pharmaceuticals, Endo International, Injex Pharma GmbH, National Medical Products (J-Tip)
- Where revenue comes from - now vs next (industry-level view)
| Period | Primary Revenue Buckets | Share | Notes |
|---|---|---|---|
| Today | New device sales (liquid, powder, depot systems) | 44% | Healthcare-led, replacement-driven purchases |
| Replacement cartridges & consumables | 26% | Ampoules, pressure cartridges, sterile tips | |
| Pharmaceutical partnerships & licensing | 18% | Drug-device combinations, co-development | |
| Service & maintenance contracts | 12% | Device servicing, patient training support | |
| Future (3-5 yrs) | Connected injection systems | 38-42% | Digital tracking, dose monitoring, compliance data |
| Consumables & cartridges | 24-28% | Recurring revenue, proprietary formats | |
| Pharmaceutical collaborations | 18-22% | Biologic delivery, specialized formulations | |
| Patient support programs | 10-14% | Training services, adherence monitoring | |
| Data analytics services | 5-8% | Usage patterns, compliance tracking | |
| Rental & subscription models | 3-6% | Device-as-a-service for chronic conditions |
The latter half (2030-2035) will witness continued growth from USD 304.1 million to USD 382.6 million, representing an addition of USD 78.5 million or 61% of the decade's expansion. This period will be defined by mass market penetration of powder injection technologies, integration with comprehensive disease management platforms, and seamless compatibility with existing healthcare infrastructure. The market trajectory signals fundamental shifts in how healthcare facilities approach drug delivery and patient safety, with participants positioned to benefit from growing demand across multiple injection types and application segments.
Regional dynamics reveal distinct adoption patterns, with developed markets leading technology advancement while emerging economies show accelerating growth through healthcare modernization programs. North American markets maintain steady expansion supported by diabetes management requirements and patient safety initiatives, while Asian markets demonstrate rapid growth driven by vaccination program expansion and healthcare infrastructure development. European markets show moderate growth influenced by patient-centric care standards and pharmaceutical innovation trends.
Healthcare industry evolution drives fundamental changes in injection delivery specifications, with healthcare providers and patients increasingly prioritizing products that combine delivery precision, safety features, and ease of use. The decade ahead will witness transformation from traditional needle-based systems toward sophisticated needle-free technologies with advanced pressure mechanisms, dose accuracy controls, and ergonomic designs that meet diverse therapeutic and preventive care requirements.
Jet Needle-free Injectors Market Key Takeaways
| Metric | Value |
|---|---|
| Market Value (2025) | USD 241.7 million |
| Market Forecast (2035) | USD 382.6 million |
| Growth Rate | 4.7% CAGR |
| Leading Injection Technology | Liquid Injections |
| Primary Application | Vaccine Delivery Segment |
The market demonstrates strong fundamentals with liquid injection systems capturing a dominant share through advanced delivery precision capabilities and pharmaceutical administration optimization. Vaccine delivery applications drive primary demand, supported by increasing immunization programs and mass vaccination requirements. Geographic expansion remains concentrated in developed markets with established healthcare infrastructure, while emerging economies show accelerating adoption rates driven by vaccination initiatives and rising patient safety standards.
Healthcare trends favor jet needle-free injectors that integrate delivery accuracy with patient comfort features, creating opportunities for manufacturers offering comprehensive product portfolios. The decade ahead presents expansion potential across vaccination programs, insulin delivery segments, and self-administration applications, with market participants positioned to capitalize on growing demand for pain-free injection solutions that meet evolving healthcare standards and patient preferences.
Imperatives for Stakeholders in Jet Needle-free Injectors Market
Design for patient safety, not just needle elimination
- Offer comprehensive training packages: devices with usage protocols, patient instruction materials, healthcare provider training, and safety documentation.
- Preconfigured workflows: sterility procedures, dose preparation protocols, injection techniques, and digital tracking systems for therapeutic administration.
Connected health readiness
- Real-time injection monitoring capabilities, dose tracking features, and electronic health record connectivity (patient data integration, adherence tracking).
Quality delivery approach
- Validated dose accuracy specifications, consistent pressure delivery systems, biocompatibility standards, and comprehensive performance documentation.
Value-based healthcare models
- Clear base device pricing with transparent service tiers (training support, replacement parts, performance guarantees); subscriptions for consumables and patient support programs.
What factors drive the dominance of the Liquid Injections and Vaccine Delivery segments in their respective categories?
Primary Classification: The market segments by injection type into powder injections, liquid injections, and depot or projectile injections, representing the evolution from basic needle-free delivery systems to sophisticated pharmaceutical administration solutions for comprehensive healthcare optimization.
Secondary Classification: Application segmentation divides the market into vaccine delivery, insulin delivery, and other therapeutic sectors, reflecting distinct requirements for dosing precision, administration convenience, and patient compliance standards.
Regional Classification: Geographic distribution covers North America, Latin America, Western Europe, Eastern Europe, East Asia, South Asia Pacific, and Middle East & Africa, with developed markets leading adoption while emerging economies show accelerating growth patterns driven by healthcare infrastructure expansion programs.
The classification structure reveals technology progression from standard needle-free mechanisms toward sophisticated delivery systems with enhanced dose accuracy and patient safety capabilities, while application diversity spans from vaccination programs to chronic disease management requiring precise pharmaceutical delivery solutions.
By Injection Type, the Liquid Injections Segment Accounts for Dominant Market Share
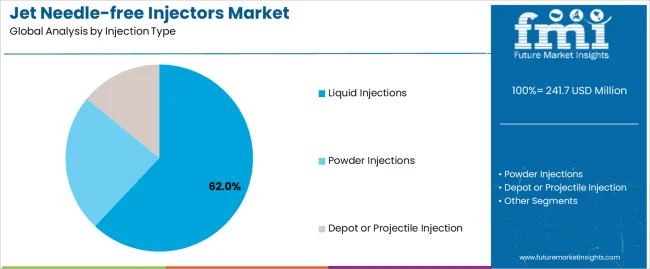
Market Position: Liquid injection systems command the leading position in the market with 62.0% market share through advanced delivery precision features, including superior dose accuracy, pharmaceutical compatibility, and healthcare administration optimization that enable medical facilities to achieve consistent therapeutic outcomes across diverse clinical and self-administration environments.
Value Drivers: The segment benefits from healthcare provider preference for versatile injection systems that provide reliable drug delivery, rapid administration capability, and patient comfort optimization without requiring complex preparation procedures. Advanced design features enable precise volume control, adjustable pressure settings, and integration with various pharmaceutical formulations, where delivery accuracy and patient acceptance represent critical treatment requirements.
Competitive Advantages: Liquid injection systems differentiate through proven delivery reliability, consistent therapeutic outcomes, and compatibility with standard pharmaceutical preparations that enhance treatment effectiveness while maintaining optimal patient safety suitable for diverse clinical applications.
Key market characteristics:
- Advanced pressure mechanisms with optimized delivery configuration and pharmaceutical compatibility capabilities
- Enhanced injection effectiveness, enabling accurate dose administration with consistent performance characteristics
- Healthcare compatibility, including sterile pathway integration, multi-dose capability, and formulation optimization for injection operations
Powder Injections Show Specialized Growth
Powder injection systems maintain a 24% market position in the jet needle-free injectors market due to their vaccine delivery advantages and storage stability properties. These systems appeal to immunization programs requiring ambient temperature storage with effective delivery for mass vaccination applications. Market growth is driven by global vaccination expansion, focusing stable formulation solutions and cold chain elimination through optimized powder delivery designs.
Depot or Projectile Injections Demonstrate Niche Growth
Depot or projectile injection systems capture 14% market share through specialized delivery requirements in long-acting medications, controlled release applications, and specialized therapeutic scenarios. These applications demand precise implantation capability capable of delivering solid drug formulations while providing sustained release characteristics and extended therapeutic duration.
By Application, the Vaccine Delivery Segment Shows Fastest Growth
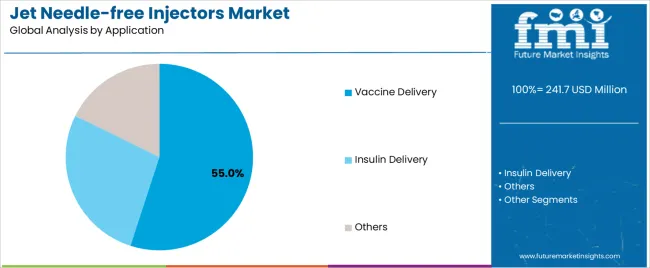
Market Context: Vaccine delivery applications demonstrate the 55.0% market share with 5.3% CAGR due to widespread adoption of mass immunization programs and increasing focus on vaccination coverage expansion, pandemic preparedness, and healthcare accessibility applications that maximize immunization efficiency while maintaining safety standards.
Appeal Factors: Healthcare administrators prioritize system efficiency, rapid deployment capability, and integration with immunization programs that enable coordinated vaccination operations across multiple population segments. The segment benefits from substantial government investment and public health initiatives that emphasize the acquisition of needle-free systems for vaccination campaigns and routine immunization applications.
Growth Drivers: Global immunization programs incorporate needle-free injectors as preferred equipment for mass vaccination, while pandemic preparedness increases demand for rapid deployment capabilities that comply with safety standards and minimize cross-contamination complexity.
Market Challenges: Varying vaccine formulations and cold chain requirements may limit system standardization across different immunization programs or regional scenarios.
Application dynamics include:
- Strong growth in mass vaccination campaigns and routine immunization requiring efficient delivery capabilities
- Increasing adoption in pandemic response and outbreak management applications for public health authorities
- Rising integration with digital tracking systems for vaccination monitoring and coverage assessment assurance
Insulin Delivery Applications Maintain Chronic Disease Focus
Insulin delivery applications capture market share through essential therapeutic requirements in diabetes management, chronic care programs, and self-administration scenarios. These applications demand reliable injection systems capable of operating with daily dosing requirements while providing consistent delivery performance and patient convenience capabilities.
What are the Drivers, Restraints, and Key Trends of the Jet Needle-free Injectors Market?
| Category | Factor | Impact | Why It Matters |
|---|---|---|---|
| Driver | Rising diabetes prevalence & insulin therapy expansion | ★★★★★ | Growing diabetic populations require convenient self-administration solutions with improved patient compliance and reduced injection anxiety across treatment regimens. |
| Driver | Needlestick injury prevention & healthcare worker safety | ★★★★★ | Occupational safety concerns transform needle-free systems from optional to essential; devices eliminating sharps injuries gain institutional adoption. |
| Driver | Mass vaccination programs & immunization initiatives | ★★★★☆ | Global immunization campaigns need efficient, safe delivery systems; demand for rapid administration and cross-contamination prevention expanding addressable market. |
| Restraint | High device costs & reimbursement limitations | ★★★★☆ | Premium pricing versus traditional syringes creates adoption barriers; limited insurance coverage slows consumer acceptance in price-sensitive markets. |
| Restraint | Pharmaceutical formulation compatibility & stability concerns | ★★★☆☆ | Not all medications suitable for needle-free delivery; formulation requirements and viscosity limitations restrict application breadth. |
| Trend | Biologic drug delivery & high-viscosity formulations | ★★★★★ | Advanced therapeutics require novel delivery methods; needle-free systems enabling biologic administration become critical value propositions. |
| Trend | Digital health integration & connected devices | ★★★★☆ | Smart injectors with dose tracking and adherence monitoring; data connectivity and patient engagement features drive competition toward integrated solutions. |
Analysis of the Jet Needle-free Injectors Market by Key Country
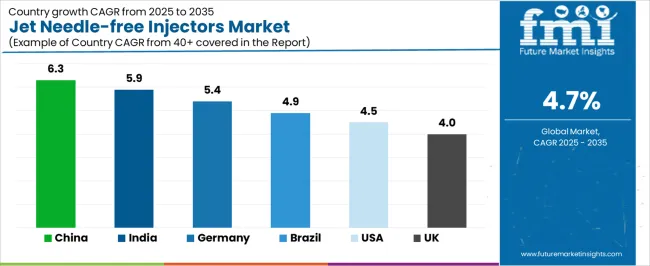
The jet needle-free injectors market demonstrates varied regional dynamics with Growth Leaders including China (6.3% growth rate) and India (5.9% growth rate) driving expansion through healthcare infrastructure initiatives and vaccination program development. Steady Performers encompass Germany (5.4% growth rate), Brazil (4.9% growth rate), and developed regions, benefiting from established healthcare systems and patient safety adoption. Mature Markets feature United States (4.5% growth rate) and United Kingdom (4.0% growth rate), where diabetes management programs and healthcare innovation support consistent growth patterns.
Regional synthesis reveals East Asian markets leading adoption through healthcare expansion and pharmaceutical development, while North American countries maintain steady expansion supported by chronic disease management and regulatory standardization requirements. European markets show moderate growth driven by patient safety applications and healthcare quality integration trends.
| Region/Country | 2025-2035 Growth | How to win | What to watch out |
|---|---|---|---|
| China | 6.3% | Focus on vaccination programs | Regulatory approval complexity |
| India | 5.9% | Offer affordable solutions | Distribution infrastructure gaps |
| Germany | 5.4% | Lead with precision systems | Reimbursement negotiations |
| Brazil | 4.9% | Value-oriented devices | Economic fluctuations |
| USA | 4.5% | Provide clinical evidence | Insurance coverage variability |
| UK | 4.0% | Push patient safety benefits | NHS procurement cycles |
Why does China drive the fastest market growth?
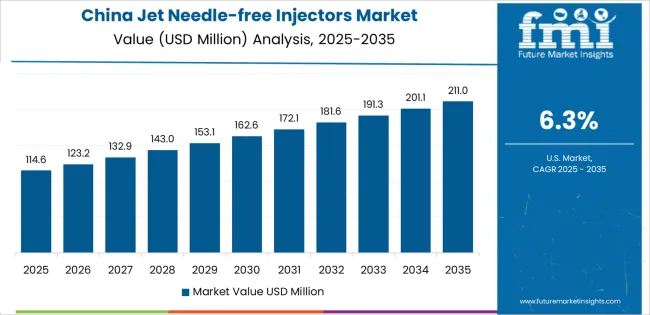
China establishes fastest market growth through aggressive healthcare modernization programs and comprehensive vaccination infrastructure development, integrating jet needle-free injectors as standard components in immunization facilities and hospital administration systems. The country's 6.3% growth rate reflects government initiatives promoting healthcare quality advancement and patient safety capabilities that mandate the use of advanced injection systems in vaccination and pharmaceutical delivery facilities. Growth concentrates in major healthcare centers, including Beijing, Shanghai, and Guangzhou, where vaccination programs showcase integrated needle-free systems that appeal to healthcare administrators seeking efficient immunization capabilities and safety enhancement applications.
Chinese manufacturers are developing cost-effective injection solutions that combine domestic production advantages with improving safety features, including contamination prevention and dose accuracy capabilities. Distribution channels through medical equipment suppliers and pharmaceutical distributors expand market access, while government support for vaccination programs supports adoption across diverse healthcare and public health segments.
Strategic Market Indicators:
- Vaccination facilities leading adoption with 68% deployment rate in immunization and mass vaccination sectors
- Government healthcare programs providing substantial funding for patient safety technology development
- Local manufacturers capturing 48% market share through competitive pricing and regulatory compliance
- International collaboration development for advanced injection technology targeting healthcare institutions
Why does India emerge as a high-growth market?
In Mumbai, Delhi, and Bangalore, healthcare facilities and vaccination centers are implementing jet needle-free injectors as standard equipment for immunization and pharmaceutical delivery applications, driven by increasing government healthcare investment and infrastructure modernization programs that emphasize the importance of patient safety capabilities. The market holds a 5.9% growth rate, supported by government immunization initiatives and healthcare infrastructure development programs that promote advanced injection systems for vaccination and therapeutic facilities. Indian healthcare providers are adopting injection systems that provide reliable delivery performance and patient acceptance features, particularly appealing in urban regions where healthcare quality and immunization coverage represent critical public health requirements.
Market expansion benefits from growing healthcare capabilities and pharmaceutical industry development that enable broader availability of needle-free injection systems for vaccination and therapeutic applications. Technology adoption follows patterns established in medical devices, where cost-effectiveness and clinical performance drive procurement decisions and operational deployment.
Market Intelligence Brief:
- Vaccination and diabetes care segments are driving initial adoption with 61% annual growth in device procurement
- Healthcare modernization programs focusing injection systems for patient safety and treatment compliance
- Healthcare facilities partnering with international manufacturers for technology access
- Immunization programs implementing needle-free systems for vaccination efficiency and coverage optimization
How does Germany maintain technology leadership?
Germany establishes technology leadership through comprehensive healthcare quality programs and advanced medical technology infrastructure, integrating jet needle-free injectors across healthcare and pharmaceutical delivery applications. The country's 5.4% growth rate reflects established healthcare relationships and mature medical device adoption that supports widespread use of precision injection systems in hospitals and diabetes care facilities. Growth concentrates in major medical centers, including Munich, Berlin, and Hamburg, where healthcare excellence showcases mature device deployment that appeals to healthcare providers seeking proven delivery precision capabilities and patient safety applications.
German manufacturers leverage engineering expertise and comprehensive validation protocols, including clinical testing and performance documentation that create product credibility and technical advantages. The market benefits from established medical device standards and patient-centric care models that support injection system use while encouraging technology advancement and safety optimization.
Market Intelligence Brief:
- Healthcare and diabetes care facilities maintaining 82% adoption rate with established device deployment
- Regulatory standards providing consistent demand for technology advancement
- Pharmaceutical partnerships capturing 57% market share through drug-device combinations and integrated solutions
- Clinical integration programs expanding needle-free delivery capabilities in therapeutic scenarios
How does Brazil show strong regional leadership?
Brazil's market expansion benefits from diverse healthcare demand, including hospital system development in São Paulo and Rio de Janeiro, vaccination program upgrades, and government healthcare initiatives that increasingly incorporate needle-free injectors for immunization applications. The country maintains a 4.9% growth rate, driven by rising healthcare activity and increasing recognition of patient safety technology benefits, including needle elimination and cross-contamination prevention.
Market dynamics focus on affordable injection systems that balance adequate performance with cost considerations important to Brazilian healthcare providers. Growing healthcare development creates continued demand for modern injection systems in new facility infrastructure and healthcare modernization projects.
Strategic Market Considerations:
- Vaccination and diabetes care segments leading growth with focus on patient safety and treatment compliance applications
- Regional healthcare requirements driving a diverse product portfolio from basic injection devices to advanced delivery systems
- Import dependency challenges offset by potential distribution partnerships with international manufacturers
- Government healthcare initiatives beginning to influence procurement standards and device requirements
How does the United States maintain market leadership?
The United States establishes market leadership through comprehensive diabetes care programs and advanced healthcare infrastructure development, integrating jet needle-free injectors across diabetes management and vaccination applications. The country's 4.5% growth rate reflects established pharmaceutical relationships and mature injection technology adoption that supports widespread use of needle-free systems in home care and clinical facilities. Growth concentrates in major healthcare markets, including major metropolitan areas and integrated health systems, where patient safety initiatives showcase mature device deployment that appeals to healthcare providers seeking proven safety capabilities and patient compliance applications.
American device manufacturers leverage established distribution networks and comprehensive support capabilities, including patient training programs and clinical evidence that create product acceptance and market advantages. The market benefits from mature regulatory standards and insurance reimbursement that support injection system use while encouraging technology advancement and therapeutic innovation.
Market Intelligence Brief:
- Diabetes care and vaccination facilities maintaining 85% awareness rate with growing device adoption
- FDA approval pathways providing consistent framework for technology advancement
- Pharmaceutical partnerships capturing 62% market share through drug delivery collaborations and co-development programs
- Digital health integration programs expanding connected injection capabilities in chronic disease scenarios
Why does the United Kingdom show established healthcare adoption?
The United Kingdom's healthcare technology market demonstrates established jet needle-free injector deployment with documented safety effectiveness in diabetes care applications and vaccination programs through integration with existing healthcare systems and patient safety infrastructure. The country maintains a 4.0% growth rate. Healthcare centers, including London, Manchester, and Birmingham, showcase quality installations where needle-free injectors integrate with comprehensive patient care platforms and electronic health systems to optimize treatment operations and safety effectiveness.
British healthcare providers prioritize patient safety and treatment adherence in injection system selection, creating demand for validated devices with proven features, including clinical effectiveness documentation and safety specifications. The market benefits from National Health Service infrastructure and patient-centric programs that provide consistent operational benefits and compliance with national healthcare standards.
Market Intelligence Brief:
- Healthcare focuses on patient outcomes and safety metrics, driving quality device adoption
- NHS partnerships providing structured procurement cycles
- Clinical collaboration between British healthcare providers and device manufacturers
- Patient education programs expanding needle-free system integration in chronic disease scenarios
What is the market split by country in Europe?
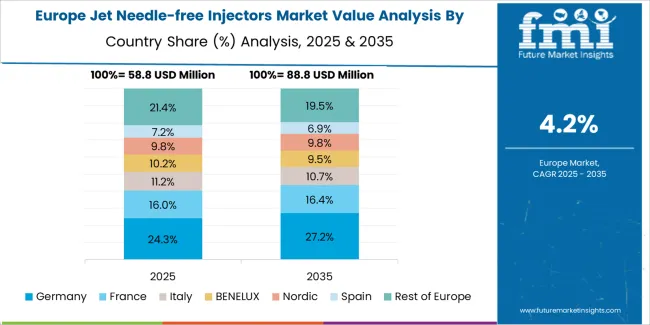
The jet needle-free injectors market in Europe is projected to grow from USD 82.9 million in 2025 to USD 196.4 million by 2035, registering a CAGR of 9.0% over the forecast period. Germany is expected to maintain its leadership position with a 33.7% market share in 2025, supported by its advanced healthcare infrastructure and major medical technology centers.
United Kingdom follows with a 22.8% share in 2025, driven by comprehensive patient safety programs and diabetes care initiatives. France holds a 18.5% share through vaccination program applications and pharmaceutical delivery requirements. Italy commands a 13.2% share, while Spain accounts for 11.8% in 2025. The Rest of Europe region is anticipated to gain momentum, expanding its collective share from 6.4% to 7.1% by 2035, attributed to increasing device adoption in Nordic countries and emerging Eastern European healthcare facilities implementing patient safety modernization programs.
How does Japan show advanced medical technology integration?
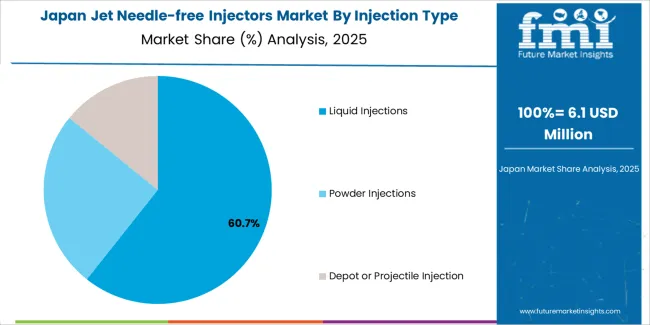
Japan demonstrates sophisticated jet needle-free injector deployment through premium healthcare infrastructure and advanced patient safety standards. The market maintains steady growth driven by diabetes management requirements and vaccination program modernization. Japanese manufacturers emphasize precision engineering and reliability features that meet rigorous medical device regulations. Technology partnerships with pharmaceutical companies enable specialized formulation development and delivery optimization suitable for Japanese healthcare priorities and demographic requirements including aging population care needs.
How does South Korea exhibit strong healthcare innovation?
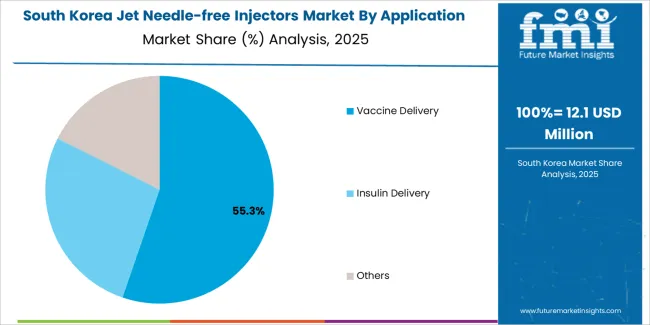
South Korea shows robust market expansion through intensive healthcare technology investment and medical device industry development programs. Seoul, major hospital systems, and diabetes care centers lead adoption with advanced needle-free injection systems incorporating digital tracking features. The market benefits from government healthcare initiatives and medical technology advancement programs that promote innovative delivery devices. Korean healthcare providers focus on patient convenience and treatment adherence features aligned with chronic disease management priorities and technological innovation objectives for future healthcare applications.
What defines the competitive landscape of the jet needle-free injectors market?
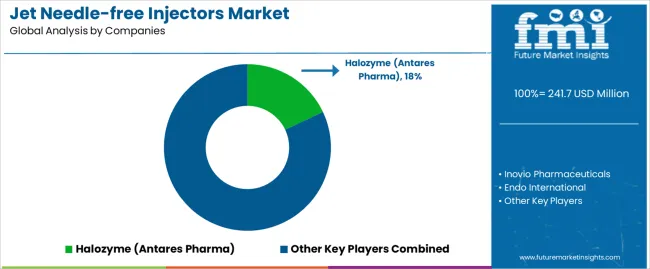
The market features approximately 15-20 specialized manufacturers, with the top 3-5 companies controlling roughly 65-70% of global revenue. Market leadership depends on pharmaceutical partnerships, clinical validation, and regulatory approval success rather than production scale advantages. Competition centers on delivery accuracy, device reliability, and formulation compatibility that drive adoption across healthcare institutions and patient populations.
Basic jet injection mechanisms and pressure delivery systems represent established technology, with manufacturers differentiating through dose precision, pharmaceutical compatibility range, and patient usability features. Margin opportunities exist in pharmaceutical collaborations offering drug-device combination products, proprietary cartridge systems, and integrated digital tracking that command premium positioning. Companies maintaining strong clinical evidence and pharmaceutical partnerships capture market advantages through product validation and therapeutic integration.
Global manufacturers leverage regulatory expertise for multi-regional approvals and pharmaceutical industry relationships, though specialized developers often compete effectively through innovative delivery mechanisms and targeted therapeutic applications. Technology innovation focuses on delivery pressure optimization, formulation compatibility enhancement, and digital connectivity integration rather than fundamental mechanism changes. Market dynamics favor participants with pharmaceutical expertise who understand drug delivery requirements and provide comprehensive clinical support alongside device offerings.
Pharmaceutical partnerships increasingly differentiate competitors, with leading manufacturers collaborating on drug-device combinations, formulation optimization, and therapeutic delivery systems that strengthen market positioning. Patient support programs and training services become important competitive factors. The market favors participants balancing device engineering with pharmaceutical collaboration effectiveness while maintaining product quality standards that meet regulatory requirements and healthcare performance expectations.
| Stakeholder | What they actually control | Typical strengths | Typical blind spots |
|---|---|---|---|
| Global platforms | Regulatory approvals, pharmaceutical partnerships, clinical validation | Proven safety, comprehensive evidence, multi-region availability | Innovation pace; therapeutic breadth |
| Technology innovators | Novel mechanisms; high-pressure systems; digital integration | Performance differentiation; patent protection; pharmaceutical interest | Market access; reimbursement challenges |
| Regional specialists | Local clinical relationships, regulatory knowledge, market access | Healthcare proximity; regional expertise; cost positioning | Technology advancement; scale limitations |
| Pharma-device ecosystems | Drug formulations, combination products, integrated solutions | Therapeutic validation; market access; prescription capture | Device manufacturing; technical service |
Key Players in the Jet Needle-free Injectors Market
- Halozyme (Antares Pharma)
- Inovio Pharmaceuticals
- Endo International
- Injex Pharma GmbH
- National Medical Products (J-Tip)
- Recipharm
- InsuJet
- Miracle Medical
- Medical International Technology (MIT)
- Akra Dermojet
- PharmaJet
- Portal Instruments
- Crossject
- Mika Medical
- Ferring Pharmaceuticals
- IntegriMedical
- Helcomin
- Nugen Medical
- Lepu Medical
- Beijing Quinovare
- Qingdao Zhonghui Health Industrial Group
Scope of the Report
| Item | Value |
|---|---|
| Quantitative Units | USD 241.7 million |
| Injection Type | Powder Injections, Liquid Injections, Depot or Projectile Injection |
| Application | Vaccine Delivery, Insulin Delivery, Others |
| Regions Covered | North America, Latin America, Western Europe, Eastern Europe, East Asia, South Asia Pacific, Middle East & Africa |
| Countries Covered | United States, China, Germany, India, United Kingdom, Japan, Brazil, France, Canada, South Korea, and 25+ additional countries |
| Key Companies Profiled | Halozyme (Antares Pharma), Inovio Pharmaceuticals, Endo International, Injex Pharma GmbH, National Medical Products (J-Tip), Recipharm, InsuJet, Miracle Medical, Medical International Technology (MIT) |
| Additional Attributes | Dollar sales by injection type and application categories, regional adoption trends across North America, East Asia, and Western Europe, competitive landscape with pharmaceutical device manufacturers and healthcare technology suppliers, healthcare provider preferences for delivery precision and patient safety, integration with pharmaceutical formulations and healthcare systems, innovations in injection technology and patient compliance enhancement, and development of connected delivery solutions with enhanced performance and healthcare optimization capabilities. |
Jet Needle-free Injectors Market by Segments
Injection Type:
- Powder Injections
- Liquid Injections
- Depot or Projectile Injection
Application:
- Vaccine Delivery
- Insulin Delivery
- Others
Region:
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Western Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Nordic
- BENELUX
- Rest of Western Europe
- Eastern Europe
- Russia
- Poland
- Rest of Eastern Europe
- East Asia
- China
- Japan
- South Korea
- South Asia Pacific
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia Pacific
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Other African Union
- Rest of Middle East & Africa
Frequently Asked Questions
How large is the needle-free injectors industry in 2026?
In 2026, the global needle-free injectors industry is expected to reach USD 21.7 billion across commercial and clinical delivery programs worldwide.
How big can the needle-free injectors industry become by 2036?
By 2036, the needle-free injectors industry is projected to reach USD 31.5 billion as repeat-dose use keeps supporting steady demand.
What growth rate is expected for the needle-free injectors industry through the forecast period?
The needle-free injectors industry is projected to expand at a CAGR of 3.8% from 2026 to 2036 during the forecast period.
Which product segment leads needle-free injectors demand by 2026?
Jet-based injectors are expected to account for 35.3% of product demand in 2026 because they retain the deepest installed base.
Which application segment leads needle-free injectors demand in 2026?
Vaccination is expected to represent 29.7% of application demand in 2026 as public programs reward fast and sharps-free dosing.
Which country is expected to post the fastest growth for needle-free injectors?
China is expected to record the fastest rise at 5.2% CAGR through 2036 as immunization scale and local device access support wider uptake.
What does the needle-free injectors market include?
The market includes sharps-free systems that deliver therapeutics through the skin by pressure or energy-assisted delivery across hospitals and self-use care.
How was the needle-free injectors forecast developed?
The forecast was developed through therapy-demand mapping and route analysis, then validated against channel spread and current company activity by region.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2020 to 2024 and Forecast, 2025 to 2035
- Historical Market Size Value (USD Million) Analysis, 2020 to 2024
- Current and Future Market Size Value (USD Million) Projections, 2025 to 2035
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2020 to 2024 and Forecast 2025 to 2035
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Injection Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Injection Type , 2020 to 2024
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Injection Type , 2025 to 2035
- Liquid Injections
- Powder Injections
- Depot or Projectile Injection
- Y to o to Y Growth Trend Analysis By Injection Type , 2020 to 2024
- Absolute $ Opportunity Analysis By Injection Type , 2025 to 2035
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application, 2020 to 2024
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application, 2025 to 2035
- Vaccine Delivery
- Insulin Delivery
- Others
- Y to o to Y Growth Trend Analysis By Application, 2020 to 2024
- Absolute $ Opportunity Analysis By Application, 2025 to 2035
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2020 to 2024
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2025 to 2035
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- USA
- Canada
- Mexico
- By Injection Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Injection Type
- By Application
- Key Takeaways
- Latin America Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Injection Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Injection Type
- By Application
- Key Takeaways
- Western Europe Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Injection Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Injection Type
- By Application
- Key Takeaways
- Eastern Europe Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Injection Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Injection Type
- By Application
- Key Takeaways
- East Asia Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- China
- Japan
- South Korea
- By Injection Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Injection Type
- By Application
- Key Takeaways
- South Asia and Pacific Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Injection Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Injection Type
- By Application
- Key Takeaways
- Middle East & Africa Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Injection Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Injection Type
- By Application
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2024
- By Injection Type
- By Application
- Canada
- Pricing Analysis
- Market Share Analysis, 2024
- By Injection Type
- By Application
- Mexico
- Pricing Analysis
- Market Share Analysis, 2024
- By Injection Type
- By Application
- Brazil
- Pricing Analysis
- Market Share Analysis, 2024
- By Injection Type
- By Application
- Chile
- Pricing Analysis
- Market Share Analysis, 2024
- By Injection Type
- By Application
- Germany
- Pricing Analysis
- Market Share Analysis, 2024
- By Injection Type
- By Application
- UK
- Pricing Analysis
- Market Share Analysis, 2024
- By Injection Type
- By Application
- Italy
- Pricing Analysis
- Market Share Analysis, 2024
- By Injection Type
- By Application
- Spain
- Pricing Analysis
- Market Share Analysis, 2024
- By Injection Type
- By Application
- France
- Pricing Analysis
- Market Share Analysis, 2024
- By Injection Type
- By Application
- India
- Pricing Analysis
- Market Share Analysis, 2024
- By Injection Type
- By Application
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2024
- By Injection Type
- By Application
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2024
- By Injection Type
- By Application
- China
- Pricing Analysis
- Market Share Analysis, 2024
- By Injection Type
- By Application
- Japan
- Pricing Analysis
- Market Share Analysis, 2024
- By Injection Type
- By Application
- South Korea
- Pricing Analysis
- Market Share Analysis, 2024
- By Injection Type
- By Application
- Russia
- Pricing Analysis
- Market Share Analysis, 2024
- By Injection Type
- By Application
- Poland
- Pricing Analysis
- Market Share Analysis, 2024
- By Injection Type
- By Application
- Hungary
- Pricing Analysis
- Market Share Analysis, 2024
- By Injection Type
- By Application
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2024
- By Injection Type
- By Application
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2024
- By Injection Type
- By Application
- South Africa
- Pricing Analysis
- Market Share Analysis, 2024
- By Injection Type
- By Application
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Injection Type
- By Application
- Competition Analysis
- Competition Deep Dive
- Halozyme (Antares Pharma)
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Inovio Pharmaceuticals
- Endo International
- Injex Pharma GmbH
- National Medical Products (J-Tip)
- Recipharm
- InsuJet
- Miracle Medical
- Medical International Technology (MIT)
- Akra Dermojet
- PharmaJet
- Portal Instruments
- Crossject
- Mika Medical
- Ferring Pharmaceuticals
- IntegriMedical
- Helcomin
- Nugen Medical
- Lepu Medical
- Beijing Quinovare
- Halozyme (Antares Pharma)
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2020 to 2035
- Table 2: Global Market Value (USD Million) Forecast by Injection Type , 2020 to 2035
- Table 3: Global Market Value (USD Million) Forecast by Application, 2020 to 2035
- Table 4: North America Market Value (USD Million) Forecast by Country, 2020 to 2035
- Table 5: North America Market Value (USD Million) Forecast by Injection Type , 2020 to 2035
- Table 6: North America Market Value (USD Million) Forecast by Application, 2020 to 2035
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2020 to 2035
- Table 8: Latin America Market Value (USD Million) Forecast by Injection Type , 2020 to 2035
- Table 9: Latin America Market Value (USD Million) Forecast by Application, 2020 to 2035
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2020 to 2035
- Table 11: Western Europe Market Value (USD Million) Forecast by Injection Type , 2020 to 2035
- Table 12: Western Europe Market Value (USD Million) Forecast by Application, 2020 to 2035
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2020 to 2035
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Injection Type , 2020 to 2035
- Table 15: Eastern Europe Market Value (USD Million) Forecast by Application, 2020 to 2035
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2020 to 2035
- Table 17: East Asia Market Value (USD Million) Forecast by Injection Type , 2020 to 2035
- Table 18: East Asia Market Value (USD Million) Forecast by Application, 2020 to 2035
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2020 to 2035
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Injection Type , 2020 to 2035
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by Application, 2020 to 2035
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2020 to 2035
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Injection Type , 2020 to 2035
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by Application, 2020 to 2035
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2020-2035
- Figure 3: Global Market Value Share and BPS Analysis by Injection Type , 2025 and 2035
- Figure 4: Global Market Y to o to Y Growth Comparison by Injection Type , 2025-2035
- Figure 5: Global Market Attractiveness Analysis by Injection Type
- Figure 6: Global Market Value Share and BPS Analysis by Application, 2025 and 2035
- Figure 7: Global Market Y to o to Y Growth Comparison by Application, 2025-2035
- Figure 8: Global Market Attractiveness Analysis by Application
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2025 and 2035
- Figure 10: Global Market Y to o to Y Growth Comparison by Region, 2025-2035
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2025-2035
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2025-2035
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2025-2035
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2025-2035
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2025-2035
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2025-2035
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2025-2035
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 20: North America Market Value Share and BPS Analysis by Injection Type , 2025 and 2035
- Figure 21: North America Market Y to o to Y Growth Comparison by Injection Type , 2025-2035
- Figure 22: North America Market Attractiveness Analysis by Injection Type
- Figure 23: North America Market Value Share and BPS Analysis by Application, 2025 and 2035
- Figure 24: North America Market Y to o to Y Growth Comparison by Application, 2025-2035
- Figure 25: North America Market Attractiveness Analysis by Application
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 27: Latin America Market Value Share and BPS Analysis by Injection Type , 2025 and 2035
- Figure 28: Latin America Market Y to o to Y Growth Comparison by Injection Type , 2025-2035
- Figure 29: Latin America Market Attractiveness Analysis by Injection Type
- Figure 30: Latin America Market Value Share and BPS Analysis by Application, 2025 and 2035
- Figure 31: Latin America Market Y to o to Y Growth Comparison by Application, 2025-2035
- Figure 32: Latin America Market Attractiveness Analysis by Application
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 34: Western Europe Market Value Share and BPS Analysis by Injection Type , 2025 and 2035
- Figure 35: Western Europe Market Y to o to Y Growth Comparison by Injection Type , 2025-2035
- Figure 36: Western Europe Market Attractiveness Analysis by Injection Type
- Figure 37: Western Europe Market Value Share and BPS Analysis by Application, 2025 and 2035
- Figure 38: Western Europe Market Y to o to Y Growth Comparison by Application, 2025-2035
- Figure 39: Western Europe Market Attractiveness Analysis by Application
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Injection Type , 2025 and 2035
- Figure 42: Eastern Europe Market Y to o to Y Growth Comparison by Injection Type , 2025-2035
- Figure 43: Eastern Europe Market Attractiveness Analysis by Injection Type
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by Application, 2025 and 2035
- Figure 45: Eastern Europe Market Y to o to Y Growth Comparison by Application, 2025-2035
- Figure 46: Eastern Europe Market Attractiveness Analysis by Application
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 48: East Asia Market Value Share and BPS Analysis by Injection Type , 2025 and 2035
- Figure 49: East Asia Market Y to o to Y Growth Comparison by Injection Type , 2025-2035
- Figure 50: East Asia Market Attractiveness Analysis by Injection Type
- Figure 51: East Asia Market Value Share and BPS Analysis by Application, 2025 and 2035
- Figure 52: East Asia Market Y to o to Y Growth Comparison by Application, 2025-2035
- Figure 53: East Asia Market Attractiveness Analysis by Application
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Injection Type , 2025 and 2035
- Figure 56: South Asia and Pacific Market Y to o to Y Growth Comparison by Injection Type , 2025-2035
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Injection Type
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by Application, 2025 and 2035
- Figure 59: South Asia and Pacific Market Y to o to Y Growth Comparison by Application, 2025-2035
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by Application
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Injection Type , 2025 and 2035
- Figure 63: Middle East & Africa Market Y to o to Y Growth Comparison by Injection Type , 2025-2035
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Injection Type
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by Application, 2025 and 2035
- Figure 66: Middle East & Africa Market Y to o to Y Growth Comparison by Application, 2025-2035
- Figure 67: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

