NTRK Fusion Gene Positive Advanced Solid Tumors Market : Global Industry Analysis 2015 - 2024 and Opportunity Assessment 2025 - 2035
NTRK Fusion Gene Positive Advanced Solid Tumors Market Size and Share Forecast Outlook 2025 to 2035
Historical Data Covered: 2015 - 2024 | Base Year: 2024 | Estimated Year: 2025 | Forecast Period: 2025 - 2035
NTRK Fusion Gene Positive Advanced Solid Tumors Market Size and Share Forecast Outlook 2025 to 2035
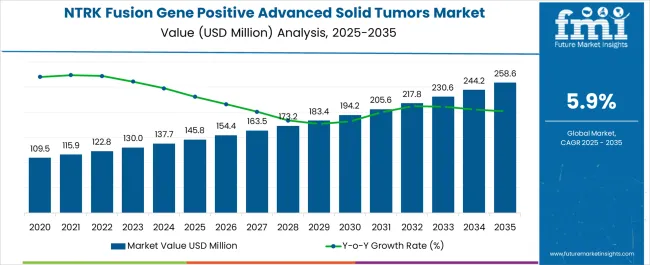
The NTRK fusion gene positive advanced solid tumors market is estimated to be valued at USD 145.8 million in 2025 and is projected to reach USD 258.6 million by 2035, registering a CAGR of 5.9 % over the forecast period.
Quick Stats for NTRK Fusion Gene Positive Advanced Solid Tumors Market
- NTRK Fusion Gene Positive Advanced Solid Tumors Market Industry Value (2025): USD 145.8 million
- NTRK Fusion Gene Positive Advanced Solid Tumors Market Forecast Value (2035): USD 258.6 million
- NTRK Fusion Gene Positive Advanced Solid Tumors Market Forecast CAGR: 5.9%
- Leading Segment in NTRK Fusion Gene Positive Advanced Solid Tumors Market in 2025: NTRK 1 Fusion Tumors (47.6%)
- Key Growth Region in NTRK Fusion Gene Positive Advanced Solid Tumors Market: North America, Asia-Pacific, Europe
- Top Players in NTRK Fusion Gene Positive Advanced Solid Tumors Market: A.G., Merck & Co., Inc., F. Hoffmann-La Roche AG, GlaxoSmithKline plc, Pfizer Inc., Plexxikon, Daiichi Sankyo Company Limited, Deciphera Pharmaceuticals, Inc., Eli Lilly and Company, Exelixis, Inc., Cephalon, Inc.
| Metric | Value |
|---|---|
| NTRK Fusion Gene Positive Advanced Solid Tumors Market Estimated Value in (2025 E) | USD 145.8 million |
| NTRK Fusion Gene Positive Advanced Solid Tumors Market Forecast Value in (2035 F) | USD 258.6 million |
| Forecast CAGR (2025 to 2035) | 5.9% |
Rationale for Segmental Growth in the NTRK Fusion Gene Positive Advanced Solid Tumors Market
Growth in this market is supported by the rising importance of precision oncology and increased identification of actionable genomic alterations in rare and aggressive tumors. Advances in genomic sequencing and companion diagnostics have enabled earlier and more reliable detection of NTRK fusions.
Pharmaceutical companies are expanding pipelines of targeted therapies aimed at TRK proteins, which is driving treatment uptake. Regulatory frameworks and accelerated approval pathways also contribute to innovation in this domain.
Hospitals and research centres are increasingly embedding genetic testing into standard oncology workflows, enabling timely treatment interventions and shifting from conventional chemotherapy toward highly targeted approaches.
Segmental Analysis
The market is segmented by Indication and End User and region. By Indication, the market is divided into NTRK 1 Fusion Tumors, NTRK 2 Fusion Tumors, and NTRK 3 Fusion Tumors. In terms of End User, the market is classified into Hospitals, Specialty Clinics, Cancer Centers, and Cancer Research Institutes. Regionally, the market is classified into North America, Latin America, Western Europe, Eastern Europe, Balkan & Baltic Countries, Russia & Belarus, Central Asia, East Asia, South Asia & Pacific, and the Middle East & Africa.
Insights into the NTRK 1 Fusion Tumors Indication Segment

The NTRK1 fusion-tumors segment is projected to represent 47.6 % of total market revenue by 2025, making it the leading indication. This is attributed to the relatively higher prevalence of NTRK1 gene fusions compared to other subtypes, combined with advances in next-generation sequencing facilitating earlier detection. Clinical evidence of durable responses and tumor-agnostic benefits of TRK inhibitors in patients with NTRK1 fusions has bolstered physician confidence. Ongoing trials and expansion of companion diagnostic testing are improving patient identification and access to therapy.
Insights into the Hospitals End User Segment
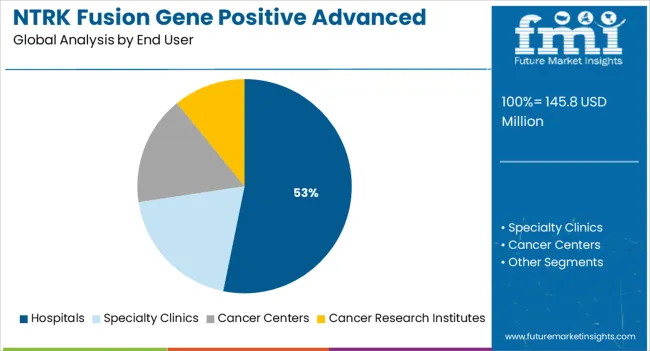
The hospitals segment is expected to account for 53.2 % of overall market revenue by 2025, establishing it as the largest end-user segment. This predominance is because hospitals house advanced oncology infrastructure, have access to genomic testing, and maintain multidisciplinary care teams. They are also the primary venues for clinical-trial enrolment, allowing early uptake of novel TRK inhibitors and combination therapies. Supportive reimbursement schemes and integrated care pathways further accelerate the uptake of NTRK-targeted treatments in hospitals. With precision oncology and comprehensive patient care becoming standard, hospitals remain the central delivery point for advanced therapeutic solutions in this market.
Revolutionizing Cancer Care: The Driving Forces behind the NTRK Fusion Gene Positive Advanced Solid Tumors Market
Advancements in precision medicine are among the main drivers of this market. Precision oncology focuses on tailoring treatment to a patient’s genetic profile, lifestyle, and medical history, enabling more effective interventions for patients with NTRK fusion-positive solid tumours. More effective targeted treatments and personalised plans drive demand for genetic testing and new therapies.
Better understanding of cancer genetics, especially NTRK fusions across multiple tumour types, has led to the development of innovative therapies targeting these alterations. As a result, patients with NTRK-fusion-positive advanced solid tumours are now able to access more efficient treatments tailored to their condition.
The increased demand for genetic testing to identify specific mutations further drives market growth. For example, in January 2025, KEYTRUDA (pembrolizumab) by Merck & Co., Inc. was approved by the U.S. Food and Drug Administration (FDA) for adult patients with stage IB, II or IIIA non-small cell lung cancer (NSCLC) after surgery and platinum-based chemotherapy; this underscores how precision treatments are shifting cancer-care paradigms.
A Pioneer in Precision: North America Leads the Way in NTRK Fusion Gene Positive Advanced Solid Tumors
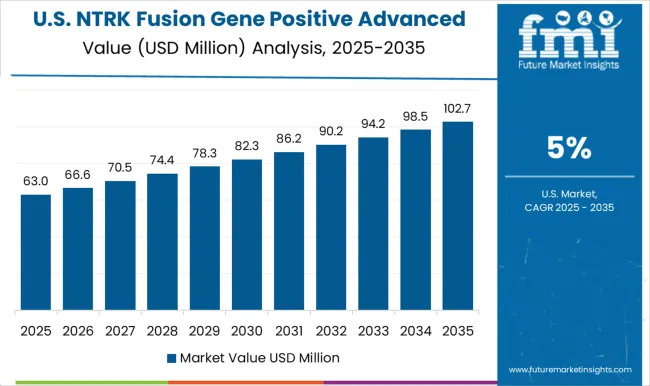
The North American market for NTRK fusion gene positive advanced solid tumors has seen significant growth in recent years. Leading pharmaceutical companies have made substantial investments in research and development, resulting in the approval of several drugs that target NTRK fusion gene positive advanced solid tumors.
As a result, North America is poised to continue its leadership role in the development of the NTRK fusion gene positive advanced solid tumors market in the coming years.
A Beacon of Hope: The Hospitals Segment Shines in the NTRK Fusion Gene Positive Advanced Solid Tumors Market
The most lucrative end user for the NTRK fusion gene positive advanced solid tumors market is the healthcare sector, specifically hospitals. With a high prevalence of advanced solid tumours and a growing focus on precision medicine, hospitals are at the forefront of the use of NTRK fusion gene positive advanced solid tumors treatments.
A patient with tumors who has mutated genes NTRK is called NTRK fusion gene positive tumors. Although NTRK fusions are rare and account to nearly 1% of solid tumors, the exact frequency is quite unclear. DNA Next Generation Sequencing (NSG) technology is a precise method to detect NTRK fusion genes. It has high specificity and sensitivity.
It can detects gene partners that may have high clinical implications in treatment. Currently Commercialized techniques such as DNA NGS panels, Foundation One may not detect NTRK fusions.
NTRK Fusion Gene Positive Advanced Solid Tumors Market: Drivers and Restraints
NTRK fusions has higher prevalence rate as they are found in most of solid cancer types. This is expected to drives NTRK fusion gene positive advanced solid tumors market. NTRK is a unique gene which has implications in many types of cancers and the development of latest drugs is expected to drive NTRK fusion gene positive advanced solid tumors market.
However, Poor healthcare infrastructure and high costs associated with genetic screening of NTRK fusion genes may hamper growth of NTRK fusion gene positive advanced solid tumors market in the future.
NTRK Fusion Gene Positive Advanced Solid Tumors Market: Overview
The global NTRK fusion gene positive advanced solid tumours market is expected to show steady growth over the forecast period due to technological advances, improved screening, and new drug launches. Early detection of NTRK fusion genes in patients contributes to market potential. The high potential is also driven by novel therapeutics in development for various tumour types with NTRK fusions. For instance, the FDA has approved TRK inhibitors such as Larotrectinib (VITRAKVI®) and Entrectinib for treatment of solid tumours with NTRK gene fusions.
NTRK Fusion Gene Positive Advanced Solid Tumors Market: Regional Overview
The global NTRK fusion gene positive advanced solid tumors market is expected to be dominated by North America owing to rising cancer clinical research and development activities. Latin America market is emerging in NTRK fusion gene positive advanced solid tumors market due to increased spending on healthcare infrastructure.
Europe market is expected to be the second most growing region in the global NTRK fusion gene positive advanced solid tumors market owing to early diagnosis and treatment adoption.
NTRK fusion gene positive advanced solid tumors market in East Asia and South Asia is expected to show significant growth due to improving healthcare infrastructure.
Middle East and Africa market is expected to be least lucrative region in the NTRK fusion gene positive advanced solid tumors market due to poor healthcare facilities and less disease screening facilities.
NTRK Fusion Gene Positive Advanced Solid Tumors Market: Key Players
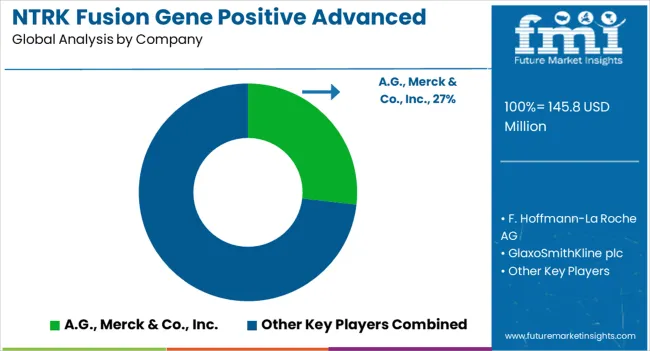
Examples of some of the market participants in NTRK fusion gene positive advanced solid tumors market identified across the value chain includes Bayer A.G., Merck & Co., Inc., F. Hoffmann-La Roche AG, Glaxosmithkline plc, Pfizer Inc., Plexxikon, Daiichi Sankyo Company Limited, Deciphera Pharmaceuticals, Inc., Eli Lilly and company, Exelixis, Inc., Cephalon, Inc., and others.
The research report on NTRK fusion gene positive advanced solid tumors market presents a comprehensive assessment of the market and contains thoughtful insights, facts, historical data, and, statistically supported and industry-validated market data. It also contains projections using a suitable set of assumptions and methodologies.
The report covers exhaust analysis on NTRK Fusion Gene Positive Advanced Solid Tumors Market
- Market Segments
- Market Dynamics
- Market Size
- Supply & Demand
- Current Trends/Issues/Challenges
- Competition & Companies involved
- Technology
- Value Chain
NTRK Fusion Gene Positive Advanced Solid Tumors Market: Segmentation
By indication:
- NTRK 1 Fusion Tumors
- NTRK 2 Fusion Tumors
- NTRK 3 Fusion Tumors
By end user:
- Hospitals
- Specialty Clinics
- Cancer Centers
- Cancer Research Institutes
By Region:
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
Frequently Asked Questions
How big is the NTRK fusion gene positive advanced solid tumors market in 2025?
The global NTRK fusion gene positive advanced solid tumors market is estimated to be valued at USD 145.8 million in 2025.
What will be the size of NTRK fusion gene positive advanced solid tumors market in 2035?
The market size for the NTRK fusion gene positive advanced solid tumors market is projected to reach USD 258.6 million by 2035.
How much will be the NTRK fusion gene positive advanced solid tumors market growth between 2025 and 2035?
The NTRK fusion gene positive advanced solid tumors market is expected to grow at a 5.9% CAGR between 2025 and 2035.
What are the key product types in the NTRK fusion gene positive advanced solid tumors market?
The key product types in NTRK fusion gene positive advanced solid tumors market are NTRK 1 fusion tumors, NTRK 2 fusion tumors and NTRK 3 fusion tumors.
Which end user segment to contribute significant share in the NTRK fusion gene positive advanced solid tumors market in 2025?
In terms of end user, hospitals segment to command 53.2% share in the NTRK fusion gene positive advanced solid tumors market in 2025.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand-side Trends
- Supply-side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2020 to 2024 and Forecast, 2025 to 2035
- Historical Market Size Value (USD Million) Analysis, 2020 to 2024
- Current and Future Market Size Value (USD Million) Projections, 2025 to 2035
- Y-o-Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2020 to 2024 and Forecast 2025 to 2035
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Indication
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Indication , 2020 to 2024
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Indication , 2025 to 2035
- NTRK 1 Fusion Tumors
- NTRK 2 Fusion Tumors
- NTRK 3 Fusion Tumors
- Y-o-Y Growth Trend Analysis By Indication , 2020 to 2024
- Absolute $ Opportunity Analysis By Indication , 2025 to 2035
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User, 2020 to 2024
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User, 2025 to 2035
- Hospitals
- Specialty Clinics
- Cancer Centers
- Cancer Research Institutes
- Y-o-Y Growth Trend Analysis By End User, 2020 to 2024
- Absolute $ Opportunity Analysis By End User, 2025 to 2035
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2020 to 2024
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2025 to 2035
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- USA
- Canada
- Mexico
- By Indication
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Indication
- By End User
- Key Takeaways
- Latin America Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Indication
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Indication
- By End User
- Key Takeaways
- Western Europe Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Indication
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Indication
- By End User
- Key Takeaways
- Eastern Europe Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Indication
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Indication
- By End User
- Key Takeaways
- East Asia Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- China
- Japan
- South Korea
- By Indication
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Indication
- By End User
- Key Takeaways
- South Asia and Pacific Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Indication
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Indication
- By End User
- Key Takeaways
- Middle East & Africa Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Indication
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Indication
- By End User
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2024
- By Indication
- By End User
- Canada
- Pricing Analysis
- Market Share Analysis, 2024
- By Indication
- By End User
- Mexico
- Pricing Analysis
- Market Share Analysis, 2024
- By Indication
- By End User
- Brazil
- Pricing Analysis
- Market Share Analysis, 2024
- By Indication
- By End User
- Chile
- Pricing Analysis
- Market Share Analysis, 2024
- By Indication
- By End User
- Germany
- Pricing Analysis
- Market Share Analysis, 2024
- By Indication
- By End User
- UK
- Pricing Analysis
- Market Share Analysis, 2024
- By Indication
- By End User
- Italy
- Pricing Analysis
- Market Share Analysis, 2024
- By Indication
- By End User
- Spain
- Pricing Analysis
- Market Share Analysis, 2024
- By Indication
- By End User
- France
- Pricing Analysis
- Market Share Analysis, 2024
- By Indication
- By End User
- India
- Pricing Analysis
- Market Share Analysis, 2024
- By Indication
- By End User
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2024
- By Indication
- By End User
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2024
- By Indication
- By End User
- China
- Pricing Analysis
- Market Share Analysis, 2024
- By Indication
- By End User
- Japan
- Pricing Analysis
- Market Share Analysis, 2024
- By Indication
- By End User
- South Korea
- Pricing Analysis
- Market Share Analysis, 2024
- By Indication
- By End User
- Russia
- Pricing Analysis
- Market Share Analysis, 2024
- By Indication
- By End User
- Poland
- Pricing Analysis
- Market Share Analysis, 2024
- By Indication
- By End User
- Hungary
- Pricing Analysis
- Market Share Analysis, 2024
- By Indication
- By End User
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2024
- By Indication
- By End User
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2024
- By Indication
- By End User
- South Africa
- Pricing Analysis
- Market Share Analysis, 2024
- By Indication
- By End User
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Indication
- By End User
- Competition Analysis
- Competition Deep Dive
- A.G., Merck & Co., Inc.
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- F. Hoffmann-La Roche AG
- GlaxoSmithKline plc
- Pfizer Inc.
- Plexxikon
- Daiichi Sankyo Company Limited
- Deciphera Pharmaceuticals, Inc.
- Eli Lilly and Company
- Exelixis, Inc.
- Cephalon, Inc.
- A.G., Merck & Co., Inc.
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2020-2035
- Table 2: Global Market Value (USD Million) Forecast by Indication , 2020-2035
- Table 3: Global Market Value (USD Million) Forecast by End User, 2020-2035
- Table 4: North America Market Value (USD Million) Forecast by Country, 2020-2035
- Table 5: North America Market Value (USD Million) Forecast by Indication , 2020-2035
- Table 6: North America Market Value (USD Million) Forecast by End User, 2020-2035
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2020-2035
- Table 8: Latin America Market Value (USD Million) Forecast by Indication , 2020-2035
- Table 9: Latin America Market Value (USD Million) Forecast by End User, 2020-2035
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2020-2035
- Table 11: Western Europe Market Value (USD Million) Forecast by Indication , 2020-2035
- Table 12: Western Europe Market Value (USD Million) Forecast by End User, 2020-2035
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2020-2035
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Indication , 2020-2035
- Table 15: Eastern Europe Market Value (USD Million) Forecast by End User, 2020-2035
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2020-2035
- Table 17: East Asia Market Value (USD Million) Forecast by Indication , 2020-2035
- Table 18: East Asia Market Value (USD Million) Forecast by End User, 2020-2035
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2020-2035
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Indication , 2020-2035
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by End User, 2020-2035
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2020-2035
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Indication , 2020-2035
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by End User, 2020-2035
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2020-2035
- Figure 3: Global Market Value (USD Million) Share and BPS Analysis by Region, 2025 and 2035
- Figure 4: Global Market Y-o-Y Growth Comparison by Region, 2025-2035
- Figure 5: Global Market Attractiveness Analysis by Region
- Figure 6: North America Market Incremental Dollar Opportunity, 2025-2035
- Figure 7: Latin America Market Incremental Dollar Opportunity, 2025-2035
- Figure 8: Western Europe Market Incremental Dollar Opportunity, 2025-2035
- Figure 9: Eastern Europe Market Incremental Dollar Opportunity, 2025-2035
- Figure 10: East Asia Market Incremental Dollar Opportunity, 2025-2035
- Figure 11: South Asia and Pacific Market Incremental Dollar Opportunity, 2025-2035
- Figure 12: Middle East & Africa Market Incremental Dollar Opportunity, 2025-2035
- Figure 13: North America Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 14: Latin America Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 15: Western Europe Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 16: Eastern Europe Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 17: East Asia Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 18: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 19: Middle East & Africa Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 20: Global Market - Tier Structure Analysis
- Figure 21: Global Market - Company Share Analysis

