Peripheral Intravenous Catheter Market : Global Industry Analysis 2016 - 2025 and Opportunity Assessment 2026 - 2036
Peripheral intravenous catheter market is segmented by product type, technology, end user, and region. Product type coverage includes short peripheral IV catheter, integrated or closed peripheral IV catheter, and others. Technology coverage includes safety PIVC and conventional PIVC. End-user coverage includes hospitals, clinics, ambulatory surgical centers, and others. Forecast is provided from 2026 to 2036.
Historical Data Covered: 2016 - 2025 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2026 - 2036
Peripheral Intravenous Catheter Market Size, Forecast and Outlook By FMI
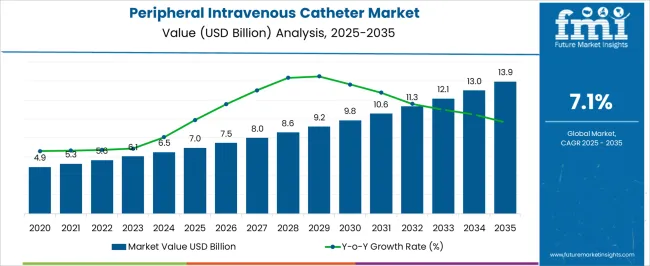
Peripheral intravenous catheter market is expected to expand from USD 7.50 billion in 2026 to USD 14.89 billion by 2036. The market is anticipated to register 7.1% CAGR during the forecast period. Short peripheral IV catheters are likely to lead with 35.1% share, while hospitals are projected to account for 54.6% of end-user demand.
Summary of the Peripheral Intravenous Catheter Market
- Demand and Growth Drivers
- Rising hospitalization volumes are increasing routine vascular access needs.
- Infection prevention goals are shifting demand toward closed catheter systems.
- Needlestick safety rules are pushing hospitals toward safety-engineered devices.
- Longer dwell-time designs are reducing repeat insertions for patients.
- Product and Segment View
- Short peripheral IV catheters lead due to routine hospital use.
- Safety PIVC demand is rising as hospitals focus on staff protection.
- Closed systems are gaining preference for blood exposure reduction.
- Antimicrobial coatings are becoming important in high-risk patient care.
- Geography and Competitive Outlook
- North America retains strong value due to premium safety device adoption.
- Asia Pacific shows faster growth through hospital capacity expansion.
- India benefits from healthcare access programs and private hospital growth.
- Leading companies compete through safety design and clinical evidence.
- Analyst Opinion
- Sabyasachi Ghosh, Principal Consultant for Healthcare at FMI, opines, “Peripheral intravenous catheters are moving from basic access products into safety and infection prevention systems. Manufacturers that document lower complication rates and reduce total treatment cost will gain stronger hospital acceptance through 2036.”
- Peripheral Intravenous Catheter Market Value Analysis
- Peripheral IV catheter demand is moving toward integrated safety systems.
- Hospitals are prioritizing closed designs that reduce blood exposure risks.
- Adoption gains from infection control needs and clinician safety standards.
- Spending will remain tied to clinical evidence and training support.
Peripheral Intravenous Catheter Market Definition
The peripheral intravenous catheter market covers the global production and supply of medical devices inserted into peripheral veins to administer fluids, medications, blood products, and parenteral nutrition, or to obtain blood samples. These catheters are typically made from medical grade polymers such as polyurethane or polytetrafluoroethylene and are available in various lengths and gauges for different clinical applications. They are used across hospitals, clinics, ambulatory surgical centers, and home care settings where intravenous access is required for patient treatment.
Market Inclusions
The report includes global and regional market size estimates in value terms, with a forecast period from 2026 to 2036. It provides segmentation by Product Type, Technology, End User, and Region. The analysis incorporates manufacturing technology trends, material science developments, safety mechanism innovations, and infection control protocol compliance. Regional demand dynamics, country level growth rates, and competitive positioning are also evaluated as per FMI methodology.
Market Exclusions
The scope excludes central venous catheters, peripherally inserted central catheters, and implantable ports designed for long term vascular access. It also excludes intravenous administration sets and infusion pumps when sold separately from catheters. Custom catheter designs for specialized research applications are not considered due to negligible market contribution.
Research Methodology
- Primary Research: Interviews were conducted with medical device manufacturers, hospital procurement managers, clinical nurse educators, and infection control specialists.
- Desk Research: Public company filings, hospital admission statistics, needlestick injury surveillance data, and infection control guideline documents were reviewed.
- Market Sizing and Forecasting: A hybrid top down and bottom up model was constructed, reconstructing demand from hospital admission figures, surgical procedure volumes, and per patient catheter utilization rates.
- Data Validation and Update Cycle: Outputs were subjected to variance testing against production shipment data, trade flow benchmarking under relevant HS codes, and structured internal review prior to publication.
Peripheral Intravenous Catheter Market Drivers, Restraints, and Opportunities
The market has historically functioned as a high volume consumables category driven by the fundamental requirement for vascular access in virtually all hospitalized patients. Its baseline scale is anchored in the millions of catheter insertions performed daily across global healthcare systems.
FMI analysts observe a bifurcation between conventional catheters purchased on price for low risk applications and advanced safety catheters adopted in settings where regulatory compliance, healthcare worker protection, and infection prevention justify premium pricing. Standard catheters face margin compression where procurement is centralized by group purchasing organizations, while growth is concentrated in products that offer documented advantages in complication reduction validated through clinical studies.
- Needlestick Safety and Prevention Act legacy: The 2000 USA federal law requiring safety engineered sharps devices created a regulatory framework that has been adopted in modified form by dozens of countries. OSHA enforcement data shows that needlestick injuries among hospital workers declined by 38% between 2001 and 2024, demonstrating the public health impact of safety device mandates and creating continuing demand for technology upgrades. [1]
- Infection control guideline evolution: The Centers for Disease Control and Prevention's 2024 update to the Guidelines for the Prevention of Intravascular Catheter Related Infections includes strengthened recommendations for antimicrobial coated catheters in patients with high infection risk and for closed system catheters to reduce blood exposure during insertion. These evidence based guidelines directly influence hospital purchasing decisions. [2]
- Extended dwell catheter development: Manufacturers have developed peripheral catheters designed for dwell times of seven to fourteen days compared to the standard 72 to 96 hours. Extended dwell catheters reduce the number of insertions required per patient admission, decreasing patient discomfort and clinician workload while increasing per catheter revenue for manufacturers. [3]
Segmental analysis
Peripheral Intravenous Catheter Market Analysis by Product Type
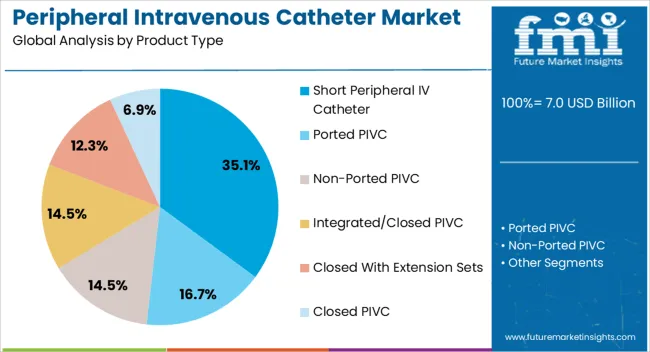
Based on FMI's peripheral intravenous catheter market report, demand for short peripheral IV catheters remains the leading product category, capturing 35.1% of the market share. This dominance is sustained by their suitability for the vast majority of routine IV therapy applications, ease of insertion by nursing staff, and lower cost compared to integrated or midline catheters. Short catheters are typically inserted in peripheral veins of the hand or forearm and remain in place for 72 to 96 hours, covering the duration of most hospital admissions.
- Closed system innovation: Manufacturers have developed integrated closed system short catheters that include a pre attached extension tube with needleless connector, eliminating the need for separate assembly and reducing blood exposure risk. Becton Dickinson's Nexiva Closed IV Catheter System, launched in India and other markets, exemplifies this trend toward combining the convenience of short catheters with safety features previously available only in premium products. [4]
- Material science advancement: Traditional polytetrafluoroethylene catheters are being replaced by polyurethane formulations that soften at body temperature, reducing vessel irritation and extending dwell time. These advanced materials command premium pricing while improving patient outcomes.
- Pediatric applications: Specialized short catheters designed for pediatric patients with smaller veins represent a growing niche within the segment, requiring precision manufacturing that favors established players with quality systems validated for sensitive populations.
Peripheral Intravenous Catheter Market Analysis by Technology
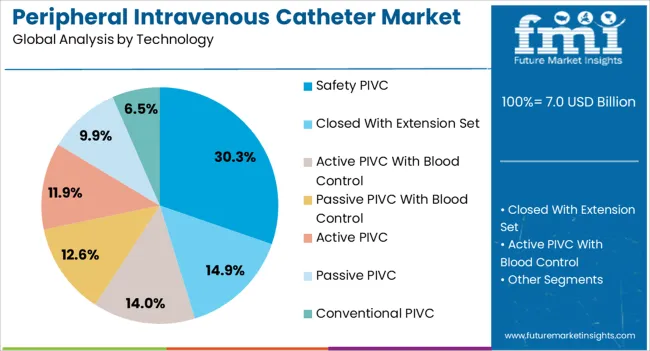
Safety PIVC technology generates maximum demand at 30.3% for peripheral intravenous catheters because healthcare facilities prioritize protection of clinical staff from needlestick injuries and patients from blood exposure. The segment's dominance is reinforced by regulatory mandates, accreditation requirements, and the documented reduction in occupational injuries achieved by safety devices.
- Passive safety mechanisms: Modern safety catheters incorporate passive safety features that activate automatically upon needle withdrawal, requiring no additional steps from clinicians. These designs address the limitation of active safety devices that require user activation and are sometimes bypassed in busy clinical settings.
- Antimicrobial technology integration: Leading manufacturers are combining safety mechanisms with antimicrobial coatings such as chlorhexidine or silver sulfadiazine that reduce bacterial colonization and catheter associated bloodstream infections. Clinical studies demonstrate that antimicrobial safety catheters reduce infection rates by 40 to 60% compared to conventional devices. [5]
- Blood control features: Closed system safety catheters incorporate valves that prevent blood exposure during insertion and removal, addressing both clinician safety and infection prevention. These features are increasingly specified in hospital protocols for patients with bloodborne pathogens.
Peripheral Intravenous Catheter Market Analysis by End User
Hospitals account for 54.6% of peripheral intravenous catheter demand because they concentrate the highest volume of patients requiring IV therapy, from emergency department admissions through surgical procedures to general ward care. The segment's dominance is reinforced by hospital procurement budgets that can accommodate premium safety devices and by infection control committees that drive technology adoption.
- Group purchasing organization influence: Hospital purchasing is increasingly centralized through group purchasing organizations that negotiate system wide contracts. These agreements favor manufacturers offering comprehensive product portfolios and documented clinical evidence, creating barriers for single product vendors. [6]
- Outpatient shift: While hospitals remain dominant, ambulatory surgical centers and home care settings represent the fastest growing end user segments. The shift of procedures to outpatient settings creates demand for catheters designed for shorter dwell times and simplified management by patients or caregivers.
- Training requirements: Hospitals investing in safety catheters must also invest in staff training to ensure proper device use. Manufacturers offering comprehensive education programs gain competitive advantage by reducing the implementation burden on hospital education departments.
Regional Analysis
The peripheral intravenous catheter market is segmented geographically across North America, Europe, Asia Pacific, Latin America, and Middle East and Africa. Growth dynamics differ based on healthcare infrastructure maturity, regulatory requirements for safety devices, hospitalization rates, and medical device reimbursement policies.
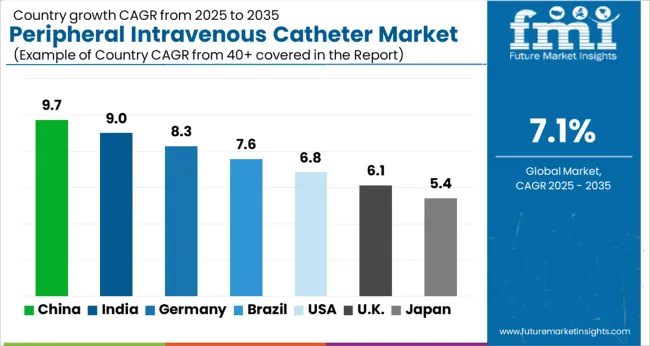
| Country | CAGR 2026 to 2036 |
|---|---|
| India | 7.5% |
| United States | 7.5% |
| China | 7.0% |
| Italy | 6.7% |
| Germany | 6.2% |
| United Kingdom | 5.9% |
| Japan | 5.5% |
Source: Future Market Insights analysis, based on proprietary forecasting model and primary research
North America Market Analysis
North America functions as the most mature regional market for peripheral intravenous catheters, characterized by near universal adoption of safety engineered devices, strong regulatory enforcement, and sophisticated group purchasing organization procurement. The region accounts for the highest average selling prices globally due to premium technology adoption.
-
United States: Demand for peripheral intravenous catheters in the United States is forecasted to advance at 7.5% CAGR through 2036. Growth is driven not by increasing patient volumes alone but by technology upgrades from conventional to safety catheters and from basic safety to closed system antimicrobial devices. The 2024 American Hospital Association Annual Survey documented 33,356,853 total admissions, representing sustained demand across emergency, surgical, and general care settings. [7]
Medicare and Medicaid reimbursement policies that incentivize infection prevention and quality outcomes support hospital investment in premium catheters. The Centers for Medicare and Medicaid Services hospital acquired condition reduction program includes catheter associated bloodstream infections among targeted complications, creating financial incentives for prevention technology adoption. [8]
FMI's analysis of peripheral intravenous catheter market in North America consists of country wise assessment that includes the United States and Canada. Readers can find OSHA enforcement tracking, group purchasing organization contract analysis, and infection prevention reimbursement trends.
Europe Market Analysis
Europe acts as a diverse regional market with variation in safety device adoption rates, healthcare system structures, and procurement approaches across countries. The region benefits from strong medical device regulation under the Medical Device Regulation framework and active health technology assessment programs that evaluate clinical evidence.
- Italy: Demand for peripheral intravenous catheters in Italy is projected to rise at 6.7% CAGR through 2036. Growth is influenced by Italy's strong healthcare infrastructure, government initiatives to improve healthcare services, and the country's position in advanced medical research. Italy's pharmaceutical industry concentration and clinical trial activity create demand for reliable vascular access devices in research settings. [9]
- Germany: Germany demonstrates a 6.2% CAGR, reflecting its robust hospital system, aging population, and medical technology leadership. German hospitals operate under diagnosis related group reimbursement that creates incentives for efficient care delivery, supporting adoption of catheters that reduce complications and extend dwell time.
United Kingdom
The United Kingdom market grows at 5.9% CAGR, shaped by National Health Service procurement priorities and infection prevention targets. The NHS England's goal to reduce healthcare associated infections by 50% by 2030 creates sustained demand for antimicrobial and closed system catheters. [10]
FMI's analysis of peripheral intravenous catheter market in Europe consists of country wise assessment that includes Germany, United Kingdom, France, Italy, and Spain. Readers can find Medical Device Regulation compliance tracking, health technology assessment outcomes, and national infection prevention target monitoring.
Asia Pacific Market Analysis
Asia Pacific remains the fastest growing regional market for peripheral intravenous catheters, driven by hospital infrastructure expansion, increasing surgical volumes, and rising chronic disease prevalence requiring IV therapy.
- India: Peripheral intravenous catheter demand in India is projected to rise at 7.5% CAGR through 2036. Growth is supported by the Ayushman Bharat scheme's expansion of hospital access, increasing medical tourism attracting patients from neighboring countries, and rising local manufacturing capacity. Key players including Becton Dickinson have launched advanced products in India, such as the Nexiva Closed IV Catheter System, addressing the growing need for safer and more efficient solutions. [4]
- India's market is distinctive for its dual structure with international brands capturing premium segments in private hospitals and domestic manufacturers serving price sensitive public sector procurement.
- China: Demand for peripheral intravenous catheters in China is projected to rise at 7.0% CAGR through 2036. Growth is driven by aging population demographics, rising chronic disease prevalence, and government investments in primary care facilities. China's concentration of medical research centers and clinical trial activity creates additional demand for reliable vascular access devices in research settings. [11]
Competitive Aligners for Market Players
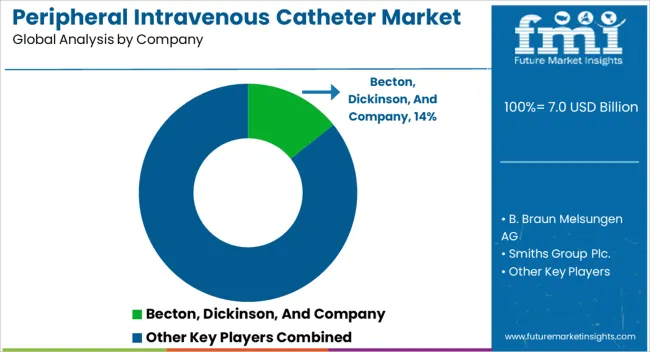
The peripheral intravenous catheter market is consolidated with Becton Dickinson and Company, B. Braun Melsungen AG, and Smiths Group Plc representing the largest global players. Terumo Corporation, Teleflex Incorporated, and NIPRO Medical Corporation maintain strong positions through regional strength and specialized product portfolios.
The primary competitive variable has shifted from basic device functionality to integrated safety systems, antimicrobial technology performance, and clinical evidence generation. Manufacturers investing in randomized controlled trials documenting reduced complication rates gain preferred status in hospital procurement decisions.
Buyer behavior in this market reflects the influence of infection control committees, nursing education departments, and group purchasing organizations. Decisions are increasingly based on total cost of care analysis rather than device acquisition cost alone.
Recent Developments
- Becton Dickinson - Product Launch: Becton Dickinson launched its Nexiva Closed IV Catheter System in India, offering advanced safety features and reducing the risk of needlestick injuries, demonstrating the company's commitment to emerging market expansion. [4]
- Teleflex Incorporated - Technology Development: Teleflex continued development of its Arrow branded safety catheters with antimicrobial surface technology for infection prevention applications.
- B. Braun Melsungen AG - Portfolio Expansion: B. Braun expanded its Introcan Safety catheter line with new sizes and configurations for specialized clinical applications.
Key Players in the Peripheral Intravenous Catheter Market
- Becton, Dickinson, and Company
- B. Braun Melsungen AG
- Smiths Group Plc.
- Terumo Corporation
- Venner Medical
- Vygon
- Teleflex Incorporated
- C. R. Bard, Inc.
- NIPRO Medical Corporation
- Argon Medical Devices, Inc.
Scope of Report
| Metric | Value |
|---|---|
| Quantitative Units | USD 7.50 billion 2026 to USD 14.89 billion 2036 at a CAGR of 7.1% |
| Market Definition | The peripheral intravenous catheter market comprises the global production and supply of medical devices inserted into peripheral veins to administer fluids, medications, blood products, and parenteral nutrition, manufactured from medical grade polymers with safety features to reduce needlestick injuries and infection risks. |
| Product Type Segmentation | Short Peripheral IV Catheter, Integrated/Closed Peripheral IV Catheter, Others |
| Technology Segmentation | Safety PIVC, Conventional PIVC |
| End User Segmentation | Hospitals, Clinics, Ambulatory Surgical Centers, Others |
| Region Segmentation | North America, Europe, Asia Pacific, Latin America, Middle East and Africa |
| Countries Covered | United States, Canada, Germany, United Kingdom, France, Italy, Spain, China, India, Japan, South Korea, Brazil and 40 plus countries |
| Key Companies Profiled | Becton, Dickinson, And Company, B. Braun Melsungen AG, Smiths Group Plc., Terumo Corporation, Venner Medical, Vygon, Teleflex Incorporated, C. R. Bard, Inc., NIPRO Medical Corporation, Argon Medical Devices, Inc. |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid top down and bottom up market modeling validated through primary interviews with medical device manufacturers, hospital procurement managers, and infection control specialists supported by trade benchmarking and hospital admission analysis |
Peripheral Intravenous Catheter Market Analysis by Segments
Product Type:
- Short Peripheral IV Catheter
- Integrated/Closed Peripheral IV Catheter
- Others
Technology:
- Safety PIVC
- Conventional PIVC
End User:
- Hospitals
- Clinics
- Ambulatory Surgical Centers
- Others
Region:
- North America (USA, Canada, Mexico)
- Latin America (Brazil, Chile, Rest of Latin America)
- Western Europe (Germany, UK, Italy, Spain, France, Nordic, BENELUX, Rest of Western Europe)
- Eastern Europe (Russia, Poland, Hungary, Balkan and Baltic, Rest of Eastern Europe)
- East Asia (China, Japan, South Korea)
- South Asia and Pacific (India, ASEAN, Australia and New Zealand, Rest of South Asia and Pacific)
- Middle East and Africa (Kingdom of Saudi Arabia, Other GCC Countries, Turkiye, South Africa, Other African Union, Rest of Middle East and Africa)
Bibliography
- [1] Occupational Safety and Health Administration, U.S. Department of Labor. (2024). Needlestick Safety and Prevention Act 25th Anniversary Impact Assessment. Washington, D.C.: OSHA.
- [2] Centers for Disease Control and Prevention. (2024). Guidelines for the Prevention of Intravascular Catheter Related Infections 2024 Update. Atlanta: CDC.
- [3] Journal of the Association for Vascular Access. (2025). Extended dwell peripheral catheters: Clinical outcomes and economic analysis. JAVA, 30(1), 45 52.
- [4] Becton Dickinson and Company. (2024). BD launches Nexiva Closed IV Catheter System in India. BD Press Release.
- [5] Infection Control and Hospital Epidemiology. (2024). Antimicrobial safety catheters for infection prevention: A multicenter study. ICHE, 45(3), 289 297.
- [6] Healthcare Supply Chain Association. (2025). Group purchasing organization impact on medical device procurement. HSCA Annual Report.
- [7] American Hospital Association. (2024). AHA Annual Survey of Hospitals FY 2024. Chicago: AHA.
- [8] Centers for Medicare and Medicaid Services. (2024). Hospital Acquired Condition Reduction Program Fiscal Year 2025. Baltimore: CMS.
- [9] Italian Ministry of Health. (2025). National Health Service Performance Report 2024. Rome: Ministero della Salute.
- [10] NHS England. (2024). NHS Infection Prevention and Control Strategy 2024 2030. London: NHS England.
- [11] National Health Commission, People's Republic of China. (2024). China Health Statistics Yearbook 2024. Beijing: NHC.
This Report Addresses
- Market intelligence to enable structured strategic decision making across medical device manufacturers, hospital procurement executives, and infection control program leaders.
- Market size estimation and 10 year revenue forecasts from 2026 to 2036 including validated CAGR and absolute dollar opportunity sizing.
- Growth opportunity mapping across Product Type, Technology, End User, and regional demand pockets with emphasis on safety engineered devices and antimicrobial catheter technologies.
- Segment and regional forecasting covering short and closed system catheters alongside demand trajectories across hospital and ambulatory care settings.
- Competition strategy assessment including safety technology development, clinical evidence generation, and clinician education programs shaping hospital purchasing decisions.
- Product and compliance tracking focused on needlestick prevention effectiveness, antimicrobial coating performance, and infection reduction validation through clinical studies.
- Regulatory impact analysis covering OSHA needlestick enforcement, CDC infection prevention guidelines, and national health system procurement policies influencing technology adoption.
- Report delivery in PDF, Excel, and presentation ready formats to support executive planning, product development alignment, and market access strategy development.
Frequently Asked Questions
How large is the demand for peripheral intravenous catheters in the global market in 2026?
Demand for peripheral intravenous catheters is estimated to be valued at USD 7.50 billion in 2026.
What will be the market size of peripheral intravenous catheters in the global market by 2036?
Market size for peripheral intravenous catheters is projected to reach USD 14.89 billion by 2036.
What is the expected demand growth for peripheral intravenous catheters between 2026 and 2036?
Demand for peripheral intravenous catheters is expected to grow at a 7.1% CAGR between 2026 and 2036.
Which product type segment is poised to lead global sales?
Short peripheral IV catheters are expected to remain the leading product type, anchored by dominance in routine short term IV therapy across all healthcare settings, holding 35.1% share in 2025.
Which technology segment dominates the peripheral intravenous catheter market?
Safety PIVC technology leads at 30.3% share in 2025, reflecting regulatory compliance requirements and hospital prioritization of healthcare worker protection.
Which end user segment leads peripheral intravenous catheter adoption?
Hospitals are the dominant end user segment at 54.6% share in 2025, driven by high patient volumes, complex treatment regimens, and comprehensive procurement budgets.
What is driving demand in India?
Expansion of hospital infrastructure under the Ayushman Bharat scheme, increasing medical tourism, and rising local manufacturing capacity with international brand product launches are driving demand in India.
What is India's growth outlook in this report?
India is projected to expand at a 7.5% CAGR during 2026 to 2036.
What is driving demand in the United States?
Technology upgrades from conventional to safety catheters, Medicare infection prevention incentives, and sustained hospitalization volumes documented at 33.4 million annual admissions are driving demand in the United States.
What is the United States growth outlook in this report?
The United States is projected to expand at a 7.5% CAGR during 2026 to 2036.
What is driving demand in China?
Aging population demographics, rising chronic disease prevalence, government investments in primary care facilities, and concentration of medical research centers are driving demand in China.
What is China's growth outlook in this report?
China is projected to expand at a 7.0% CAGR during 2026 to 2036.
Does the report cover Italy in its regional analysis?
Yes, Italy is included within Europe and is covered in the country wise assessment.
What is Italy's growth outlook in this report?
Italy is projected to expand at a 6.7% CAGR during 2026 to 2036.
Does the report cover Germany in its regional analysis?
Yes, Germany is included within Europe under the regional coverage framework.
What is Germany's growth outlook in this report?
Germany is projected to expand at a 6.2% CAGR during 2026 to 2036.
Does the report cover the United Kingdom in its regional analysis?
Yes, the United Kingdom is included within Europe under the regional coverage framework.
What is the United Kingdom's growth outlook in this report?
The United Kingdom is projected to expand at a 5.9% CAGR during 2026 to 2036.
Does the report cover Japan in its regional analysis?
Yes, Japan is included within East Asia under the regional scope of analysis.
What is Japan's growth outlook in this report?
Japan is projected to expand at a 5.5% CAGR during 2026 to 2036.
What are peripheral intravenous catheters and what are they mainly used for?
Peripheral intravenous catheters are medical devices inserted into peripheral veins to administer fluids, medications, blood products, and parenteral nutrition, or to obtain blood samples. They are mainly used in hospitals, clinics, and ambulatory settings for patients requiring intravenous therapy.
What does the peripheral intravenous catheter market mean in this report?
It refers to the global production and supply of peripheral intravenous catheters by product type, technology, end user, and region, measured in value terms at the manufacturer shipment layer.
What is included in the scope of this peripheral intravenous catheter market report?
Scope includes market sizing and forecasting for 2026 to 2036, segmentation by product type, technology, end user, and region, along with competitive assessment, regulatory impact analysis, and country wise outlook.
What is excluded from the scope of this report?
The scope excludes central venous catheters, peripherally inserted central catheters, implantable ports, intravenous administration sets and infusion pumps sold separately, and custom research catheters.
How are regulatory developments shaping the peripheral intravenous catheter market?
Regulatory developments including the Needlestick Safety and Prevention Act framework, OSHA enforcement, CDC infection prevention guidelines, and national health system procurement policies are shifting competitive advantage from conventional devices to safety engineered catheters with documented complication reduction validated through clinical studies.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product Type , 2026 to 2036
- Short Peripheral IV Catheter
- Integrated/Closed Peripheral IV Catheter
- Short Peripheral IV Catheter
- Y to o to Y Growth Trend Analysis By Product Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Product Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Technology
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Technology, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Technology, 2026 to 2036
- Safety PIVC
- Conventional PIVC
- Safety PIVC
- Y to o to Y Growth Trend Analysis By Technology, 2021 to 2025
- Absolute $ Opportunity Analysis By Technology, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End Use
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End Use, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End Use, 2026 to 2036
- Hospitals
- Hospitals
- Y to o to Y Growth Trend Analysis By End Use, 2021 to 2025
- Absolute $ Opportunity Analysis By End Use, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product Type
- By Technology
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Technology
- By End Use
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product Type
- By Technology
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Technology
- By End Use
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product Type
- By Technology
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Technology
- By End Use
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product Type
- By Technology
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Technology
- By End Use
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product Type
- By Technology
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Technology
- By End Use
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product Type
- By Technology
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Technology
- By End Use
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product Type
- By Technology
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Technology
- By End Use
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Technology
- By End Use
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Technology
- By End Use
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Technology
- By End Use
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Technology
- By End Use
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Technology
- By End Use
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Technology
- By End Use
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Technology
- By End Use
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Technology
- By End Use
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Technology
- By End Use
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Technology
- By End Use
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Technology
- By End Use
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Technology
- By End Use
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Technology
- By End Use
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Technology
- By End Use
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Technology
- By End Use
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Technology
- By End Use
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Technology
- By End Use
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Technology
- By End Use
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Technology
- By End Use
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Technology
- By End Use
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Technology
- By End Use
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Technology
- By End Use
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product Type
- By Technology
- By End Use
- Competition Analysis
- Competition Deep Dive
- Becton, Dickinson, and Company
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- B. Braun Melsungen AG
- Smiths Group Plc.
- Terumo Corporation
- Venner Medical
- Vygon
- Becton, Dickinson, and Company
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 10: Latin America Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 13: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Western Europe Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 15: Western Europe Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 17: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 18: Eastern Europe Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 19: Eastern Europe Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 20: Eastern Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 21: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: East Asia Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 23: East Asia Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 24: East Asia Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 25: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: South Asia and Pacific Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 27: South Asia and Pacific Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 28: South Asia and Pacific Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 29: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 30: Middle East & Africa Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 31: Middle East & Africa Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 32: Middle East & Africa Market Value (USD Million) Forecast by End Use, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Product Type
- Figure 6: Global Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Technology
- Figure 9: Global Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by End Use
- Figure 12: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Region
- Figure 15: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 20: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 21: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 23: North America Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 25: North America Market Attractiveness Analysis by Product Type
- Figure 26: North America Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 27: North America Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 28: North America Market Attractiveness Analysis by Technology
- Figure 29: North America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 31: North America Market Attractiveness Analysis by End Use
- Figure 32: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 33: Latin America Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 34: Latin America Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 35: Latin America Market Attractiveness Analysis by Product Type
- Figure 36: Latin America Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 37: Latin America Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 38: Latin America Market Attractiveness Analysis by Technology
- Figure 39: Latin America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 40: Latin America Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 41: Latin America Market Attractiveness Analysis by End Use
- Figure 42: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 43: Western Europe Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 44: Western Europe Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 45: Western Europe Market Attractiveness Analysis by Product Type
- Figure 46: Western Europe Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 47: Western Europe Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 48: Western Europe Market Attractiveness Analysis by Technology
- Figure 49: Western Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 50: Western Europe Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 51: Western Europe Market Attractiveness Analysis by End Use
- Figure 52: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 53: Eastern Europe Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 54: Eastern Europe Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 55: Eastern Europe Market Attractiveness Analysis by Product Type
- Figure 56: Eastern Europe Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 57: Eastern Europe Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 58: Eastern Europe Market Attractiveness Analysis by Technology
- Figure 59: Eastern Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 60: Eastern Europe Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 61: Eastern Europe Market Attractiveness Analysis by End Use
- Figure 62: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 63: East Asia Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 64: East Asia Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 65: East Asia Market Attractiveness Analysis by Product Type
- Figure 66: East Asia Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 67: East Asia Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 68: East Asia Market Attractiveness Analysis by Technology
- Figure 69: East Asia Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 70: East Asia Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 71: East Asia Market Attractiveness Analysis by End Use
- Figure 72: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 73: South Asia and Pacific Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 74: South Asia and Pacific Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 75: South Asia and Pacific Market Attractiveness Analysis by Product Type
- Figure 76: South Asia and Pacific Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 77: South Asia and Pacific Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 78: South Asia and Pacific Market Attractiveness Analysis by Technology
- Figure 79: South Asia and Pacific Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 80: South Asia and Pacific Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 81: South Asia and Pacific Market Attractiveness Analysis by End Use
- Figure 82: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 83: Middle East & Africa Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 84: Middle East & Africa Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 85: Middle East & Africa Market Attractiveness Analysis by Product Type
- Figure 86: Middle East & Africa Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 87: Middle East & Africa Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 88: Middle East & Africa Market Attractiveness Analysis by Technology
- Figure 89: Middle East & Africa Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 90: Middle East & Africa Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 91: Middle East & Africa Market Attractiveness Analysis by End Use
- Figure 92: Global Market - Tier Structure Analysis
- Figure 93: Global Market - Company Share Analysis

