Self Testing Market
The self testing market is segmented by Test Type (Glucose Monitoring, Pregnancy Tests, Cholesterol Testing, HIV Testing, Others), Sample Type (Blood-based Tests, Urine-based Tests, Saliva-based Tests, Others), and End-Use Application (Home Healthcare, Chronic Disease Management, Preventive Care, Clinical Settings, Others). Forecast for 2026 to 2036.
Self Testing Market Size, Market Forecast and Outlook By FMI
The self testing market is expected to increase from USD 29.4 billion in 2026 to USD 52.7 billion by 2036. The market is anticipated to register a 6.1% CAGR during the forecast period. Glucose monitoring is likely to lead with a 34.8% share, and blood-based tests are projected to account for 58.6% of sample type demand in 2026.
Summary of the Self Testing Market
- Demand and Growth Drivers
- Consumer preference for private health checks is expected to raise adoption across chronic disease monitoring and preventive care settings.
- Rising healthcare costs are likely to increase interest in at-home diagnostic products for routine monitoring and early screening.
- Digital health tools are anticipated to improve user engagement through connected results and easier health trend tracking.
- Product and Segment View
- Glucose monitoring is expected to lead the test type segment as diabetes management requires frequent and repeatable testing.
- Blood-based tests are likely to stay important as many diagnostic applications need accurate biomarker readings and reliable results.
- Home healthcare applications are anticipated to gain stronger use as patients seek convenient testing outside clinical facilities.
- Geography and Competitive Outlook
- India is expected to lead country-level expansion as low-cost kits reach pharmacies across smaller cities and rural locations.
- The USA is likely to record steady demand as digital health platforms improve connected self-testing for chronic care.
- Companies with stronger device accuracy and app integration are anticipated to strengthen their position in self-testing products.
- Analyst Opinion
- Sabyasachi Ghosh, Principal Consultant for Healthcare at FMI, suggests, “The self testing market is expected to move toward connected diagnostic tools able to combine convenience with reliable clinical readings. Manufacturers offering accurate devices and simple digital reporting are likely to gain stronger acceptance across home healthcare and preventive care use cases.”
- Self Testing Market Value Analysis
- The self testing market is moving from basic home checks toward connected diagnostic products used in daily health management.
- Demand is likely to increase as users seek faster answers for glucose monitoring and other common screening needs.
- Adoption is expected to gain traction from app-enabled devices able to record results and guide repeat testing.
- Spending is anticipated to be backed by chronic disease monitoring needs and stronger consumer comfort with home diagnostics.
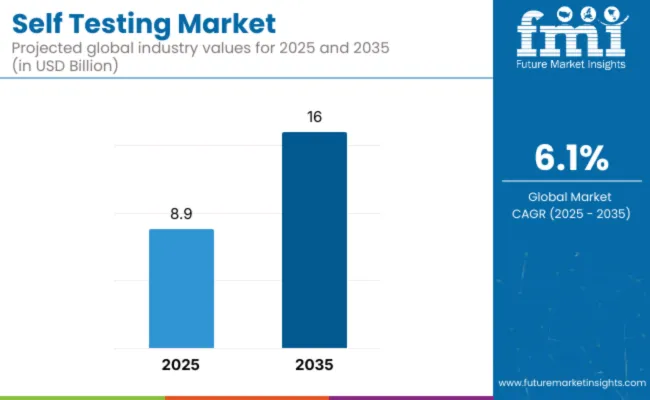
Self Testing Market Key Takeaways
| Metric | Value |
|---|---|
| Market Value (2026) | USD 29.4 billion |
| Market Forecast Value (2036) | USD 52.7 billion |
| Forecast CAGR (2026-2036) | 6.1% |
Source: Future Market Insights’ proprietary forecasting model and primary research
Key Shifts in the Self Testing Market From 2026 to 2036
Based on Future Market Insights’ analysis, pre‑2026, the self‑testing market is dominated by basic lateral flow devices for limited conditions, such as pregnancy, glucose, and a small set of infectious markers. Products were standalone, siloed, and often lacked integration with broader care pathways, limiting actionable value for users.
In 2026, regulatory frameworks and digital health standards elevated analytical rigour and connectivity requirements for self‑tests. Regulatory bodies in major markets adopted harmonized validation criteria and digital reporting guidelines, which removed ambiguity around performance claims. This shift moved the market from passive detection toward clinically validated, digitally enabled self‑testing tools that support early intervention and care continuity.
Consumer engagement has changed dramatically. Traditional self‑tests focused on simple yes/no results delivered on device. From 2026-2036, self‑testing solutions integrate with health apps, telehealth platforms, and wearable ecosystems. Real‑time insights, personalized guidance, and remote clinician support transformed self‑tests into interactive health touchpoints rather than isolated products. Especially, the at-home cholesterol monitoring products have gained immense traction among the middle-class income group.
The production and delivery model also evolved. The previous decade’s rigid mass production could not keep pace with demand for diverse test panels and personalized solutions. AI‑driven assay design, modular manufacturing lines, and demand forecasting tools enabled rapid iteration and small‑batch customization, reducing waste and enhancing supply responsiveness.
What are the Various Segments of the Self Testing Market?
Self testing plays a critical role in delivering diagnostic accuracy, early disease detection, and patient convenience across health monitoring, chronic disease management, and preventive care applications. Adoption is influenced by accuracy requirements, cost considerations, regulatory compliance, and end-use performance criteria. Segmentation by test type, sample type, and end-use application highlights how healthcare providers select specific testing solutions to meet diagnostic standards, cost targets, and patient preferences across diverse healthcare categories.
Which Test Type Holds the Largest Share in Self Testing?
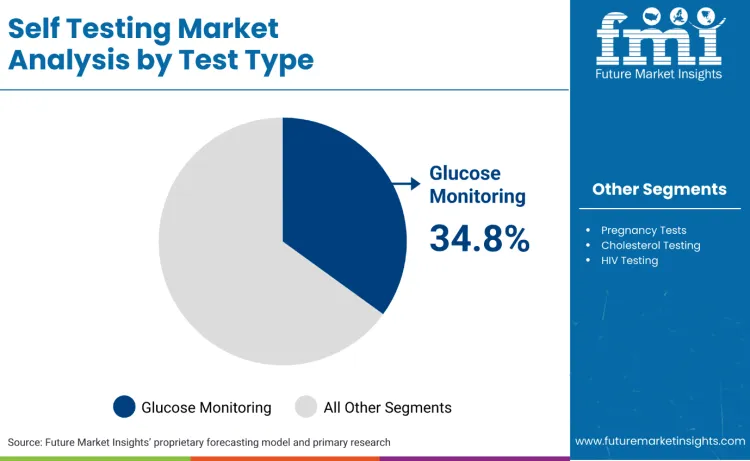
Glucose monitoring accounts for 34.8%, driven by extensive use in diabetes management, blood sugar tracking, and metabolic monitoring applications. Pregnancy tests hold 18.7%, supporting reproductive health monitoring, family planning, and early detection applications. Cholesterol testing represents 15.2%, favored for cardiovascular health assessment, lipid monitoring, and preventive care applications. HIV testing contributes 12.4%, used in disease screening and early detection scenarios. Other test types account for 18.9%, used in specialized applications and niche medical segments.
Key Points
- Glucose monitoring dominates diabetes management and metabolic health applications.
- Pregnancy tests support reproductive health and family planning markets.
- Test selection depends on accuracy requirements and regulatory considerations.
How Do Sample Type Requirements Influence Testing Selection?
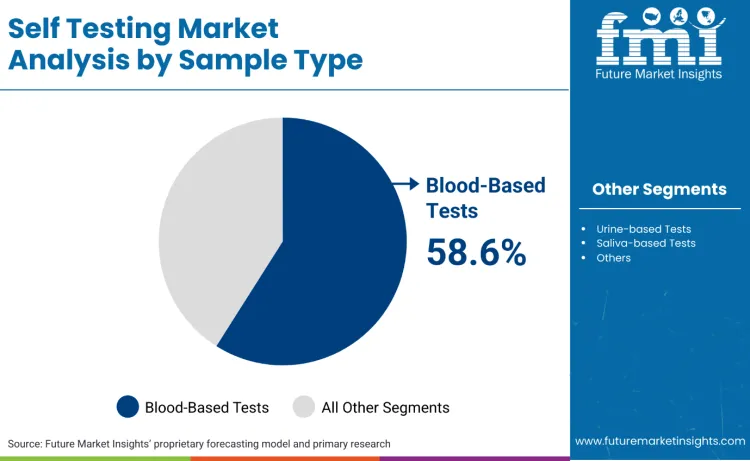
Blood-based tests represent 58.6%, reflecting priority on applications requiring high accuracy and comprehensive biomarker analysis. Urine-based tests account for 28.3%, suitable for screening applications requiring non-invasive sample collection. Saliva-based tests hold 9.8%, used for genetic testing and hormone monitoring requiring simple collection methods. Other sample types contribute 3.3%, favored for specialized testing and research applications.
Key Points
- Blood-based tests remain the primary diagnostic segment.
- Urine tests serve screening and non-invasive monitoring applications.
- Sample selection depends on diagnostic requirements and user convenience specifications.
How Smart Technology is Taking the Self Testing Space by Storm?
Smart technology is reshaping the self‑testing market by making at‑home diagnostics more accurate, connected, and easy to use. Modern devices, such as blood sugar testers often include sensors that automatically capture data and companion apps that guide users through testing and display results in real time. Integration with cloud platforms allows users to store results, track trends over time, and share information with healthcare providers when needed.
Artificial intelligence and machine learning are improving the interpretation of test data and helping reduce user error. A clear example is the Cue Health Monitoring System from Cue Health, which pairs a portable diagnostic reader with a smartphone app to deliver rapid, lab‑quality results for conditions such as respiratory infections. The smart connectivity of this product enables seamless result reporting and trend monitoring. As smart technology continues to evolve, self‑testing tools are becoming more reliable, user friendly, and capable of supporting proactive personal health management.
How Is the Self Testing Market Evolving Globally?
Global demand for self testing is increasing as healthcare systems expand diagnostic capacity while addressing patient convenience, diagnostic accuracy, and cost efficiency application requirements. Growth reflects rising use of digital connectivity features, advanced biosensor technologies, and regulatory-optimized device specifications across healthcare facilities, home care environments, and preventive medicine programs. Product selection focuses on diagnostic accuracy, user interface design, and cost performance under various healthcare scenarios. India records 7.8% CAGR, USA records 6.5% CAGR, Germany records 5.9% CAGR, Japan records 5.4% CAGR, and UK records 5.1% CAGR. Adoption remains driven by diagnostic requirements and cost efficiency rather than volume expansion alone.
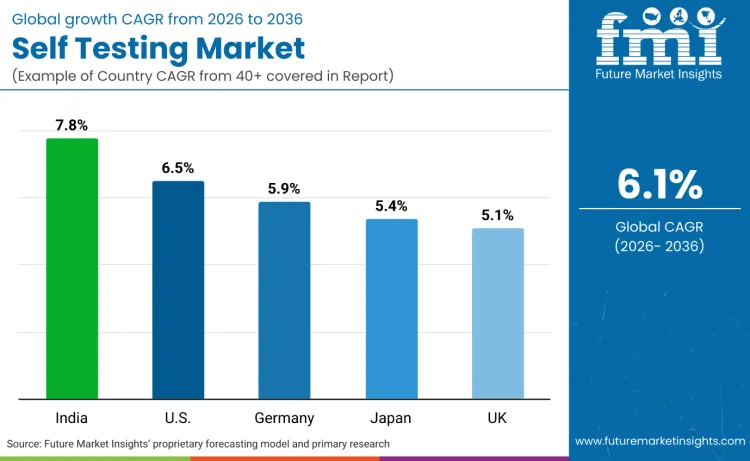
| Country | CAGR (2026-2036) |
|---|---|
| India | 7.8% |
| USA | 6.5% |
| Germany | 5.9% |
| Japan | 5.4% |
| UK | 5.1% |
Source: Future Market Insights’ proprietary forecasting model and primary research
India: How is the DIY Diagnostics Concept Driving Rural Market Entry?
The primary driver in India is the rapid adoption of the "Do-it-Yourself" (DIY) testing concept among the growing middle class. A key 2026 development is the CDSCO's implementation of the "Strengthening of Pharmaceutical Industry" (SPI) scheme, which has incentivized domestic MSMEs to manufacture low-cost, high-sensitivity self-test kits for blood glucose and infectious diseases. This local production push has drastically reduced prices, making these kits accessible via retail pharmacy networks in Tier-2 and Tier-3 cities. Propelled by this democratization of healthcare, the Indian self-testing market is set to grow at a 7.8% CAGR during the study period.
USA: Will FDA Support for Digital Health Devices Standardize At-Home Results?
In the USA, the market is being transformed by the integration of self-tests with digital health platforms. A major 2026 driver is the launch of the FDA’s "TEMPO for Digital Health Devices" pilot program, which streamlines the approval of self-testing kits that use smartphone apps to interpret and transmit results to physicians. This allows for real-world data collection and immediate clinical intervention, particularly for chronic disease management. Reflecting this shift toward connected, personalized healthcare, the USA self-testing market is projected to grow at a 6.5% CAGR during the study period.
Germany: Can the "DiGA" Fast-Track Procedure Boost Clinical Self-Testing?
Germany remains a leader in evidence-based self-care through its BfArM "DiGA" (Digital Health Applications) fast-track process. A critical driver in 2026 is the inclusion of "companion self-test kits" within the DiGA directory, where doctors can prescribe at-home diagnostic kits alongside health apps, with the costs fully covered by statutory health insurance. This has legitimized self-testing as a clinical tool rather than a consumer novelty. Within this insurance-backed landscape, the German self-testing market is expected to grow at a 5.9% CAGR during the study period.
Japan: Is "Self-Inspection" Regulatory Reform Improving Elderly Care?
In Japan, the market is driven by the world's most rapidly aging population and a focus on geriatric monitoring. A major development in early 2026 is the PMDA’s new notifications on "Self-Inspection for In-Vitro Diagnostics," which simplifies the labeling and package insert requirements for blood glucose and infectious disease kits to make them more "senior-friendly." This regulatory pivot is designed to reduce the diagnostic burden on Japan’s shrinking medical workforce. Supported by this demographic necessity, the Japanese self-testing market is set to grow at a 5.4% CAGR during the study period.
UK: How are Public Health Initiatives Reducing the Stigma of STD Testing?
The UK market is undergoing a shift toward "discreet diagnostics" to improve public health outcomes. A key driver is the 2025-2026 expansion of National HIV and STD Testing Week initiatives, funded by the Department of Health and Social Care (DHSC). These programs provide free, online-ordered self-test kits to high-risk groups, significantly reducing the stigma and logistical barriers associated with clinic visits. Supported by these aggressive government-backed awareness campaigns, the UK self-testing market is set to grow at a 5.1% CAGR during the study period.
Who are the Major Players Active in the Self Testing Market?
The 2026 competitive landscape for the self-testing market is defined by a strategic pivot from pandemic-era crisis response to the management of chronic conditions and AI-integrated diagnostics. Abbott Laboratories maintains its market leadership through the aggressive expansion of its FreeStyle Libre ecosystem, notably launching the next-generation FreeStyle Libre 2 Plus in mid-2025, which provides continuous glucose readings every minute.
In a significant portfolio restructuring, Abbott announced a definitive agreement to acquire Exact Sciences in November 2025 (slated to close in Q2 2026), a move designed to integrate at-home cancer screening into its diagnostic core. Roche Holding AG has shifted its focus toward decentralized molecular testing, receiving a CLIA waiver in December 2025 for its first point-of-care Bordetella infection test, while simultaneously launching new PCR panels to enhance self-sampling accuracy for women’s health.
Key Players in the Self Testing Market
- Abbott Laboratories
- Roche Holding AG
- Johnson & Johnson
- Siemens Healthineers AG
- Becton Dickinson and Company
- Quidel Corporation
- OraSure Technologies Inc.
- Trinity Biotech plc
- Meridian Bioscience Inc.
- Chembio Diagnostics Inc.
Scope of the Report
| Items | Values |
|---|---|
| Quantitative Units | USD billion |
| Test Type | Glucose Monitoring; Pregnancy Tests; Cholesterol Testing; HIV Testing; Others |
| Sample Type | Blood-based Tests; Urine-based Tests; Saliva-based Tests; Others |
| End-Use Application | Home Healthcare; Chronic Disease Management; Preventive Care; Clinical Settings; Others |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Countries Covered | India, USA, Germany, Japan, UK, and 40+ countries |
| Key Companies Profiled | Abbott Laboratories; Roche Holding AG; Johnson & Johnson; Siemens Healthineers AG; Becton Dickinson and Company; Others |
| Additional Attributes | Dollar sales by test type, sample type, and end-use application; performance in diagnostic accuracy and cost efficiency across home healthcare, chronic disease monitoring, and preventive care matrices; device quality improvement, user interface enhancement, and regulatory compliance under healthcare operations; impact on diagnostic reliability, supply consistency, and cost management during healthcare processes; compatibility with healthcare systems and quality control targets; procurement dynamics driven by healthcare expansion, quality improvement programs, and long-term diagnostic supply partnerships. |
Self Testing Market by Segment
Test Type:
- Glucose Monitoring
- Pregnancy Tests
- Cholesterol Testing
- HIV Testing
- Others
Sample Type:
- Blood-based Tests
- Urine-based Tests
- Saliva-based Tests
- Others
End-Use Application:
- Home Healthcare
- Chronic Disease Management
- Preventive Care
- Clinical Settings
- Others
Region:
- Asia Pacific
- China
- Japan
- South Korea
- India
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Nordic
- BENELUX
- Rest of Europe
- North America
- USA
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Other African Union
- Rest of Middle East & Africa
Bibliography
- International Self Testing Association. (2023). Device Standards for Home Diagnostics: Technical Guidelines. ISTA.
- Global Healthcare Diagnostics Foundation. (2023). Quality Specifications for Self Testing Devices: Accuracy and Safety Requirements. GHDF.
- Society of Diagnostic Technology. (2024). SDT Guidelines for Device Selection in Healthcare Applications (updated accuracy specifications for diagnostic and monitoring applications). SDT.
- Williams, M. K., et al. (2023). Diagnostic accuracy effects of advanced self testing devices on healthcare delivery efficiency: A comprehensive analysis of glucose monitoring and screening applications. Journal of Home Healthcare Technology, 28, 45-62.
- Peterson, L. R., et al. (2023). Fifth annual self testing report: Special focus on regulatory compliance and cost efficiency in healthcare applications. International Diagnostics Review, 31, 18-35.
Frequently Asked Questions
How big is the self testing market in 2026?
The global self testing market is estimated to be valued at USD 29.4 billion in 2026.
What will be the size of the self testing market in 2036?
The market size for the self testing market is projected to reach USD 52.7 billion by 2036.
How much will the self testing market grow between 2026 and 2036?
The self testing market is expected to grow at a 6.1% CAGR between 2026 and 2036.
What are the key test types in the self testing market?
The key test types in the self testing market include glucose monitoring, pregnancy tests, cholesterol testing, HIV testing, and other specialized diagnostic applications.
Which test segment will contribute a significant share in the self testing market in 2026?
In terms of test type, the glucose monitoring segment is set to command a 34.8% share in the self testing market in 2026.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand Side Trends
- Supply Side Trends
- Technological and Regulatory Roadmap
- Strategic Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Diagnostic Method Drivers and Technological Shifts
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Expert Input and Fieldwork (Primary Evidence)
- Tooling, Models, and Reference Databases
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunities
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Regulatory and Certification Landscape
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036
- Historical Market Size Analysis (USD Billion), 2021 to 2025
- Market Size Forecast (USD Billion), 2026 to 2036
- Y-o-Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Analysis by Test Type
- Glucose Monitoring
- Pregnancy Tests
- Cholesterol Testing
- HIV Testing
- Other Tests
- Global Market Analysis by Sample Type
- Blood-based Tests
- Urine-based Tests
- Saliva-based Tests
- Other Sample Types
- Global Market Analysis by End-Use
- Home Healthcare
- Chronic Disease Management
- Preventive Care
- Clinical Settings
- Other End-Uses
- Regional Market Analysis
- Asia Pacific
- Europe
- North America
- Latin America
- Middle East & Africa
- Market Structure and Competitive Landscape
- Key Players Analysis
- Scope of the Report
- Bibliography
- FAQs
List of Tables
- Self Testing Market Key Takeaways
- Global Self Testing Market Size (USD Billion), 2021-2036
- Global Self Testing Market Y-o-Y Growth (%), 2021-2036
- Global Self Testing Market Absolute $ Opportunity (USD Billion), 2026-2036
- Global Self Testing Market Pricing Analysis, 2021-2036
- Global Self Testing Market Size by Test Type, 2021-2036
- Global Self Testing Market Size by Sample Type, 2021-2036
- Global Self Testing Market Size by End-Use, 2021-2036
- Global Self Testing Market Size by Region, 2021-2036
- Asia Pacific Self Testing Market Size by Country, 2021-2036
- Europe Self Testing Market Size by Country, 2021-2036
- North America Self Testing Market Size by Country, 2021-2036
List of Figures
- Self Testing Market Size (USD Billion), 2021-2036
- Self Testing Market Y-o-Y Growth (%), 2021-2036
- Self Testing Market Absolute $ Opportunity Analysis, 2026-2036
- Self Testing Market Value Chain Analysis
- Self Testing Market Supply Chain Analysis
- Self Testing Market Investment Feasibility Matrix
- Self Testing Market PESTLE Analysis
- Self Testing Market Porter’s Five Forces Analysis
- Self Testing Market Product Life Cycle Analysis
- Self Testing Market Opportunity Map Analysis
- Self Testing Market Scenario Forecast Analysis
- Global Self Testing Market Share by Test Type, 2026
- Global Self Testing Market Share by Sample Type, 2026
- Global Self Testing Market Share by End-Use, 2026
- Global Self Testing Market Share by Region, 2026
- Self Testing Market Competitive Landscape
- Self Testing Market Structure Analysis
- Abbott Laboratories-Company Profile
- Roche Holding AG-Company Profile
- Johnson & Johnson-Company Profile
- Siemens Healthineers AG-Company Profile
- Becton Dickinson and Company-Company Profile
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

