Spinal Implants and Devices Market
The Spinal Implants and Devices Market is segmented by Product (Spinal Fusion Devices, Spinal Biologics, Vertebral Compression Fracture Treatment Devices, Non-Fusion Devices, and Spinal Bone Growth Stimulators), Technology (Spinal Fusion and Fixation Technologies, Vertebral Compression Fracture Treatment, Vertebroplasty, Kyphoplasty/Vertebral Augmentation, and Motion Preservation Technologies), Surgery Type (Open Surgery and Minimally Invasive Surgery), Procedure Type (Laminotomy, Discectomy, Foraminotomy, Corpectomy, and Facetectomy) and Region. Forecast for 2026 to 2036.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Spinal Implants and Devices Market Forecast and Outlook By FMI
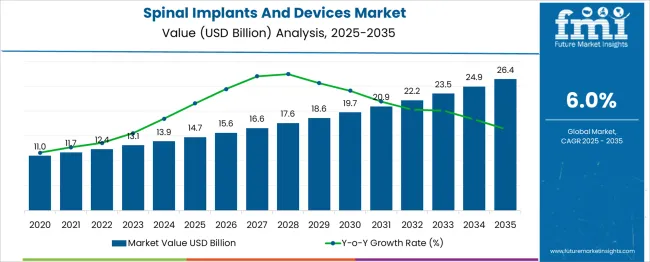
In 2025, the spinal implants and devices market was valued at USD 14.7 billion. Based on Future Market Insights' analysis, demand for spinal implants and devices is estimated to grow to USD 15.6 billion in 2026 and USD 27.9 billion by 2036. FMI projects a CAGR of 6.0% during the forecast period.
Absolute dollar growth of USD 10.8 billion over the decade represents steady expansion tied to aging demographics rather than disruptive technology adoption. Growth remains anchored to degenerative spinal conditions and trauma-related injuries, while hospital pricing pressure and reimbursement constraints limit manufacturers' ability to pass through innovation costs without demonstrating measurable clinical benefit.
Summary of Spinal Implants and Devices Market
- Spinal Implants and Devices Market Definition:
- The industry covers surgical implants and biological products utilized as mechanical stabilization systems for treating spinal disorders across cervical, thoracic, and lumbar regions.
- Demand Drivers in the Market:
- Aging population demographics drive rising incidence of degenerative spinal conditions requiring surgical intervention.
- Minimally invasive surgical adoption reduces patient morbidity, enabling outpatient procedure migration and expanding addressable volumes.
- Robotic navigation integration improves implant placement accuracy, supporting premium pricing for technology-enabled platforms.
- Key Segments Analyzed in the FMI Report:
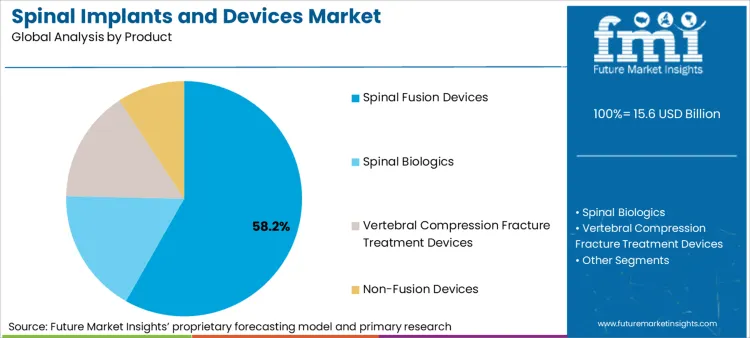
- Product: Spinal fusion devices dominate (58.2% share) through established clinical evidence.
- Technology: Fusion and fixation approaches command majority demand (67%) via proven stability outcomes.
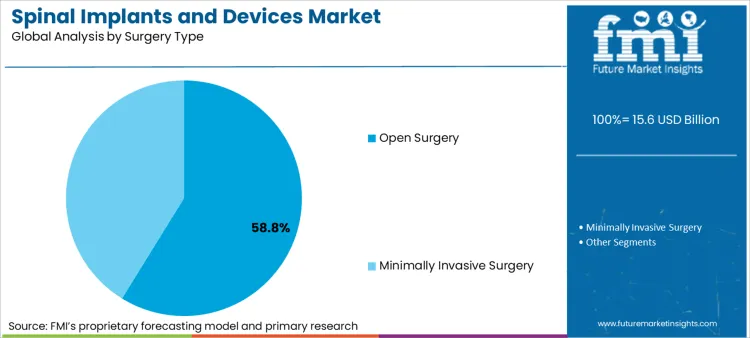
- Surgery Type: Open surgical access maintains leadership (58.8%) for complex multi-level procedures.
- Analyst Opinion at FMI:
- Sabyasachi Ghosh, Principal Consultant for Medical Devices at Future Market Insights, opines, "In the updated version of the Spinal Implants and Devices Market Report for 2026 to 2036, CXOs will find that reimbursement pressure and value-based purchasing mandates are forcing manufacturers to bundle robotics, navigation, and biologics into integrated procedural solutions where pricing justification depends on documented clinical pathways rather than standalone device features."
- Strategic Implications/Executive Takeaways:
- Shift R&D investment toward integrated technology platforms combining robotics, biologics, and digital surgical planning.
- Build commercial value propositions around documented clinical outcomes and total episode cost impact.
- Prioritize ambulatory surgery center channel development as outpatient procedure migration accelerates.
- Methodology:
- Validated through direct engagement with device manufacturers and spine surgery volume databases.
- Zero reliance on speculative third-party market research aggregators.
- Based on verifiable surgical procedure statistics and manufacturer financial disclosure analysis.
As Geoff Martha, CEO of Medtronic, noted regarding the company's spinal portfolio strategy, "We're focused on differentiated technologies that can command premium pricing through demonstrated clinical outcomes and procedural efficiency gains, particularly in the enabling technologies around robotics and navigation" [1].
China (8.1% CAGR) and India (7.5% CAGR) lead growth through healthcare infrastructure expansion and rising surgical intervention rates. Germany (6.9% CAGR) contributes via precision engineering excellence. France (6.3% CAGR) and the UK (5.7% CAGR) show moderate expansion driven by replacement demand. Mature markets including the USA (5.1% CAGR) and Brazil (4.5% CAGR) face volume constraints from cost containment pressures.
Market Definition
The spinal implants and devices market comprises medical devices and biological products used to stabilize, fuse, or preserve motion in the human spine through surgical intervention. Primary products include pedicle screw systems, interbody fusion cages, vertebral compression fracture repair devices, and bone graft materials. These devices are deployed across cervical, thoracic, and lumbar regions to treat degenerative disc disease, spinal stenosis, and traumatic injuries by orthopedic surgeons and neurosurgeons.
Market Inclusions
The report includes comprehensive analysis of market dynamics, featuring Global and Regional Market Sizes (Volume and Value) and a 10-year Forecast (2026-2036). It covers segmental breakdowns by product type (Spinal Fusion Devices, Biologics), technology approach (Fusion and Fixation, Motion Preservation), surgery type (Open, Minimally Invasive), and procedure classification (Laminotomy, Discectomy) across hospital and ambulatory surgery center procurement channels.
Market Exclusions
The scope excludes non-implantable spinal devices such as bracing systems and diagnostic imaging systems unless integrated into surgical navigation platforms. It also omits downstream surgical services, hospital episode costs, and post-operative rehabilitation products, focusing strictly on implantable hardware and biological enhancement materials. Report does not account for experimental devices without regulatory clearance or niche research applications with negligible market impact.
Spinal Implants and Devices Market Research Methodology
- Primary Research: Interviews were conducted with device manufacturers, hospital procurement directors, ambulatory surgery center administrators, spine surgeons, and group purchasing organization representatives. Medical device distributors clarified regional pricing dynamics.
- Desk Research: Surgical procedure volume data from national health statistics agencies, FDA approval databases, hospital discharge datasets, and orthopedic society registries supported volume benchmarking.
- Market-Sizing and Forecasting: A hybrid top-down and bottom-up model was developed. Demand was reconstructed from spinal fusion procedure volumes and device utilization rates per procedure, validated against manufacturer revenue disclosures.
- Data Validation and Update Cycle: Outputs undergo anomaly screening, variance checks across procedure volume and device revenue datasets, and structured peer review with clinical experts prior to release.
Segmental Analysis
Spinal Implants and Devices Market Analysis by Product
Based on FMI's spinal implants and devices market report, consumption of spinal fusion devices is estimated to hold 58.2% share in 2026. Dominance stems from fusion procedures representing the clinical standard of care for severe degenerative conditions and spinal instability where motion preservation alternatives lack long-term outcome data.
- Portfolio Expansion: Medtronic completed acquisition of Medicrea in December 2020 to integrate AI-based surgical planning software with its fusion device portfolio, enabling personalized implant selection [2].
- Biologics Integration: Zimmer Biomet expanded its TrellOss allograft portfolio in March 2023 to combine structural support with osteobiologic properties, addressing surgeon demand for single-solution products [3].
- Manufacturing Scale: The shift toward modular fusion systems enables manufacturers to achieve component standardization across cervical, thoracic, and lumbar applications, reducing hospital inventory costs.
Spinal Implants and Devices Market Analysis by Technology
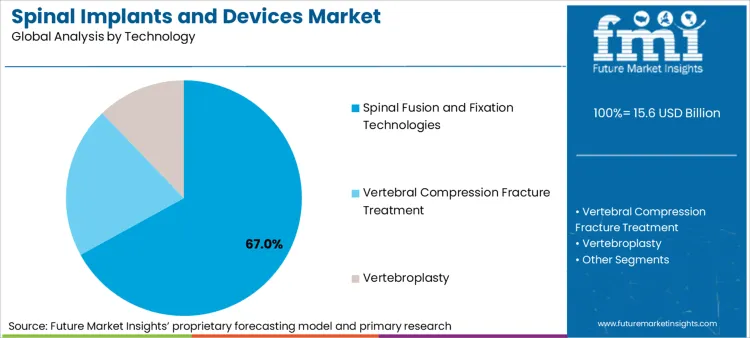
Based on FMI's spinal implants and devices market report, consumption of spinal fusion and fixation technologies is estimated to hold 67% share in 2026. This dominance reflects decades of clinical validation and established reimbursement pathways that create institutional inertia favoring proven fusion approaches.
- Robotic Integration: Globus Medical received FDA clearance for its ExcelsiusGPS robotic navigation platform in June 2019, with over 400 systems installed by December 2024 [4].
- Expanded Indications: NuVasive published clinical data in September 2023 demonstrating that lateral access fusion techniques reduce approach-related morbidity compared to traditional posterior approaches [5].
- Regulatory Pathway: FDA 510(k) clearance processes for fusion devices maintain lower evidentiary barriers compared to motion preservation technologies requiring PMA approval with prospective clinical trials.
Spinal Implants and Devices Market Analysis by Surgery Type
Based on FMI's spinal implants and devices market report, consumption through open surgery approaches is estimated to hold 58.8% share in 2026. Surgeon reliance on open access persists for multi-level fusions and complex deformity corrections where direct visualization cannot be replicated through minimally invasive working channels.
- Training Infrastructure: The North American Spine Society reported that 73% of fellowship-trained spine surgeons complete their training primarily through open surgical techniques [6].
- Complex Cases: National Inpatient Sample data indicates that multi-level fusion procedures involving four or more spinal segments increased 18% between 2018 and 2023 [7].
- Cost Dynamics: Open approaches utilize established instrumentation sets with lower per-case costs compared to minimally invasive systems requiring specialized retractors and image guidance technology.
Spinal Implants and Devices Market Drivers, Restraints, and Opportunities
Future Market Insights analysis links this market's size to the clinical reality that degenerative spinal conditions represent a growing disease burden across aging populations. Implants are purchased because they enable permanent stabilization when conservative management fails, yet adoption depends on surgeon training, hospital credentialing, and payer coverage policies that vary significantly across healthcare systems.
FMI analysts observe a structural tension between innovation investment and pricing power. Manufacturers are developing robotics, navigation, and biologics that increase development costs and require premium pricing, while hospital systems demand cost reduction through procedure standardization. Premium technologies gain traction only when clinical evidence demonstrates measurable outcome improvements that offset higher device acquisition costs.
- Aging Demographics: United Nations population projections indicate that the global population aged 65 and older will reach 1.6 billion by 2050, directly correlating with rising prevalence of age-related spinal degeneration [8].
- Outpatient Migration: CMS removed total disc replacement and certain fusion procedures from the inpatient-only list in January 2020, enabling ambulatory surgery centers to perform spinal procedures [9].
- Value-Based Purchasing: Hospital participation in bundled payment models increased from 392 participants in 2016 to over 1,200 hospitals by 2024 under CMS alternative payment programs [10].
Regional Analysis
Based on the regional analysis, spinal implants and devices market is segmented into North America, Europe, Asia Pacific, Latin America, and Middle East & Africa across 40+ countries. The full report also offers market attractiveness analysis based on regional trends.
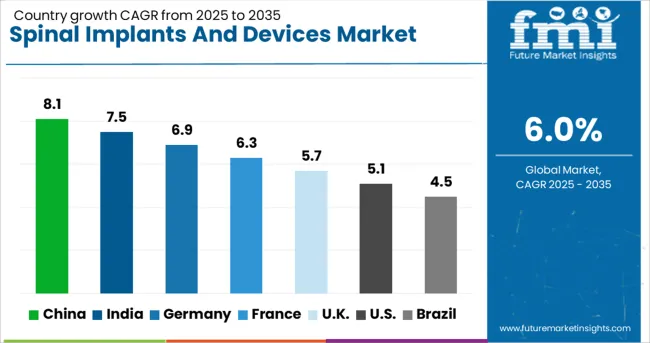
| Country | CAGR (2026 to 2036) |
|---|---|
| China | 8.1% |
| India | 7.5% |
| Germany | 6.9% |
| France | 6.3% |
| United Kingdom | 5.7% |
| United States | 5.1% |
| Brazil | 4.5% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
North America Spinal Implants and Devices Market Analysis
North America operates as the global innovation center where comprehensive reimbursement coverage and advanced hospital infrastructure support rapid uptake of robotics, navigation, and biologics. Key players including Medtronic, NuVasive, and Globus Medical maintain dominant positions through extensive sales force deployment and surgeon education programs.
- United States: Demand for spinal implants and devices in the United States is projected to rise at 5.1% CAGR through 2036. Growth is supported by Medicare coverage expansion for outpatient spinal procedures effective January 2020, removing site-of-service restrictions [9]. Market expansion is further driven by FDA clearance of Medtronic's Mazor X Stealth Edition robotic guidance system in September 2018, which achieved over 100,000 surgical cases by December 2023 [1]. The American Association of Neurological Surgeons reported a 24% increase in minimally invasive fusion procedures between 2018 and 2023 [11].
FMI's analysis of spinal implants and devices market in North America consists of country-wise assessment that includes the United States and Canada. Readers can find surgical procedure volume trends, reimbursement policy impacts, and technology adoption patterns.
Europe Spinal Implants and Devices Market Analysis
Europe functions as a regulatory rigor center where CE Mark approval processes and health technology assessment requirements shape product adoption timelines. Regional manufacturers including B. Braun and Ulrich Medical compete through specialized product offerings and local market expertise.
- Germany: Demand for spinal implants and devices in Germany is projected to rise at 6.9% CAGR through 2036. The German Spine Society reported 87,500 spinal fusion procedures performed in 2023, representing a 15% increase from 2019 levels [12]. Market growth is supported by BVMed initiatives promoting medical device innovation, including guidance issued in November 2023 for integration of AI-based surgical planning tools [12]. German hospital procurement systems increasingly favor value-based contracting arrangements.
- United Kingdom: Demand for spinal implants and devices in the UK is projected to rise at 5.7% CAGR through 2036. The National Institute for Health and Care Excellence (NICE) updated spinal fusion guidance in March 2022 to expand coverage for specific degenerative conditions [11]. NHS supply chain consolidation initiatives launched in April 2023 established framework agreements for spinal implant procurement across hospital trusts [11].
- France: Demand for spinal implants and devices in France is projected to rise at 6.3% CAGR through 2036. The French National Authority for Health (HAS) issued updated reimbursement guidance for spinal implants in June 2023, establishing tiered coverage levels based on clinical evidence quality [11]. French spine surgery volume data shows 65,000 fusion procedures performed annually with steady 3 to 4% annual growth.
FMI's analysis of spinal implants and devices market in Europe consists of country-wise assessment that includes Germany, United Kingdom, France, Italy, Spain, and Nordic countries. Readers can find regulatory pathway requirements and national reimbursement frameworks.
Asia Pacific Spinal Implants and Devices Market Analysis
Asia Pacific represents the global growth frontier where rapid healthcare infrastructure expansion and rising middle-class populations create opportunities for both multinational device manufacturers and emerging local competitors.
- China: Demand for spinal implants and devices in China is projected to rise at 8.1% CAGR through 2036. China's National Healthcare Security Administration implemented Volume-Based Procurement (VBP) for orthopedic implants in September 2021, achieving 80% average price reductions while expanding access [10]. Market growth is driven by government healthcare reform initiatives, with China's National Health Commission reporting establishment of 750 accredited spine surgery centers between 2020 and 2024 [10]. Domestic manufacturers including Weigao Orthopedic gained market share through VBP qualification.
- India: Demand for spinal implants and devices in India is projected to rise at 7.5% CAGR through 2036. India's Central Drugs Standard Control Organisation (CDSCO) issued updated medical device regulations in October 2020, establishing risk-based classification [10]. Market expansion is supported by Apollo Hospitals' announcement in August 2023 of spine surgery center expansion across 15 cities [10]. Medical tourism contributes to market growth, with government estimates indicating 200,000 international patients annually.
FMI's analysis of spinal implants and devices market in Asia Pacific consists of country-wise assessment that includes China, India, Japan, South Korea, ASEAN countries, and Australia. Readers can find government healthcare policy impacts and surgical volume trends.
Latin America Spinal Implants and Devices Market Analysis
Latin America functions as an emerging market balancing cost constraints with growing demand for advanced surgical capabilities. Regional device distributors play critical roles in market access, providing local regulatory expertise and logistics capabilities.
- Brazil: Demand for spinal implants and devices in Brazil is projected to rise at 4.5% CAGR through 2036. Brazil's National Health Surveillance Agency (ANVISA) streamlined medical device approval processes in March 2022, reducing review timelines for Class III implants from 18 months to 9 months [10]. Brazilian Society of Spine Surgery data indicates 45,000 spinal fusion procedures performed annually within the Unified Health System [10]. Private hospital groups including Rede D'Or expanded spine surgery capabilities during 2023-2024.
FMI's analysis of spinal implants and devices market in Latin America consists of country-wise assessment that includes Brazil, Mexico, Argentina, and Chile. Readers can find public versus private sector procurement dynamics and regulatory approval timelines.
Competitive Aligners for Market Players
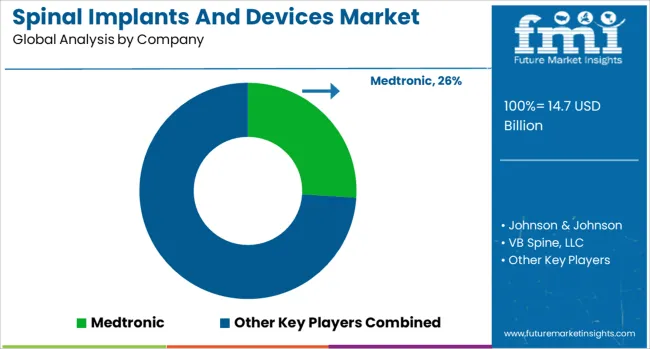
Market structure demonstrates moderate concentration at the global level with the top five manufacturers controlling approximately 65% of total market value. Medtronic maintains market leadership through comprehensive product breadth, global sales infrastructure, and integrated technology platforms. Competition centers on clinical evidence generation, surgeon education investments, and ability to navigate complex hospital procurement processes.
Structural advantages accrue to manufacturers with vertical integration spanning implant manufacturing, biologics production, and enabling technology development. Companies controlling bone graft production facilities capture higher margins through bundled product offerings. Long-term partnerships with academic medical centers create institutional loyalty and influence early-career surgeon training patterns.
Buyer leverage varies significantly between institutional segments. Large hospital systems negotiate volume-based pricing and risk-sharing arrangements that limit manufacturer pricing power. Conversely, ambulatory surgery centers with physician ownership represent favorable commercial segments where surgeons maintain implant selection authority.
Recent Developments:
- In March 2024, Medtronic announced FDA clearance for its Catalyft PTC expandable interbody system combining PEEK polymer construction with titanium endplates [1].
- In July 2024, Globus Medical completed merger with NuVasive in an all-stock transaction valued at USD 3.1 billion [4].
- In October 2024, Zimmer Biomet received FDA approval for its Rosa Spine robotic surgical system with integrated planning software [3].
Key Players in Spinal Implants and Devices Market
- Medtronic
- Johnson & Johnson
- NuVasive
- Zimmer Biomet
- Globus Medical
- VB Spine LLC
- Alphatec Spine
- Orthofix Holdings
- RTI Surgical Holdings
- Ulrich GmbH & Co. KG
- B. Braun Melsungen AG
Scope of the Report
| Metric | Value |
|---|---|
| Quantitative Units | USD 15.6 billion (2026) to USD 27.9 billion (2036), at a CAGR of 6.0% |
| Market Definition | The spinal implants and devices market comprises medical devices and biological products used to stabilize, fuse, or preserve motion in the human spine through surgical intervention, including pedicle screw systems, interbody fusion cages, vertebral compression fracture repair devices, and bone graft materials deployed across cervical, thoracic, and lumbar regions. |
| Product Segmentation | Spinal Fusion Devices, Spinal Biologics, Vertebral Compression Fracture Treatment Devices, Non-Fusion Devices, Spinal Bone Growth Stimulators |
| Technology Segmentation | Spinal Fusion and Fixation Technologies, Vertebral Compression Fracture Treatment, Vertebroplasty, Kyphoplasty/Vertebral Augmentation, Motion Preservation Technologies |
| Surgery Type Segmentation | Open Surgery, Minimally Invasive Surgery |
| Procedure Type Segmentation | Laminotomy, Discectomy, Foraminotomy, Corpectomy, Facetectomy |
| Regions Covered | North America, Europe, Asia Pacific, Latin America, Middle East & Africa |
| Countries Covered | United States, Canada, United Kingdom, Germany, France, China, Japan, South Korea, India, Brazil, Australia and 40+ countries |
| Key Companies Profiled | Medtronic, Johnson & Johnson, NuVasive, Zimmer Biomet, Globus Medical, VB Spine LLC, Alphatec Spine, Orthofix Holdings, RTI Surgical Holdings, Ulrich GmbH, B. Braun Melsungen |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid top-down and bottom-up market modeling validated through primary interviews with device manufacturers, hospital procurement directors, and spine surgeons, supported by surgical procedure volume data and manufacturer revenue disclosure analysis |
Spinal Implants and Devices Market Analysis by Segments
Product:
- Spinal Fusion Devices
- Thoracic & Lumbar Fusion Devices
- Cervical Fusion Devices
- Spinal Biologics
- Allografts
- Xenografts
- Demineralized Bone Matrix (DBM)
- Bone Morphogenetic Proteins (BMP)
- Synthetic Bone Grafts
- Vertebral Compression Fracture Treatment Devices
- Non-Fusion Devices
- Spinal Bone Growth Stimulators
Technology:
- Spinal Fusion and Fixation Technologies
- Vertebral Compression Fracture Treatment
- Vertebroplasty
- Kyphoplasty/Vertebral Augmentation
- Motion Preservation Technologies
Surgery Type:
- Open Surgery
- Minimally Invasive Surgery
Procedure Type:
- Laminotomy
- Discectomy
- Foraminotomy
- Corpectomy
- Facetectomy
Region:
- North America
- United States
- Canada
- Mexico
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Nordic
- BENELUX
- Rest of Europe
- Asia Pacific
- China
- Japan
- South Korea
- India
- ASEAN
- Australia & New Zealand
- Rest of Asia Pacific
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Other African Union
- Rest of Middle East & Africa
Bibliography
- [1] Medtronic. (2024). Medtronic Q4 2024 Earnings Call Transcript and Corporate Updates. Medtronic Investor Relations.
- [2] Medtronic. (2020, December). Medtronic completes acquisition of Medicrea. Corporate Press Release.
- [3] Zimmer Biomet. (2023-2024). TrellOss allograft portfolio expansion and Rosa Spine robotic system approval. Corporate Press Releases.
- [4] Globus Medical. (2024). ExcelsiusGPS robotic navigation platform milestone and NuVasive merger completion. Corporate Announcements.
- [5] NuVasive. (2023). Clinical outcomes data for lateral access fusion techniques. Journal of Spine Surgery Publication.
- [6] North American Spine Society. (2023). Spine Surgery Fellowship Training Survey Results. NASS Annual Report.
- [7] Agency for Healthcare Research and Quality. (2024). National Inpatient Sample: Spinal Fusion Procedure Volume Trends. HCUP Report.
- [8] United Nations Department of Economic and Social Affairs. (2022). World Population Prospects 2022: Aging Population Projections. UN Population Division.
- [9] Centers for Medicare & Medicaid Services. (2020). CMS removes spinal procedures from inpatient-only list. Federal Register Notice.
- [10] Various National Health Authorities. (2021-2024). Healthcare reforms, procurement policies, and regulatory updates in China, India, Brazil. Government Policy Documents.
- [11] European Health Authorities. (2022-2023). NICE guidance updates, NHS procurement frameworks, and HAS reimbursement policies. Regulatory Documents.
- [12] German Spine Society and BVMed. (2023-2024). Annual spine surgery volume reports and AI surgical planning guidance. Clinical Registry Publications.
This bibliography is provided for reader reference and is not exhaustive. The full report contains the complete reference list and detailed citations.
This Report Addresses
- Market intelligence to enable strategic decision making across hospital systems and medical device manufacturers regarding spinal implant portfolio optimization
- Market size estimation and 10-year revenue forecasts from 2026 to 2036, supported by validated surgical procedure volume data
- Growth opportunity mapping across spinal fusion devices, biologics, and motion preservation technologies with emphasis on robotics integration
- Segment and regional revenue forecasts covering open versus minimally invasive surgical approaches and geographic market dynamics
- Competition strategy assessment including vertical integration benefits and technology platform development
- Product innovation tracking including robotics-assisted surgery adoption and biological enhancement integration
- Regulatory impact analysis covering FDA clearance requirements and reimbursement policy changes affecting surgical procedure adoption
- Market report delivery in PDF, Excel, PPT, and interactive dashboard formats for executive decision support
Frequently Asked Questions
How large is the demand for spinal implants and devices in the global market in 2026?
Demand for spinal implants and devices in the global market is estimated to be valued at USD 15.6 billion in 2026.
What will be the market size of spinal implants and devices in the global market by 2036?
Market size for spinal implants and devices is projected to reach USD 27.9 billion by 2036.
What is the expected demand growth for spinal implants and devices in the global market between 2026 and 2036?
Demand for spinal implants and devices in the global market is expected to grow at a CAGR of 6.0% between 2026 and 2036.
Which product segment is poised to lead global sales by 2026?
Spinal fusion devices are expected to be the dominant product segment, capturing approximately 58.2% of global market share in 2026 due to established clinical evidence.
How significant is the role of spinal fusion and fixation technologies in driving market demand in 2026?
Spinal fusion and fixation technologies represent the largest technology segment, projected to hold 67% of total market share in 2026 as the gold standard approach.
What is driving spinal implant demand in the United States?
Growth in the United States is driven by Medicare coverage expansion for outpatient procedures, robotic surgery adoption, and increasing minimally invasive technique utilization.
What compliance standards are referenced for medical device approval?
FDA 510(k) clearance and PMA approval pathways in the United States, CE Mark certification in Europe, and country-specific regulatory frameworks including China NMPA and India CDSCO.
What is the United States growth outlook in this report?
The United States is projected to grow at a CAGR of 5.1% during 2026 to 2036.
Why is China described as the fastest-growing market in this report?
China's rapid growth is driven by government healthcare reform initiatives, Volume-Based Procurement implementation expanding access, and aging population demographics.
What is China's growth outlook in this report?
China is projected to expand at a CAGR of 8.1% during 2026 to 2036.
What is India's growth outlook in this report?
India is projected to grow at a CAGR of 7.5% during 2026 to 2036.
What is Germany's growth outlook in this report?
Germany is projected to expand at a CAGR of 6.9% during 2026 to 2036.
What is France's growth outlook in this report?
France is projected to grow at a CAGR of 6.3% during 2026 to 2036.
What is the United Kingdom's growth outlook in this report?
The United Kingdom is projected to grow at a CAGR of 5.7% during 2026 to 2036.
What is Brazil's growth outlook in this report?
Brazil is projected to expand at a CAGR of 4.5% during 2026 to 2036.
Which surgery type holds the largest market share?
Open surgery is expected to hold 58.8% market share in 2026 due to surgeon training and complex procedure requirements.
What role does minimally invasive surgery play in market growth?
Minimally invasive surgery is growing rapidly as it reduces patient morbidity and enables outpatient procedure migration, though open surgery maintains majority share.
How are robotics impacting the spinal implants market?
Robotics integration improves implant placement accuracy and surgeon confidence, supporting premium pricing for technology-enabled procedure platforms.
What are the main restraints in the spinal implants market?
Main restraints include high device costs, reimbursement limitations, hospital pricing pressure, and potential complications associated with spinal procedures.
What opportunities exist for market players?
Opportunities include ambulatory surgery center channel development, integrated technology platforms combining robotics and biologics, and value-based care solution development.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product , 2026 to 2036
- Spinal Fusion Devices
- Spinal Biologics
- Vertebral Compression Fracture Treatment Devices
- Non-Fusion Devices
- Spinal Fusion Devices
- Y to o to Y Growth Trend Analysis By Product , 2021 to 2025
- Absolute $ Opportunity Analysis By Product , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Technology
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Technology, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Technology, 2026 to 2036
- Spinal Fusion and Fixation Technologies
- Vertebral Compression Fracture Treatment
- Vertebroplasty
- Spinal Fusion and Fixation Technologies
- Y to o to Y Growth Trend Analysis By Technology, 2021 to 2025
- Absolute $ Opportunity Analysis By Technology, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Surgery Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Surgery Type, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Surgery Type, 2026 to 2036
- Open Surgery
- Minimally Invasive Surgery
- Open Surgery
- Y to o to Y Growth Trend Analysis By Surgery Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Surgery Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product
- By Technology
- By Surgery Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Technology
- By Surgery Type
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product
- By Technology
- By Surgery Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Technology
- By Surgery Type
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product
- By Technology
- By Surgery Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Technology
- By Surgery Type
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product
- By Technology
- By Surgery Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Technology
- By Surgery Type
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product
- By Technology
- By Surgery Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Technology
- By Surgery Type
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product
- By Technology
- By Surgery Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Technology
- By Surgery Type
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product
- By Technology
- By Surgery Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Technology
- By Surgery Type
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Technology
- By Surgery Type
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Technology
- By Surgery Type
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Technology
- By Surgery Type
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Technology
- By Surgery Type
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Technology
- By Surgery Type
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Technology
- By Surgery Type
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Technology
- By Surgery Type
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Technology
- By Surgery Type
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Technology
- By Surgery Type
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Technology
- By Surgery Type
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Technology
- By Surgery Type
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Technology
- By Surgery Type
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Technology
- By Surgery Type
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Technology
- By Surgery Type
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Technology
- By Surgery Type
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Technology
- By Surgery Type
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Technology
- By Surgery Type
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Technology
- By Surgery Type
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Technology
- By Surgery Type
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Technology
- By Surgery Type
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Technology
- By Surgery Type
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Technology
- By Surgery Type
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product
- By Technology
- By Surgery Type
- Competition Analysis
- Competition Deep Dive
- Medtronic
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Johnson & Johnson
- NuVasive
- Zimmer Biomet
- Globus Medical
- VB Spine LLC
- Alphatec Spine
- Medtronic
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Surgery Type, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Surgery Type, 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 10: Latin America Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by Surgery Type, 2021 to 2036
- Table 13: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Western Europe Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 15: Western Europe Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by Surgery Type, 2021 to 2036
- Table 17: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 18: Eastern Europe Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 19: Eastern Europe Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 20: Eastern Europe Market Value (USD Million) Forecast by Surgery Type, 2021 to 2036
- Table 21: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: East Asia Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 23: East Asia Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 24: East Asia Market Value (USD Million) Forecast by Surgery Type, 2021 to 2036
- Table 25: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: South Asia and Pacific Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 27: South Asia and Pacific Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 28: South Asia and Pacific Market Value (USD Million) Forecast by Surgery Type, 2021 to 2036
- Table 29: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 30: Middle East & Africa Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 31: Middle East & Africa Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 32: Middle East & Africa Market Value (USD Million) Forecast by Surgery Type, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Product
- Figure 6: Global Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Technology
- Figure 9: Global Market Value Share and BPS Analysis by Surgery Type, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Surgery Type, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Surgery Type
- Figure 12: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Region
- Figure 15: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 20: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 21: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 23: North America Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 25: North America Market Attractiveness Analysis by Product
- Figure 26: North America Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 27: North America Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 28: North America Market Attractiveness Analysis by Technology
- Figure 29: North America Market Value Share and BPS Analysis by Surgery Type, 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Surgery Type, 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Surgery Type
- Figure 32: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 33: Latin America Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 34: Latin America Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 35: Latin America Market Attractiveness Analysis by Product
- Figure 36: Latin America Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 37: Latin America Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 38: Latin America Market Attractiveness Analysis by Technology
- Figure 39: Latin America Market Value Share and BPS Analysis by Surgery Type, 2026 and 2036
- Figure 40: Latin America Market Y-o-Y Growth Comparison by Surgery Type, 2026-2036
- Figure 41: Latin America Market Attractiveness Analysis by Surgery Type
- Figure 42: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 43: Western Europe Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 44: Western Europe Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 45: Western Europe Market Attractiveness Analysis by Product
- Figure 46: Western Europe Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 47: Western Europe Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 48: Western Europe Market Attractiveness Analysis by Technology
- Figure 49: Western Europe Market Value Share and BPS Analysis by Surgery Type, 2026 and 2036
- Figure 50: Western Europe Market Y-o-Y Growth Comparison by Surgery Type, 2026-2036
- Figure 51: Western Europe Market Attractiveness Analysis by Surgery Type
- Figure 52: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 53: Eastern Europe Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 54: Eastern Europe Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 55: Eastern Europe Market Attractiveness Analysis by Product
- Figure 56: Eastern Europe Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 57: Eastern Europe Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 58: Eastern Europe Market Attractiveness Analysis by Technology
- Figure 59: Eastern Europe Market Value Share and BPS Analysis by Surgery Type, 2026 and 2036
- Figure 60: Eastern Europe Market Y-o-Y Growth Comparison by Surgery Type, 2026-2036
- Figure 61: Eastern Europe Market Attractiveness Analysis by Surgery Type
- Figure 62: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 63: East Asia Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 64: East Asia Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 65: East Asia Market Attractiveness Analysis by Product
- Figure 66: East Asia Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 67: East Asia Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 68: East Asia Market Attractiveness Analysis by Technology
- Figure 69: East Asia Market Value Share and BPS Analysis by Surgery Type, 2026 and 2036
- Figure 70: East Asia Market Y-o-Y Growth Comparison by Surgery Type, 2026-2036
- Figure 71: East Asia Market Attractiveness Analysis by Surgery Type
- Figure 72: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 73: South Asia and Pacific Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 74: South Asia and Pacific Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 75: South Asia and Pacific Market Attractiveness Analysis by Product
- Figure 76: South Asia and Pacific Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 77: South Asia and Pacific Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 78: South Asia and Pacific Market Attractiveness Analysis by Technology
- Figure 79: South Asia and Pacific Market Value Share and BPS Analysis by Surgery Type, 2026 and 2036
- Figure 80: South Asia and Pacific Market Y-o-Y Growth Comparison by Surgery Type, 2026-2036
- Figure 81: South Asia and Pacific Market Attractiveness Analysis by Surgery Type
- Figure 82: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 83: Middle East & Africa Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 84: Middle East & Africa Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 85: Middle East & Africa Market Attractiveness Analysis by Product
- Figure 86: Middle East & Africa Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 87: Middle East & Africa Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 88: Middle East & Africa Market Attractiveness Analysis by Technology
- Figure 89: Middle East & Africa Market Value Share and BPS Analysis by Surgery Type, 2026 and 2036
- Figure 90: Middle East & Africa Market Y-o-Y Growth Comparison by Surgery Type, 2026-2036
- Figure 91: Middle East & Africa Market Attractiveness Analysis by Surgery Type
- Figure 92: Global Market - Tier Structure Analysis
- Figure 93: Global Market - Company Share Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

