Swab and Viral Transport Medium Market
This report offers detailed coverage of the swab and viral transport medium market, encompassing market sizing, revenue forecasting, competitor analysis, demand evaluation, growth factors, challenges, product trends, supply chain review, and future market directions.
Swab and Viral Transport Medium Market Size, Market Forecast and Outlook by FMI
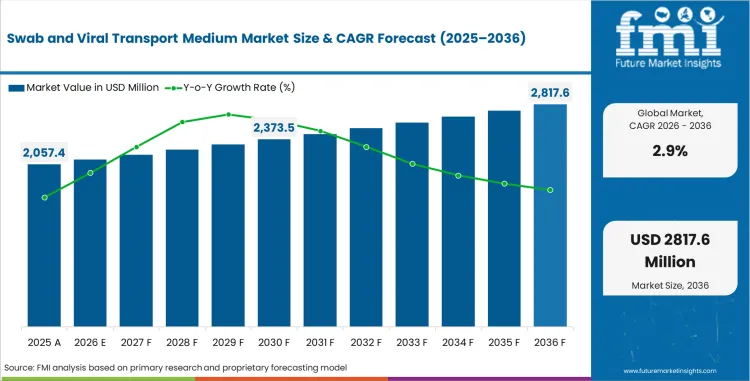
In 2026, the swab and viral transport medium market was valued at USD 1,999.4 million. Based on Future Market Insights analysis, demand for swab and viral transport medium is estimated to grow to USD 2,817.6 million by 2036. FMI projects a CAGR of 2.9% during the forecast period.
Summary of Swab and Viral Transport Medium Market
- Swab and Viral Transport Medium Market Definition
- The industry covers specimen collection swabs and stabilizing transport media used to preserve viral samples across hospitals, clinics, and diagnostic laboratory testing workflows.
- Demand Drivers in the Market
- Public health respiratory virus surveillance programs are driving recurring procurement across microbiology laboratories, hospitals, and decentralized testing environments.
- Seasonal influenza and RSV testing cycles are driving predictable demand peaks for standardized swab and transport medium kits.
- Expansion in point-of-care and decentralized testing is influencing SKU standardization favoring suppliers with scalable workflow-compatible specimen collection kits, as reported by FMI.
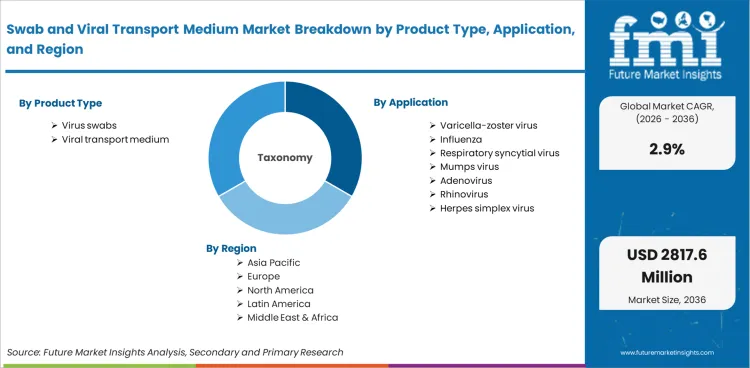
- Key Segments Analyzed in the FMI Report
- By Product Type: Virus Swabs lead at 59.0% share in 2026, followed by Viral Transport Medium at 41.0% share.
- By Application: Varicella-Zoster Virus leads at 32.6% share in 2026, followed by Influenza and Respiratory Syncytial Virus in the next tier of demand.
- Geography: India and China show the fastest growth at 5.2% and 4.2% CAGR, supported by expanding diagnostic infrastructure and preparedness-focused procurement.
- Analyst Opinion at FMI
- Sabyasachi Ghosh, Principal Consultant at Future Market Insights, opines, ‘Buyer preference is being shaped by specimen stability, standardized product performance, and uninterrupted fulfillment discipline, as healthcare systems move toward steady-state surveillance procurement and reduce tolerance for variability in routine testing workflows.’
- Strategic Implications and Executive Takeaways
- Strengthen institutional contracting with documentation-ready quality systems, validated specimen preservation claims, and dependable replenishment support aligned to laboratory workflows.
- Build growth plans around expanding diagnostic networks where recurring viral testing volumes require tighter inventory reliability and standardized product use.
- Protect approved supplier positions through continuity of supply commitments, since validated collection and transport products are not switched easily once embedded into routine protocols.
- Methodology
- As per FMI, sizing and segment splits were reconciled using company primary disclosures and demand mapping by laboratory, hospital, and decentralized testing settings.
- Product and end-user shares were validated by linking specimen collection demand patterns to surveillance intensity and routine testing throughput.
- Pricing sensitivity was stress tested under institutional tender and replenishment scenarios to confirm revenue response under realistic procurement conditions.
Absolute dollar growth of USD 799.2 million over the decade signals steady expansion rather than a demand reset. As per FMI, procurement is expected to remain supported by respiratory virus surveillance programs, recurring seasonal testing cycles, and routine specimen collection requirements across hospitals, public health laboratories, and decentralized care settings. At the same time, post-pandemic normalization, standardized purchasing, and price-sensitive institutional tenders are expected to keep value growth measured across mature health systems. “Today, beyond the accuracy of diagnostic results, patients seek greater comfort in testing. The COVID-19 pandemic has especially underscored the importance of patient well-being in healthcare settings, the opportunity to provide the same quality and performance with a less invasive sample with our Biofire Spotfire R/ST Panel Mini truly illustrates our mission to make an impact on patients’ health.” Said Dr Charles K Cooper, executive vice president and chief medical officer at bioMérieux. [1]
Based on FMI's report, India (5.2% CAGR) and China (4.2% CAGR) are expected to lead growth due to expanding diagnostic infrastructure and continued preparedness-oriented procurement. France (2.8% CAGR) and Germany (2.5% CAGR) are expected to maintain stable expansion through structured public health testing systems. Mature markets such as the USA (2.1%) and the UK (2.2%) are expected to contribute mainly through replacement demand, with growth moderated by established surveillance capacity and standardized replenishment cycles.
Swab and Viral Transport Medium Market Definition
The market includes specimen collection swabs and stabilizing viral transport media used to preserve viral material during transit from the point of collection to the diagnostic laboratory. These products are sold to hospitals, ambulatory and clinic-based testing settings, specialty care sites, and diagnostic laboratories to support viral testing across respiratory, dermatological, and mucosal specimen workflows where sample integrity must be maintained. The market includes virus swabs and viral transport medium products used for routine diagnostic use. The market is driven by specimen collection volumes, surveillance intensity, laboratory workflow requirements, and institutional procurement protocols. The revenue stream is derived from sales of swabs and matched transport systems to healthcare and laboratory end users.
Swab and Viral Transport Medium Market Inclusions
The report provides global and regional market sizing, as well as a 10-year forecast from 2026 to 2036. The report also provides market sizing by product type, application, and end user, with country-level CAGR comparisons across key markets. The report also provides analysis on competitive positioning of key suppliers, procurement dynamics across institutional buyers, and the influence of product quality, regulatory compliance, and distribution continuity on supplier choice, as per FMI.
Swab and Viral Transport Medium Market Exclusions
The scope does not include bacterial culture swabs and transport systems that are not formulated for viral specimen preservation. Molecular extraction kits, PCR reagents, assay platforms, and diagnostic instruments are also excluded as separate product categories. The scope also does not include broader laboratory consumables that are not used as viral collection and transport products, but only swabs and viral transport medium products used for patient and laboratory viral testing workflows.
Swab and Viral Transport Medium Market Research Methodology
- Primary research: Sample size of 320 Interviews were carried out with hospital microbiology laboratory directors, public health surveillance coordinators, diagnostic procurement managers, and specimen collection product distributors to confirm purchasing patterns and replacement demand drivers.
- Desk research: Public data from regulatory bodies, surveillance agencies, epidemiology publications, company materials, and healthcare system sources were analyzed to compare usage requirements and procurement standards.
- Market sizing and forecasting: A hybrid approach was created based on product-level demand mapping, end-user consumption patterns, and regional testing intensity, and then harmonized through volume and price triangulation.
- Data validation and update cycle: Results were filtered for variance, tested for internal consistency, and assessed through structured peer review before distribution.
Segmental analysis
Swab and Viral Transport Medium Market Analysis by Product Type
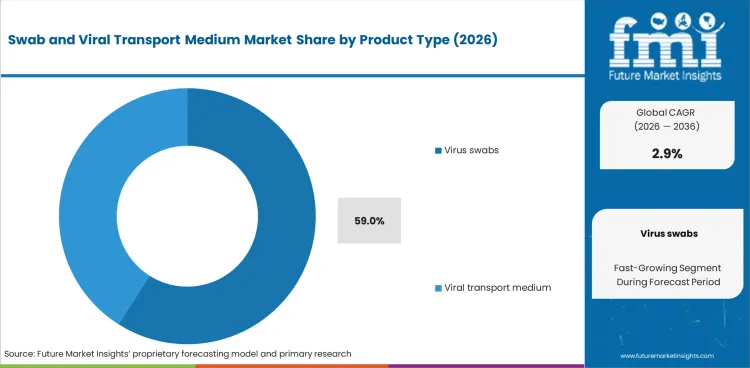
Based on FMI’s report, virus swabs are estimated to hold 59.0% share in 2026. This lead position is supported by their indispensable role in first-step specimen capture across respiratory, throat, nasal, and mucosal testing workflows, which keeps them embedded in routine viral diagnostic protocols and procurement catalogs. Viral transport medium accounts for the remaining share, reflecting its paired but dependent role within specimen preservation and transit workflows.
- Front-end testing requirement: Viral testing begins with specimen collection, and CDC states that for current respiratory viral diagnosis, an upper respiratory specimen should be collected using sterile swabs, making swabs the entry-point product consumed across routine hospital and laboratory testing workflows. [2]
- Molecular assay sample reliability: Recent peer-reviewed evidence continues to treat nasopharyngeal swab PCR samples as the benchmark for respiratory virus detection, while another 2024 study found midturbinate swabs yielded 53% lower viral load than nasopharyngeal swabs for influenza, reinforcing preference for high-quality swab-based collection. [3]
- Routine diagnostic starting point: Virus swabs hold the highest share because specimen collection is the first and unavoidable step in most viral testing workflows, making swabs a routine-use consumable across hospitals, diagnostic laboratories, surveillance programs, and outpatient testing settings.
Swab and Viral Transport Medium Market Analysis by Application
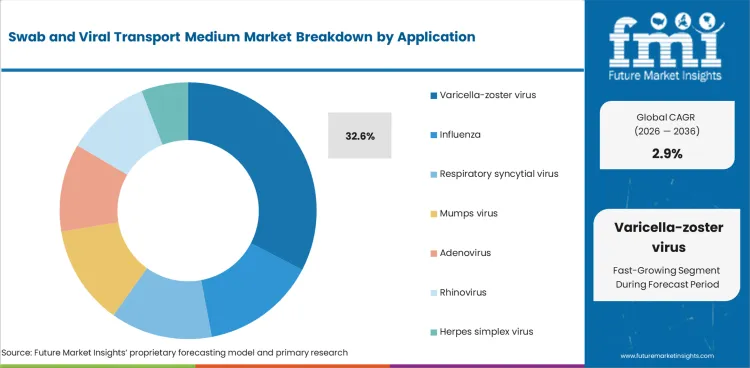
Varicella-zoster virus accounts for 32.6% share in 2026, based on FMI’s report, reflecting its established role within viral diagnostic workflows where timely and accurate confirmation supports treatment decisions and infection management. Influenza and respiratory syncytial virus continue to generate recurring demand through seasonal cycles, while other viral applications contribute through steady but narrower testing requirements. The leading position of varicella-zoster virus is supported by diagnostic relevance, patient management needs, and dependable specimen preservation requirements.
- High recurring disease burden: Recent global burden analysis reported that combined varicella and herpes zoster cases rose from 72.8 million in 1990 to 86.7 million in 2021, indicating a large and persistent diagnostic base that supports continued specimen collection demand for VZV-related testing. [4]
- Older adults drive testing: A 2025 burden study found incidence in adults aged 65 years and older increased from 2.83 million to 6.52 million between 1992 and 2021, supporting sustained VZV testing need as aging populations raise shingles-related diagnostic volumes.[5]
- Confirmatory testing sustains demand: Varicella-zoster virus holds a leading application share because suspected chickenpox and shingles cases often require laboratory confirmation, and guidance continues to favor PCR on lesion-based swab specimens, keeping VZV tied to routine swab collection and transport workflows in clinical practice.
Swab and Viral Transport Medium Market Drivers, Restraints, and Opportunities
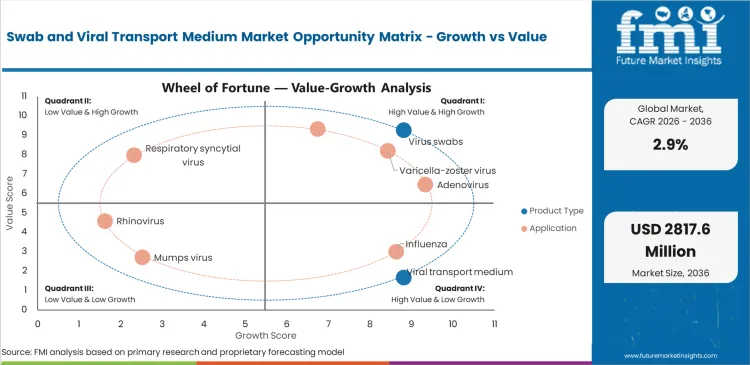
Future Market Insights analysis indicates that historical patterns point to a surveillance-driven diagnostic consumables category where demand is anchored in year-round specimen collection and transport needs for respiratory and other viral pathogens. Estimated valuation in 2026 is being supported by public health monitoring programs, routine hospital and laboratory testing workflows, and growing use of decentralized collection pathways that require standardized swab and transport products, as per FMI.
While category demand is being restrained by post-pandemic normalization, commoditization in standard product formats, and tender-led price discipline in mature markets, value is being supported by broader testing access, preparedness-oriented procurement, and greater reliance on molecular diagnostic workflows that depend on front-end specimen integrity. Based on FMI’s report, the forecast trajectory reflects a market where hospitals and diagnostic laboratories remain core demand centers, while suppliers are differentiated by product consistency, compliance readiness, and dependable fulfillment execution.
- Surveillance-backed procurement: Demand is being supported as buyers continue to replenish standardized specimen collection products for recurring public health and seasonal testing programs.
- Tender-pricing: As products remain standardized, pricing power is limited, and competition is filtered through institutional procurement and replenishment contracts.
- Decentralized testing expansion: Demand is being supported as more viral testing is routed through clinics, specialty settings, and distributed care environments requiring reliable collection workflows.
Regional Analysis
Based on the regional analysis, swab and viral transport medium market is segmented into North America, Latin America, Western Europe, Eastern Europe, East Asia, South Asia & Pacific, and Middle East & Africa across 40+ countries. Regional performance is assessed using country-level demand signals tied to surveillance intensity, diagnostic infrastructure, procurement discipline, and regulatory readiness, as per FMI. The full report also offers market attractiveness analysis based on regional trends.
.webp)
| Country | CAGR |
|---|---|
| United States | 2.1% |
| United Kingdom | 2.2% |
| Germany | 2.5% |
| France | 2.8% |
| China | 4.2% |
| India | 5.2% |
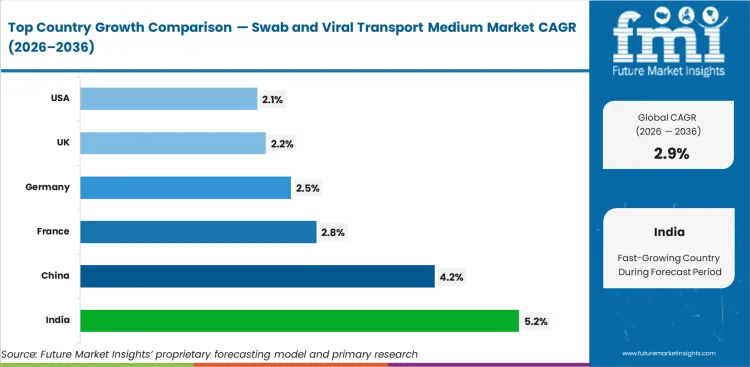
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
North America Swab and Viral Transport Medium Market Analysis
North America is shaped by surveillance continuity, laboratory standardization, and procurement discipline, where product choice is influenced by microbiology workflow compatibility and institutional compliance requirements across hospitals and diagnostic networks. Becton, Dickinson and Company holds strong placement through portfolio breadth, broad distribution reach, and established laboratory relationships. Cardinal Health, Inc. and McKesson Laboratories benefit from institutional supply access and distribution strength across healthcare procurement channels. Based on FMI’s report, demand remains anchored in recurring testing and replacement cycles.
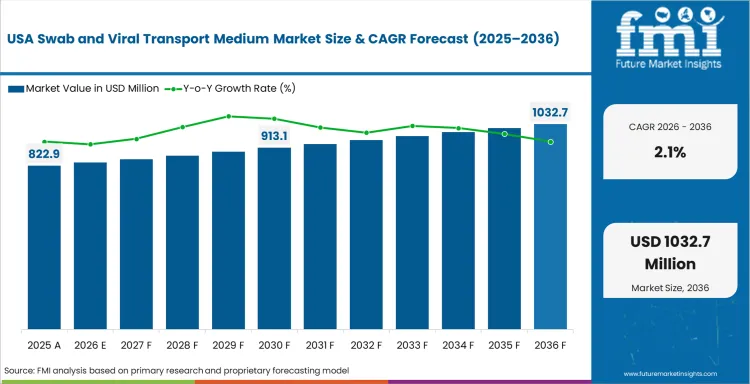
- United States: The demand for swab and viral transport medium in the United States is expected to grow at a CAGR of 2.1% during the forecast period of 2036. This is driven by high disease testing rates, broad laboratory access, and continued use of surveillance-linked specimen collection workflows, although this is tempered by market maturity and standardized purchasing behavior.
FMI’s report includes a detailed analysis of growth in the North American region, along with a country-wise assessment that includes the United States. Readers can also find regional trends, regulations, and market growth based on different segments and countries in the North America region.
Asia Pacific Swab and Viral Transport Medium Market Analysis
Asia Pacific remains the market for demand growth, driven by increases in diagnostic infrastructure development, viral testing penetration, and readiness-driven procurement within hospital and laboratory infrastructures. Becton, Dickinson and Company has significant institutional presence owing to their multinational scale and product availability. Competition for Thermo Fisher Scientific, Quidel Corporation, and regional suppliers is based on their reach, compatibility, and price support for public and private sector diagnostic systems. According to FMI’s report, demand is driven by increases in testing capacity and infrastructure formalization.
- China: The swab and viral transport medium market in China has been projected to grow at a CAGR of 4.2% over the forecast period of 2036. This growth will be attributed to the high investments in pandemics, expansion in laboratories, and funding for public health.
- India: Demand for swab and viral transport medium in India is projected to rise at 5.2% CAGR through 2036. Growth is being supported by diagnostic infrastructure development, higher testing penetration, and increasing use of standardized specimen collection products across urban and secondary care environments.
The full report examines the market for swab and viral transport media in East Asia and South Asia from 2021 to 2036, including pricing trends and growth drivers in China, Japan, South Korea, India, Indonesia, Malaysia, and Thailand. This assessment identifies trends that influence the demand and procurement in the region.
Europe Swab and Viral Transport Medium Market Analysis
Europe is compliance-led and surveillance-oriented, where procurement is shaped by laboratory quality standards, public health testing structures, and documentation expectations across hospital and diagnostic settings. Becton, Dickinson and Company holds strong institutional access across large laboratory networks. Deltalab and VIRCELL S.L. compete through focused diagnostic consumables portfolios and established regional distribution relationships. As per FMI, replacement demand and seasonal testing cycles continue to govern purchasing rhythm across the region.
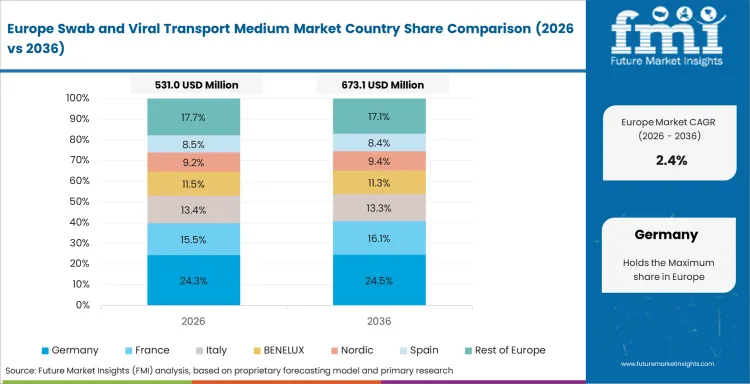
- Germany: Swab and viral transport medium demand in Germany is forecasted to grow at a CAGR of 2.5% through 2036, owing to robust public health campaigns, reliable testing mechanisms, and the continued importance of prompt diagnosis. Demand is managed through institutional channels and is based on compliance.
- France: The market for swab and viral transport medium in France is anticipated to grow at a CAGR of 2.8% during the forecast period of 2036. This is because of the existing procurement mechanisms, stable diagnostic processes, and the consistent use of compliant specimen collection devices in surveillance and clinical environments.
- United Kingdom: This Market in the United Kingdom has been projected to grow at a CAGR of 2.2% over the forecast period of 2036. The public health demand and regular use in laboratories in the United Kingdom contribute to the growth of the Swab & Viral Transport Medium market. However, the presence of mature testing infrastructure and regular replacement cycles help in controlling the growth.
FMI’s analysis of swab and viral transport medium market in Europe consists of country-wise assessment that includes Germany, France, the UK, Italy, Spain, Nordic countries, BENELUX, and Rest of Europe. Readers can know various regulations and latest trends in the regional market.
Competitive Aligners for Market Players
The state of the market structure remains moderately fragmented. The state of competition remains effective in the hands of suppliers who can ensure consistency in the product offerings, large volume fulfillment, and compliant manufacturing for routine specimen collection and transport products. The product portfolio mix indicates the nature of the product in which the demand share for virus swabs remains larger compared to the product. The state of competition remains not in terms of innovation but in the ability to ensure product performance.
Firms with developed manufacturing infrastructures, extensive distribution channels, and developed quality control mechanisms have structural advantages since the qualification process is based on documentation, workflow acceptance, and continuity. Scale players have advantages based on catalog size, multi-channel access, and the ability to serve hospitals, laboratories, and decentralized testing sites under a single procurement umbrella. Suppliers with lower distribution coverage usually compete based on access to specific regions, pricing flexibility, and/or specific customer relationships, which may compromise their ability to compete effectively in a rebid process.
Customer concentration supports customer leverage. Recurring demand is driven by hospitals, diagnostic labs, and public health programs. Specialty and decentralized locations are the next tier for product consumption. Large procurement networks maintain active preferred supplier lists and favor validated products that are interchangeable and can be replenished quickly during surges in demand. There is limited price power for standard products, but there is measurable advantage for products where reliability, kit standardization, or distribution creates operational preference, according to Future Market Insights.
Recent Developments
- In September 2025, bioMérieux has received US Food and Drug Administration (FDA) 510(k) clearance and CLIA waiver for anterior nasal swab specimens as an additional sample type for use with the Biofire Spotfire Respiratory/Sore Throat Panel Mini, specifically for respiratory testing applications. [1]
- In February 2025, Hologic, Inc announced that the USA Food and Drug Administration (FDA) has granted 510(k) clearance for the company’s Aptima SARS-CoV-2 assay, which was first granted Emergency Use Authorization (EUA) status in May 2020. The Aptima SARS-CoV-2 Assay is an in vitro diagnostic test that detects SARS-CoV-2 RNA from nasopharyngeal (NP) or nasal swab samples obtained from people exhibiting signs and symptoms of a respiratory tract infection. [6]
Key Players in Swab and Viral Transport Medium Market
- Becton, Dickinson and Company
- McKesson Laboratories
- Cardinal Health, Inc.
- Thermo Fisher Scientific
- Quidel Corporation
- Copan Diagnostic, Inc.
- Deltalab
- VIRCELL S.L.
- Titan Biotech, Inc.
- Medical Wire & Equipment (MWE)
Scope of the Report
| Metric | Value |
|---|---|
| Quantitative Units | USD 1,999.4 million (2026) to USD 2,817.6 million (2036), at a CAGR of 2.9% |
| Market Definition | The swab and viral transport medium market comprises specimen collection swabs and stabilizing transport media used to preserve viral pathogen viability during transit from collection point to diagnostic laboratory, where demand is shaped by surveillance programs, laboratory workflow compatibility, and regulatory-compliant product use across healthcare settings. |
| Product Segmentation | Virus swabs, Viral transport medium |
| Application Segmentation | Varicella-zoster virus, Influenza, Respiratory syncytial virus, Mumps virus, Adenovirus, Rhinovirus, Herpes simplex virus |
| End User Segmentation | Hospitals & ASCs, Specialty clinics, Diagnostic laboratories, Others |
| Regions Covered | North America, Latin America, East Asia, South Asia & Pacific, Western Europe, Eastern Europe, and Middle East & Africa |
| Countries Covered | United States, United Kingdom, Germany, France, China, India, and 40+ countries |
| Key Companies Profiled | Becton, Dickinson and Company, McKesson Laboratories, Cardinal Health, Inc., Thermo Fisher Scientific, Quidel Corporation, Copan Diagnostic, Inc., Deltalab, VIRCELL S.L., Titan Biotech, Inc., Medical Wire & Equipment (MWE) |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid top down and bottom up market modeling validated through primary interviews with laboratory and procurement stakeholders, supported by demand triangulation across surveillance intensity, specimen collection workflows, and product adoption patterns, as per FMI. |
Swab and Viral Transport Medium Market Analysis by Segments
Product Type
- Virus swabs
- Viral transport medium
Application
- Varicella-zoster virus
- Influenza
- Respiratory syncytial virus
- Mumps virus
- Adenovirus
- Rhinovirus
- Herpes simplex virus
End User
- Hospitals & ASCs
- Specialty clinics
- Diagnostic laboratories
- Others
Region
- North America
- USA
- Canada
- Mexico
- Latin America
- Brazil
- Argentina
- Rest of Latin America
- East Asia
- China
- Japan
- South Korea
- South Asia & Pacific
- India
- ASEAN Countries
- Australia & New Zealand
- Rest of South Asia
- Western Europe
- Germany
- UK
- France
- Italy
- Spain
- BENELUX
- Nordic Countries
- Rest of Western Europe
- Eastern Europe
- Russia
- Hungary
- Poland
- Rest of Eastern Europe
- Middle East & Africa
- Saudi Arabia
- Türkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
Bibliography
- [1] bioMérieux (2025) bioMérieux Receives FDA Clearance for Anterior Nasal Swab Testing on Spotfire Platform.
- [2] CDC (COVID- 19). (2024) Interim Guidelines for Collecting and Handling of Clinical Specimens for COVID-19 Testing
- [3] PubMed Central (2025) Saliva and Anterior Nasal Samples for Detecting Respiratory Viruses in Children
- [4] PubMed Central (2025) Global burden of varicella and herpes zoster across 204 countries, 1990-2021: a temporal trend analysis in the era of the COVID-19 pandemic and projections to 2036
- [5] PubMed Central. (2025) The global burden of varicella and herpes zoster in adults aged 65 years and older: a comprehensive analysis based on the global burden of disease 2021
- [6] Hologic, Inc. (2025) Hologic Announces FDA Clearance of Aptima SARS-CoV-2 Assay.
This Report Addresses
- Market intelligence to enable structured strategic decision making across viral specimen collection planning and diagnostic workflow continuity
- Market size estimation and 10-year revenue forecasts from 2026 to 2036 for swab and viral transport medium demand
- Growth opportunity mapping across product type classes and application-wise diagnostic demand pools
- Segment and regional revenue forecasts aligned to the stated taxonomy and covered geographies
- Competition strategy assessment including supplier positioning, product fit, and distribution coverage benchmarking
- Product and compliance tracking focused on specimen integrity expectations, laboratory standards, and routine replenishment requirements
- Regulatory impact analysis covering quality-led product selection and institution-level protocol adoption in regulated care settings
- Market report delivery in PDF, Excel, PPT, and interactive dashboard formats for executive and operational use
Frequently Asked Questions
How large is the demand for Swab and Viral Transport Medium in the global market in 2026?
Demand for swab and viral transport medium in the global market is estimated to be valued at USD 1,999.4 million in 2026, as per FMI.
What will be the market size of Swab and Viral Transport Medium in the global market by 2036?
Market size for swab and viral transport medium is projected to reach 2,817.6 million by 2036.
What is the expected demand growth for Swab and Viral Transport Medium in the global market between 2026 and 2036?
Demand for swab and viral transport medium in the global market is expected to grow at a CAGR of 2.9% between 2026 and 2036.
Which product type is poised to lead global sales by 2026?
Virus swabs are expected to be the dominant product type, capturing 59.0% share in 2026.
How significant is the role of varicella-zoster virus in driving adoption in 2026?
Varicella-zoster virus is projected to hold 32.6% share of application demand in 2026.
Which country is expected to record the fastest growth during the forecast period?
India is projected to grow at a CAGR of 5.2% during 2026 to 2036 among the listed countries.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product Type , 2026 to 2036
- Virus swabs
- Viral transport medium
- Virus swabs
- Y to o to Y Growth Trend Analysis By Product Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Product Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application, 2026 to 2036
- Varicella-zoster virus
- Influenza
- Respiratory syncytial virus
- Mumps virus
- Adenovirus
- Rhinovirus
- Herpes simplex virus
- Varicella-zoster virus
- Y to o to Y Growth Trend Analysis By Application, 2021 to 2025
- Absolute $ Opportunity Analysis By Application, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product Type
- By Application
- Competition Analysis
- Competition Deep Dive
- Becton, Dickinson and Company
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- McKesson Laboratories
- Cardinal Health, Inc.
- Thermo Fisher Scientific
- Quidel Corporation
- Copan Diagnostic, Inc.
- Deltalab
- VIRCELL S.L.
- Titan Biotech, Inc.
- Medical Wire & Equipment (MWE)
- Becton, Dickinson and Company
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by Application, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Product Type
- Figure 6: Global Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Application
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 21: North America Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 22: North America Market Attractiveness Analysis by Product Type
- Figure 23: North America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 25: North America Market Attractiveness Analysis by Application
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 28: Latin America Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 29: Latin America Market Attractiveness Analysis by Product Type
- Figure 30: Latin America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 31: Latin America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 32: Latin America Market Attractiveness Analysis by Application
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 35: Western Europe Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 36: Western Europe Market Attractiveness Analysis by Product Type
- Figure 37: Western Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 38: Western Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 39: Western Europe Market Attractiveness Analysis by Application
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 42: Eastern Europe Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by Product Type
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 45: Eastern Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by Application
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 49: East Asia Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 50: East Asia Market Attractiveness Analysis by Product Type
- Figure 51: East Asia Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 52: East Asia Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 53: East Asia Market Attractiveness Analysis by Application
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 56: South Asia and Pacific Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Product Type
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 59: South Asia and Pacific Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by Application
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 63: Middle East & Africa Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Product Type
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 66: Middle East & Africa Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

