Andersen-Tawil Syndrome Treatment Market
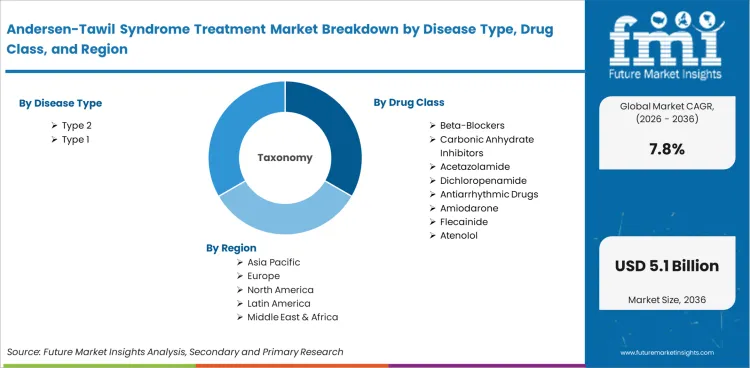
Andersen-Tawil Syndrome Treatment Market is segmented by Disease Type (Type 2, Type 1), Drug Class (Beta-Blockers, Carbonic Anhydrate Inhibitors, Acetazolamide, Dichloropenamide, Antiarrhythmic Drugs, Amiodarone, Flecainide, Atenolol), Distribution Channel (Hospital Pharmacy, Retail Pharmacy, Online Pharmacy), and Region. Forecast for 2026 to 2036.
Andersen-Tawil Syndrome Treatment Market Size, Market Forecast and Outlook By FMI
Summary of the Andersen-Tawil Syndrome Treatment Market
- Demand and Growth Drivers
- Increasing diagnostic awareness and genetic testing accessibility are shaping demand for Andersen-Tawil syndrome treatments, as healthcare systems improve identification of rare channelopathies requiring long-term pharmacological management.
- Beta-blockers account for 34.8% of the drug class segment, reflecting their established role in managing cardiac arrhythmias associated with Andersen-Tawil syndrome.
- Hospital pharmacies hold 49.2% of distribution share, confirming institutional healthcare settings as the primary dispensing channel for rare disease treatments.
- Product and Segment View
- Type 2 disease is expected to account for 62.5% of the disease type segment in 2026, reflecting higher diagnostic prevalence and established treatment pathways for KCNJ2-related presentations.
- Beta-blockers hold 34.8% of drug class share, confirming their frontline position in arrhythmia management for Andersen-Tawil patients.
- Hospitals represent 46.8% of end-use share, indicating that specialized cardiac and genetics centers are the primary treatment delivery settings.
- Geography and Competitive Outlook
- The USA (8.2%) and South Korea (8.0%) are expected to lead growth, driven by advanced diagnostic infrastructure and rare disease treatment frameworks.
- Japan (7.4%) reflects strong rare disease healthcare programs and genetic testing capabilities.
- Teva Pharmaceuticals Ltd holds an estimated 18.0% market share, leveraging its generic pharmaceutical portfolio serving rare cardiac conditions.
- Analyst Opinion
- The Andersen-Tawil syndrome treatment market is evolving from symptomatic management toward more targeted therapeutic approaches as understanding of KCNJ2 and KCNJ5 gene mutations improves.
- Growth reflects expanding diagnostic capabilities that identify more patients requiring treatment, combined with increasing healthcare spending on rare disease management.
- Pharmaceutical companies with established rare disease portfolios and experience navigating orphan drug regulatory pathways are positioned to capture long-term market share.
- Pricing dynamics are shaped by orphan drug designation frameworks that provide market exclusivity and reimbursement advantages in developed markets.
- Companies investing in gene-targeted therapies and personalized treatment approaches are expected to reshape the competitive landscape beyond 2030.
Andersen-Tawil Syndrome Treatment Market Definition
The Andersen-Tawil Syndrome Treatment Market encompasses pharmaceutical products and therapeutic interventions used in the management of Andersen-Tawil syndrome, a rare genetic channelopathy characterized by periodic paralysis, cardiac arrhythmias, and distinctive physical features. The market covers beta-blockers, carbonic anhydrase inhibitors, antiarrhythmic drugs, and related therapies.
Andersen-Tawil Syndrome Treatment Market Inclusions
Market scope includes all commercially traded treatments categorized by disease type (Type 1, Type 2), drug class (beta-blockers, carbonic anhydrase inhibitors, acetazolamide, dichloropenamide, antiarrhythmic drugs, amiodarone, flecainide, atenolol), distribution channel (hospital pharmacy, retail pharmacy, online pharmacy), and end use (hospitals, specialty clinics, home care). The revenue range spans 2026 to 2036.
Andersen-Tawil Syndrome Treatment Market Exclusions
The scope does not include general cardiac arrhythmia treatments not specific to Andersen-Tawil syndrome, general electrolyte management products, or experimental gene therapies not yet commercially available.
Andersen-Tawil Syndrome Treatment Market Research Methodology
- Primary Research: FMI analysts conducted interviews with industry participants, product specialists, and distribution channel experts across key markets.
- Desk Research: Combined industry publications, regulatory filings, trade association data, and manufacturer disclosures.
- Market Sizing and Forecasting: Bottom-up aggregation across product segments and regional adoption curves, validated through top-down cross-referencing.
- Data Validation: Cross-checked quarterly against industry production data and manufacturer-reported figures.
Why is the Andersen-Tawil Syndrome Treatment Market Growing?
- Genetic testing accessibility is improving, leading to earlier and more accurate diagnosis of Andersen-Tawil syndrome, which expands the identified patient population requiring pharmacological management.
- Rare disease treatment frameworks in the USA, EU, and East Asia provide regulatory incentives and reimbursement support that encourage pharmaceutical investment in orphan indications.
- The USA (8.2%) leads growth, reflecting advanced diagnostic infrastructure, established rare disease healthcare pathways, and strong reimbursement frameworks for orphan drugs.
Demand for Andersen-Tawil syndrome treatments reflects improving diagnostic capabilities that identify a growing number of patients with this rare channelopathy. Genetic panel testing and next-generation sequencing are making diagnosis more accessible, particularly in developed markets where healthcare systems invest in rare disease identification programs. This expanded diagnostic reach translates into a larger treated patient population requiring long-term pharmacological management.
Type 2 disease (62.5% of disease type share) accounts for the majority of diagnosed cases, with KCNJ2 gene mutations being the most commonly identified genetic basis. Treatment protocols for Type 2 are better established, with beta-blockers, carbonic anhydrase inhibitors, and antiarrhythmic agents forming the standard pharmacological approach. Type 1 presentations, while less common, often require more complex multi-drug regimens.
Orphan drug regulatory frameworks in the USA (Orphan Drug Act), EU (European Medicines Agency orphan designation), and Japan (Ministry of Health, Labour and Welfare rare disease programs) provide market exclusivity periods, regulatory fast-tracking, and reimbursement advantages that incentivize pharmaceutical investment in treatments for rare conditions like Andersen-Tawil syndrome.
Market Segmentation Analysis
- Type 2 disease holds 62.5% of disease type share, reflecting higher diagnostic prevalence and established treatment protocols.
- Beta-blockers account for 34.8% of drug class share, confirming their frontline role in cardiac arrhythmia management for affected patients.
- Hospital pharmacies represent 49.2% of distribution, indicating institutional dispensing dominance for rare disease treatments.
The Andersen-Tawil Syndrome Treatment Market is segmented by disease type (Type 1, Type 2), drug class (beta-blockers, carbonic anhydrase inhibitors, acetazolamide, dichloropenamide, antiarrhythmic drugs, amiodarone, flecainide, atenolol), distribution channel (hospital pharmacy, retail pharmacy, online pharmacy), and end use (hospitals, specialty clinics, home care).
Insights into the Type 2 Disease Segment
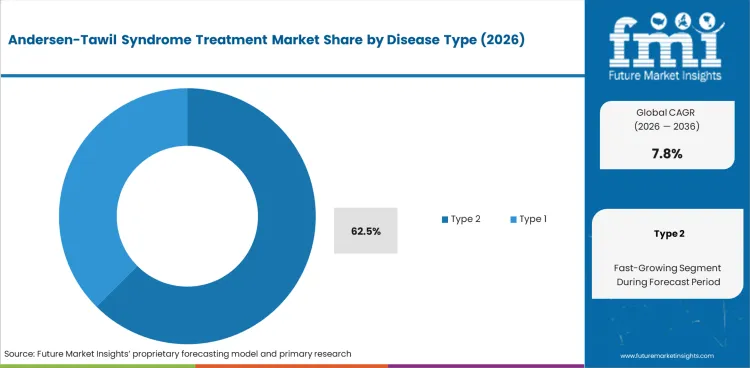
In 2026, Type 2 is expected to represent 62.5% of the disease type segment. Type 2 Andersen-Tawil syndrome, associated with KCNJ2 mutations, presents with a more defined clinical profile that facilitates diagnosis and treatment protocol standardization. Patients typically require long-term beta-blocker therapy combined with periodic paralysis management.
Diagnostic advances in genetic testing are improving identification rates for Type 2 presentations, expanding the treatable patient population.
Insights into the Hospital Pharmacy Distribution
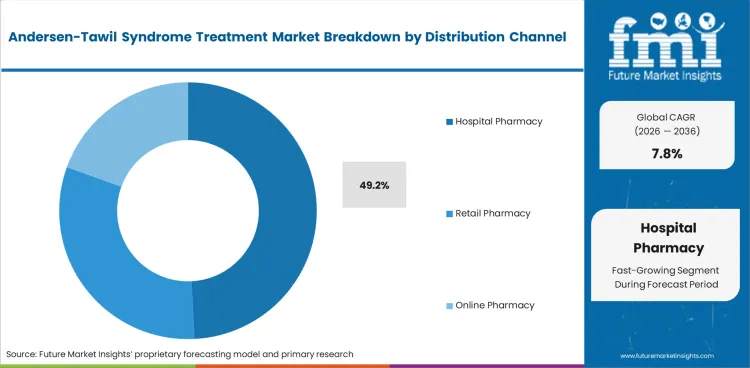
Hospital pharmacies account for 49.2% of distribution share. Rare disease treatments are predominantly initiated and managed within hospital settings where specialist cardiac and genetics teams oversee diagnosis, treatment initiation, and ongoing monitoring.
Retail and online pharmacy channels serve maintenance prescriptions for stable patients transitioning to outpatient management.
Andersen-Tawil Syndrome Treatment Market Drivers, Restraints, and Opportunities
- Improving genetic diagnostic capabilities are expanding the identified patient population requiring Andersen-Tawil syndrome treatment.
- Limited patient population size constrains pharmaceutical investment in dedicated therapies, resulting in reliance on repurposed medications from broader cardiac and neurological indications.
- Gene-targeted therapeutic approaches offer long-term opportunities to develop Andersen-Tawil-specific treatments that address underlying channelopathy mechanisms.
The Andersen-Tawil Syndrome Treatment Market benefits from improving diagnostics and rare disease regulatory frameworks. Small patient populations and treatment complexity present constraints, while gene therapy and personalized medicine offer growth opportunities.
Diagnostic Capability Expansion
Demand is shaped by improving access to genetic testing and rare disease diagnostic programs. Next-generation sequencing panels that include channelopathy genes are becoming standard in cardiac genetics workups, improving identification of Andersen-Tawil patients who previously went undiagnosed.
Limited Patient Population and Investment Constraints
Growth is constrained by the small patient population, which limits the commercial return on dedicated drug development. Most treatments are repurposed from broader cardiac and neurological indications, limiting the availability of Andersen-Tawil-specific therapeutic options.
Orphan Drug Regulatory Incentives
Adoption is supported by orphan drug designation frameworks that provide market exclusivity, regulatory fast-tracking, and reimbursement advantages. These incentives encourage pharmaceutical companies to invest in rare disease indications despite limited patient populations.
Gene-Targeted Therapy Development
Growth reflects emerging research into gene-targeted approaches for channelopathies. KCNJ2 and KCNJ5 gene correction strategies, while still in early development, represent long-term opportunities to develop treatments that address the underlying genetic cause rather than managing symptoms.
Analysis of Andersen-Tawil Syndrome Treatment Market By Key Countries
.webp)
| Country | CAGR |
|---|---|
| USA | 8.2% |
| South Korea | 8% |
| EU | 7.8% |
| UK | 7.6% |
| Japan | 7.4% |
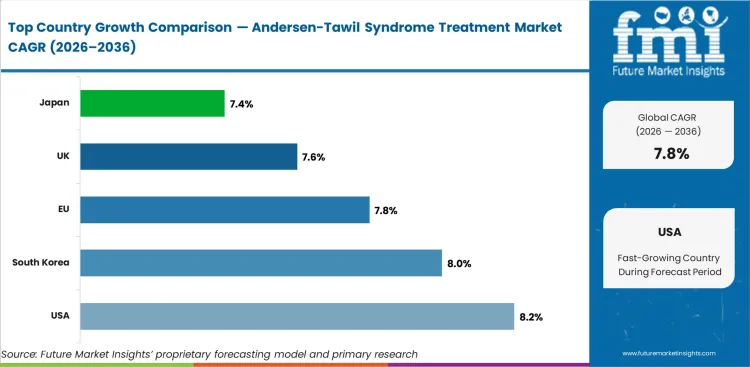
- The USA leads growth at 8.2% CAGR, reflecting advanced diagnostic infrastructure and established rare disease treatment frameworks.
- South Korea (8.0%) and the EU (7.8%) sustain growth through expanding genetic testing programs and rare disease healthcare investment.
- Japan (7.4%) supports demand through dedicated rare disease healthcare programs and genetic testing capabilities.
The global Andersen-Tawil Syndrome Treatment Market is expected to expand at a CAGR of 7.8% from 2026 to 2036. The analysis covers key markets including the USA, South Korea, EU, UK, and Japan.
Demand Outlook for Andersen-Tawil Syndrome Treatment Market in USA
The USA is expected to lead growth at 8.2% through 2036, driven by advanced genetic diagnostic infrastructure, Orphan Drug Act incentives, and strong reimbursement for rare disease treatments.
- Genetic testing infrastructure supports early diagnosis.
- Orphan Drug Act provides regulatory and commercial incentives.
- Specialized rare disease centers concentrate treatment expertise.
Future Outlook for Andersen-Tawil Syndrome Treatment Market in South Korea
South Korea is expected to grow at 8.0% through 2036, reflecting advanced healthcare infrastructure, expanding genetic testing programs, and strong government support for rare disease management.
- Genetic testing program expansion improves diagnostic rates.
- Government rare disease support frameworks enhance treatment access.
- Advanced cardiac care capabilities support specialist treatment delivery.
Opportunity Analysis of Andersen-Tawil Syndrome Treatment Market in EU
The EU is expected to grow at 7.8% through 2036, supported by EMA orphan drug designation, cross-border rare disease networks, and expanding genetic diagnostic capabilities.
- EMA orphan designation provides regulatory incentives.
- European Reference Networks improve rare disease care coordination.
- Genetic testing accessibility improves diagnostic identification.
In-depth Analysis of Andersen-Tawil Syndrome Treatment Market in UK
The UK is expected to grow at 7.6% through 2036, reflecting NHS rare disease programs, expanding genetic testing through Genomics England, and specialist cardiac care infrastructure.
- NHS rare disease programs support structured treatment access.
- Genomics England improves rare channelopathy identification.
- Specialist cardiac genetics centers deliver concentrated expertise.
Sales Analysis of Andersen-Tawil Syndrome Treatment Market in Japan
Japan is expected to grow at 7.4% through 2036, driven by dedicated rare disease healthcare programs, advanced genetic testing infrastructure, and strong pharmaceutical reimbursement frameworks.
- Rare disease programs support treatment access and reimbursement.
- Advanced genetic testing capabilities improve diagnostic rates.
- Pharmaceutical reimbursement frameworks incentivize treatment provision.
Competitive Landscape and Strategic Positioning
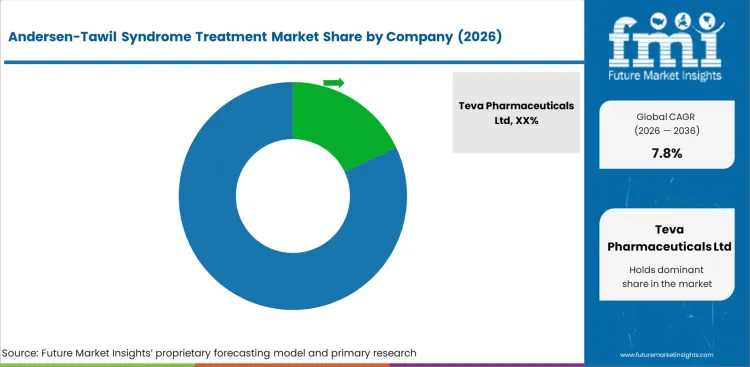
- Teva Pharmaceuticals Ltd leads with an estimated 18.0% market share, leveraging its generic pharmaceutical portfolio to serve rare cardiac conditions.
- Novartis AG and Pfizer Inc. hold positions through broad cardiovascular and rare disease pharmaceutical portfolios.
- Generic pharmaceutical manufacturers dominate the market given the reliance on repurposed medications from broader cardiac indications.
Teva Pharmaceuticals Ltd maintains market leadership through its generic pharmaceutical portfolio, which includes beta-blockers, carbonic anhydrase inhibitors, and antiarrhythmic agents commonly prescribed for Andersen-Tawil syndrome management. The company's global distribution network and cost-competitive pricing support market access.
Novartis AG and Pfizer Inc. participate through branded and generic cardiovascular pharmaceutical portfolios that include treatments used in Andersen-Tawil management. Both companies maintain global regulatory and distribution capabilities.
Sun Pharmaceuticals, Zydus Pharmaceuticals, and Aurobindo Pharma serve the market through cost-effective generic formulations of beta-blockers and antiarrhythmic agents, particularly in price-sensitive markets.
The competitive landscape is shaped by generic pharmaceutical competition, with limited branded product differentiation. Future competitive dynamics may shift as gene-targeted and personalized medicine approaches progress through clinical development.
Key Companies in the Andersen-Tawil Syndrome Treatment Market
Key global companies leading the andersen-tawil syndrome treatment market include:
- Teva Pharmaceuticals Ltd (Israel), Novartis AG (Switzerland), Pfizer Inc. (USA), and Mylan N.V. (USA) maintain strong positions through broad cardiovascular pharmaceutical portfolios.
- Sun Pharmaceuticals Industries Ltd. (India), Zydus Pharmaceuticals (India), and Aurobindo Pharma (India) hold positions through cost-effective generic formulations.
- Advanz Pharmaceuticals (UK), Dr. Reddy's Laboratories (India), and Viatris Inc. (USA) represent additional competition through targeted rare disease and generic drug portfolios.
Competitive Benchmarking: Andersen-Tawil Syndrome Treatment Market
| Company | Cardiac Drug Range | Rare Disease Focus | Distribution Reach | Regulatory Portfolio |
|---|---|---|---|---|
| Teva Pharmaceuticals | Broad | Low | Global | Global |
| Novartis AG | Broad | Moderate | Global | Global |
| Pfizer Inc. | Broad | Moderate | Global | Global |
| Mylan N.V. | Broad | Low | Global | Global |
| Sun Pharmaceuticals | Moderate | Low | Global | Global |
| Zydus Pharmaceuticals | Moderate | Low | Regional | Regional |
| Aurobindo Pharma | Moderate | Low | Global | Global |
| Advanz Pharmaceuticals | Narrow | Moderate | Regional | Europe |
| Dr. Reddy's Laboratories | Moderate | Low | Global | Global |
| Viatris Inc. | Broad | Low | Global | Global |
Source: Future Market Insights competitive analysis, 2026.
Key Developments in Andersen-Tawil Syndrome Treatment Market
- In 2025, Novartis AG initiated a clinical evaluation of a targeted potassium channel modulator for Andersen-Tawil syndrome patients in a Phase 2 trial across European centers.
- In 2025, Teva Pharmaceuticals expanded its generic acetazolamide distribution to additional rare disease pharmacy networks in North America and Europe.
Key Players in the Andersen-Tawil Syndrome Treatment Market
Major Global Players
- Teva Pharmaceuticals Ltd
- Novartis AG
- Pfizer Inc.
- Mylan N.V.
- Sun Pharmaceuticals Industries Ltd.
- Zydus Pharmaceuticals, Inc.
- Aurobindo Pharma
Emerging Players/Startups
- Advanz Pharmaceuticals
- Dr. Reddy's Laboratories Ltd.
- Viatris Inc.
- Amneal Pharmaceuticals
- Cipla Ltd.
- Lupin Limited
- Hikma Pharmaceuticals
- Sandoz International
- Torrent Pharmaceuticals
Report Scope and Coverage
| Parameter | Details |
|---|---|
| Quantitative Units | USD 2.39 billion to USD 5.07 billion, at a CAGR of 7.8% |
| Market Definition | The Andersen-Tawil syndrome treatment market encompasses pharmaceutical products for managing this rare genetic channelopathy. |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia and Pacific, Middle East and Africa |
| Countries Covered | USA, South Korea, EU, UK, Japan, 30 plus countries |
| Key Companies Profiled | Teva Pharmaceuticals, Novartis AG, Pfizer Inc., Mylan N.V., Sun Pharmaceuticals, Zydus Pharmaceuticals, Aurobindo Pharma, Advanz Pharmaceuticals |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid bottom-up and top-down methodology starting with verified transaction data, projecting adoption velocity across segments and regions. |
Market Segmentation Analysis
Andersen-Tawil Syndrome Treatment Market Market Segmented by Disease Type:
- Type 2
- Type 1
Andersen-Tawil Syndrome Treatment Market Market Segmented by Drug Class:
- Beta-Blockers
- Carbonic Anhydrate Inhibitors
- Acetazolamide
- Dichloropenamide
- Antiarrhythmic Drugs
- Amiodarone
- Flecainide
- Atenolol
Andersen-Tawil Syndrome Treatment Market Market Segmented by Distribution Channel:
- Hospital Pharmacy
- Retail Pharmacy
- Online Pharmacy
Andersen-Tawil Syndrome Treatment Market Market Segmented by End Use:
- Hospitals
- Specialty Clinics
- Home Care
Andersen-Tawil Syndrome Treatment Market Market by Region:
- North America
- USA
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Western Europe
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- Eastern Europe
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- East Asia
- China
- Japan
- South Korea
- South Asia and Pacific
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
Research Sources and Bibliography
- 1. World Health Organization. (2025). WHO Rare Disease Global Health Estimates. WHO.
- 2. National Institutes of Health. (2024). NIH Genetic and Rare Diseases Information Center: Andersen-Tawil Syndrome. NIH.
- 3. European Medicines Agency. (2025). EMA Orphan Drug Designation Annual Report. EMA.
- 4. USA Food and Drug Administration. (2025). FDA Orphan Drug Designations Database. FDA.
- 5. National Organization for Rare Disorders. (2024). NORD Rare Disease Database. NORD.
This bibliography is provided for reader reference.
This Report Answers
- Estimating the size of the market and revenue projections from 2026 to 2036.
- Segmentation by disease type, drug class, distribution channel, and end use.
- Regional and country-level demand analysis across key markets.
- Competitive landscape assessment and company benchmarking.
- Analysis of genetic diagnostic trends and their impact on patient identification.
- Regulatory landscape for orphan drug designation across major jurisdictions.
- Identification of growth opportunities in gene-targeted therapy development.
- Supply chain and pharmaceutical distribution dynamics.
Frequently Asked Questions
What is the global market size for Andersen-Tawil Syndrome Treatment in 2026?
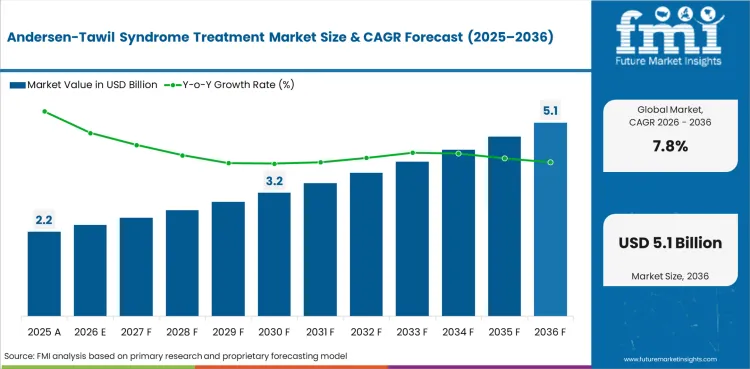
In 2026, the global Andersen-Tawil Syndrome Treatment Market is expected to be valued at USD 2.39 billion.
How large will the Andersen-Tawil Syndrome Treatment Market be in 2036?
By 2036, the Andersen-Tawil Syndrome Treatment Market is expected to reach USD 5.07 billion.
What is the projected CAGR from 2026 to 2036?
The Andersen-Tawil Syndrome Treatment Market is projected to expand at a CAGR of 7.8% from 2026 to 2036.
Which disease type is expected to lead in 2026?
Type 2 is expected to account for 62.5% of the disease type segment in 2026, reflecting higher diagnostic prevalence and established treatment protocols.
What is driving demand in the USA?
The USA is expected to grow at 8.2% through 2036, driven by advanced genetic diagnostic infrastructure, Orphan Drug Act incentives, and strong reimbursement frameworks.
What is driving demand in South Korea?
South Korea is expected to grow at 8.0% through 2036, supported by expanding genetic testing programs and government rare disease management support.
What does this report mean by 'Andersen-Tawil Syndrome Treatment Market'?
The Andersen-Tawil Syndrome Treatment Market includes pharmaceutical products and therapeutic interventions for managing Andersen-Tawil syndrome, a rare genetic channelopathy.
How does FMI develop and validate the forecast?
Forecasting uses a hybrid bottom-up and top-down approach, starting with verified pharmaceutical transaction data and projecting adoption across clinical segments and regions.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Disease Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Disease Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Disease Type , 2026 to 2036
- Type 2
- Type 1
- Type 2
- Y to o to Y Growth Trend Analysis By Disease Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Disease Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Drug Class
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Drug Class, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Drug Class, 2026 to 2036
- Beta-Blockers
- Carbonic Anhydrate Inhibitors
- Acetazolamide
- Dichloropenamide
- Antiarrhythmic Drugs
- Amiodarone
- Flecainide
- Atenolol
- Beta-Blockers
- Y to o to Y Growth Trend Analysis By Drug Class, 2021 to 2025
- Absolute $ Opportunity Analysis By Drug Class, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Distribution Channel
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Distribution Channel, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Distribution Channel, 2026 to 2036
- Hospital Pharmacy
- Retail Pharmacy
- Online Pharmacy
- Hospital Pharmacy
- Y to o to Y Growth Trend Analysis By Distribution Channel, 2021 to 2025
- Absolute $ Opportunity Analysis By Distribution Channel, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End Use
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End Use, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End Use, 2026 to 2036
- Hospitals
- Specialty Clinics
- Home Care
- Hospitals
- Y to o to Y Growth Trend Analysis By End Use, 2021 to 2025
- Absolute $ Opportunity Analysis By End Use, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Disease Type
- By Drug Class
- By Distribution Channel
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Disease Type
- By Drug Class
- By Distribution Channel
- By End Use
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Disease Type
- By Drug Class
- By Distribution Channel
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Disease Type
- By Drug Class
- By Distribution Channel
- By End Use
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Disease Type
- By Drug Class
- By Distribution Channel
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Disease Type
- By Drug Class
- By Distribution Channel
- By End Use
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Disease Type
- By Drug Class
- By Distribution Channel
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Disease Type
- By Drug Class
- By Distribution Channel
- By End Use
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Disease Type
- By Drug Class
- By Distribution Channel
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Disease Type
- By Drug Class
- By Distribution Channel
- By End Use
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Disease Type
- By Drug Class
- By Distribution Channel
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Disease Type
- By Drug Class
- By Distribution Channel
- By End Use
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Disease Type
- By Drug Class
- By Distribution Channel
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Disease Type
- By Drug Class
- By Distribution Channel
- By End Use
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Disease Type
- By Drug Class
- By Distribution Channel
- By End Use
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Disease Type
- By Drug Class
- By Distribution Channel
- By End Use
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Disease Type
- By Drug Class
- By Distribution Channel
- By End Use
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Disease Type
- By Drug Class
- By Distribution Channel
- By End Use
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Disease Type
- By Drug Class
- By Distribution Channel
- By End Use
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Disease Type
- By Drug Class
- By Distribution Channel
- By End Use
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Disease Type
- By Drug Class
- By Distribution Channel
- By End Use
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Disease Type
- By Drug Class
- By Distribution Channel
- By End Use
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Disease Type
- By Drug Class
- By Distribution Channel
- By End Use
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Disease Type
- By Drug Class
- By Distribution Channel
- By End Use
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Disease Type
- By Drug Class
- By Distribution Channel
- By End Use
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Disease Type
- By Drug Class
- By Distribution Channel
- By End Use
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Disease Type
- By Drug Class
- By Distribution Channel
- By End Use
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Disease Type
- By Drug Class
- By Distribution Channel
- By End Use
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Disease Type
- By Drug Class
- By Distribution Channel
- By End Use
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Disease Type
- By Drug Class
- By Distribution Channel
- By End Use
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Disease Type
- By Drug Class
- By Distribution Channel
- By End Use
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Disease Type
- By Drug Class
- By Distribution Channel
- By End Use
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Disease Type
- By Drug Class
- By Distribution Channel
- By End Use
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Disease Type
- By Drug Class
- By Distribution Channel
- By End Use
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Disease Type
- By Drug Class
- By Distribution Channel
- By End Use
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Disease Type
- By Drug Class
- By Distribution Channel
- By End Use
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Disease Type
- By Drug Class
- By Distribution Channel
- By End Use
- Competition Analysis
- Competition Deep Dive
- Teva Pharmaceuticals Ltd
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Zydus Pharmaceuticals, Inc.
- Sun Pharmaceuticals Industries Ltd.
- Advanz Pharmaceuticals
- Novartis AG
- Mylan N.V.
- Aurobindo Pharma
- Dr. Reddy’s Laboratories Ltd.
- Viatris Inc.
- Pfizer Inc.
- Teva Pharmaceuticals Ltd
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Disease Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Drug Class, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Disease Type , 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Drug Class, 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by Disease Type , 2021 to 2036
- Table 13: Latin America Market Value (USD Million) Forecast by Drug Class, 2021 to 2036
- Table 14: Latin America Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: Western Europe Market Value (USD Million) Forecast by Disease Type , 2021 to 2036
- Table 18: Western Europe Market Value (USD Million) Forecast by Drug Class, 2021 to 2036
- Table 19: Western Europe Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 20: Western Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 21: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: Eastern Europe Market Value (USD Million) Forecast by Disease Type , 2021 to 2036
- Table 23: Eastern Europe Market Value (USD Million) Forecast by Drug Class, 2021 to 2036
- Table 24: Eastern Europe Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 25: Eastern Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 26: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 27: East Asia Market Value (USD Million) Forecast by Disease Type , 2021 to 2036
- Table 28: East Asia Market Value (USD Million) Forecast by Drug Class, 2021 to 2036
- Table 29: East Asia Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 30: East Asia Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 31: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 32: South Asia and Pacific Market Value (USD Million) Forecast by Disease Type , 2021 to 2036
- Table 33: South Asia and Pacific Market Value (USD Million) Forecast by Drug Class, 2021 to 2036
- Table 34: South Asia and Pacific Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 35: South Asia and Pacific Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 36: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 37: Middle East & Africa Market Value (USD Million) Forecast by Disease Type , 2021 to 2036
- Table 38: Middle East & Africa Market Value (USD Million) Forecast by Drug Class, 2021 to 2036
- Table 39: Middle East & Africa Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 40: Middle East & Africa Market Value (USD Million) Forecast by End Use, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Disease Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Disease Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Disease Type
- Figure 6: Global Market Value Share and BPS Analysis by Drug Class, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Drug Class, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Drug Class
- Figure 9: Global Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Distribution Channel
- Figure 12: Global Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by End Use
- Figure 15: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 17: Global Market Attractiveness Analysis by Region
- Figure 18: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 20: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 21: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 23: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 24: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 25: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 26: North America Market Value Share and BPS Analysis by Disease Type , 2026 and 2036
- Figure 27: North America Market Y-o-Y Growth Comparison by Disease Type , 2026-2036
- Figure 28: North America Market Attractiveness Analysis by Disease Type
- Figure 29: North America Market Value Share and BPS Analysis by Drug Class, 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Drug Class, 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Drug Class
- Figure 32: North America Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 34: North America Market Attractiveness Analysis by Distribution Channel
- Figure 35: North America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 37: North America Market Attractiveness Analysis by End Use
- Figure 38: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 39: Latin America Market Value Share and BPS Analysis by Disease Type , 2026 and 2036
- Figure 40: Latin America Market Y-o-Y Growth Comparison by Disease Type , 2026-2036
- Figure 41: Latin America Market Attractiveness Analysis by Disease Type
- Figure 42: Latin America Market Value Share and BPS Analysis by Drug Class, 2026 and 2036
- Figure 43: Latin America Market Y-o-Y Growth Comparison by Drug Class, 2026-2036
- Figure 44: Latin America Market Attractiveness Analysis by Drug Class
- Figure 45: Latin America Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 46: Latin America Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 47: Latin America Market Attractiveness Analysis by Distribution Channel
- Figure 48: Latin America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 49: Latin America Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 50: Latin America Market Attractiveness Analysis by End Use
- Figure 51: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 52: Western Europe Market Value Share and BPS Analysis by Disease Type , 2026 and 2036
- Figure 53: Western Europe Market Y-o-Y Growth Comparison by Disease Type , 2026-2036
- Figure 54: Western Europe Market Attractiveness Analysis by Disease Type
- Figure 55: Western Europe Market Value Share and BPS Analysis by Drug Class, 2026 and 2036
- Figure 56: Western Europe Market Y-o-Y Growth Comparison by Drug Class, 2026-2036
- Figure 57: Western Europe Market Attractiveness Analysis by Drug Class
- Figure 58: Western Europe Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 59: Western Europe Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 60: Western Europe Market Attractiveness Analysis by Distribution Channel
- Figure 61: Western Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 62: Western Europe Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 63: Western Europe Market Attractiveness Analysis by End Use
- Figure 64: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 65: Eastern Europe Market Value Share and BPS Analysis by Disease Type , 2026 and 2036
- Figure 66: Eastern Europe Market Y-o-Y Growth Comparison by Disease Type , 2026-2036
- Figure 67: Eastern Europe Market Attractiveness Analysis by Disease Type
- Figure 68: Eastern Europe Market Value Share and BPS Analysis by Drug Class, 2026 and 2036
- Figure 69: Eastern Europe Market Y-o-Y Growth Comparison by Drug Class, 2026-2036
- Figure 70: Eastern Europe Market Attractiveness Analysis by Drug Class
- Figure 71: Eastern Europe Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 72: Eastern Europe Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 73: Eastern Europe Market Attractiveness Analysis by Distribution Channel
- Figure 74: Eastern Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 75: Eastern Europe Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 76: Eastern Europe Market Attractiveness Analysis by End Use
- Figure 77: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 78: East Asia Market Value Share and BPS Analysis by Disease Type , 2026 and 2036
- Figure 79: East Asia Market Y-o-Y Growth Comparison by Disease Type , 2026-2036
- Figure 80: East Asia Market Attractiveness Analysis by Disease Type
- Figure 81: East Asia Market Value Share and BPS Analysis by Drug Class, 2026 and 2036
- Figure 82: East Asia Market Y-o-Y Growth Comparison by Drug Class, 2026-2036
- Figure 83: East Asia Market Attractiveness Analysis by Drug Class
- Figure 84: East Asia Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 85: East Asia Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 86: East Asia Market Attractiveness Analysis by Distribution Channel
- Figure 87: East Asia Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 88: East Asia Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 89: East Asia Market Attractiveness Analysis by End Use
- Figure 90: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 91: South Asia and Pacific Market Value Share and BPS Analysis by Disease Type , 2026 and 2036
- Figure 92: South Asia and Pacific Market Y-o-Y Growth Comparison by Disease Type , 2026-2036
- Figure 93: South Asia and Pacific Market Attractiveness Analysis by Disease Type
- Figure 94: South Asia and Pacific Market Value Share and BPS Analysis by Drug Class, 2026 and 2036
- Figure 95: South Asia and Pacific Market Y-o-Y Growth Comparison by Drug Class, 2026-2036
- Figure 96: South Asia and Pacific Market Attractiveness Analysis by Drug Class
- Figure 97: South Asia and Pacific Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 98: South Asia and Pacific Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 99: South Asia and Pacific Market Attractiveness Analysis by Distribution Channel
- Figure 100: South Asia and Pacific Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 101: South Asia and Pacific Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 102: South Asia and Pacific Market Attractiveness Analysis by End Use
- Figure 103: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 104: Middle East & Africa Market Value Share and BPS Analysis by Disease Type , 2026 and 2036
- Figure 105: Middle East & Africa Market Y-o-Y Growth Comparison by Disease Type , 2026-2036
- Figure 106: Middle East & Africa Market Attractiveness Analysis by Disease Type
- Figure 107: Middle East & Africa Market Value Share and BPS Analysis by Drug Class, 2026 and 2036
- Figure 108: Middle East & Africa Market Y-o-Y Growth Comparison by Drug Class, 2026-2036
- Figure 109: Middle East & Africa Market Attractiveness Analysis by Drug Class
- Figure 110: Middle East & Africa Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 111: Middle East & Africa Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 112: Middle East & Africa Market Attractiveness Analysis by Distribution Channel
- Figure 113: Middle East & Africa Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 114: Middle East & Africa Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 115: Middle East & Africa Market Attractiveness Analysis by End Use
- Figure 116: Global Market - Tier Structure Analysis
- Figure 117: Global Market - Company Share Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

