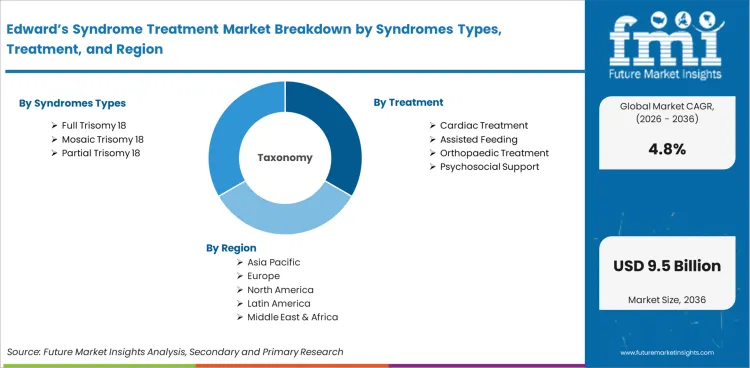
Edward’s Syndrome Treatment Market is segmented by Syndromes Types (Full Trisomy 18, Mosaic Trisomy 18, Partial Trisomy 18), Treatment (Cardiac Treatment, Assisted Feeding, Orthopaedic Treatment, Psychosocial Support), End User (Hospitals, Clinics, Ambulatory Surgical Centres), and Region. Forecast for 2026 to 2036.
The Edward's syndrome treatment market covers pharmaceutical, surgical, and supportive interventions used in managing trisomy 18 (Edward's syndrome), a chromosomal condition affecting multiple organ systems. Treatments include cardiac surgery, respiratory support, nutritional management, and palliative care distributed through hospital and specialty care channels.
Market scope covers all commercially available Edward's syndrome treatments categorized by syndrome type (full trisomy 18, mosaic trisomy 18, partial trisomy 18), treatment (cardiac treatment, respiratory support, nutritional management, palliative care, others), end user (hospitals, specialty centres, homecare), and patient age group (infants, children, adults). Revenue coverage spans 2026 to 2036.
The scope does not include prenatal screening and diagnostic testing services, genetic counseling, or general neonatal intensive care not specifically related to trisomy 18 management.
Demand for Edward's syndrome treatment reflects the evolving clinical philosophy regarding active intervention for trisomy 18. Historically managed primarily through comfort care, an increasing number of medical centres are offering cardiac surgery and other active treatments to selected trisomy 18 infants, particularly those with mosaic or partial trisomy presentations. This philosophical shift is expanding treatment demand and per-patient healthcare expenditure.
Adoption of cardiac treatment leads at 34.8% of treatment demand, reflecting the high prevalence of congenital heart defects in trisomy 18 patients. Ventricular septal defects, atrial septal defects, and patent ductus arteriosus are common cardiac findings that, when surgically addressed, can significantly extend survival in appropriately selected patients.
Pricing reflects the intensive hospital-based care required for trisomy 18 management, including NICU stays, cardiac surgical procedures, and ongoing supportive care. The per-patient cost of active treatment significantly exceeds comfort care approaches, creating healthcare resource allocation considerations that vary across institutional and national policies.
The Edward's syndrome treatment market is segmented by syndrome type, treatment, end user, and patient age group. Demand patterns reflect the evolving clinical approach to trisomy 18 management, the concentration of care in hospital settings, and the age-dependent treatment requirements across the limited but growing survival population.
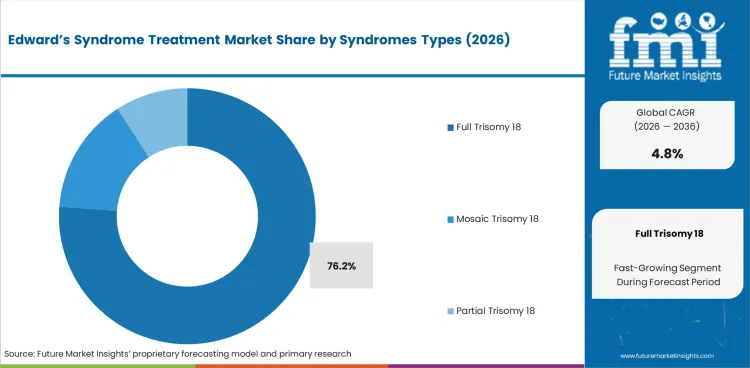
Full trisomy 18 represents 76.2% of syndrome type demand, reflecting the predominance of complete chromosomal trisomy in the diagnosed population. Full trisomy 18 presents with the most severe clinical features and historically the poorest prognosis, though evolving treatment approaches are extending survival for selected patients.
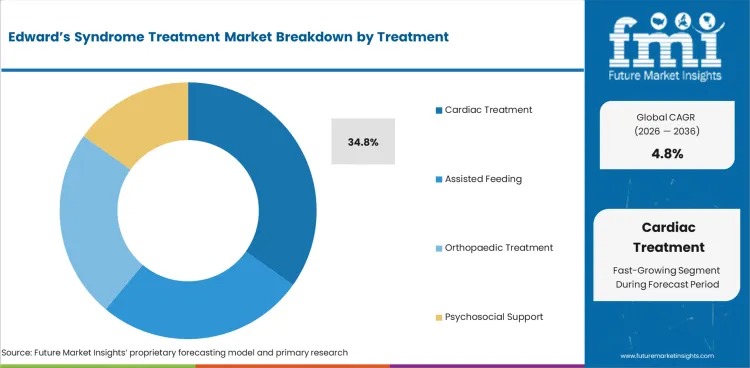
Cardiac treatment leads at 34.8% due to the high prevalence of congenital heart defects in trisomy 18 patients. Surgical correction of cardiac anomalies represents the treatment intervention with the most significant impact on extending survival beyond the neonatal period.
The edward’s syndrome treatment market is shaped by regulatory, technology, and procurement dynamics.
Demand is shaped by the evolving clinical approach that offers active surgical and medical intervention to selected trisomy 18 patients, expanding treatment beyond the historical comfort-care-only approach.
Growth reflects improving prenatal screening accuracy through cell-free DNA testing that increases confirmed trisomy 18 identification and supports earlier perinatal care planning.
Adoption of active treatment is increasing as clinical experience demonstrates variable outcomes particularly in mosaic and partial trisomy 18 presentations where survival potential is greater.
The intensive resource requirements for active trisomy 18 treatment create institutional and policy-level allocation decisions that constrain treatment adoption in resource-limited settings.
.webp)
| Country | CAGR |
|---|---|
| USA | 4.9% |
| South Korea | 4.9% |
| EU | 4.8% |
| Japan | 4.8% |
| UK | 4.7% |
The global edward’s syndrome treatment market is expected to grow at 4.8% per year from 2026 to 2036.
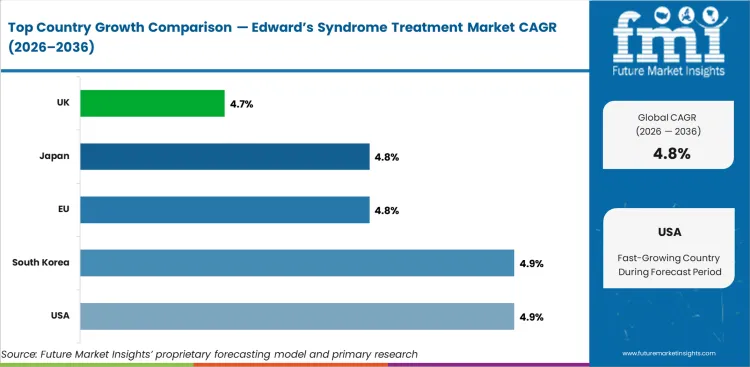
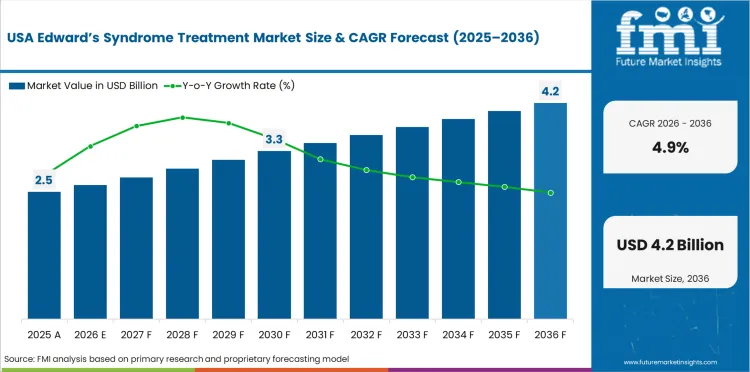
The USA is expected to grow at 4.9% through 2036, driven by children's hospital cardiac surgery programmes, evolving treatment philosophy, and comprehensive insurance coverage for neonatal intensive care.
South Korea is expected to grow at 4.9% through 2036, supported by advanced neonatal care infrastructure, prenatal diagnostic capabilities, and growing adoption of active treatment approaches.
Opportunity Analysis of Edward's Syndrome Treatment Market in the European Union
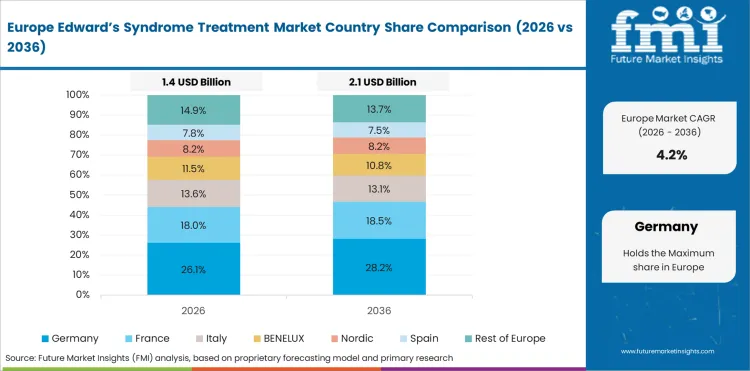
The EU is expected to grow at 4.7% through 2036, supported by cross-border paediatric referral networks, advanced prenatal screening programmes, and evolving clinical guidelines.
Japan is expected to grow at 4.6% through 2036, driven by advanced neonatal intensive care, paediatric cardiac surgery expertise, and comprehensive healthcare coverage.
The UK is expected to grow at 4.5% through 2036, supported by NHS paediatric services, evolving clinical approaches, and specialist children's hospital networks.
Abbott Laboratories leads through its diagnostic and cardiac device portfolio supporting prenatal detection and surgical management of congenital heart defects in trisomy 18 patients.
F. Hoffmann-La Roche Ltd. and PerkinElmer, Inc. maintain positions through prenatal screening platforms that enable early trisomy 18 identification and care planning.
Quest Diagnostics Incorporated and Illumina, Inc. compete on genetic testing and genomic sequencing technologies supporting trisomy 18 diagnosis and prognosis assessment.
Entry barriers include the specialized neonatal and paediatric cardiac expertise required, the small patient population, and the diagnostic technology investment needed for accurate prenatal screening.
Key Companies in the Edward’s Syndrome Treatment Market
| Company | Diagnostic Portfolio | Cardiac Devices | Screening Technology | Geographic Reach |
|---|---|---|---|---|
| Abbott Laboratories | High | High | High | Global |
| F. Hoffmann-La Roche Ltd. | High | Low | High | Global |
| PerkinElmer, Inc. | Medium | Low | High | Global |
| Quest Diagnostics | Medium | Low | High | N. America |
| Illumina, Inc. | Medium | Low | High | Global |
| Natera, Inc. | Low | Low | High | N. America |
Source: Future Market Insights competitive analysis, 2026.
Key Developments in Edward’s Syndrome Treatment Market
Major Global Players
Emerging Players/Startups
| Parameter | Details |
|---|---|
| Quantitative Units | USD 5.93 billion to USD 9.48 billion, at a CAGR of 4.8% |
| Market Definition | The Edward's syndrome treatment market covers pharmaceutical, surgical, and supportive interventions used in managing trisomy 18 (Edward's syndrome), a chromosomal condition affecting multiple organ systems. Treatments include cardiac surgery, respiratory support, nutritional management, and palliative care distributed through hospital and specialty care channels. |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia and Pacific, Middle East and Africa |
| Countries Covered | USA, UK, France, Germany, Italy, South Korea, Japan, China, India, 30 plus countries |
| Key Companies Profiled | Abbott Laboratories, F. Hoffmann-La Roche Ltd., PerkinElmer, Inc., Quest Diagnostics Incorporated, Illumina, Inc., GE Healthcare, Natera, Inc., Thermo Fisher Scientific Inc., Agilent Technologies, Inc., Bio-Rad Laboratories, Inc. |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid bottom-up and top-down methodology starting with verified transaction data, projecting adoption velocity across segments and regions. |
This bibliography is provided for reader reference. The full Future Market Insights report contains the complete reference list with publication dates, URLs, and supporting data for all cited works.
What is the global market demand for Edward’s Syndrome Treatment in 2026?
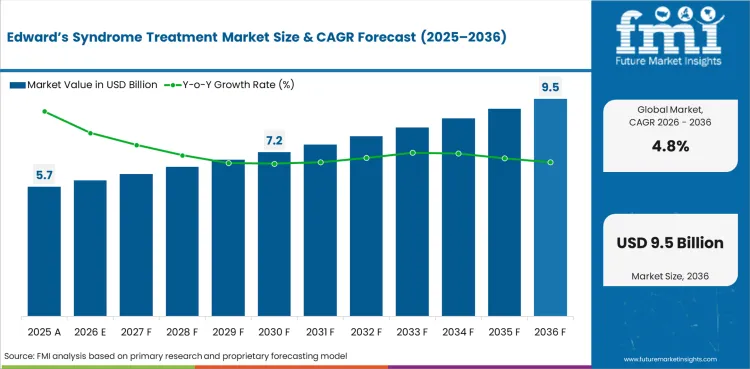
In 2026, the global market is expected to be worth USD 5.93 billion.
How big will the Edward’s Syndrome Treatment Market be in 2036?
By 2036, the market is expected to be worth USD 9.48 billion.
How much is demand expected to grow between 2026 and 2036?
Between 2026 and 2036, demand is expected to grow at a CAGR of 4.8%.
Which Syndromes Types segment is expected to lead in 2026?
Full Trisomy 18 is expected to account for 76.2% of the segment in 2026.
What is causing demand to rise in USA?
USA is expected to grow at 4.9% through 2036.
What is causing demand to rise in South Korea?
South Korea is expected to grow at 4.9% through 2036.
What does this report mean by Edward’s Syndrome Treatment Market definition?
The Edward's syndrome treatment market covers pharmaceutical, surgical, and supportive interventions used in managing trisomy 18 (Edward's syndrome), a chromosomal condition affecting multiple organ s.
How does FMI make the forecast?
Forecasting uses a hybrid bottom-up and top-down methodology starting with verified transaction data, projecting adoption velocity across segments and regions.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.