Pre-treatment Cleaning Market : Global Industry Analysis 2016 - 2025 and Opportunity Assessment 2026 - 2036
Pre-treatment Cleaning Market Size and Share Forecast Outlook 2026 to 2036
Historical Data Covered: 2016 - 2025 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2026 - 2036
Pre-treatment Cleaning Market Forecast and Outlook 2026 to 2036
The pre-treatment cleaning market is forecasted to reach USD 2,255.4 million in 2026 and expand to USD 4,605.4 million by 2036, reflecting a 7.4% CAGR over the forecast period. Value creation is closely linked to its role as a prerequisite step in coating, plating, sterilization, and precision manufacturing workflows. Demand is reinforced by rising quality thresholds in automotive, medical device, electronics, and industrial components, where surface contamination directly affects downstream yield and product durability. Revenue stability is supported by recurring chemical consumption, bath maintenance cycles, and replacement demand rather than discrete capital investment waves.
Demand formation is governed by surface performance sensitivity, regulatory cleanliness thresholds, and integration with automated production lines. Cycle standardization is accelerating investment in endoscope reprocessing device platforms that reduce manual handling steps. Utilization intensifies where adhesion failure, corrosion risk, or sterility compromise create high downstream cost exposure. Environmental compliance requirements influence chemistry selection, wastewater treatment integration, and process validation intensity. Adoption remains moderated by validation complexity, chemistry management overhead, and cost-performance tradeoffs in mixed-material production environments. Market expansion remains process-driven, supported by quality assurance priorities and regulatory discipline, rather than broad-based industrial capacity additions.
Quick Stats for Pre-treatment Cleaning Market
- Pre-treatment Cleaning Market Value (2026): USD 2,255.4 million
- Pre-treatment Cleaning Market Forecast Value (2036): USD 4,605.4 million
- Pre-treatment Cleaning Market Forecast CAGR 2026 to 2036: 7.4%
- Leading Cleaning Solution by Demand Share: Enzymatic detergents
- Fastest-Growing Countries: India, Brazil, China, USA, Germany
- Top Players in Global Demand: STERIS, Getinge, Ecolab, ASP, Cantel
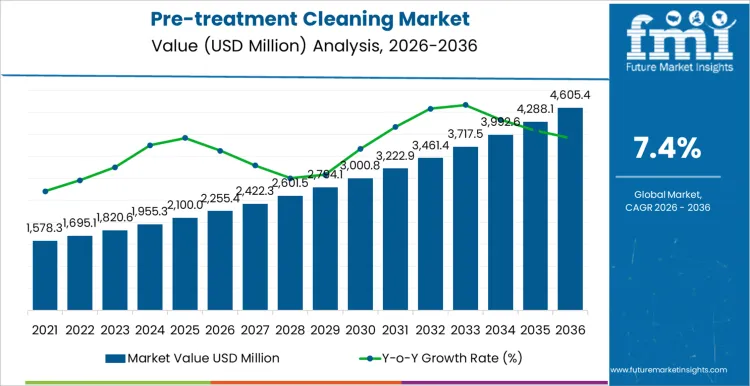
Pre-treatment Cleaning Market Key Takeaways
| Metric | Value |
|---|---|
| Market Value (2026) | USD 2,255.4 million |
| Market Forecast Value (2036) | USD 4,605.4 million |
| Forecast CAGR 2026 to 2036 | 7.4% |
Why is the Demand for Pre-treatment Cleaning Growing?
Demand for pre-treatment cleaning solutions is rising as manufacturers and process engineers emphasize surface integrity prior to coatings, plating, and bonding operations that determine final product quality and longevity. Pre-treatment cleaning removes oils, particulates, oxides, and process residues that can disrupt adhesion, accelerate corrosion, or cause coating defects in sectors such as automotive, aerospace, appliances, and industrial equipment. Facilities often rationalize spend under broader industrial cleaners budgets while keeping pretreatment specs locked to coating validation needs. Engineers specify chemistries and systems tailored to substrate materials and downstream processes because cleaning efficacy directly impacts surface energy, wetting behavior, and coating uniformity in high-performance finished goods. Procurement teams evaluate product compatibility with existing pretreatment lines, waste handling infrastructure, and environmental discharge limits to ensure solutions meet operational requirements and regulatory expectations. Performance criteria such as consistent contaminant removal, minimal surface etching, and stable formulation during extended production runs are critical for facilities operating at high throughput and tight quality tolerances.
Growth in advanced surface finishing technologies and adoption of lightweight materials reinforces uptake of targeted pre-treatment cleaning as an essential step in integrated production workflows. Manufacturers and integrators coordinate cleaning selection with rinse staging, drying protocols, and coating application methods to reduce rework, improve first-pass yield, and extend service life of coated components. Environmental compliance drivers around effluent quality and worker safety encourage transition to low-emission and low-toxicity cleaning chemistries that align with permitting requirements without sacrificing performance. Many plants are shifting legacy solvents to low-VOC degreasers to reduce exposure limits while keeping throughput stable. Training for operations staff on monitoring cleaning bath condition and waste minimization supports cost control and consistent output across shifts and product variants. These application-specific quality, process integration, and regulatory considerations are contributing to sustained demand growth in the pre-treatment cleaning market.
How Is the Pre-treatment Cleaning Industry Segmented by Cleaning Solutions, Application Areas, and End Users?
Demand for pre-treatment cleaning is driven by infection control standards, complexity of reusable instruments, and regulatory expectations before disinfection or sterilization. Healthcare facilities and life science operators prioritize effective soil removal to protect downstream sterilization efficacy. Usage patterns reflect rising procedural volumes, increased endoscope utilization, and stricter audit requirements. Product selection focuses on cleaning efficacy, material compatibility, and workflow integration within reprocessing cycles. Purchasing teams increasingly evaluate endoscope detergents and disinfectants as a combined protocol decision to reduce mismatched chemistry across steps. Segmentation highlights how formulation type, use environment, and operational responsibility influence adoption across healthcare and industrial cleaning pathways.
Which Cleaning Solution Category Accounts for the Largest Share in Pre-treatment Cleaning?
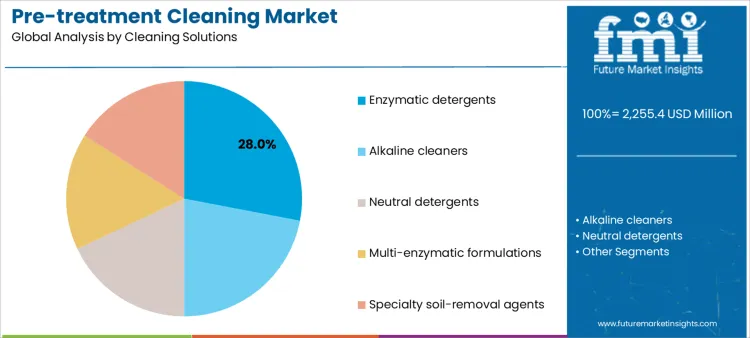
Enzymatic detergents hold 28.0%, reflecting widespread use for breaking down protein, lipid, and carbohydrate soils on complex instruments. Procurement teams often benchmark performance claims against the broader enzymatic cleaning segment when selecting detergents for complex soil loads. These formulations are preferred for lumened devices and delicate materials requiring low-temperature cleaning. Alkaline cleaners at 22.0% address heavy organic and inorganic residues in robust instruments. Neutral detergents at 18.0% support routine cleaning where material sensitivity is critical. Multi-enzymatic formulations at 16.0% offer broader soil coverage within single-step processes. Specialty soil-removal agents at 16.0% address niche contamination challenges. Solution segmentation indicates preference for enzymatic efficacy balanced with material protection.
Key Points
- Enzymatic detergents dominate due to broad organic soil removal.
- Alkaline cleaners manage heavy contamination loads.
- Neutral and specialty solutions protect sensitive instruments.
How Do Application Areas Shape Demand for Pre-treatment Cleaning Solutions?
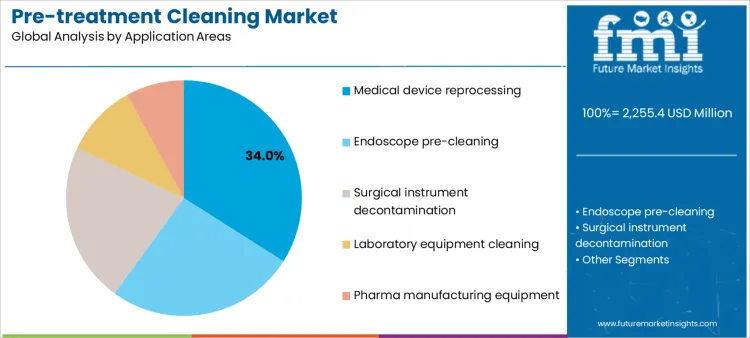
Medical device reprocessing represents 34.0%, driven by mandatory cleaning prior to high-level disinfection or sterilization. Endoscope pre-cleaning at 26.0% reflects immediate soil removal needs for complex internal channels. High channel complexity is increasing demand for validated endoscope reprocessing chemicals that maintain efficacy under variable water quality. Surgical instrument decontamination at 22.0% supports infection prevention across reusable surgical tools. Laboratory equipment cleaning at 10.0% focuses on residue control for analytical reliability. Pharma manufacturing equipment at 8.0% applies pre-treatment cleaning to maintain process hygiene. Application segmentation shows higher utilization where device complexity and contamination risk require validated pre-cleaning protocols.
Key Points
- Device reprocessing generates consistent cleaning demand.
- Endoscopes require rapid and thorough soil removal.
- Manufacturing environments apply targeted hygiene control.
Which End User Groups Drive Utilization of Pre-treatment Cleaning Solutions?
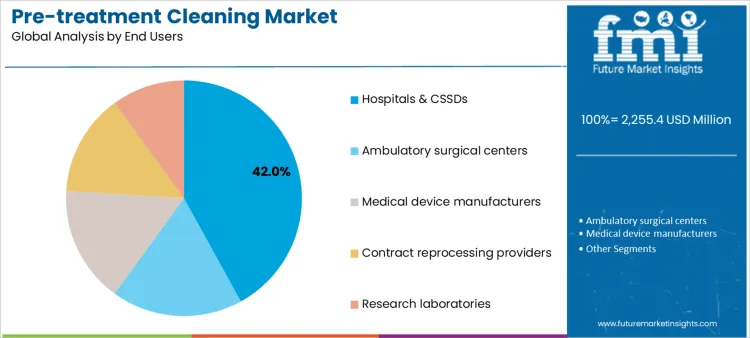
Hospitals and CSSDs account for 42.0%, reflecting centralized responsibility for instrument cleaning and infection prevention. Central sterile departments increasingly standardize instrument reprocessing detergents to reduce residue risk and audit variability. Ambulatory surgical centers at 18.0% emphasize efficient cleaning compatible with rapid case turnover. Medical device manufacturers at 16.0% integrate pre-treatment cleaning into production and validation workflows. Contract reprocessing providers at 14.0% manage cleaning across diverse client instrument sets. Research laboratories at 10.0% apply cleaning solutions within controlled experimental settings. End-user segmentation highlights concentration where compliance accountability, throughput, and process control define purchasing behavior.
Key Points
- Hospitals lead due to centralized reprocessing operations.
- ASCs prioritize speed and workflow efficiency.
- Manufacturers and service providers require validated cleaning.
What are the Key Dynamics in the Pre-treatment Cleaning Market?
Demand for pre-treatment cleaning reflects requirement to remove contaminants before downstream manufacturing, coating, sterilization, or analytical processes. Adoption concentrates in medical device manufacturing, pharmaceuticals, electronics, automotive components, metals processing, and surface finishing operations. Global scope aligns with quality assurance and process yield protection. Usage centers on aqueous, solvent-based, ultrasonic, and automated cleaning systems applied prior to coating, assembly, sterilization, or packaging stages.
How do downstream process sensitivity and quality compliance requirements shape adoption?
Surface contamination directly affects adhesion, sterility, electrical performance, and dimensional accuracy. Demand increases where residues such as oils, particulates, bio-burden, or machining fluids compromise subsequent processing steps. Medical device and pharmaceutical producers rely on validated pre-treatment cleaning to meet regulatory cleanliness thresholds before sterilization or packaging. Electronics manufacturers require precision cleaning to prevent ionic contamination that causes signal drift or failure. Metal finishing and coating operations depend on uniform surface preparation to ensure coating integrity and corrosion resistance. Cleaning process selection depends on substrate material, contaminant type, and allowable residues. Integration with automated lines reduces variability and rework. Adoption reflects operational focus on preventing downstream defects rather than correcting failures after value addition.
How do process validation burden, chemistry management, and cost exposure influence market scalability?
Pre-treatment cleaning effectiveness depends on tightly controlled parameters including chemistry concentration, temperature, and dwell time. Demand sensitivity rises where validation and documentation requirements increase operational overhead. Wastewater treatment and solvent recovery add environmental compliance cost. Chemistry compatibility limits flexibility across mixed-material production lines. Equipment footprint and energy consumption affect facility planning. Frequent bath monitoring and replacement increase labor intensity. Capital investment competes with outsourcing in low-volume production. Residue testing adds analytical cost. Scalability remains constrained by validation complexity, environmental management obligations, and cost-performance tradeoffs tied to increasingly stringent cleanliness expectations.
How Is Demand for Pre-treatment Cleaning Evolving Globally?
Demand for pre-treatment cleaning is expanding globally as manufacturing processes emphasize surface preparation, contamination control, and downstream process reliability. Metal finishing, coating, semiconductor, and medical device workflows depend on consistent cleaning outcomes before critical steps. Environmental regulations influence chemistry selection and wastewater management. Automation and inline systems increase throughput consistency. Many hospitals pair chemistry upgrades with new medical cleaning devices to improve repeatability and reduce manual rework. Growth rates in India at 9.9%, Brazil at 9.4%, China at 9.0%, the USA at 5.8%, and Germany at 5.7% indicate sustained expansion driven by industrial quality requirements, compliance pressures, and process efficiency improvements rather than capacity additions alone.
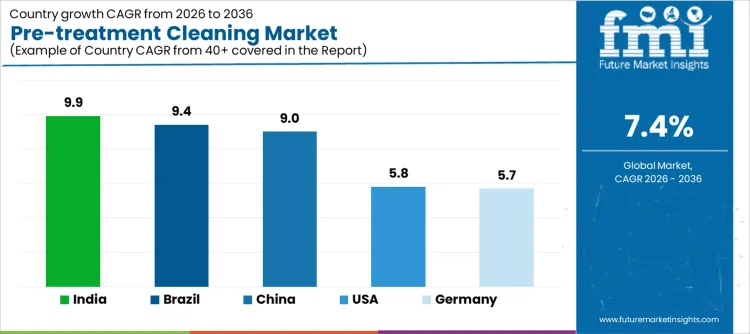
| Country | CAGR (%) |
|---|---|
| India | 9.9% |
| Brazil | 9.4% |
| China | 9.0% |
| USA | 5.8% |
| Germany | 5.7% |
What Is Driving Strong Growth of Pre-treatment Cleaning Demand in India?
Pre-treatment cleaning demand in India is growing at a CAGR of 9.9%, supported by expansion across automotive components, metal fabrication, and electronics assembly. Surface preparation requirements increase as manufacturers supply export-oriented customers with stricter quality specifications. Adoption of powder coating and advanced plating elevates reliance on consistent degreasing and rinsing stages. Regulatory scrutiny on effluent discharge accelerates transition toward optimized chemistries and closed-loop systems. Equipment suppliers expand localized service support. Growth reflects quality compliance, export readiness, and environmental alignment rather than basic industrial output growth.
- Expansion of export-oriented manufacturing
- Increased adoption of powder coating processes
- Rising effluent compliance requirements
- Growth of localized equipment and service support
Why Is Brazil Experiencing Rapid Uptake of Pre-treatment Cleaning Solutions?
Pre-treatment cleaning demand in Brazil is expanding at a CAGR of 9.4%, driven by modernization of industrial finishing lines. Automotive and appliance manufacturers upgrade surface preparation to reduce rework and defect rates. Increased use of corrosion protection coatings raises cleaning precision requirements. Environmental enforcement encourages efficient chemical usage and wastewater treatment integration. Regional manufacturers invest in turnkey pre-treatment systems. Growth reflects process modernization and compliance-driven upgrades rather than expansion of heavy industrial capacity.
- Modernization of industrial finishing lines
- Higher corrosion protection performance requirements
- Environmental enforcement influencing chemical choice
- Investment in integrated pre-treatment systems
How Is China Sustaining Momentum in Pre-treatment Cleaning Adoption?
Pre-treatment cleaning demand in China is growing at a CAGR of 9.0%, aligned with scale of manufacturing and quality standardization. High-volume production increases sensitivity to surface defects, elevating cleaning consistency needs. Export manufacturing requires adherence to international coating and assembly standards. Regulatory oversight tightens control of chemical discharge and energy usage. Domestic suppliers offer scalable systems compatible with automated lines. Growth reflects volume-driven quality control and regulatory enforcement rather than diversification into new manufacturing sectors.
- High-volume manufacturing quality sensitivity
- Export compliance with international standards
- Tightened chemical discharge regulations
- Availability of scalable domestic systems
What Factors Are Supporting Pre-treatment Cleaning Demand in United States?
Pre-treatment cleaning demand in the United States is expanding at a CAGR of 5.8%, supported by mature manufacturing and process optimization initiatives. Aerospace, medical device, and precision components require validated surface cleanliness. Replacement of legacy systems with automated, low-chemical solutions sustains demand. Environmental compliance drives upgrades in wastewater handling and chemistry control. Emphasis on yield improvement influences investment decisions. Growth reflects technology refresh cycles and compliance adherence rather than manufacturing footprint expansion.
- Precision manufacturing surface validation needs
- Replacement of legacy cleaning systems
- Environmental compliance-driven upgrades
- Focus on yield and defect reduction
Why Is Germany Showing Measured Expansion in Pre-treatment Cleaning?
Pre-treatment cleaning demand in Germany is growing at a CAGR of 5.7%, shaped by engineering precision and regulatory discipline. Automotive and industrial equipment manufacturers emphasize reproducible surface preparation. Strict environmental standards influence selection of low-emission and recyclable chemistries. Incremental automation upgrades improve consistency and traceability. Procurement prioritizes long-term reliability over rapid capacity growth. Expansion remains measured, reflecting optimization of existing lines and compliance stability rather than aggressive industrial expansion.
- Precision-driven surface preparation standards
- Strict environmental chemistry regulations
- Incremental automation and traceability upgrades
- Reliability-focused procurement practices
What is the competitive landscape of demand for the pre-treatment cleaning market globally?
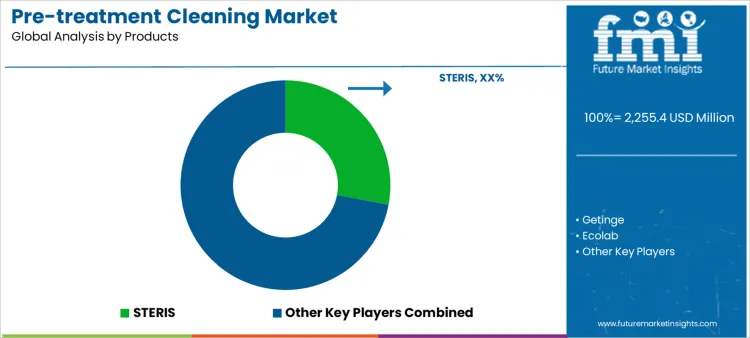
Demand for pre-treatment cleaning is driven by infection prevention standards, complex device reprocessing needs, and compliance expectations before sterilization. Sterile processing teams evaluate soil removal efficacy, material compatibility, enzymatic activity, foam control, and performance across lumened instruments. Buyer assessment includes cycle time impact, dosing accuracy, residue risk, water quality tolerance, and staff safety. Procurement behavior reflects standardized protocols, recurring consumable purchasing, validation documentation, and training support. Trend in the pre-treatment cleaning market shows increased use of automated washers and validated chemistries to improve consistency and reduce manual variability across reprocessing workflows.
STERIS leads competitive positioning through comprehensive pre-treatment chemistries integrated with equipment, workflows, and compliance programs. Getinge competes with detergents aligned to its reprocessing platforms and global service infrastructure. Ecolab supports demand with enzymatic cleaners and monitoring tools emphasizing standardization and staff safety. Advanced Sterilization Products maintains relevance via low-temperature compatibility and device-focused cleaning solutions. Cantel participates with specialty detergents supporting endoscope and complex instrument reprocessing. Competitive differentiation centers on cleaning efficacy, validation support, workflow integration, and supplier capability to sustain audited reprocessing environments.
Key Players in the Pre-treatment Cleaning Market
- STERIS
- Getinge
- Ecolab
- ASP
- Cantel
- Others
Scope of the Report
| Items | Values |
|---|---|
| Quantitative Units | USD million |
| Cleaning Solutions | Enzymatic detergents; Alkaline cleaners; Neutral detergents; Multi-enzymatic formulations; Specialty soil-removal agents |
| Application Areas | Medical device reprocessing; Endoscope pre-cleaning; Surgical instrument decontamination; Laboratory equipment cleaning; Pharma manufacturing equipment |
| End Users | Hospitals & CSSDs; Ambulatory surgical centers; Medical device manufacturers; Contract reprocessing providers; Research laboratories |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Countries Covered | India, Brazil, China, USA, Germany, and 40+ countries |
| Key Companies Profiled | STERIS; Getinge; Ecolab; ASP; Cantel; Others |
| Additional Attributes | Dollar sales by cleaning solution type, application area, and end user; soil removal efficiency across protein, lipid, and biofilm residues in pre-treatment workflows; material compatibility with delicate instruments and endoscopes; role of enzymatic and multi-enzymatic formulations in reducing manual cleaning burden; compliance with reprocessing guidelines and infection control standards; procurement dynamics driven by hospital CSSD volumes, device complexity, and outsourced reprocessing demand. |
Pre-treatment Cleaning Market by Segment
Cleaning Solutions:
- Enzymatic detergents
- Alkaline cleaners
- Neutral detergents
- Multi-enzymatic formulations
- Specialty soil-removal agents
Application Areas:
- Medical device reprocessing
- Endoscope pre-cleaning
- Surgical instrument decontamination
- Laboratory equipment cleaning
- Pharma manufacturing equipment
End Users:
- Hospitals & CSSDs
- Ambulatory surgical centers
- Medical device manufacturers
- Contract reprocessing providers
- Research laboratories
Region:
- Asia Pacific
- China
- Japan
- South Korea
- India
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Nordic
- BENELUX
- Rest of Europe
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Other African Union
- Rest of Middle East & Africa
Bibliography
- International Organization for Standardization. (2023). ISO 15883‑1: Washer-disinfectors — Part 1: General requirements, terms and definitions, and tests. ISO.
- European Commission. (2024). Best available techniques (BAT) reference document for surface treatment using chemicals and solvents (Industrial Emissions Directive update). Publications Office of the European Union.
- United States Food and Drug Administration. (2023). Reprocessing medical devices in health care settings: Validation methods and cleaning effectiveness guidance. U.S. Department of Health and Human Services.
Frequently Asked Questions
How big is the pre-treatment cleaning market in 2026?
The global pre-treatment cleaning market is estimated to be valued at USD 2,255.4 million in 2026.
What will be the size of pre-treatment cleaning market in 2036?
The market size for the pre-treatment cleaning market is projected to reach USD 4,605.4 million by 2036.
How much will be the pre-treatment cleaning market growth between 2026 and 2036?
The pre-treatment cleaning market is expected to grow at a 7.4% CAGR between 2026 and 2036.
What are the key product types in the pre-treatment cleaning market?
The key product types in pre-treatment cleaning market are enzymatic detergents, alkaline cleaners, neutral detergents, multi-enzymatic formulations and specialty soil-removal agents.
Which application areas segment to contribute significant share in the pre-treatment cleaning market in 2026?
In terms of application areas, medical device reprocessing segment to command 34.0% share in the pre-treatment cleaning market in 2026.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand-side Trends
- Supply-side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y-o-Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Cleaning Solutions
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Cleaning Solutions , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Cleaning Solutions , 2026 to 2036
- Enzymatic detergents
- Alkaline cleaners
- Neutral detergents
- Multi-enzymatic formulations
- Specialty soil-removal agents
- Enzymatic detergents
- Y-o-Y Growth Trend Analysis By Cleaning Solutions , 2021 to 2025
- Absolute $ Opportunity Analysis By Cleaning Solutions , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application Areas
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application Areas, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application Areas, 2026 to 2036
- Medical device reprocessing
- Endoscope pre-cleaning
- Surgical instrument decontamination
- Laboratory equipment cleaning
- Pharma manufacturing equipment
- Medical device reprocessing
- Y-o-Y Growth Trend Analysis By Application Areas, 2021 to 2025
- Absolute $ Opportunity Analysis By Application Areas, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End Users
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End Users, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End Users, 2026 to 2036
- Hospitals & CSSDs
- Ambulatory surgical centers
- Medical device manufacturers
- Contract reprocessing providers
- Research laboratories
- Hospitals & CSSDs
- Y-o-Y Growth Trend Analysis By End Users, 2021 to 2025
- Absolute $ Opportunity Analysis By End Users, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Cleaning Solutions
- By Application Areas
- By End Users
- By Country
- Market Attractiveness Analysis
- By Country
- By Cleaning Solutions
- By Application Areas
- By End Users
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Cleaning Solutions
- By Application Areas
- By End Users
- By Country
- Market Attractiveness Analysis
- By Country
- By Cleaning Solutions
- By Application Areas
- By End Users
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Cleaning Solutions
- By Application Areas
- By End Users
- By Country
- Market Attractiveness Analysis
- By Country
- By Cleaning Solutions
- By Application Areas
- By End Users
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Cleaning Solutions
- By Application Areas
- By End Users
- By Country
- Market Attractiveness Analysis
- By Country
- By Cleaning Solutions
- By Application Areas
- By End Users
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Cleaning Solutions
- By Application Areas
- By End Users
- By Country
- Market Attractiveness Analysis
- By Country
- By Cleaning Solutions
- By Application Areas
- By End Users
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Cleaning Solutions
- By Application Areas
- By End Users
- By Country
- Market Attractiveness Analysis
- By Country
- By Cleaning Solutions
- By Application Areas
- By End Users
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Cleaning Solutions
- By Application Areas
- By End Users
- By Country
- Market Attractiveness Analysis
- By Country
- By Cleaning Solutions
- By Application Areas
- By End Users
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Cleaning Solutions
- By Application Areas
- By End Users
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Cleaning Solutions
- By Application Areas
- By End Users
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Cleaning Solutions
- By Application Areas
- By End Users
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Cleaning Solutions
- By Application Areas
- By End Users
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Cleaning Solutions
- By Application Areas
- By End Users
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Cleaning Solutions
- By Application Areas
- By End Users
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Cleaning Solutions
- By Application Areas
- By End Users
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Cleaning Solutions
- By Application Areas
- By End Users
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Cleaning Solutions
- By Application Areas
- By End Users
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Cleaning Solutions
- By Application Areas
- By End Users
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Cleaning Solutions
- By Application Areas
- By End Users
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Cleaning Solutions
- By Application Areas
- By End Users
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Cleaning Solutions
- By Application Areas
- By End Users
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Cleaning Solutions
- By Application Areas
- By End Users
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Cleaning Solutions
- By Application Areas
- By End Users
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Cleaning Solutions
- By Application Areas
- By End Users
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Cleaning Solutions
- By Application Areas
- By End Users
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Cleaning Solutions
- By Application Areas
- By End Users
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Cleaning Solutions
- By Application Areas
- By End Users
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Cleaning Solutions
- By Application Areas
- By End Users
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Cleaning Solutions
- By Application Areas
- By End Users
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Cleaning Solutions
- By Application Areas
- By End Users
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Cleaning Solutions
- By Application Areas
- By End Users
- Competition Analysis
- Competition Deep Dive
- STERIS
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Getinge
- Ecolab
- ASP
- Cantel
- Others
- STERIS
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Cleaning Solutions , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Application Areas, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by End Users, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Cleaning Solutions , 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Application Areas, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by End Users, 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 10: Latin America Market Value (USD Million) Forecast by Cleaning Solutions , 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by Application Areas, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by End Users, 2021 to 2036
- Table 13: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Western Europe Market Value (USD Million) Forecast by Cleaning Solutions , 2021 to 2036
- Table 15: Western Europe Market Value (USD Million) Forecast by Application Areas, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by End Users, 2021 to 2036
- Table 17: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 18: Eastern Europe Market Value (USD Million) Forecast by Cleaning Solutions , 2021 to 2036
- Table 19: Eastern Europe Market Value (USD Million) Forecast by Application Areas, 2021 to 2036
- Table 20: Eastern Europe Market Value (USD Million) Forecast by End Users, 2021 to 2036
- Table 21: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: East Asia Market Value (USD Million) Forecast by Cleaning Solutions , 2021 to 2036
- Table 23: East Asia Market Value (USD Million) Forecast by Application Areas, 2021 to 2036
- Table 24: East Asia Market Value (USD Million) Forecast by End Users, 2021 to 2036
- Table 25: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: South Asia and Pacific Market Value (USD Million) Forecast by Cleaning Solutions , 2021 to 2036
- Table 27: South Asia and Pacific Market Value (USD Million) Forecast by Application Areas, 2021 to 2036
- Table 28: South Asia and Pacific Market Value (USD Million) Forecast by End Users, 2021 to 2036
- Table 29: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 30: Middle East & Africa Market Value (USD Million) Forecast by Cleaning Solutions , 2021 to 2036
- Table 31: Middle East & Africa Market Value (USD Million) Forecast by Application Areas, 2021 to 2036
- Table 32: Middle East & Africa Market Value (USD Million) Forecast by End Users, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021 to 2036
- Figure 3: Global Market Value Share and BPS Analysis by Cleaning Solutions , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Cleaning Solutions , 2026 to 2036
- Figure 5: Global Market Attractiveness Analysis by Cleaning Solutions
- Figure 6: Global Market Value Share and BPS Analysis by Application Areas, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Application Areas, 2026 to 2036
- Figure 8: Global Market Attractiveness Analysis by Application Areas
- Figure 9: Global Market Value Share and BPS Analysis by End Users, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by End Users, 2026 to 2036
- Figure 11: Global Market Attractiveness Analysis by End Users
- Figure 12: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Region, 2026 to 2036
- Figure 14: Global Market Attractiveness Analysis by Region
- Figure 15: North America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 16: Latin America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 17: Western Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 18: Eastern Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 19: East Asia Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 20: South Asia and Pacific Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 21: Middle East & Africa Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 22: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 23: North America Market Value Share and BPS Analysis by Cleaning Solutions , 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Cleaning Solutions , 2026 to 2036
- Figure 25: North America Market Attractiveness Analysis by Cleaning Solutions
- Figure 26: North America Market Value Share and BPS Analysis by Application Areas, 2026 and 2036
- Figure 27: North America Market Y-o-Y Growth Comparison by Application Areas, 2026 to 2036
- Figure 28: North America Market Attractiveness Analysis by Application Areas
- Figure 29: North America Market Value Share and BPS Analysis by End Users, 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by End Users, 2026 to 2036
- Figure 31: North America Market Attractiveness Analysis by End Users
- Figure 32: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 33: Latin America Market Value Share and BPS Analysis by Cleaning Solutions , 2026 and 2036
- Figure 34: Latin America Market Y-o-Y Growth Comparison by Cleaning Solutions , 2026 to 2036
- Figure 35: Latin America Market Attractiveness Analysis by Cleaning Solutions
- Figure 36: Latin America Market Value Share and BPS Analysis by Application Areas, 2026 and 2036
- Figure 37: Latin America Market Y-o-Y Growth Comparison by Application Areas, 2026 to 2036
- Figure 38: Latin America Market Attractiveness Analysis by Application Areas
- Figure 39: Latin America Market Value Share and BPS Analysis by End Users, 2026 and 2036
- Figure 40: Latin America Market Y-o-Y Growth Comparison by End Users, 2026 to 2036
- Figure 41: Latin America Market Attractiveness Analysis by End Users
- Figure 42: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 43: Western Europe Market Value Share and BPS Analysis by Cleaning Solutions , 2026 and 2036
- Figure 44: Western Europe Market Y-o-Y Growth Comparison by Cleaning Solutions , 2026 to 2036
- Figure 45: Western Europe Market Attractiveness Analysis by Cleaning Solutions
- Figure 46: Western Europe Market Value Share and BPS Analysis by Application Areas, 2026 and 2036
- Figure 47: Western Europe Market Y-o-Y Growth Comparison by Application Areas, 2026 to 2036
- Figure 48: Western Europe Market Attractiveness Analysis by Application Areas
- Figure 49: Western Europe Market Value Share and BPS Analysis by End Users, 2026 and 2036
- Figure 50: Western Europe Market Y-o-Y Growth Comparison by End Users, 2026 to 2036
- Figure 51: Western Europe Market Attractiveness Analysis by End Users
- Figure 52: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 53: Eastern Europe Market Value Share and BPS Analysis by Cleaning Solutions , 2026 and 2036
- Figure 54: Eastern Europe Market Y-o-Y Growth Comparison by Cleaning Solutions , 2026 to 2036
- Figure 55: Eastern Europe Market Attractiveness Analysis by Cleaning Solutions
- Figure 56: Eastern Europe Market Value Share and BPS Analysis by Application Areas, 2026 and 2036
- Figure 57: Eastern Europe Market Y-o-Y Growth Comparison by Application Areas, 2026 to 2036
- Figure 58: Eastern Europe Market Attractiveness Analysis by Application Areas
- Figure 59: Eastern Europe Market Value Share and BPS Analysis by End Users, 2026 and 2036
- Figure 60: Eastern Europe Market Y-o-Y Growth Comparison by End Users, 2026 to 2036
- Figure 61: Eastern Europe Market Attractiveness Analysis by End Users
- Figure 62: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 63: East Asia Market Value Share and BPS Analysis by Cleaning Solutions , 2026 and 2036
- Figure 64: East Asia Market Y-o-Y Growth Comparison by Cleaning Solutions , 2026 to 2036
- Figure 65: East Asia Market Attractiveness Analysis by Cleaning Solutions
- Figure 66: East Asia Market Value Share and BPS Analysis by Application Areas, 2026 and 2036
- Figure 67: East Asia Market Y-o-Y Growth Comparison by Application Areas, 2026 to 2036
- Figure 68: East Asia Market Attractiveness Analysis by Application Areas
- Figure 69: East Asia Market Value Share and BPS Analysis by End Users, 2026 and 2036
- Figure 70: East Asia Market Y-o-Y Growth Comparison by End Users, 2026 to 2036
- Figure 71: East Asia Market Attractiveness Analysis by End Users
- Figure 72: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 73: South Asia and Pacific Market Value Share and BPS Analysis by Cleaning Solutions , 2026 and 2036
- Figure 74: South Asia and Pacific Market Y-o-Y Growth Comparison by Cleaning Solutions , 2026 to 2036
- Figure 75: South Asia and Pacific Market Attractiveness Analysis by Cleaning Solutions
- Figure 76: South Asia and Pacific Market Value Share and BPS Analysis by Application Areas, 2026 and 2036
- Figure 77: South Asia and Pacific Market Y-o-Y Growth Comparison by Application Areas, 2026 to 2036
- Figure 78: South Asia and Pacific Market Attractiveness Analysis by Application Areas
- Figure 79: South Asia and Pacific Market Value Share and BPS Analysis by End Users, 2026 and 2036
- Figure 80: South Asia and Pacific Market Y-o-Y Growth Comparison by End Users, 2026 to 2036
- Figure 81: South Asia and Pacific Market Attractiveness Analysis by End Users
- Figure 82: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 83: Middle East & Africa Market Value Share and BPS Analysis by Cleaning Solutions , 2026 and 2036
- Figure 84: Middle East & Africa Market Y-o-Y Growth Comparison by Cleaning Solutions , 2026 to 2036
- Figure 85: Middle East & Africa Market Attractiveness Analysis by Cleaning Solutions
- Figure 86: Middle East & Africa Market Value Share and BPS Analysis by Application Areas, 2026 and 2036
- Figure 87: Middle East & Africa Market Y-o-Y Growth Comparison by Application Areas, 2026 to 2036
- Figure 88: Middle East & Africa Market Attractiveness Analysis by Application Areas
- Figure 89: Middle East & Africa Market Value Share and BPS Analysis by End Users, 2026 and 2036
- Figure 90: Middle East & Africa Market Y-o-Y Growth Comparison by End Users, 2026 to 2036
- Figure 91: Middle East & Africa Market Attractiveness Analysis by End Users
- Figure 92: Global Market - Tier Structure Analysis
- Figure 93: Global Market - Company Share Analysis

