Endoscope Reprocessing Device Market
Endoscope Reprocessing Device Market Analysis - Size, Share, and Forecast Outlook 2026 to 2036
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Endoscope Endoscope Reprocessing Device Market Forecast and Outlook 2026 to 2036
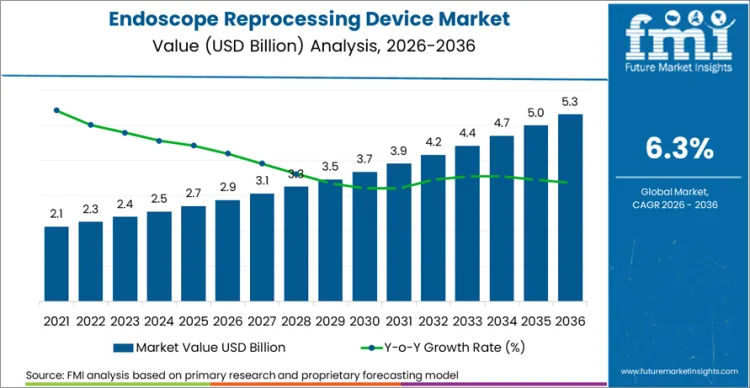
The global endoscope reprocessing device sector is on track to achieve a valuation of USD 5.30 billion by 2036, accelerating from USD 2.88 billion in 2026 at a CAGR of 6.3%. As per Future Market Insights, expansion is structurally underpinned by the growing volume of endoscopic procedures globally and the persistent regulatory pressure to prevent healthcare-associated infections from contaminated flexible endoscopes. The USA Food and Drug Administration issued supplemental guidance in 2024 on the reprocessing of duodenoscopes with elevator channels, reinforcing the need for validated high-level disinfection and sterilization protocols.
This regulatory pressure compels hospitals and ambulatory surgery centers to invest in automated endoscope reprocessors (AERs) that provide documented, traceable reprocessing cycles. Simultaneously, the industry is shifting from manual reprocessing to fully automated systems with digital compliance tracking.
The Olympus Press Office stated: 'India, endoscopic procedures already outnumber those in Japan and are expected to grow further, and infection control systems are still evolving.' In October 2024, Olympus received CE-mark approval under the Medical Device Regulation (MDR) for three cloud-based AI medical devices (CADDIE, CADU, and SMARTIBD), forming the foundation of its Intelligent Endoscopy Ecosystem. FMI opines that Olympus's AI ecosystem strategy, which connects hardware, AI software, and data services, will reshape the reprocessing market by creating an integrated value chain where endoscope cleaning verification is digitally linked to clinical outcome data.
The operational reality for endoscope reprocessing suppliers is defined by the need to balance infection control stringency with procedure turnaround time in high-volume endoscopy suites. As per FMI, the endoscope reprocessing market is structurally driven by the growing gap between rising procedure volumes (particularly colonoscopy screening driven by colorectal cancer guidelines) and the limited number of reusable endoscopes available per facility. FMI is of the opinion that single-use endoscope adoption, while growing, will not replace reusable platforms within the forecast period due to cost and sustainability concerns, which sustains the demand for high-throughput AER systems with rapid cycle times.
Summary of Endoscope Reprocessing Device Market
What Is Growth Outlook for Endoscope Reprocessing Device Market as per Future Market Insights Projection?
Future Market Insights projects the endoscope reprocessing device market to expand at a CAGR of 6.3% from 2026 to 2036, increasing from USD 2.88 Billion in 2026 to USD 5.30 Billion by 2036.
FMI Research Approach: FMI proprietary forecasting model based on endoscopic procedure volumes and AER installed base replacement cycles.
How Do FMI Analysts Perceive Endoscope Reprocessing Device Market to Evolve?
FMI analysts perceive the market evolving toward digitally verified sterility assurance where automated reprocessors integrate with cloud-based AI endoscopy ecosystems to provide traceable compliance documentation.
FMI Research Approach: Olympus AI endoscopy ecosystem CE-mark (October 2024) and FDA duodenoscope reprocessing guidance (2024).
Which Country Holds Largest Share in Global Endoscope Reprocessing Device Market?
The United States holds a significant share, supported by the highest endoscopic procedure volume per capita, stringent FDA reprocessing guidelines, and the largest AER installed base.
FMI Research Approach: FMI country-level revenue modeling by endoscopy procedure volumes and AER procurement data.
How Large Will Endoscope Reprocessing Device Market Be by 2036?
The global endoscope reprocessing device market is projected to reach USD 5.30 Billion by 2036.
FMI Research Approach: FMI long-term revenue forecast derived from colorectal cancer screening guideline expansion and AER replacement cycles.
What Is Definition of Endoscope Reprocessing Device Market?
The endoscope reprocessing device market includes revenue from automated endoscope reprocessors, manual cleaning accessories, sterility verification systems, and digital compliance tracking platforms used to decontaminate flexible and semi-rigid endoscopes.
FMI Research Approach: FMI market taxonomy aligned with FDA and MDR endoscope reprocessing classifications.
What Are Globally Unique Trends Shaping Endoscope Reprocessing Device Market?
Globally unique trends include the integration of AI-connected endoscopy ecosystems with reprocessing workflows, tightening FDA duodenoscope reprocessing requirements, and the rapid growth of endoscopic procedure volumes in India surpassing Japan.
FMI Research Approach: Olympus AI ecosystem CE-mark (October 2024) and FDA duodenoscope guidance (2024).
Endoscope Reprocessing Device Market Key Takeaways
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 2.88 Billion |
| Industry Value (2036) | USD 5.30 Billion |
| CAGR (2026 to 2036) | 6.3% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Stringent Regulations Encourage Medical Facilities to Adopt Endoscope Reprocessing Devices
Regulatory bodies around the world have introduced stringent measures for the reprocessing of medical devices. Endoscopes come within that purview, necessitating the increased use of reprocessing devices in healthcare facilities.
Non-compliance with these regulations has the ability to incur severe punishments, including penalties, loss of accreditation, and reputational damage, not to mention harm to patients. Hence, healthcare facilities are diligently adopting reprocessing devices.
The following table provides the various regulatory bodies that mandate the use of reprocessing devices.
| Regulatory Body | Recommendations |
|---|---|
| Centers for Disease Control and Prevention (CDC) | Use of high-level disinfection and sterilization for semi-critical devices like endoscopes |
| Association for the Advancement of Medical Instrumentation (AAMI) | Standards like AAMI ST91 for reprocessing endoscopes |
| Food and Drug Administration (FDA) | Manufacturers are required to provide data that reprocessing has been effective for endoscopes |
| European Union Medical Device Regulation (EU MDR) | Stringent rules regarding the use of endoscopes in Europe |
The following table highlights some of the recent innovations and their impact.
| Innovation | Impact |
|---|---|
| Modern automatic endoscope reprocessors (AERs) | Handling of multiple scopes simultaneously. |
| Hydrogen Peroxide Vapor or Ozone-Based Systems | Faster, safer, and eco-friendlier working. |
| Barcode Scanning and Digital Record-Keeping | Tracking of every step in the reprocessing cycle. |
| Eco-Friendly Disinfectants | Adding to more sustainability oriented healthcare settings. |
High Costs Hinder Progress
Advanced automatic endoscope reprocessors and sterilization systems are costly and require a high initial investment. Most healthcare facilities, especially in developing regions, cannot afford to spend such large amounts of money.
Regular maintenance of the reprocessing device is necessary for its efficacy and compliance with the standards. This increases cost in the form of service charges and maintenance contracts. Consumables used in the reprocessing activity, like high-level disinfectants, enzymatic detergents, sterile water, and filters, can also be cost-intensive.
Quite often, sophisticated devices for reprocessing require upgrading infrastructures such as dedicated spaces, water supply, and ventilation. Installation and operation are prohibitive for small hospitals and clinics.
Adequate training of the staff operating these devices and following the reprocessing protocols is needed. Training programs are expensive and take a long time to initiate, especially in institutions with rapid turnover rates among the personnel. Hence, the labor costs associated with the devices is also high.
Segment-wise Analysis
Large Number of Endoscopies Performed in Hospitals Leading to Greater Use of Endoscopy Reprocessing Devices
By End Use
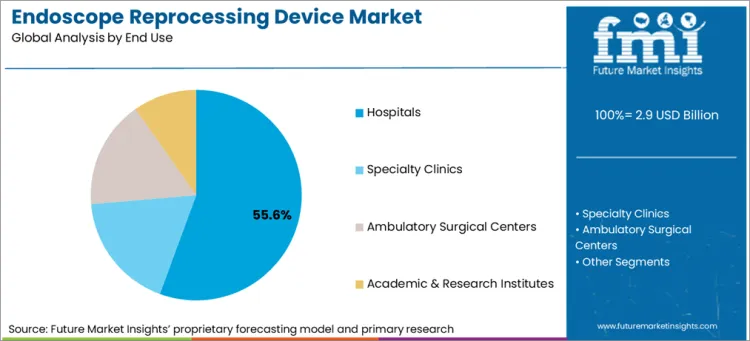
| Attributes | Details |
|---|---|
| Top End Use | Hospitals |
| Market Share in 2026 | 55.6% |
Hospitals are the primary end users of endoscopy reprocessing devices. This segment is likely to account for 55.6% of the value share in 2026.
Most endoscopy procedures are performed in hospitals. Patients and their relatives have an implicit trust in hospitals and their staff. As a result, the number of endoscopies is increasing substantially in hospitals. This is leading to more recourse towards endoscopy reprocessing devices.
Safety and Efficiency of Automated Endoscope Reprocessors Preferred by End Users
By Product
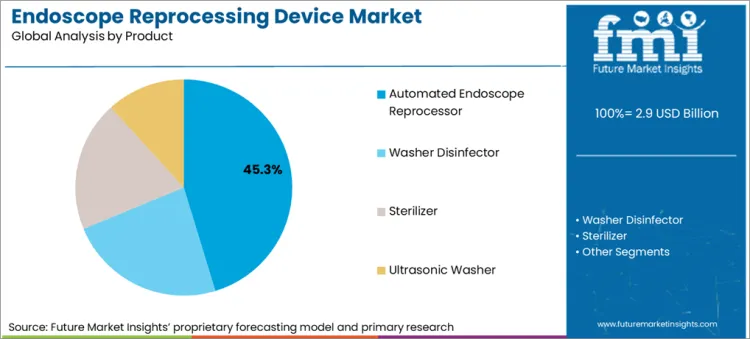
| Attributes | Details |
|---|---|
| Top Product | Automated Endoscope Reprocessor |
| Market Share in 2026 | 45.3% |
Endoscopy reprocessing devices are majorly used in the form of automated endoscope reprocessors. These reprocessors is likely to account for 45.3% of the value share in 2026.
The automated endoscope reprocessor segment is the most significant in the market for endoscopy reprocessing devices, mainly attributed to its efficiency and safety. Automated cleaning and disinfection by AERs significantly reduce processing time, enabling high throughput, which is necessary for busy healthcare settings. It minimizes human contact with contaminated instruments, thus lowering the risk of cross-contamination and ensuring consistent adherence to protocols.
Besides, AERs meet high regulatory requirements by offering automated tracking and documentation which further increases compliance and quality assurance. Moreover, advances in technology have allowed them to integrate with hospital systems and make user-friendly features that enhance their reliability and usability.
With the increased focus on infection control and patient safety, AERs are being adopted by more and more healthcare facilities. Though the initial investment is higher, the long-term savings in labor costs and reduced equipment damage make AERs a cost-effective choice.
Increased Efficiency Provided by Automatic Devices Help End Users
Automatic endoscopy processing devices are preferred over semi-automatic devices because they are efficient, safe, and reliable. They fully automate the cleaning and disinfection processes, reducing the time required for reprocessing, which also minimizes the risk of human error.
Moreover, automatic devices provide integrated tracking and documentation features, which help meet regulatory standards. Overall, their ability to streamline workflows and improve safety makes automatic endoscopy processing devices the favored choice in healthcare settings.
Mobile Reprocessing Devices Provide Enhanced Flexibility
Mobile processing devices are more favored than flat-top ones due to the advantage of portability, which facilitates easy shifting from one place to another in healthcare facilities. These provide easy reprocessing throughout the facility with minimal need for dedicated spaces, especially in busy environments, thus optimizing workflow and maximizing space utilization.
Mobile devices also can be readily mobilized to meet variable demands and improve operational efficiency. Overall, the portability and usability of mobile endoscopy processing devices make them a more viable option for healthcare settings.
Region-wise Analysis
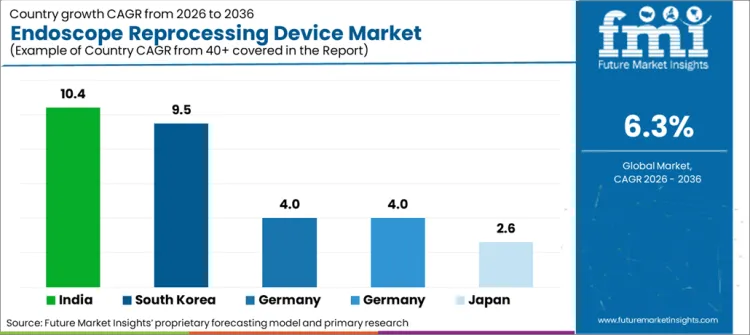
| Countries | CAGR 2026 to 2036 |
|---|---|
| United States | 4% |
| Japan | 2.6% |
| India | 10.4% |
| Germany | 4% |
| South Korea | 9.5% |
Japan Endoscope Reprocessing Device Industry Outlook
The Japan endoscope reprocessing device market is estimated to advance at a CAGR of 2.6% over the 2026 to 2036 period. In Japan, a high number of patients are preferring minimally invasive surgeries. These surgeries require precise operation of tools and reprocessing devices enable this smooth operation.
Japanese manufacturers are prominent not only locally but worldwide. Japanese companies like Olympus are leaders, providing novel products to the worldwide market. While there is concern over the limited budget available to hospitals in rural areas of Japan, there are still a plethora of opportunities in the country for companies.
Germany Endoscope Reprocessing Device Industry Analysis
The endoscope reprocessing device market in Germany is expected to register a CAGR of 4% from 2026 to 2036. Germany has one of the leading healthcare sectors in Europe. As a result, smart reprocessing systems with AI and machine learning capabilities are becoming popular in the country.
In Germany, rules mandated by various regulatory authorities make reprocessing devices necessary in healthcare settings. The European Union Medical Device Regulation (EU MDR) and Robert Koch Institute (RKI) guidelines are two such standards mandating strict infection protection protocols, necessitating the use of reprocessing devices.
South Korea Endoscopy Reprocessing Device Indsutry Outlook
The endoscope reprocessing device market in South Korea is expected to register a CAGR of 9.5% from 2026 to 2036. The South Korean healthcare sector is benefitting from increased government investment in medical facilities. Schemes like the Korea Health Industry Development Institute (KHIDI) increase the adoption of apparatus like endoscopy reprocessing devices.
Another notable factor in South Korea is the rising medical tourism in the country. The proliferation of 5G in the country is enabling healthcare facilities to have advanced equipment, including IoT-enabled endoscopy reprocessing devices. As a result, tourists from all over the world are traveling to South Korea for procedures like endoscopies.
United States Endoscope Reprocessing Device Industry Analysis
The endoscope reprocessing device market in the United States is set to progress at a CAGR of 4% from 2026 to 2036. The healthcare system in the United States is robust and provides impetus for novel products. As a result, reprocessing devices that enable integration with Electronic Health Records (EHR) as well as IoT-enabled devices are becoming more popular in the country.
Regulatory authorities like the Centers for Disease Control and Prevention (CDC), FDA, and the Association for the Advancement of Medical Instrumentation (AAMI) make it mandatory for endoscopy devices to be kept in proper order. Hence, endoscope reprocessing device sales are shooting up.
Another key factor shaping the landscape in the country is the rising number of the geriatric population. This segment of the population requires endoscopy frequently, leading to more use of reprocessing devices.
Competitive Landscape
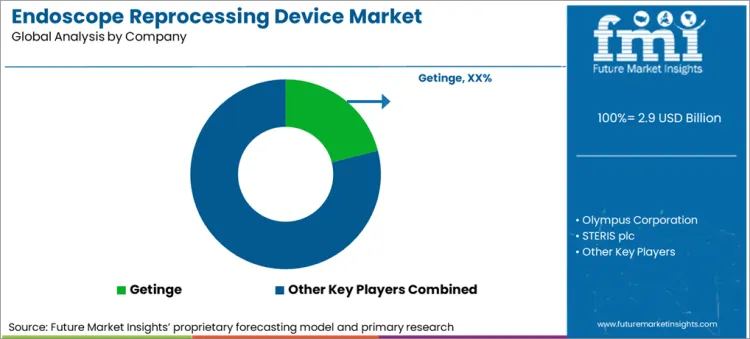
The striving for technological progress in the healthcare sector is also reflected in the endoscopy reprocessing device market. With the help of technology, players aim to keep the product line moving and endoscope reprocessing device sales consistent.
One of the notable product launches was Getinge AB launching Enhanced ED-Flow AER in June 2022. It was an updated version of the ED-Flow Automated Endoscope Reprocessor (AER), making significant strides in digital connectivity and data management.
Recognizing the rising importance of endoscopy reprocessing devices, several healthcare companies are entering or expanding their presence in the market. For instance, HOYA Corporation acquired Wassenburg Medical B.V. in November 2023. Wassenburg is a prominent manufacturer of these devices and thus HOYA expanded its footprint in the market through the acquisition.
Recent Developments:
- In October 2024, Olympus received CE-mark approval under MDR for three cloud-based AI devices (CADDIE, CADU, SMARTIBD) forming its Intelligent Endoscopy Ecosystem.
- The USA FDA issued supplemental guidance in 2024 on duodenoscope reprocessing with elevator channels, reinforcing validated high-level disinfection requirements.
Key Companies
- Getinge
- Olympus Corporation
- STERIS plc
- ASP India Private Ltd.
- Steelco S.p.A.
- Arc Healthcare Solutions
- Creo Medical GmbH - Endo-Technik
- D&D ELECTRONICS CO., LTD
- Detro Healthcare
- DGM Pharma-Apparate Handel
- HEFEI JINNICK MEDICAL TECHNOLOGY CO., LTD.
- Hefei Qianbaijian Medical Equipment
- Jiangsu Huaxi Medical Equipment
- Medonica
Market Definition
The endoscope reprocessing device market represents revenue from equipment and systems used to clean, disinfect, and sterilize flexible and semi-rigid endoscopes between patient uses in hospitals, ambulatory surgery centers, and endoscopy clinics.
Inclusions cover automated endoscope reprocessors (AERs), manual cleaning brushes and detergents, high-level disinfectants, sterility verification test strips, and digital reprocessing compliance tracking software.
Exclusions include endoscope manufacturing and sales, single-use disposable endoscopes, and general hospital sterilization equipment not specific to endoscope reprocessing. Operating room instrument sterilizers are outside scope.
Scope of Report
| Items | Values |
|---|---|
| Quantitative Units (2026) | USD 2.88 Billion |
| Product Type | Automated Endoscope Reprocessors, Manual Cleaning Accessories, Disinfectants, Digital Compliance Systems |
| End User | Hospitals, Ambulatory Surgery Centers, Endoscopy Clinics |
| Regions Covered | North America, Europe, Asia Pacific, Latin America, Middle East and Africa |
| Key Companies Profiled | Olympus, Cantel Medical (Medivators), Getinge, Steelco (SteelcoBelimed), Wassenburg Medical |
Endoscope Reprocessing Device Market Segmentation
By Product:
The market is segmented into automated endoscope reprocessors, washer disinfectors, sterilizers, ultrasonic washers, and drying & storage cabinets based on the product.
By Operation Mode:
Based on the operation mode, the market is bifurcated into semiautomatic and automatic.
By Modality:
In terms of modality, the market is segmented into floor standing/bench top and mobile.
By End User:
In terms of end users, the market is segmented into hospitals, specialty clinics, ambulatory surgical centers, academic & research institutes, and the Central Sterile Services Department (CSSD).
By Region:
Based on the region, the market is segmented into North America, Latin America, Europe, South Asia, East Asia, Middle East & Africa, and Oceania.
Bibliography
- Olympus Corporation. (2024, October). CE-mark approval for AI endoscopy devices under MDR. Olympus Press Releases.
- USA Food and Drug Administration. (2024). Supplemental guidance on duodenoscope reprocessing. FDA.
Frequently Asked Questions
What is the current global market size for Endoscope Reprocessing Device?
The global market is valued at USD 2.88 Billion in 2026, driven by rising endoscopic procedure volumes globally and heightened regulatory scrutiny on flexible endoscope contamination and infection control.
What is the projected Compound Annual Growth Rate (CAGR) for the market over the next 10 years?
The market is projected to grow at a CAGR of 6.3% from 2026 to 2036.
Which regions are experiencing the fastest expansion?
Asia Pacific leads with India's endoscopic procedure volumes surpassing Japan, followed by North America driven by FDA reprocessing compliance requirements and colorectal cancer screening expansion.
What are the primary market drivers?
FDA duodenoscope reprocessing guidelines and the integration of AI-connected endoscopy ecosystems requiring digitally verified sterility assurance are the primary growth catalysts.
Who are the leading suppliers in the industry?
Olympus, Cantel Medical (Medivators), Getinge, SteelcoBelimed, and Wassenburg Medical are key players, differentiating through AI-connected reprocessing ecosystems and automated high-throughput AER platforms.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End Use
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End Use , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End Use , 2026 to 2036
- Hospitals
- Specialty Clinics
- Ambulatory Surgical Centers
- Academic & Research Institutes
- Hospitals
- Y to o to Y Growth Trend Analysis By End Use , 2021 to 2025
- Absolute $ Opportunity Analysis By End Use , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product, 2026 to 2036
- Automated Endoscope Reprocessor
- Washer Disinfector
- Sterilizer
- Ultrasonic Washer
- Automated Endoscope Reprocessor
- Y to o to Y Growth Trend Analysis By Product, 2021 to 2025
- Absolute $ Opportunity Analysis By Product, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By End Use
- By Product
- By Country
- Market Attractiveness Analysis
- By Country
- By End Use
- By Product
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By End Use
- By Product
- By Country
- Market Attractiveness Analysis
- By Country
- By End Use
- By Product
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By End Use
- By Product
- By Country
- Market Attractiveness Analysis
- By Country
- By End Use
- By Product
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By End Use
- By Product
- By Country
- Market Attractiveness Analysis
- By Country
- By End Use
- By Product
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By End Use
- By Product
- By Country
- Market Attractiveness Analysis
- By Country
- By End Use
- By Product
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By End Use
- By Product
- By Country
- Market Attractiveness Analysis
- By Country
- By End Use
- By Product
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By End Use
- By Product
- By Country
- Market Attractiveness Analysis
- By Country
- By End Use
- By Product
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By End Use
- By Product
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By End Use
- By Product
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By End Use
- By Product
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By End Use
- By Product
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By End Use
- By Product
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By End Use
- By Product
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By End Use
- By Product
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By End Use
- By Product
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By End Use
- By Product
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By End Use
- By Product
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By End Use
- By Product
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By End Use
- By Product
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By End Use
- By Product
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By End Use
- By Product
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By End Use
- By Product
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By End Use
- By Product
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By End Use
- By Product
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By End Use
- By Product
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By End Use
- By Product
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By End Use
- By Product
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By End Use
- By Product
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By End Use
- By Product
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By End Use
- By Product
- Competition Analysis
- Competition Deep Dive
- Getinge
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Olympus Corporation
- STERIS plc
- ASP India Private Ltd.
- Steelco S.p.A.
- Arc Healthcare Solutions
- Getinge
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by End Use , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by End Use , 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by End Use , 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by End Use , 2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by End Use , 2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by End Use , 2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by End Use , 2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by End Use , 2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by Product, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by End Use , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by End Use , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by End Use
- Figure 6: Global Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Product, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Product
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by End Use , 2026 and 2036
- Figure 21: North America Market Y-o-Y Growth Comparison by End Use , 2026-2036
- Figure 22: North America Market Attractiveness Analysis by End Use
- Figure 23: North America Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Product, 2026-2036
- Figure 25: North America Market Attractiveness Analysis by Product
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by End Use , 2026 and 2036
- Figure 28: Latin America Market Y-o-Y Growth Comparison by End Use , 2026-2036
- Figure 29: Latin America Market Attractiveness Analysis by End Use
- Figure 30: Latin America Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 31: Latin America Market Y-o-Y Growth Comparison by Product, 2026-2036
- Figure 32: Latin America Market Attractiveness Analysis by Product
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by End Use , 2026 and 2036
- Figure 35: Western Europe Market Y-o-Y Growth Comparison by End Use , 2026-2036
- Figure 36: Western Europe Market Attractiveness Analysis by End Use
- Figure 37: Western Europe Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 38: Western Europe Market Y-o-Y Growth Comparison by Product, 2026-2036
- Figure 39: Western Europe Market Attractiveness Analysis by Product
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by End Use , 2026 and 2036
- Figure 42: Eastern Europe Market Y-o-Y Growth Comparison by End Use , 2026-2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by End Use
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 45: Eastern Europe Market Y-o-Y Growth Comparison by Product, 2026-2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by Product
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by End Use , 2026 and 2036
- Figure 49: East Asia Market Y-o-Y Growth Comparison by End Use , 2026-2036
- Figure 50: East Asia Market Attractiveness Analysis by End Use
- Figure 51: East Asia Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 52: East Asia Market Y-o-Y Growth Comparison by Product, 2026-2036
- Figure 53: East Asia Market Attractiveness Analysis by Product
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by End Use , 2026 and 2036
- Figure 56: South Asia and Pacific Market Y-o-Y Growth Comparison by End Use , 2026-2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by End Use
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 59: South Asia and Pacific Market Y-o-Y Growth Comparison by Product, 2026-2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by Product
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by End Use , 2026 and 2036
- Figure 63: Middle East & Africa Market Y-o-Y Growth Comparison by End Use , 2026-2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by End Use
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 66: Middle East & Africa Market Y-o-Y Growth Comparison by Product, 2026-2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by Product
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis

