Endoscope Protective Barrier Covers Market
This report provides a rigorous examination of the endoscope protective barrier covers market, spanning market size analysis, revenue predictions, competitive structure, demand forecasts, key drivers, industry challenges, Applications developments, End User developments, supply chain trends, and future growth avenues.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Endoscope Protective Barrier Covers Market Size, Market Forecast and Outlook By FMI
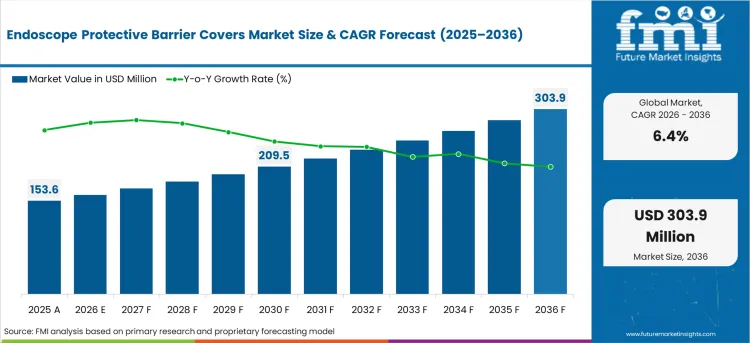
The endoscope protective barrier covers market was valued at USD 153.60 million in 2025, projected to reach USD 163.43 million in 2026, and is forecast to expand to USD 303.91 million by 2036 at a 6.4% CAGR. As per FMI, growing infection prevention awareness, rising endoscope reprocessing compliance costs, and increasing adoption of single-use barrier solutions in outpatient ENT and gastroenterology settings are sustaining demand for disposable sterile covers that eliminate cross-contamination risk during endoscopic procedures. Barrier covers provide a cost-effective infection prevention layer that reduces the reprocessing burden on sterile processing departments while maintaining endoscope optical and functional performance.
Summary of Endoscope Protective Barrier Covers Market
- Market snapshot
- The endoscope protective barrier covers market is valued at USD 153.60 million in 2025 and is projected to reach USD 303.91 million by 2036.
- The industry is expected to grow at a 6.4% CAGR from 2026 to 2036, creating an incremental opportunity of USD 140.48 million.
- The market is an infection prevention consumable category where reprocessing cost avoidance, cross-contamination risk elimination, and outpatient procedural throughput economics define procurement decisions.
- Demand and growth drivers
- Demand is rising as infection prevention protocols and reprocessing compliance costs drive adoption of single-use barrier covers that eliminate cross-contamination risk.
- ENT office-based and clinic settings adopt barrier covers to enable sequential procedures without intermediate reprocessing, increasing per-session throughput.
- Ambulatory surgical centres are minimising reprocessing capital investment by adopting barrier-based infection prevention for compatible endoscope types.
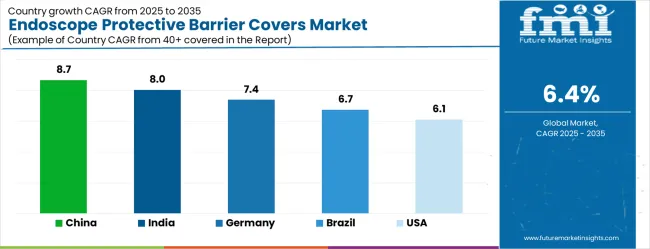
- Among key countries, China leads at 8.7% CAGR, followed by India at 8.0%, Germany at 7.4%, Brazil at 6.7%, the USA at 6.1%, the UK at 5.5%, and Japan at 4.8%.
- Product and segment view
- Endoscope protective barrier covers are single-use, sterile sheaths designed to encase endoscope insertion tubes during procedures, preventing direct contact between the endoscope surface and patient tissue.
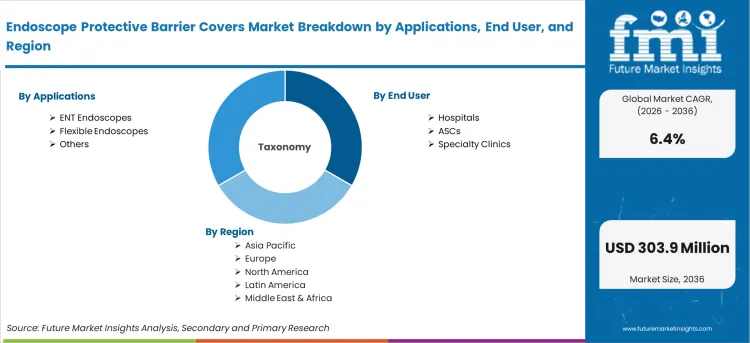
- Applications include ENT endoscopes, flexible endoscopes, and other rigid and semi-rigid endoscope types.
- ENT Endoscopes leads by Applications with 45.3% share in 2026 as otolaryngology office-based procedures represent the highest-volume barrier cover use case.
- Hospitals leads by End User with 50.7% share in 2026 as inpatient endoscopy suites integrate barrier covers into infection prevention protocols alongside standard reprocessing.
- Scope includes disposable sterile endoscope barrier covers; excludes reusable endoscope sheaths, endoscope reprocessing chemicals, and single-use endoscopes.
- Geography and competitive outlook
- China and India are the fastest-growing markets due to endoscopy infrastructure expansion and infection control compliance adoption. The USA and Germany anchor mature, guideline-driven demand.
- Competition is shaped by optical clarity preservation, material biocompatibility, and compatibility across endoscope OEM platforms.
- Key players include Cover S.r.l, Cogentix Medical, Exact Medical, HIWIN Healthcare, Locamed, Fairmont Medical Products, Femcare-Nikomed, MetroMed Healthcare, and Plasti-MedScientific.
- Analyst opinion at FMI
- Sabyasachi Ghosh, Principal Consultant for Healthcare and Medical Devices, opines: 'As per FMI, endoscope protective barrier covers are transitioning from a niche infection prevention accessory to a standard-of-care consumable in outpatient ENT and ASC settings. Facilities that rely solely on reprocessing without barrier cover supplementation face compounding sterile processing labour costs and audit risk from incomplete channel cleaning documentation. Barrier cover suppliers without validated compatibility across the installed base of major endoscope OEM platforms will experience limited formulary inclusion as procurement committees standardise approved accessory lists.'
- Strategic implications / Executive takeaways
- ENT practice managers must evaluate barrier cover adoption as a reprocessing cost avoidance strategy that simultaneously increases per-session procedural throughput in office-based settings.
- Infection control officers should integrate barrier covers into endoscopy suite protocols as a supplementary contamination prevention layer for procedures where full reprocessing turnaround creates scheduling bottlenecks.
- Procurement teams must verify barrier cover compatibility across the facility's installed endoscope OEM platforms before formulary standardisation.
Endoscope Protective Barrier Covers Market key takeaways
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 163.43 million |
| Industry Value (2036) | USD 303.91 million |
| CAGR (2026 to 2036) | 6.4% |
Source: Future Market Insights, 2026
FMI analysts observe that ENT endoscopes dominate the application segment because otolaryngology procedures frequently occur in clinic and office-based settings where automated reprocessing infrastructure is limited or absent. Disposable barrier covers enable ENT practitioners to perform sequential nasal, laryngeal, and otologic examinations without intermediate reprocessing steps, reducing patient wait times and increasing per-session procedural throughput. The parallel growth of ambulatory surgical centre (ASC) volumes is creating incremental demand as these facilities seek to minimise capital investment in reprocessing equipment by adopting barrier-based infection prevention for compatible endoscope types.
China leads with an 8.7% CAGR through 2036 as hospital construction programmes expand endoscopy suites and infection control standards tighten. India follows at 8.0%, driven by outpatient ENT clinic proliferation and growing NABH accreditation compliance. Germany registers a 7.4% pace as EU MDR reprocessing validation costs push facilities toward barrier-based alternatives. Brazil expands at 6.7% on the back of ambulatory surgical centre growth. The United States tracks at 6.1%, reflecting mature hospital adoption with incremental growth from ASC and office-based ENT procedure expansion. The United Kingdom maintains a 5.5% trajectory driven by NHS decontamination cost reduction initiatives. Japan expands at 4.8%, anchored by its ageing population driving ENT and gastroenterology procedure volumes.
Endoscope Protective Barrier Covers Market definition
Endoscope protective barrier covers are single-use, sterile polymer sheaths designed to encase endoscope insertion tubes during diagnostic and therapeutic procedures. These covers prevent direct contact between the endoscope surface and patient tissue, reducing cross-contamination risk and supplementing or replacing full reprocessing cycles in outpatient, office-based, and ambulatory surgical centre settings.
Endoscope Protective Barrier Covers Market inclusions
Market scope includes disposable sterile barrier covers for ENT endoscopes, flexible endoscopes, and other compatible endoscope types. The report covers global and regional market sizes for 2026 to 2036 with segment breakdowns by application and end user.
Endoscope Protective Barrier Covers Market exclusions
The scope excludes reusable endoscope sheaths, endoscope reprocessing chemicals and equipment, fully single-use disposable endoscopes, and endoscope repair and maintenance services.
Endoscope Protective Barrier Covers Market research methodology
- Primary research: Analysts engaged with ENT practice managers, sterile processing directors, and infection control officers to map barrier cover adoption triggers and procurement decision criteria.
- Desk research: Data collection aggregated infection prevention accreditation standards, EU MDR reprocessing cost analyses, and published endoscopic procedure volume statistics by care setting.
- Market sizing and forecasting: Baseline values derive from a bottom-up aggregation of barrier cover consumption per endoscopic procedure, applying procedure volume growth curves and outpatient setting penetration rates.
- Data validation and update cycle: Projections are cross-validated against published medical device company accessory revenue disclosures and endoscopy facility survey data.
Why is the Endoscope Protective Barrier Covers Market Growing?
The Endoscope Protective Barrier Covers market is witnessing robust growth, driven by the increasing adoption of minimally invasive procedures and heightened focus on infection control in healthcare facilities. Rising awareness of hospital-acquired infections and stringent hygiene regulations are prompting healthcare providers to implement protective barriers for endoscopic procedures. The market is being further supported by advancements in materials technology, enabling covers that offer enhanced durability, flexibility, and compatibility with various endoscope types.
Growing emphasis on patient safety, combined with the need to reduce sterilization costs and turnaround time, has increased the adoption of disposable protective barrier covers. Hospitals and outpatient centers are increasingly investing in these solutions to maintain compliance with healthcare standards and improve procedural efficiency.
The rising prevalence of diagnostic and surgical endoscopic procedures across ENT, gastroenterology, and urology specialties is contributing to market expansion As hospitals and clinics continue to prioritize hygiene, patient safety, and operational efficiency, the Endoscope Protective Barrier Covers market is expected to sustain significant growth in the coming years.
Segmental Analysis
The endoscope protective barrier covers market is segmented by applications, end user, and geographic regions. By applications, endoscope protective barrier covers market is divided into ENT Endoscopes, Flexible Endoscopes, and Others. In terms of end user, endoscope protective barrier covers market is classified into Hospitals, ASCs, and Specialty Clinics. Regionally, the endoscope protective barrier covers industry is classified into North America, Latin America, Western Europe, Eastern Europe, Balkan & Baltic Countries, Russia & Belarus, Central Asia, East Asia, South Asia & Pacific, and the Middle East & Africa.
Insights into the ENT Endoscopes Applications Segment
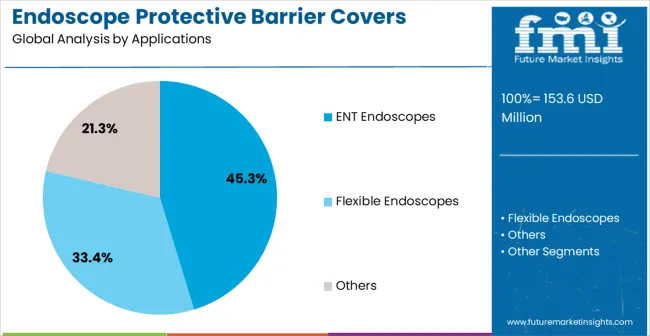
The ENT endoscopes application segment is projected to hold 45.3% of the market revenue in 2026, establishing it as the leading application area. Growth in this segment is being driven by the rising number of ear, nose, and throat procedures requiring high levels of hygiene and infection prevention. Protective barrier covers for ENT endoscopes minimize cross-contamination risks and reduce the need for repeated sterilization, enhancing procedural efficiency and patient safety.
Compatibility with different endoscope models and advancements in cover materials, including high-quality plastics and polymers, have strengthened adoption. Hospitals and specialty clinics are increasingly deploying ENT endoscope covers to comply with infection control protocols and improve workflow efficiency.
The segment’s expansion is further supported by rising patient awareness of hygiene standards and regulatory guidelines emphasizing single-use or disposable barriers As ENT procedures continue to increase globally, the application segment is expected to maintain its leadership, driven by the need for reliable, safe, and cost-effective protective solutions.
Insights into the Hospitals End User Segment
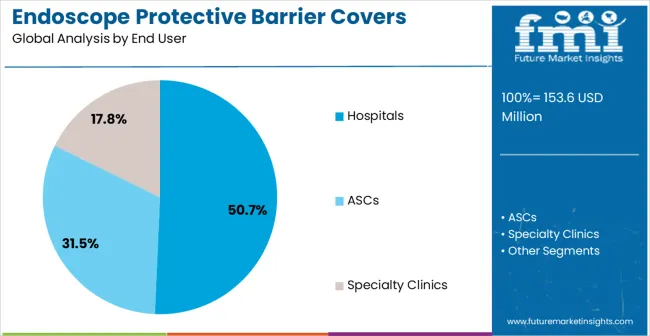
The hospitals end user segment is expected to account for 50.7% of the market revenue in 2026, making it the leading end-use category. Growth is being driven by hospitals’ increasing focus on patient safety, hygiene standards, and operational efficiency. Protective barrier covers for endoscopes reduce cross-contamination risks, improve turnaround times for procedures, and ensure compliance with infection control protocols.
The adoption of disposable and high-quality materials that offer flexibility and durability has further accelerated usage in hospital settings. Rising investments in healthcare infrastructure, growing prevalence of endoscopic procedures, and stringent regulatory guidelines are reinforcing the segment’s leadership.
Hospitals are leveraging these solutions to manage high patient volumes efficiently while maintaining hygiene standards As the demand for minimally invasive procedures and infection prevention continues to grow, the hospital segment is expected to remain the largest contributor to market revenue, supported by continuous innovation in barrier cover materials, design, and procedural compatibility.
Market Overview
Endoscope Protective Barrier Covers Market Overview
The endoscope protective barrier covers act as a shielding fence to the endoscope, mainly prevents from cross-contamination and the spread of dangerous microorganisms. These endoscope covers have the properties of flexibility and antimicrobial protective mechanism. Endoscope covers are totally sterile, a product with an integrated working channel between the patient and the endoscope.
The disposable endoscope protective barrier comprises of an operative channel for suction, irrigation, and tool passageway, doctors may use the covers during a full range of therapeutic & diagnostic procedures. The endoscope cover is available in numerous functioning channel sizes, letting the physician to acclimatize the possibility to fit the diagnostic procedure.
By using the endoscope barrier cover, it eliminates the need for conventional high-level disinfection/sterilization routines between each procedure. Instead, the physician get a safe, sterile protective barrier for each patient and rapid equipment turnaround for the practice. The endoscope protective barrier covers market has evolved gradually and comprises of different varieties of sizes used during endoscopy procedures.
The market is anticipated to grow with fastest CAGR due to increase in the endoscopy diagnostic and therapeutic procedures coupled with few cases with cross contamination in the USA and Europe.
Analysis of Endoscope Protective Barrier Covers Market By Key Countries
.webp)
| Country | CAGR |
|---|---|
| China | 8.7% |
| India | 8.0% |
| Germany | 7.4% |
| Brazil | 6.7% |
| USA | 6.1% |
| UK | 5.5% |
| Japan | 4.8% |
Source: FMI analysis based on primary research and proprietary forecasting model
Country-wise Analysis
The Endoscope Protective Barrier Covers Market is expected to register a CAGR of 6.4% during the forecast period, exhibiting varied country level momentum. China leads with the highest CAGR of 8.7%, followed by India at 8.0%. Developed markets such as Germany, France, and the UK continue to expand steadily, while the USA is likely to grow at consistent rates. Japan posts the lowest CAGR at 4.8%, yet still underscores a broadly positive trajectory for the global Endoscope Protective Barrier Covers Market. In 2024, Germany held a dominant revenue in the Western Europe market and is expected to grow with a CAGR of 7.4%. The USA Endoscope Protective Barrier Covers Market is estimated to be valued at USD 53.0 million in 2026 and is anticipated to reach a valuation of USD 53.0 million by 2036. Sales are projected to rise at a CAGR of 0.0% over the forecast period between 2026 and 2036. While Japan and South Korea markets are estimated to be valued at USD 8.1 million and USD 4.5 million respectively in 2026.
Key Players in the Endoscope Protective Barrier Covers Market
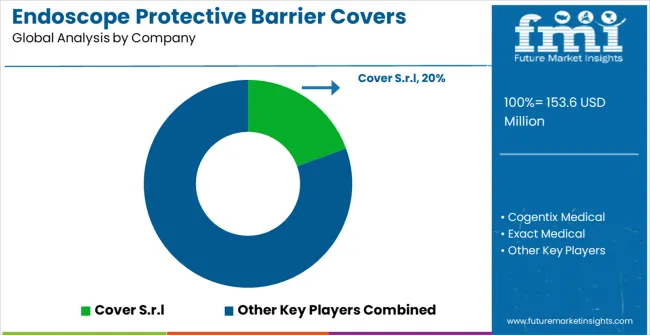
- Cover S.r.l
- Cogentix Medical
- Exact Medical
- HIWIN Healthcare
- Locamed
- Fairmont Medical Products
- Femcare-Nikomed
- MetroMed Healthcare
- Plasti-MedScientific
Scope of the report
| Metric | Value |
|---|---|
| Quantitative Units | USD 163.43 million to USD 303.91 million, at a CAGR of 6.4% |
| Market Definition | Endoscope protective barrier covers are single-use sterile sheaths that prevent cross-contamination during endoscopic procedures by encasing insertion tubes. |
| Applications Segmentation | ENT Endoscopes, Flexible Endoscopes, Others |
| End User Segmentation | Hospitals, ASCs, Specialty Clinics |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia, Oceania, Middle East & Africa |
| Countries Covered | China, India, Germany, Brazil, USA, UK, Japan, and 40 plus countries |
| Key Companies Profiled | Cover S.r.l, Cogentix Medical, Exact Medical, HIWIN Healthcare, Locamed, Fairmont Medical Products, Femcare-Nikomed, MetroMed Healthcare, Plasti-MedScientific |
| Forecast Period | 2026 to 2036 |
| Approach | Bottom-up methodology starting with barrier cover consumption per procedure, projecting procedure volume growth and outpatient setting penetration. |
Endoscope Protective Barrier Covers Market by Segments
Applications:
- ENT Endoscopes
- Flexible Endoscopes
- Others
End User:
- Hospitals
- ASCs
- Specialty Clinics
Region:
- North America
- USA
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Western Europe
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- Eastern Europe
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- East Asia
- China
- Japan
- South Korea
- South Asia and Pacific
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
Bibliography
- 1. World Health Organization. (2024). Decontamination and reprocessing of medical devices for healthcare facilities. WHO.
- 2. European Commission. (2024). EU Medical Device Regulation 2017/745: Single-use device and reprocessing requirements. European Commission.
- 3. United States Food and Drug Administration. (2024). Reprocessing of reusable medical devices: Guidance for barrier cover use. FDA.
- 4. National Accreditation Board for Hospitals, India. (2024). NABH infection control standards: Endoscope handling requirements. NABH.
- 5. Society of Gastroenterology Nurses and Associates. (2024). Standards of infection prevention in reprocessing. SGNA.
- 6. Organisation for Economic Co-operation and Development. (2024). Health at a Glance 2024: Ambulatory procedure volume trends. OECD.
This bibliography is provided for reader reference. The full Future Market Insights report contains the complete reference list with primary research documentation.
Frequently Asked Questions
How large is the demand for endoscope protective barrier covers in 2026?
Demand for endoscope protective barrier covers in the global market is estimated to be valued at USD 163.43 million in 2026.
What will be the market size by 2036?
Market size for endoscope protective barrier covers is projected to reach USD 303.91 million by 2036.
What is the expected demand growth between 2026 and 2036?
Demand for endoscope protective barrier covers is expected to grow at a CAGR of 6.4% between 2026 and 2036.
Which Application leads in 2026?
ENT Endoscopes accounts for 45.3% in 2026 as otolaryngology office-based procedures represent the highest-volume barrier cover use case.
Which End User leads in 2026?
Hospitals accounts for 50.7% in 2026 as inpatient endoscopy suites integrate barrier covers into infection prevention protocols.
What is driving demand in China?
Hospital construction programmes expanding endoscopy suites and tightening infection control standards are increasing barrier cover consumption.
What is included in the scope?
The scope covers disposable sterile barrier covers; excludes reusable sheaths, reprocessing chemicals, and single-use endoscopes.
Table of Content
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Applications
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Applications , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Applications , 2026 to 2036
- ENT Endoscopes
- Flexible Endoscopes
- Others
- ENT Endoscopes
- Y to o to Y Growth Trend Analysis By Applications , 2021 to 2025
- Absolute $ Opportunity Analysis By Applications , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User, 2026 to 2036
- Hospitals
- ASCs
- Specialty Clinics
- Hospitals
- Y to o to Y Growth Trend Analysis By End User, 2021 to 2025
- Absolute $ Opportunity Analysis By End User, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Applications
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Applications
- By End User
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Applications
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Applications
- By End User
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Applications
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Applications
- By End User
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Applications
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Applications
- By End User
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Applications
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Applications
- By End User
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Applications
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Applications
- By End User
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Applications
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Applications
- By End User
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Applications
- By End User
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Applications
- By End User
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Applications
- By End User
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Applications
- By End User
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Applications
- By End User
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Applications
- By End User
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Applications
- By End User
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Applications
- By End User
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Applications
- By End User
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Applications
- By End User
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Applications
- By End User
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Applications
- By End User
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Applications
- By End User
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Applications
- By End User
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Applications
- By End User
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Applications
- By End User
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Applications
- By End User
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Applications
- By End User
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Applications
- By End User
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Applications
- By End User
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Applications
- By End User
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Applications
- By End User
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Applications
- By End User
- Competition Analysis
- Competition Deep Dive
- Cover S.r.l
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Cogentix Medical
- Exact Medical
- HIWIN Healthcare
- Locamed
- Fairmont Medical Products
- Femcare-Nikomed
- MetroMed Healthcare
- Plasti-MedScientific
- Cover S.r.l
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Applications , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Applications , 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by Applications , 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by Applications , 2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Applications , 2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by Applications , 2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Applications , 2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Applications , 2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by End User, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Applications , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Applications , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Applications
- Figure 6: Global Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by End User
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by Applications , 2026 and 2036
- Figure 21: North America Market Y-o-Y Growth Comparison by Applications , 2026-2036
- Figure 22: North America Market Attractiveness Analysis by Applications
- Figure 23: North America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 25: North America Market Attractiveness Analysis by End User
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by Applications , 2026 and 2036
- Figure 28: Latin America Market Y-o-Y Growth Comparison by Applications , 2026-2036
- Figure 29: Latin America Market Attractiveness Analysis by Applications
- Figure 30: Latin America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 31: Latin America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 32: Latin America Market Attractiveness Analysis by End User
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by Applications , 2026 and 2036
- Figure 35: Western Europe Market Y-o-Y Growth Comparison by Applications , 2026-2036
- Figure 36: Western Europe Market Attractiveness Analysis by Applications
- Figure 37: Western Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 38: Western Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 39: Western Europe Market Attractiveness Analysis by End User
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Applications , 2026 and 2036
- Figure 42: Eastern Europe Market Y-o-Y Growth Comparison by Applications , 2026-2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by Applications
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 45: Eastern Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by End User
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by Applications , 2026 and 2036
- Figure 49: East Asia Market Y-o-Y Growth Comparison by Applications , 2026-2036
- Figure 50: East Asia Market Attractiveness Analysis by Applications
- Figure 51: East Asia Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 52: East Asia Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 53: East Asia Market Attractiveness Analysis by End User
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Applications , 2026 and 2036
- Figure 56: South Asia and Pacific Market Y-o-Y Growth Comparison by Applications , 2026-2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Applications
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 59: South Asia and Pacific Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by End User
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Applications , 2026 and 2036
- Figure 63: Middle East & Africa Market Y-o-Y Growth Comparison by Applications , 2026-2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Applications
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 66: Middle East & Africa Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by End User
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis

