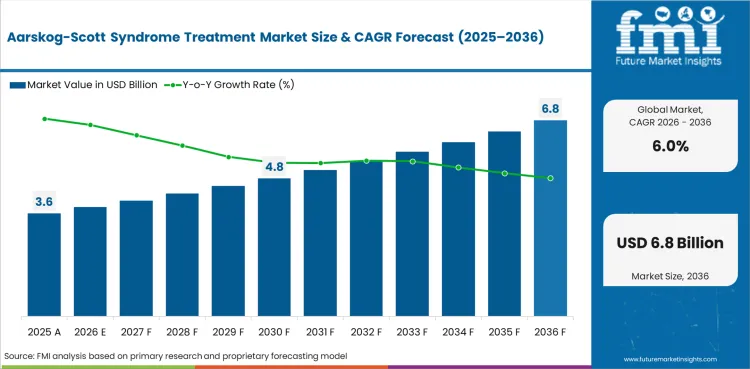
The Aarskog-Scott syndrome treatment market covers pharmaceutical and therapeutic interventions used in managing the symptoms of Aarskog-Scott syndrome, a rare X-linked genetic disorder affecting facial features, skeletal development, and growth. Treatments include growth hormone therapy, surgical interventions, orthodontic management, and supportive therapies.
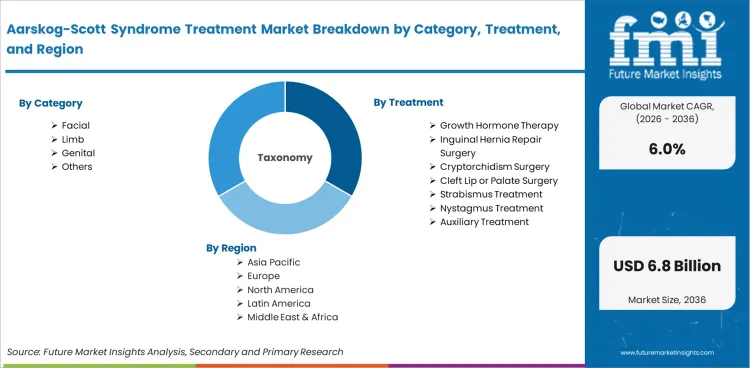
Market scope covers all commercially available Aarskog-Scott syndrome treatments categorized by category (facial, skeletal, growth, genital, others), treatment (growth hormone therapy, surgical intervention, orthodontic management, supportive therapy, others), end use (hospitals, clinics, homecare, others), and diagnosis (genetic testing, clinical assessment, imaging, others). Revenue coverage spans 2026 to 2036.
The scope does not include general pediatric growth monitoring services, genetic counseling services billed separately, or educational support services.
Demand for Aarskog-Scott syndrome treatment reflects improving genetic diagnostic capabilities and the growing recognition of rare genetic conditions in pediatric clinical practice. The disorder presents with variable severity, and treatment approaches are tailored to individual symptom profiles including growth deficiency, facial dysmorphology, and skeletal abnormalities.
Adoption of growth hormone therapy represents 29.6% of treatment demand, reflecting its role in addressing the short stature that is a common feature of the syndrome. Growth hormone treatment protocols follow established pediatric endocrinology guidelines, with treatment typically initiated in early childhood and continuing through the growth period.
Pricing reflects the combination of growth hormone therapy costs, surgical intervention fees, and supportive therapy expenses. Growth hormone therapy represents the most significant recurring pharmaceutical cost, while surgical and orthodontic interventions are typically one-time or limited-duration expenses within the overall treatment pathway.
The aarskog-scott syndrome treatment market is segmented by category, treatment, end use, and diagnosis.
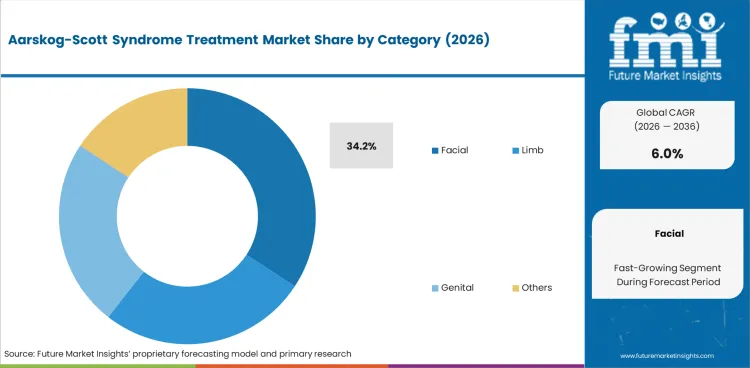
Facial manifestations represent 34.2% of category demand, reflecting the clinical importance of managing craniofacial features including broad forehead, widely spaced eyes, and small nose that characterize the syndrome. Surgical and orthodontic interventions address functional and aesthetic aspects of facial involvement.
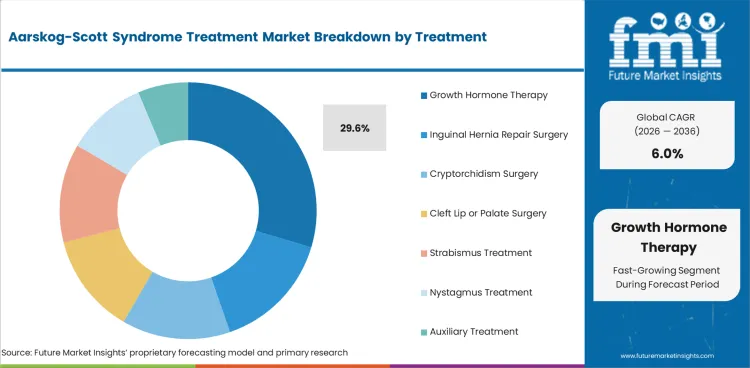
Growth hormone therapy represents 29.6% of treatment demand, addressing the short stature that is a common clinical feature. Treatment follows pediatric endocrinology guidelines with monitoring of growth velocity, bone age, and hormonal response to optimize growth outcomes.
The aarskog-scott syndrome treatment market is shaped by regulatory, technology, and procurement dynamics.
Demand is shaped by expanding genetic testing capabilities that increase Aarskog-Scott syndrome case identification. Whole exome sequencing and targeted FGD1 gene testing enable definitive molecular diagnosis that supports treatment initiation.
Growth reflects the expansion of rare disease treatment programs that improve pharmaceutical access and insurance coverage for orphan conditions including Aarskog-Scott syndrome.
Adoption of growth hormone therapy is supported by established pediatric endocrinology treatment standards that provide clinical guidelines for managing growth deficiency in genetic short stature conditions.
The small patient population limits commercial incentives for condition-specific drug development. Treatment relies on therapeutic approaches developed for broader indications, including growth hormone therapy originally developed for growth hormone deficiency.
.webp)
| Country | CAGR |
|---|---|
| USA | 6.2% |
| South Korea | 6.1% |
| EU | 6.0% |
| UK | 5.9% |
| Japan | 5.9% |
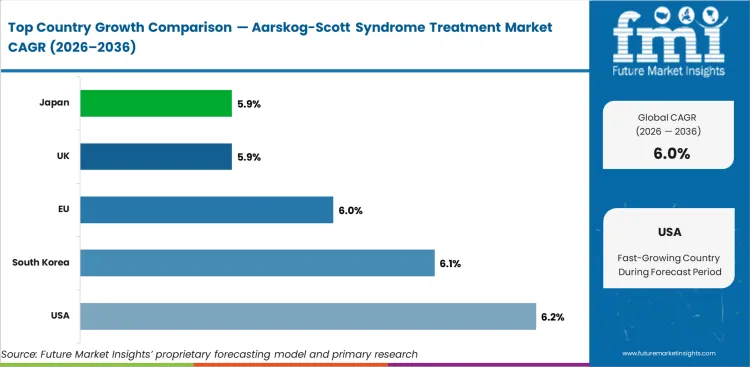
The global aarskog-scott syndrome treatment market is expected to grow at 6.0% per year from 2026 to 2036.
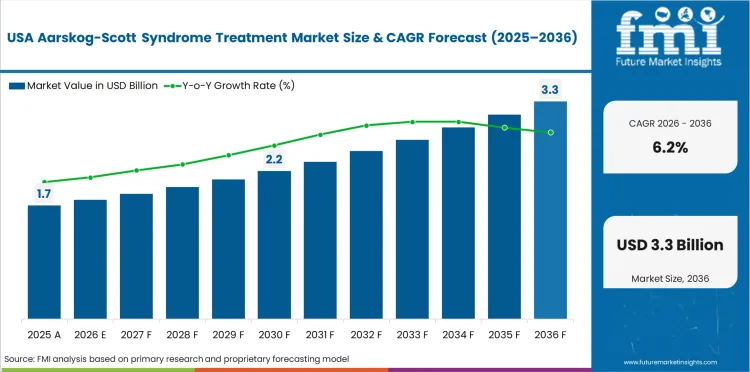
The USA is expected to grow at 6.2% through 2036, driven by advanced genetic diagnostic infrastructure, established pediatric endocrinology networks, and insurance coverage for rare disease treatments.
South Korea is expected to grow at 6.1% through 2036, supported by advanced genetic testing capabilities, growing rare disease awareness, and government health insurance coverage for orphan disease treatments.
Opportunity Analysis of Aarskog-Scott Syndrome Treatment Market in the European Union
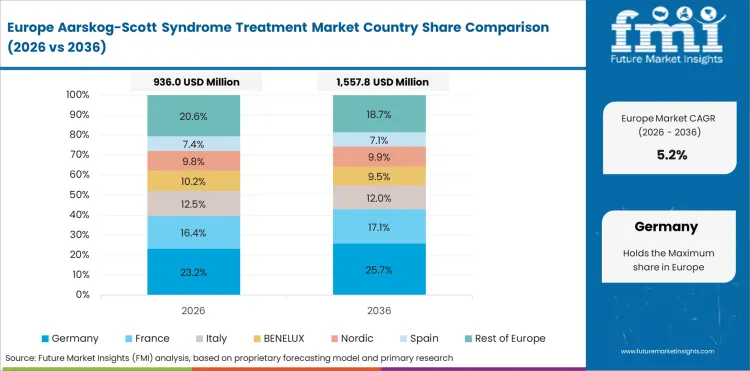
The EU is expected to grow at 6.0% through 2036, supported by EU rare disease policy frameworks, cross-border rare disease treatment networks, and genetic diagnostic standardization.
The UK is expected to grow at 5.8% through 2036, supported by NHS genomic medicine services, rare disease clinical networks, and NICE-guided treatment pathways.
Japan is expected to grow at 5.9% through 2036, driven by advanced genetic medicine infrastructure, pediatric specialty treatment centres, and government rare disease support programs.
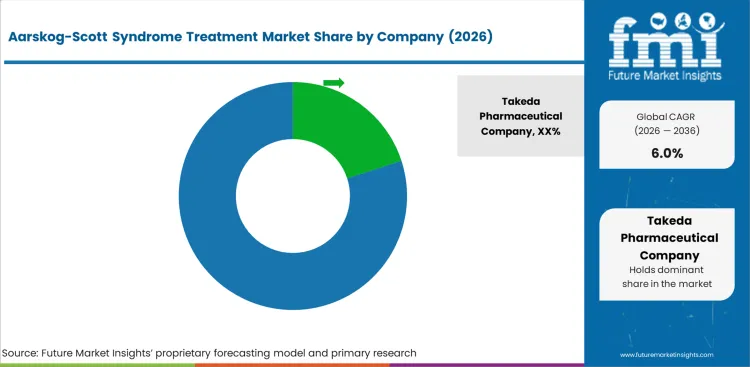
Takeda Pharmaceutical Company leads through its rare disease portfolio and established relationships with pediatric treatment centres that serve genetic condition management.
Sanofi Genzyme and Pfizer Inc. maintain positions through growth hormone therapy portfolios and rare disease treatment programs that include Aarskog-Scott syndrome supportive care.
Novartis AG and Orphan Biovitrum AB compete on rare disease pharmaceutical development and orphan drug access programs.
Entry barriers include the specialized rare disease clinical knowledge required, small patient populations that limit commercial return, and the established prescribing relationships between pediatric endocrinologists and incumbent growth hormone manufacturers.
Key global companies leading the aarskog-scott syndrome treatment market include:
| Company | Rare Disease Portfolio | Growth Hormone Products | Patient Access Programs | Geographic Reach |
|---|---|---|---|---|
| Takeda Pharmaceutical Company | High | Medium | Strong | Global |
| Sanofi Genzyme | High | High | Strong | Global |
| Pfizer Inc. | Medium | High | Strong | Global |
| Novartis AG | High | Medium | Strong | Global |
| Orphan Biovitrum AB | Medium | Low | Moderate | Europe |
| Novo Nordisk A/S | Low | High | Strong | Global |
Source: Future Market Insights competitive analysis, 2026.
Key Developments in Aarskog-Scott Syndrome Treatment Market
Major Global Players
Emerging Players/Startups
| Parameter | Details |
|---|---|
| Quantitative Units | USD 3.82 billion to USD 6.83 billion, at a CAGR of 6.0% |
| Market Definition | The Aarskog-Scott syndrome treatment market covers pharmaceutical and therapeutic interventions used in managing the symptoms of Aarskog-Scott syndrome, a rare X-linked genetic disorder affecting facial features, skeletal development, and growth. Treatments include growth hormone therapy, surgical interventions, orthodontic management, and supportive therapies. |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia and Pacific, Middle East and Africa |
| Countries Covered | USA, UK, France, Germany, Italy, South Korea, Japan, China, India, 30 plus countries |
| Key Companies Profiled | Takeda Pharmaceutical Company, Sanofi Genzyme, Pfizer Inc., Novartis AG, Orphan Biovitrum AB, BioMarin Pharmaceutical, Ultragenyx Pharmaceutical, Rare Disease Therapeutics, Medtronic, Shire Pharmaceuticals, Other Key Players |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid bottom-up and top-down methodology starting with verified transaction data, projecting adoption velocity across segments and regions. |
This bibliography is provided for reader reference. The full Future Market Insights report contains the complete reference list with publication dates, URLs, and supporting data for all cited works.
What is the global market demand for Aarskog-Scott Syndrome Treatment in 2026?
In 2026, the global market is expected to be worth USD 3.82 billion.
How big will the Aarskog-Scott Syndrome Treatment Market be in 2036?
By 2036, the market is expected to be worth USD 6.83 billion.
How much is demand expected to grow between 2026 and 2036?
Between 2026 and 2036, demand is expected to grow at a CAGR of 6.0%.
Which Category segment is expected to lead in 2026?
Facial is expected to account for 34.2% of the segment in 2026.
What is causing demand to rise in USA?
USA is expected to grow at 6.2% through 2036.
What is causing demand to rise in South Korea?
South Korea is expected to grow at 6.1% through 2036.
What does this report mean by Aarskog-Scott Syndrome Treatment Market definition?
The Aarskog-Scott syndrome treatment market covers pharmaceutical and therapeutic interventions used in managing the symptoms of Aarskog-Scott syndrome, a rare X-linked genetic disorder affecting faci.
How does FMI make the forecast?
Forecasting uses a hybrid bottom-up and top-down methodology starting with verified transaction data, projecting adoption velocity across segments and regions.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.