Thoracentesis and Paracentesis Market
Thoracentesis and Paracentesis Market Forecast and Outlook 2026 to 2036
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Thoracentesis and Paracentesis Market Forecast and Outlook 2026 to 2036
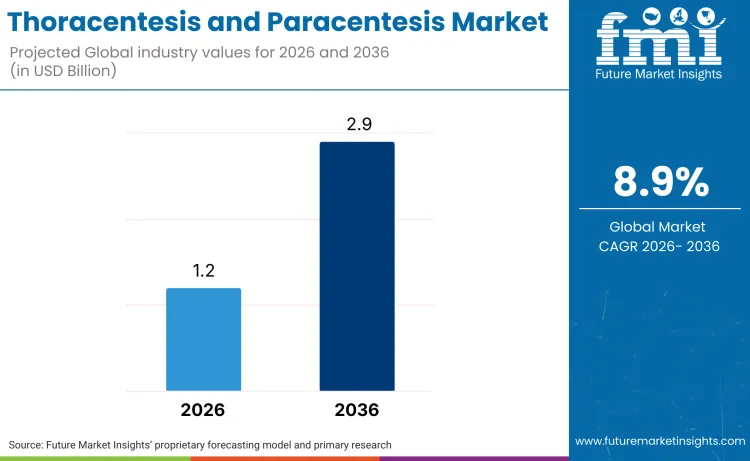
The global thoracentesis and paracentesis market is predicted to reach USD 1.2 billion in 2026, at 8.9% CAGR. The thoracentesis and paracentesis market is projected to reach USD 2.9 billion as adoption shifts toward automated, closed-loop systems and handheld robotics that improve precision in complex GI and oncological interventions. Expansion is fundamentally driven by the escalating prevalence of chronic liver conditions, where the Centers for Disease Control and Prevention reported in 2024 that 4.5 million adults in the United States alone have been diagnosed with liver disease. Such high morbidity rates create a continuous baseline demand for reliable drainage procedures and closed loop centesis systems that ensure patient safety during fluid removal.
Manufacturers are responding by integrating interventional tools with advanced monitoring capabilities to manage these complex cases. Tom Polen, CEO of BD, validated this strategic direction in June 2024: "Critical Care expands BD's portfolio of smart connected care solutions with its growing set of leading monitoring technologies, advanced AI-enabled clinical decision tools and robust innovation pipeline. We believe the combination unlocks multiple new avenues for growth and value creation."
Rising complications such as ascites, which now affects approximately 60,000 per 100,000 individuals worldwide according to National Institutes of Health data from January 2025, are forcing a shift toward minimally invasive micro-access tools. Clinical requirements for managing malignant ascites are evolving rapidly, pushing hospitals to procure devices that minimize infection risks in outpatient settings. Merit Medical responded to this specific clinical need by launching the Micro ACE Advanced Micro-Access System in 2024, creating a new standard for precision in soft tissue interventions.
Companies that can deliver enhanced safety profiles while reducing procedural time are securing contracts with major ambulatory surgery centers. FMI analysis indicates that the integration of safety-engineered needles and catheter kits will determine market leadership as healthcare providers prioritize infection control over unit cost in the coming decade.
Summary of Thoracentesis and Paracentesis Market
- The thoracentesis and paracentesis market comprises thoracentesis kits, paracentesis kits, and drainage catheters used for therapeutic and diagnostic pleural and peritoneal fluid removal across hospital and outpatient settings.
- The defined scope is structured under FMI taxonomy with indication mapping across ascites, pleural effusion, empyema, and related conditions, excluding capital imaging systems and non device procedural revenue.
- The thoracentesis and paracentesis market is projected to grow at a CAGR of 8.9% from 2026 to 2036, expanding from USD 1.2 billion in 2026 to USD 2.9 billion by 2036, based on FMI proprietary bottom up revenue modeling, historical procedure volume analysis, and product mix forecasting.
- The sector is transitioning toward procedure specific, safety engineered, and image compatible centesis systems integrated into outpatient workflows, supported by primary interviews with manufacturers and clinicians and scenario modeling aligned with safety device penetration.
- Competition is concentrated among major players such as BD, Teleflex, and Merit Medical, with consolidation and strategic acquisitions strengthening portfolios, expanding bundled centesis offerings, and reinforcing entry barriers across hospital and ambulatory procurement channels.
- India is projected to record the fastest growth at 11.7% CAGR through 2036, while the United States retains the largest value share supported by favorable outpatient reimbursement frameworks and high chronic liver disease and oncology related procedural volumes.
Thoracentesis and Paracentesis Market Key Takeaways
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 1.2 billion |
| Industry Value (2036) | USD 2.9 billion |
| CAGR (2026-2036) | 8.9% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Which Factors Drive Thoracentesis And Paracentesis Market Growth?
Chronic liver disease is fundamentally reshaping the market, manifesting as a critical need for recurrent fluid management solutions. Companies such as Cook Medical validated this trend by launching its Interventional MRI (iMRI) division in October 2024, signaling that precise, image-guided intervention is becoming the standard of care for complex fluid collections. Structural support for this growth is robust, cementing the link between hepatology caseloads and device utilization.
How is the Thoracentesis And Paracentesis Market Segmented?
The thoracentesis and paracentesis market is segmented by product type, procedure, end user, and indication. By product, the market includes paracentesis kits, thoracentesis kits, and specialized drainage catheters designed for specific anatomical sites. By procedure, segmentation covers therapeutic and diagnostic interventions, reflecting the dual purpose of fluid removal for symptom relief and pathology analysis. By end user, adoption spans hospitals, ambulatory surgery centers, and specialty clinics, highlighting the migration of procedures to lower-cost outpatient environments. Segmentation analysis reveals a distinct preference for procedure-specific kits over individual components, driven by the need for efficiency in high-volume settings treating malignant pleural effusion.
Why do Paracentesis Kits hold a dominant share?
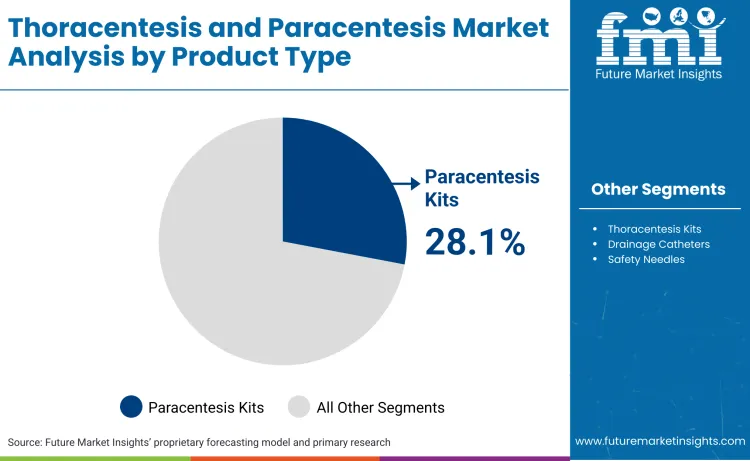
Paracentesis kits command a 28.1% market share, driven by the escalating burden of decompensated cirrhosis and the resulting fluid accumulation in the abdomen. A 2024 longitudinal study published in the National Institutes of Health database revealed a 50.5% increase in hospital admissions for ascites in surveyed regions compared to the previous period, validating the surge in kit procurement. The American Association for the Study of Liver Diseases (AASLD) 2024 guidelines emphasize large-volume paracentesis as the standard for refractory ascites, necessitating robust peritoneal catheter systems. This clinical necessity ensures sustained procurement of large-volume drainage kits in hospital settings, as providers seek all-in-one solutions that reduce preparation time and minimize sterility breaches during critical interventions.
Why does Ascites Drainage hold a Leading Share?
Ascites drainage applications command a leading 35.1% share in 2026 of the procedural market, driven by the chronic nature of liver decompensation which requires recurrent therapeutic intervention. This sustained demand stems directly from the high global prevalence of cirrhosis, where the National Institutes of Health confirmed in January 2025 that ascites affects nearly 60,000 per 100,000 individuals worldwide. Such widespread morbidity compels healthcare systems to adopt standardized malignant ascites management protocols, ensuring that drainage procedures remain the primary line of palliative care for end-stage liver disease. The shift toward frequent, volume-based intervention is further evidenced by Merit Medical's strategic focus on the Micro ACE system in 2024, which was specifically engineered to facilitate safer access for these high-frequency abdominal procedures. Manufacturers are prioritizing the development of infection-resistant drainage systems to support the lifelong management needs of hepatology patients, cementing this segment's market dominance.
Why Is Image Guidance Transforming Interventional Procedures?
Shift toward ultrasound and MRI-guided procedures is reducing complications like pneumothorax, effectively making blind insertion techniques obsolete. Cook Medical launched its Interventional MRI (iMRI) division in October 2024, partnering with Siemens Healthineers to enable real-time visualization during procedures involving image guided systems. Integrating visualization directly with drainage tools ensures higher success rates in complex anatomies. Pete Yonkman, President of Cook Medical, stated: "This division was born from that same source of insights, where our devices can partner with better visualization and precision guidance to improve patient outcomes." Adoption of these guided workflows forces smaller manufacturers to upgrade their portfolios or risk exclusion from premium hospital tenders.
How is Market Consolidation Influencing Critical Care Portfolios?
Major players are acquiring specialized assets to bundle drainage tools with comprehensive monitoring platforms. Becton Dickinson (BD) acquired Edwards Lifesciences' Critical Care group for USD 4.2 billion in June 2024, consolidating central venous catheter technology with interventional drainage assets. This strategic move indicates a landscape where standalone drainage tools are being absorbed into wider critical care ecosystems to improve hospital contracting efficiency. Tom Polen, CEO of BD, confirmed: "The transaction is expected to be immediately accretive to all key financial measures, including revenue growth, adjusted gross margin, adjusted operating margin and adjusted EPS, and enhances BD's portfolio of smart connected care solutions." Such consolidation creates high barriers to entry for niche players lacking broad distribution networks.
Why is the Industry expanding into Gastrointestinal Interventions?
Leading manufacturers are crossing over into GI and soft tissue markets to address complex fluid collections associated with abdominal cancers. Merit Medical acquired EndoGastric Solutions for USD 105 million in July 2024, expanding its footprint in the GI space and complementing its existing radiology drainage catheters. The NIH highlighted in 2025 that peritoneal carcinomatosis and GI malignancies account for 10% of ascites cases, creating a specific crossover niche for GI-focused interventional tools. By diversifying into these adjacent anatomical applications, companies buffer themselves against stagnation in standard procedure volumes. FMI analysis suggests that this cross-specialty expansion will define the next phase of product development, focusing on versatile tools capable of navigating complex abdominal structures.
How Will Thoracentesis and Paracentesis Market Expansion Unfold Across Key Global Regions?
Developed markets like the US and Germany are prioritizing outpatient efficiency and reimbursement optimization, while emerging economies are driven by raw procedural volume growth. India leads the global expansion with an 11.7% CAGR, followed closely by Brazil at 10.6%, outpacing the global average of 8.9%. Western markets are characterized by the rapid adoption of chest drainage system innovations that facilitate home care, whereas regions like South Asia are heavily investing in basic hospital infrastructure to manage rising non-communicable disease burdens.

| Country | CAGR (2026 to 2036) |
|---|---|
| India | 11.7% |
| Brazil | 10.6% |
| United States | 7.1% |
| China | 9.8% |
| Germany | 6.5% |
Source: FMI historical analysis and forecast data.
Why is India Emerging as a High-volume Hub?
The thoracentesis and paracentesis industry in India is poised to register a commanding 11.7% CAGR through 2036. This robust growth is directly fueled by the critical burden of liver disease and non-communicable conditions, which compels public health networks to upgrade their interventional infrastructure for fluid management. The Department of Pharmaceuticals Annual Report 2023-24 validated this momentum by highlighting a surge in investment opportunities specifically targeting chronic disease management, creating an acute demand for cost-effective catheter solutions. The Ministry of Health and Family Welfare further catalyzed this expansion in 2024 through active collaborations aimed at enhancing medical device access across tier-2 cities. These large-scale public health investments position India as the primary global volume engine for basic drainage consumables.
What Fuels the Public Sector Demand in Brazil?
Thoracentesis and paracentesis demand in Brazil is projected to expand at a 10.6% CAGR, outpacing regional averages due to a specific public health crisis regarding hepatology. This expansion is driven by the high prevalence of alcohol-related liver disease, which generates a consistent volume of hospital admissions requiring immediate ascites drainage. Manufacturers that can successfully navigate public tender procurements for reliable drainage catheter supplies will secure long-term revenue stability as the government prioritizes essential care access.
Why is the USA pivoting to outpatient reimbursement?
The USA remains the key market, projected to grow at a steady 7.1% CAGR. Growth is sustained by favorable regulatory adjustmentsthat incentivize the shift of interventional procedures to lower-cost ambulatory settings. This reimbursement change forces clinics to adopt efficient, all-in-one systems that streamline United States interventional radiology workflows and reduce bed turnaround times. Fred Lampropoulos, CEO of Merit Medical, noted the impact of this operational focus: “In 2024, Merit introduced the Continued Growth Initiatives (CGI) Program to support financial targets and build upon the success of the Foundations for Growth Program. As a result, we continue to strengthen profitability, deliver top-line growth, and drive continuous innovation in the marketplace.” Continued alignment with these evolving CPT codes will remain the single most critical factor for commercial success in this region.
How is the Competition in the Thoracentesis and Paracentesis Market?
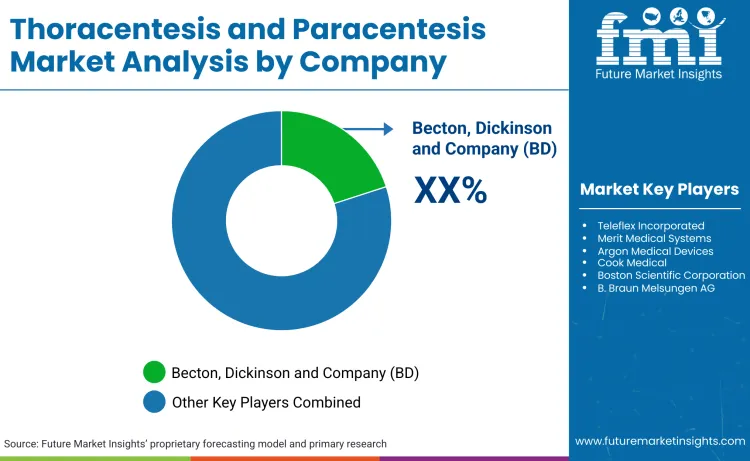
The thoracentesis and paracentesis market is highly concentrated, with BD commanding 15.4% share, followed by Teleflex and Merit Medical. Dominance is driven by scale, where BD's USD 4.2 billion acquisition of Edwards' Critical Care unit in 2024 reinforces its ability to bundle drainage kits with hemodynamic monitoring. Market consolidation is accelerating, as evidenced by Merit Medical's acquisition of both EndoGastric Solutions and Cook Medical's Lead Management portfolio in 2024, effectively squeezing out mid-tier competitors. Large incumbents leverage these comprehensive portfolios to secure hospital-wide contracts that cover everything from aspiration and biopsy needles to complex drainage systems.
Leading players are shifting toward "Smart Connected Care" and minimally invasive ecosystems. BD's 2024 strategy focuses on integrating AI-driven monitoring with physical intervention, moving beyond simple hardware. Merit Medical responded by expanding its specialized toolset with the launch of the Micro ACE system in 2024, targeting the high-precision needs of ultrasound guided breast biopsy and soft tissue access.
The advancements represented by BD and Merit Medical reflect a broader trend in the industry, where technology and care converge to create smart, connected ecosystems. As these companies and others innovate, the healthcare sector is poised to transition towards a future where minimally invasive techniques and intelligent monitoring are the norms, raising the standard of care delivered to patients globally.
Key Players in Thoracentesis and Paracentesis Market
- Becton, Dickinson and Company (BD)
- Teleflex Incorporated
- Merit Medical Systems
- Argon Medical Devices
- Cook Medical
- Boston Scientific Corporation
- B. Braun Melsungen AG
- Cardinal Health
- Rocket Medical
- Medtronic
Market Definition
The thoracentesis and paracentesis market represents revenue generated from the sale of medical devices, kits, and catheters specifically designed for the drainage of fluid from the pleural (thoracentesis) and peritoneal (paracentesis) cavities. As operationally defined in the article, the market measures commercial consumption of manual and vacuum-assisted drainage systems, safety needles, and procedure-specific trays used in therapeutic and diagnostic interventions. Market sizing reflects the value of consumables and disposable devices sold to healthcare facilities, analyzed by product type, indication, and end user, and expressed in USD billion.
The market includes comprehensive procedural kits, drainage catheters, collection bags, and safety-engineered needles explicitly segmented in the article. It covers applications for managing ascites, pleural effusion, empyema, and hemothorax, driven by underlying conditions such as liver cirrhosis, oncology, and heart failure. Revenue generated from sales to hospitals, ambulatory surgery centers, and specialty clinics is included. The scope counts adoption across key regions including North America, Europe, Asia Pacific, Latin America, and the Middle East & Africa.
The market excludes capital equipment such as ultrasound machines or MRI scanners used for image guidance, unless sold as an integral, non-separable part of a single-use drainage kit. Professional fees for physicians, hospital stay charges, and reimbursement amounts for the procedure itself are not counted as market revenue. General-purpose surgical tools not specifically packaged or marketed for centesis procedures are excluded. Pharmaceutical agents used in conjunction with drainage are outside the scope.
Recent Developments:
- In January 2025, Argon Medical Devices acquired the SeQure and DraKon microcatheters from Interventional Systems, expanding their interventional oncology offerings. These microcatheters enhance precise delivery in vascular procedures, potentially supporting advanced drainage and aspiration applications in thoracentesis and paracentesis markets.
- In April 2025, Cook Medical announced a collaboration with Mendaera Inc. to develop solutions for needle-based interventions using handheld robotics, initially focused on urological procedures but with potential applications in fluid aspiration techniques like thoracentesis and paracentesis. This partnership combines Cook's needles with Mendaera's robotic platform to improve precision and accessibility in interventional procedures.
- In July 2025, Argon Medical Devices opened a new Distribution & Education Center in Derby, United Kingdom, to streamline product distribution and provide training for Europe, Asia, and Africa. This facility supports faster access to interventional devices, including those used in paracentesis and thoracentesis, boosting market growth in these regions.
Scope of Report
| Items | Values |
|---|---|
| Quantitative Units (2026) | USD 1.2 billion |
| Product Type | Paracentesis Kits, Thoracentesis Kits, Drainage Catheters, Safety Needles |
| Procedure Type | Therapeutic, Diagnostic |
| Indication | Ascites, Pleural Effusion, Empyema, Hemothorax |
| End User | Hospitals, Ambulatory Surgery Centers, Specialty Clinics |
| Regions Covered | North America, Europe, Asia Pacific, Latin America, Middle East & Africa |
| Countries Covered | United States, Germany, United Kingdom, India, China, Brazil, and 40+ countries |
| Key Companies Profiled | BD, Teleflex, Merit Medical, Argon Medical, Cook Medical, Boston Scientific, B. Braun |
| Additional Attributes | Revenue analysis by product type and procedure, adoption trends across acute and outpatient care settings, regulatory and compliance landscape (FDA, CE marking), procedural volume impact analysis, safety-engineered device penetration, pricing and reimbursement considerations, and competitive positioning analysis |
Source: FMI historical analysis and forecast data.
Thoracentesis and Paracentesis Market by Segments
-
By Product Type:
- Paracentesis Kits
- Thoracentesis Kits
- Drainage Catheters
- Safety Needles
-
By Indication:
- Ascites
- Pleural Effusion
- Other Indications
-
By End User:
- Hospitals
- Ambulatory Surgery Centers
- Specialty Clinics
-
By Region:
- North America
- Europe
- Asia Pacific
- Latin America
- Middle East & Africa
Bibliography
- Becton, Dickinson and Company. (2024, June 3). BD to acquire Edwards Lifesciences’ Critical Care product group. BD Investor Relations.
- National Center for Biotechnology Information. (2025). Ascites. StatPearls Publishing.
- Centers for Medicare & Medicaid Services. (2024, January). MM13488 - Hospital Outpatient Prospective Payment System update. CMS.
- World Health Organization. (2024). Global hepatitis report 2024. WHO.
- American Cancer Society. (2024). Cancer statistics 2024. ACS.
- Jepsen, P., et al. (2023). Hospital admissions for cirrhosis and complications including ascites: population-based trends. PubMed Central.
- Merit Medical Systems. (2024). Annual report 2024. Merit Medical Investor Relations.
Frequently Asked Questions
What is the current global market size for Thoracentesis and Paracentesis?
The global industry is valued at USD 1.2 billion in 2026, driven by rising liver disease prevalence and oncology caseloads.
What is the projected CAGR for the Thoracentesis and Paracentesis market?
Market revenues are projected to grow at an 8.9% CAGR from 2026 to 2036.Which segment currently dominates the industry?
Which segment currently dominates the industry?
Paracentesis kits hold the dominant share of 28.1%, supported by the high volume of recurrent ascites drainage procedures.
What are the primary risks or barriers to adoption?
Procedural risks such as infection or organ perforation remain barriers, though adoption of image-guided and safety-engineered devices is mitigating these concerns.
Who are the leading players in the Thoracentesis and Paracentesis space?
BD, Teleflex, and Merit Medical lead the sector through comprehensive product portfolios and recent strategic acquisitions.
Table of Content
- Executive Summary
- Thoracentesis and Paracentesis Market Introduction
- Market Definition
- Market Scope
- Market Taxonomy
- Research Methodology
- Study Framework
- Assumptions and Limitations
- Data Sources
- Market Estimation and Forecasting Approach
- Thoracentesis and Paracentesis Market Dynamics
- Drivers
- Restraints
- Opportunities
- Trends
- Global Thoracentesis and Paracentesis Market Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Thoracentesis and Paracentesis Market Analysis by Product Type
- Global Thoracentesis and Paracentesis Market Analysis by Procedure Type
- Global Thoracentesis and Paracentesis Market Analysis by Indication
- Global Thoracentesis and Paracentesis Market Analysis by End User
- Global Thoracentesis and Paracentesis Market Analysis by Region
- North America Thoracentesis and Paracentesis Market Analysis
- United States
- Canada
- Latin America Thoracentesis and Paracentesis Market Analysis
- Brazil
- Rest of Latin America
- Europe Thoracentesis and Paracentesis Market Analysis
- Germany
- United Kingdom
- France
- Italy
- Spain
- Rest of Europe
- Asia Pacific Thoracentesis and Paracentesis Market Analysis
- China
- India
- Japan
- Rest of Asia Pacific
- Middle East & Africa Thoracentesis and Paracentesis Market Analysis
- GCC
- South Africa
- Rest of Middle East & Africa
- Competitive Landscape Analysis
- Key Players Profiled
- Market Definition
- Market Segmentation
- Bibliography
- Frequently Asked Questions
List of Tables
- Global Thoracentesis and Paracentesis Market Size (USD Billion), 2021-2036
- Global Thoracentesis and Paracentesis Market Y-o-Y Growth (%), 2021-2036
- Global Thoracentesis and Paracentesis Market Absolute $ Opportunity (USD Billion), 2026-2036
- Global Thoracentesis and Paracentesis Market Size by Product Type, 2021-2036
- Global Thoracentesis and Paracentesis Market Size by Procedure Type, 2021-2036
- Global Thoracentesis and Paracentesis Market Size by Indication, 2021-2036
- Global Thoracentesis and Paracentesis Market Size by End User, 2021-2036
- Global Thoracentesis and Paracentesis Market Size by Region, 2021-2036
- North America Thoracentesis and Paracentesis Market Size by Country, 2021-2036
- Latin America Thoracentesis and Paracentesis Market Size by Country, 2021-2036
- Europe Thoracentesis and Paracentesis Market Size by Country, 2021-2036
- Asia Pacific Thoracentesis and Paracentesis Market Size by Country, 2021-2036
- Middle East & Africa Thoracentesis and Paracentesis Market Size by Country, 2021-2036
- Global Thoracentesis and Paracentesis Market Share (%) by Product Type, 2025
- Global Thoracentesis and Paracentesis Market Share (%) by Procedure Type, 2025
- Global Thoracentesis and Paracentesis Market Share (%) by End User, 2025
- Global Thoracentesis and Paracentesis Market Share (%) by Region, 2025
List of Figures
- Global Thoracentesis and Paracentesis Market Growth Framework
- Thoracentesis and Paracentesis Market Value Chain Analysis
- Thoracentesis and Paracentesis Market Product and Technology Landscape
- Thoracentesis and Paracentesis Market Opportunity Map
- Thoracentesis and Paracentesis Market Scenario Forecast Model
- Global Thoracentesis and Paracentesis Market Share by Product Type, 2025
- Global Thoracentesis and Paracentesis Market Share by Procedure Type, 2025
- Global Thoracentesis and Paracentesis Market Share by End User, 2025
- Global Thoracentesis and Paracentesis Market Share by Region, 2025
- North America Thoracentesis and Paracentesis Market Structure
- Latin America Thoracentesis and Paracentesis Market Structure
- Europe Thoracentesis and Paracentesis Market Structure
- Asia Pacific Thoracentesis and Paracentesis Market Structure
- Middle East & Africa Thoracentesis and Paracentesis Market Structure
- Thoracentesis and Paracentesis Market Competitive Landscape Mapping
- Thoracentesis and Paracentesis Market Strategic Positioning Matrix
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

