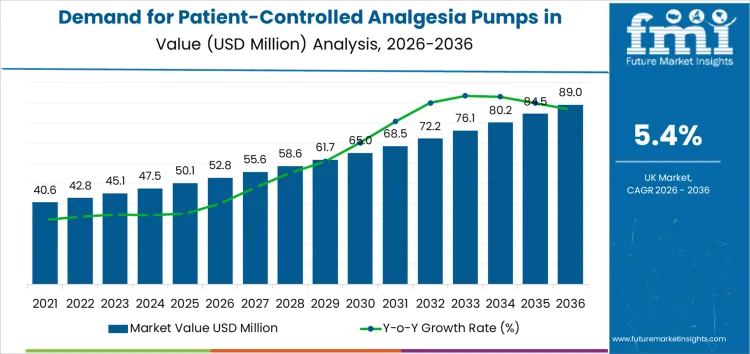
Demand for patient-controlled analgesia pumps in the United Kingdom is projected to grow from USD 52.8 million in 2026 to USD 89.0 million by 2036, at a CAGR of 5.4%. Capital intensity in this market will be significant, as investment in high-tech medical devices, coupled with regulatory compliance and safety standards, requires substantial up-front capital. Return timelines will be relatively extended due to the long product life cycles and the slow pace of adoption in some healthcare settings, where purchasing decisions are often driven by budgets and patient needs. Selectivity in investment will play a crucial role as companies navigate competition and regulatory hurdles. Investors will need to prioritize companies with strong R&D pipelines and operational efficiency, as those able to innovate while controlling costs will be best positioned for long-term success. Market players that secure contracts with hospitals and healthcare providers will have an edge in capturing sustained demand for these critical devices.
Demand for patient-controlled analgesia (PCA) pumps in the United Kingdom is expected to expand steadily throughout the forecast period, supported by the healthcare system’s growing emphasis on structured, patient-centric pain management. Adoption is rising as hospitals increasingly prioritize better post-operative recovery outcomes, improved patient comfort, and reduced reliance on repeated manual dosing by nursing staff. PCA pumps are becoming more common in surgical wards and critical care settings where controlled analgesic delivery and consistent pain control are essential.
The market outlook remains positive, driven by broader hospital uptake and continuous improvements in device technology, including enhanced safety mechanisms, dosing precision, and monitoring capabilities. Growth is expected to be stable and predictable, reflecting the ongoing clinical need for reliable pain management tools across both elective and emergency procedures. Over time, rising procedure volumes, strengthened pain management protocols, and a greater focus on patient autonomy in treatment decisions will continue to support demand. Overall, the PCA pump category benefits from its clear clinical value and its role in modernizing pain control practices within the UK healthcare system.
| Metric | Value |
|---|---|
| Industry Sales Value (2026) | USD 52.8 million |
| Industry Forecast Value (2036) | USD 89 million |
| Industry Forecast CAGR (2026 to 2036) | 5.4% |
Demand for patient controlled analgesia pumps in the United Kingdom has strengthened as clinical care models and pain management protocols evolved across hospital and outpatient settings. Historically, postoperative and acute pain relief relied on nurse administered injections or fixed infusion regimens, which limited responsiveness to individual patient needs and placed additional burden on clinical staff. UK healthcare providers increasingly adopted patient controlled analgesia pumps that allow individuals to self administer controlled doses of analgesic medication within prescribed safety limits. This shift supported improvements in patient comfort and perceived autonomy, especially following major surgical procedures where pain intensity can vary significantly among patients. Surgical units and anesthesia teams valued PCA pumps for their ability to standardize dosing parameters while reducing delays associated with manual administration. Procurement and clinical engineering teams responded by integrating these devices into perioperative care pathways, aligning with broader goals to optimize pain control, reduce length of stay, and improve resource utilization in acute care wards.
Looking ahead, demand for patient controlled analgesia pumps in the United Kingdom is expected to continue rising as healthcare organizations prioritize personalized care and operational efficiency. Growth will be influenced by demographic changes, such as an ageing population with higher incidence of surgical interventions and chronic pain conditions requiring tailored analgesic strategies. NHS trusts and private hospital groups weighing capital investments will evaluate PCA pump options based on device reliability, ease of programming, integration with electronic medical records, and safety features that minimize overdose risk. Suppliers with offerings that include robust training, service support, and data capture capabilities will gain traction with clinical and procurement stakeholders focused on long term value beyond initial acquisition cost. Expanded use of PCA pumps in community healthcare settings and post acute care could further support demand as care models evolve toward early discharge with remote monitoring support. Continued emphasis on evidence based pain management, patient satisfaction metrics, and care standardization positions patient controlled analgesia pumps as a key technology in the UK healthcare landscape.
Demand for patient-controlled analgesia (PCA) pumps in the United Kingdom is driven by product type and end-user sector. PCA pumps lead the product segment with 75%, while hospitals dominate the end-user segment with 65%. Other products, such as pump accessories, and end-users including ambulatory surgical centers, home care settings, and long-term care centers, also contribute significantly to the market. These trends reflect the growing demand for precise, patient-controlled pain management solutions in various healthcare settings.
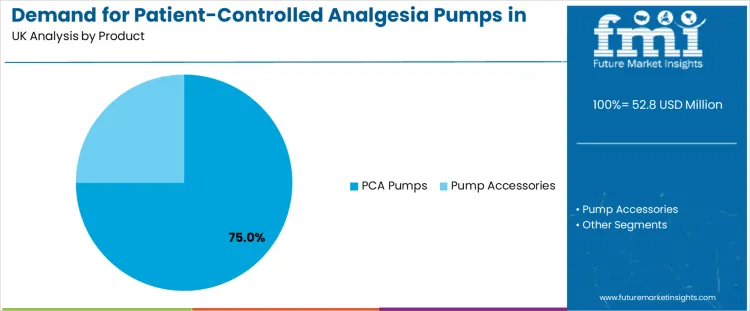
The product segment for patient-controlled analgesia pumps in the United Kingdom is primarily led by PCA pumps, which account for 75% of the market share. PCA pumps are widely used in hospital settings to provide pain relief for patients recovering from surgery, injury, or certain medical conditions. These pumps allow patients to self-administer a controlled amount of pain medication, typically opioids, at set intervals, giving them more control over their pain management and improving overall comfort.
The growing preference for PCA pumps is driven by their ability to provide effective and tailored pain relief, reducing the need for healthcare providers to manually administer medication and improving patient satisfaction and recovery time. The demand for PCA pumps is particularly high in post-operative care, where pain management is critical to recovery. Pump accessories follow as a smaller but important segment, supporting the overall functionality and maintenance of PCA pumps. These accessories, such as infusion sets, connectors, and reservoirs, are essential for ensuring the proper operation of PCA pumps and are necessary for ongoing patient care. The accessory market benefits from the widespread use of PCA pumps in various healthcare environments, driving demand for related products to support pain management and pump functionality.
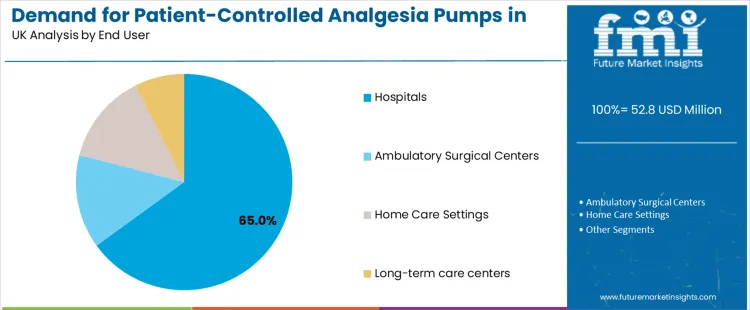
The end-user segment for patient-controlled analgesia pumps in the United Kingdom is primarily driven by hospitals, which account for 65% of the market share. Hospitals are the largest users of PCA pumps, as they are critical in managing pain for patients undergoing surgery, those in intensive care units (ICUs), and those requiring long-term pain management. PCA pumps offer hospitals a way to provide patients with effective pain control while also reducing the burden on nursing staff. The widespread adoption of PCA pumps in hospitals is driven by their efficiency, safety, and the ability to improve patient outcomes by allowing for more individualized pain management. Ambulatory surgical centers (ASCs) follow as another key end-user segment, particularly for outpatient surgeries.
PCA pumps are used in ASCs to manage post-operative pain for patients undergoing minor surgeries, allowing them to control their pain levels during recovery without requiring prolonged hospital stays. Home care settings are becoming an increasingly important market for PCA pumps, driven by the growing trend toward at-home recovery and the need for effective pain management in patients recovering from surgery or managing chronic pain. The use of PCA pumps in home care settings is supported by technological advancements that allow for safe and controlled medication delivery in a home environment, offering patients more autonomy in managing their pain. Long-term care centers, including nursing homes and rehabilitation facilities, represent a smaller but significant market for PCA pumps, as these settings care for patients with chronic pain or those recovering from major procedures. As the demand for personalized, effective pain management solutions continues to rise, the market for PCA pumps is expected to grow across hospitals, ambulatory surgical centers, home care settings, and long-term care centers, with hospitals remaining the largest segment.
Demand for patient controlled analgesia (PCA) pumps in the United Kingdom reflects the clinical focus on personalised pain management for acute and post operative care. PCA pumps allow patients to self administer controlled doses of analgesics, improving comfort and reducing variability in pain control. Procurement occurs across NHS trusts, private hospitals, surgical centres, and ambulatory care units where standards for pain protocols and recovery benchmarks are established. Purchase volumes are influenced by surgical case load, trauma admissions, enhanced recovery pathways, and hospital capital planning cycles for clinical equipment replacement.
Several factors are driving demand for PCA pumps in the United Kingdom. Clinical emphasis on patient safety, pain control, and recovery outcomes supports their use in orthopaedic, general, and thoracic surgery. Protocols that integrate multimodal analgesia position PCA as a tool to reduce reliance on fixed dosing and improve patient autonomy. NHS procurement frameworks and clinical commissioning groups increasingly recognise evidence that PCA can shorten average length of stay and reduce complications associated with under treatment of pain. Technology improvements, such as dose limiting safeguards and digital logging, increase clinician confidence and support adoption across acute care settings.
Despite clinical benefits, several restraints affect PCA pump demand in the United Kingdom. Budget pressures within NHS trusts and private healthcare providers can defer capital expenditure on advanced infusion pumps, especially when competing with other priority equipment needs. Training requirements for safe programming, monitoring, and troubleshooting increase operational costs and require allocation of staff time. Variability in clinical guidelines across facilities may limit consistent adoption, as some units favour alternative pain management modalities. Supply chain considerations, including lead times for replacement parts and consumables such as prefilled syringes, add complexity to inventory and service planning.
Key trends influencing PCA pump demand in the United Kingdom include integration of smart pump technologies that offer connectivity with electronic health records and automated dose documentation. Devices with enhanced safety features, such as barcode medication administration and dose error reduction systems, align with broader patient safety initiatives. There is growing interest in ambulatory PCA solutions that support pain management following day case surgery, reducing need for extended hospital stays. Data analytics from connected devices inform quality improvement programmes and support clinical audit. Collaborative procurement and framework agreements across hospital networks aim to standardise equipment and reduce unit costs.
The demand for patient-controlled analgesia (PCA) pumps in the United Kingdom varies across regions, with England leading the market. The increasing use of PCA pumps for pain management in hospitals, particularly for post-operative care and chronic pain conditions, is driving this demand. England’s larger healthcare infrastructure and more extensive use of advanced medical technologies contribute significantly to its dominant market share. Scotland, Wales, and Northern Ireland also show steady demand, driven by the rising adoption of PCA pumps in pain management protocols, growing patient awareness, and improved healthcare facilities. As patient autonomy in pain management becomes increasingly important, the demand for PCA pumps is expected to grow across all regions, with England maintaining the highest share of the market.
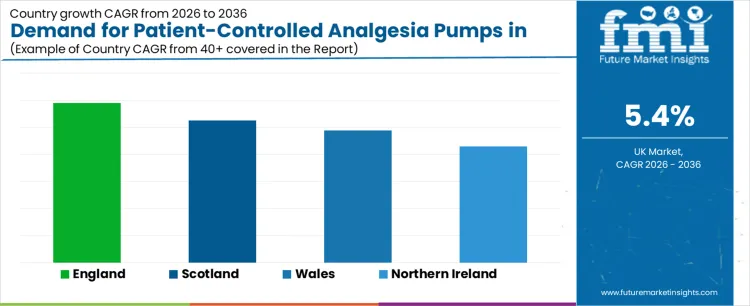
| Country | CAGR (2026 to 2036) |
|---|---|
| England | 5.9% |
| Scotland | 5.3% |
| Wales | 4.9% |
| Northern Ireland | 4.3% |
The demand for patient-controlled analgesia (PCA) pumps in England is projected to grow at a CAGR of 5.9% from 2026 to 2036. The primary drivers of this growth are the increasing incidence of surgical procedures, the growing need for efficient pain management solutions, and the rising preference for patient autonomy in pain control. PCA pumps offer patients the ability to self-administer pain relief within prescribed limits, which improves patient satisfaction and reduces the need for nursing intervention. The rising adoption of PCA pumps in hospitals, particularly in post-operative care and palliative settings, is fueling demand. Additionally, the growing awareness of the benefits of PCA pumps in improving pain management outcomes, along with advancements in pump technology, such as more compact, user-friendly devices, is contributing to market growth in England. The NHS’s continued investment in pain management technologies ensures a sustained demand for these devices.
In Scotland, the demand for patient-controlled analgesia pumps is expected to grow at a CAGR of 5.3% from 2026 to 2036. The healthcare sector in Scotland is increasingly adopting PCA pumps to enhance pain management in both surgical and palliative care settings. The rising number of surgical procedures and growing awareness of effective pain relief methods are key drivers of this demand. Scottish hospitals are increasingly implementing PCA pumps in pain management protocols to improve patient outcomes, reduce recovery times, and increase patient satisfaction. The region’s healthcare system is also focusing on improving patient autonomy and reducing reliance on nursing staff for pain management, which further supports the adoption of PCA pumps. As patient care in Scotland continues to evolve with a focus on personalized medicine and pain management, the market for PCA pumps is expected to grow steadily.
The demand for PCA pumps in Wales is projected to grow at a CAGR of 4.9% through 2036. The growing focus on efficient pain management, especially for post-surgical patients and those with chronic conditions, is driving the adoption of PCA pumps in Welsh hospitals. PCA pumps allow patients to have more control over their pain relief, leading to higher satisfaction and better clinical outcomes. The increasing number of surgeries performed, coupled with the demand for advanced pain management solutions, is fueling the growth of this market. Additionally, the Welsh healthcare system’s focus on improving quality of care and patient autonomy, as well as the growing awareness of PCA pumps’ benefits, is contributing to the expansion of their use. As healthcare providers in Wales continue to invest in modern pain management technologies, demand for PCA pumps is expected to rise.
Northern Ireland’s demand for patient-controlled analgesia pumps is expected to grow at a CAGR of 4.3% from 2026 to 2036. Although Northern Ireland represents the smallest market for PCA pumps in the UK, the demand is steadily increasing, driven by the region’s growing focus on effective pain management solutions in hospitals. As more patients seek improved pain relief options following surgery or during palliative care, PCA pumps are becoming an integral part of pain management protocols. The adoption of PCA pumps is further supported by Northern Ireland’s efforts to enhance patient care and improve clinical outcomes through the use of advanced medical technologies. With increasing awareness among healthcare professionals and patients about the benefits of PCA pumps in managing pain effectively and safely, the demand for these devices is expected to continue growing steadily in the region.
Demand for patient controlled analgesia (PCA) pumps in the United Kingdom is driven by hospital pain management protocols, postoperative care pathways, and demand for controlled opioid delivery in acute care settings. NHS procurement teams and hospital pharmacy departments evaluate PCA systems on documented delivery accuracy, documented safety interlocks, documented software reliability, and documented compliance with UK medical device standards. Becton Dickinson and Company is positioned as a leading supplier with documented product literature that details pump architecture, documented infusion precision metrics, and documented clinical safety features. These materials are used by clinical engineers and procurement leads when aligning device selection with hospital quality assurance criteria and departmental budgets. Brochures supplied by Becton Dickinson list documented alarm performance, documented audit trail capabilities, and documented service support options that are referenced during tender evaluations and device lifecycle planning.
B.Braun Melsungen AG competes with documented PCA systems that include technical sheets on pump durability, documented battery performance, and documented drug library integrations. Buyers in the United Kingdom consult these specifications to ensure that chosen devices align with formulary practices and ward workflow patterns. Moog Inc. supplies infusion platforms with documentation on pump modularity, documented flow rate control data, and documented calibration procedures that assist biomedical teams in mapping device capabilities to clinical protocols. Smiths Group/ICU Medical offers documented PCA pumps with specification guides covering occlusion detection thresholds, documented user interface standards, and documented training resources for clinical staff. Fresenius SE & Co. KGaA provides documented systems with literature outlining documented maintenance cycles, documented compatibility with electronic health record interfaces, and documented service coverage metrics. Procurement decisions incorporate documented lead times, documented spare parts availability, and documented post market support commitments. Technical reviewers and clinical stakeholders examine documented performance data to ensure selected systems support pain management goals, reduce risk of programming errors, and maintain operational uptime in acute care units. Competitive behavior in the UK PCA pump segment reflects emphasis on measurable technical specifications and documented support commitments that assist hospitals during evaluation and selection processes.
| Items | Values |
|---|---|
| Quantitative Units (2026) | USD Million |
| Product | PCA Pumps, Pump Accessories |
| Application | Pain Management, Antibiotic/Antiviral Therapy, Chemotherapy, Others |
| End User | Hospitals, Ambulatory Surgical Centers, Home Care Settings, Long-term Care Centers |
| Companies | Becton Dickinson and Company, B.Braun Melsungen AG, Moog Inc., Smiths Group/ ICU Medical, Fresenius SE & Co. KGaA |
| Regions Covered | United Kingdom |
| Countries Covered | England, Scotland, Wales, Northern Ireland |
| Additional Attributes | Dollar by sales by product, application, end user, and region. Includes market trends towards patient comfort, ease of use, and the role of PCA pumps in effective pain management across various healthcare settings. |
How big is the demand for patient-controlled analgesia pumps in United Kingdom in 2026?
The demand for patient-controlled analgesia pumps in United Kingdom is estimated to be valued at USD 52.8 million in 2026.
What will be the size of patient-controlled analgesia pumps in United Kingdom in 2036?
The market size for the patient-controlled analgesia pumps in United Kingdom is projected to reach USD 89.0 million by 2036.
How much will be the demand for patient-controlled analgesia pumps in United Kingdom growth between 2026 and 2036?
The demand for patient-controlled analgesia pumps in United Kingdom is expected to grow at a 5.4% CAGR between 2026 and 2036.
What are the key product types in the patient-controlled analgesia pumps in united kingdom?
The key product types in patient-controlled analgesia pumps in United Kingdom are PCA pumps and pump accessories.
Which product segment is expected to contribute significant share in the patient-controlled analgesia pumps in united kingdom in 2026?
In terms of product, PCA pumps segment is expected to command 75.0% share in the patient-controlled analgesia pumps in United Kingdom in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.