United Kingdom Respiratory Inhaler Devices Market
Demand for Respiratory Inhaler Devices in UK Size and Share Forecast Outlook 2026 to 2036
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Respiratory Inhaler Devices Outlook in the UK 2026 to 2036
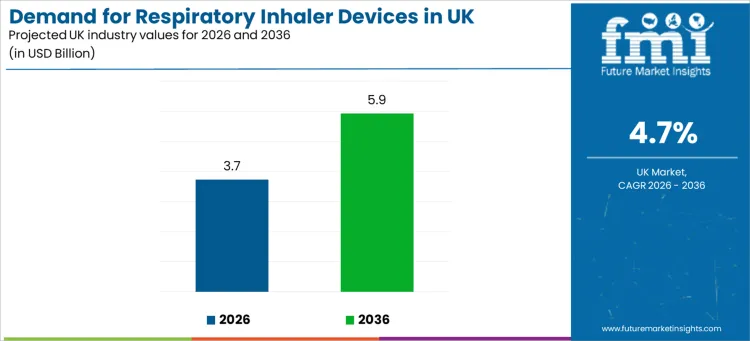
Demand for respiratory inhaler devices in the UK is projected at USD 3.7 billion in 2026 and is expected to reach USD 5.9 billion by 2036, expanding at a 4.7% CAGR. This growth is anchored in everyday disease control needs across asthma, chronic obstructive pulmonary disease, and specialist indications that rely on consistent inhaled drug delivery.
Inhaler devices are no longer viewed as simple accessories to therapy. They act as the practical interface between prescription intent and real-world outcomes, shaping dose delivery, patient adherence, symptom relief, and the need for urgent care escalation. These devices include dry powder inhalers, metered dose inhalers, nebulizers, and other delivery formats used across community settings and hospital care.
Quick Stats for Respiratory Inhaler Devices Demand in the UK
- Respiratory Inhaler Devices Valuation in the UK (2026): USD 3.7 billion
- Respiratory Inhaler Devices Forecast Valuation in the UK (2036): USD 5.9 billion
- Forecast CAGR 2026 to 2036: 4.7%
- Leading Regional Growth: England (5.2%)
- Leading Product: Dry Powder Inhalers (40.3% share)
- Leading Technology: Manually Operated Inhaler Devices (72.5% share)
- Leading Disease Indication: Asthma (42.0% share)
- Key Companies Profiled: GlaxoSmithKline plc (GSK); AstraZeneca plc; Boehringer Ingelheim International GmbH; Teva Pharmaceutical Industries Ltd.; Novartis AG
Selection priorities are built around dose reliability, ease of use, inspiratory flow requirements, portability, and patient training burden. Decision-makers also evaluate device families through procurement resilience, long-term serviceability, device handling errors, and environmental considerations linked to propellants.
For senior leaders in pharmaceutical and device businesses, growth is tied to three fundamentals: persistent respiratory disease burden, tighter focus on technique-driven outcomes, and rising expectations for responsible device design. For care providers and service partners, value sits in patient education models, pharmacy-led inhaler checks, digital adherence support, and product portfolios that fit diverse patient needs without creating device confusion.
Respiratory Inhaler Devices in the UK Key Takeaways
| Metric | Value |
|---|---|
| Industry Value (2026) | USD 3.7 billion |
| Industry Forecast Value (2036) | USD 5.9 billion |
| Forecast CAGR (2026 to 2036) | 4.7% |
Why is the UK Building Sustained Demand for Respiratory Inhaler Devices?
UK demand is reinforced by how respiratory care is delivered at scale. Inhaled therapy remains central because it targets the lungs directly while limiting systemic exposure for many medicines. Clinical teams rely on device choice to match patient capability, symptom patterns, and risk profile. When a patient struggles with inhaler technique, the therapy can fail even when the drug is appropriate. That creates a strong operational push toward device optimisation rather than medication changes alone.
Guideline direction supports disciplined diagnosis and long-term control planning. NICE guidance covers diagnosing, monitoring, and managing asthma across age groups, with a focus on accuracy of diagnosis and reducing attack risk. COPD management guidance also frames a structured care approach where treatment choice and ongoing review are designed to reduce symptoms and support long-term health stability. These structures indirectly increase device demand by reinforcing chronic therapy pathways that require dependable supply and repeat prescribing cycles.
Asthma remains the largest disease indication share at 42.0%, reflecting the scale of controller and reliever use across primary care and specialist clinics. Device selection is influenced by patient age, coordination capability, inspiratory flow, and the need for fast symptom relief. Many care systems prioritise reducing device variation for patients, limiting training burden and improving adherence stability.
Technology direction is also shaping demand. Manually operated devices hold a 72.5% share, reflecting the scale advantages and widespread familiarity of traditional delivery formats. Digitally operated inhaler devices continue to gain attention in targeted care models where adherence visibility and technique prompts support better control, even though routine use is not always prioritised in broad pathways. Recent UK-aligned clinical guidance also notes that digital inhalers are not recommended for routine use in asthma.
Environmental priorities now influence procurement conversations. NHS sustainability work highlights inhaler emissions and the role of propellants in carbon impact, reinforcing interest in clinically appropriate low-carbon options where suitable. This adds a new layer to device strategy, especially in high-volume prescribing environments. Teams aligning respiratory portfolios across device families often map decisions against broader device categories such as respiratory inhaler devices and connected-care options such as smart inhalers when planning long-term product positioning.
How is Respiratory Inhaler Devices Demand Segmented by Product, Technology, and Disease Indication?
Segmentation reflects how device mechanics interact with patient behaviour, clinical setting, and disease pattern. It also reflects operational realities such as training burden, refill cadence, and how quickly a device can be adopted at scale without creating technique errors.
Why do dry powder inhalers hold the leading product share in UK prescribing patterns?
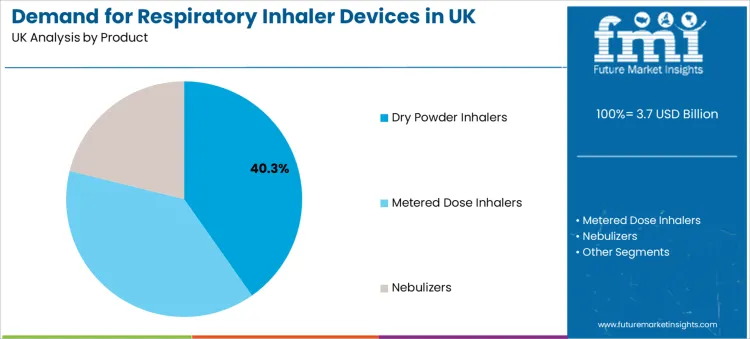
Dry powder inhalers lead with a 40.3% share, positioning them as the largest product type. Their advantage often sits in breath-actuated delivery, removing the coordination challenge seen in some pressurised devices. For many users, simpler actuation improves consistency and reduces technique-driven dose loss. DPIs also align with sustainability discussions because they avoid certain propellant-driven emissions, making them attractive in settings where clinical suitability and environmental targets both matter.
From a procurement lens, DPIs can support repeatable rollouts across large patient groups, especially when training materials are standardised. Device design factors such as dose counters, tactile feedback, and resistance profiles influence which DPI formats scale well across community prescribing. Stakeholders comparing DPI design evolution often keep close alignment with dry powder inhalers as portfolios broaden and combination therapies mature.
How do metered dose inhalers sustain demand through rescue use and broad familiarity?
Metered dose inhalers remain central in everyday respiratory care due to portability, rapid actuation, and wide familiarity across patient populations. They support rescue therapy needs and can be integrated into both chronic control and acute symptom relief pathways. The main operational challenge lies in coordination technique. That can drive use of spacers or repeated inhaler education, especially for older adults and children.
Another decision factor is sustainability. NHS guidance highlights that the propellant used in metered dose inhalers contributes substantially to inhaler-related emissions, pushing greater attention toward clinically appropriate low-carbon options and prescribing improvement programs. This dynamic does not remove demand, but it does influence product strategy and lifecycle planning.
Why do nebulizers stay relevant for patients who need assisted delivery and controlled dosing?
Nebulizers maintain a role where patients have difficulty using handheld devices, where dose delivery needs longer administration time, or where treatment settings require controlled delivery under supervision. They are often used in homecare, acute care, and specialist pathways depending on indication and patient capability. Their relevance increases when inspiratory effort is limited or when coordination barriers make handheld technique unreliable.
Care model designers tracking homecare respiratory support often align device planning with adjacent equipment areas such as nebuliser, especially where portability and patient independence shape adoption choices.
Why do manually operated inhaler devices dominate technology selection in the UK?
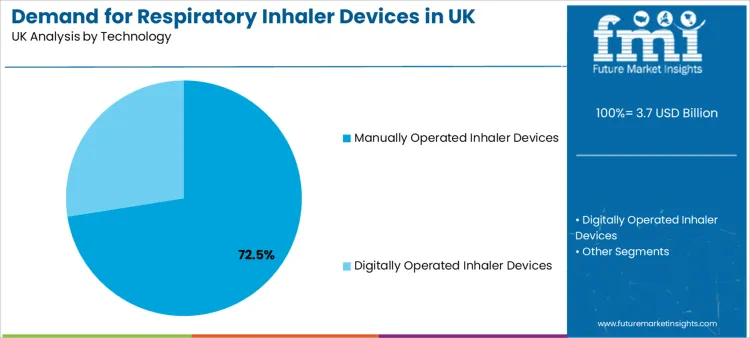
Manually operated inhaler devices lead with a 72.5% share, reflecting scale economics, established prescribing habits, and patient familiarity. They work within conventional pharmacy supply systems and do not require software onboarding, battery management, or digital data workflows. That reduces friction for large prescribing populations.
Manual dominance also reflects the reality that many care teams prioritise standardised patient education and routine technique checks over device digitisation. This model supports stable outcomes when training is consistent and review cycles are embedded in routine care.
How are disease indication needs shaping device selection across asthma, COPD, and specialist cases?
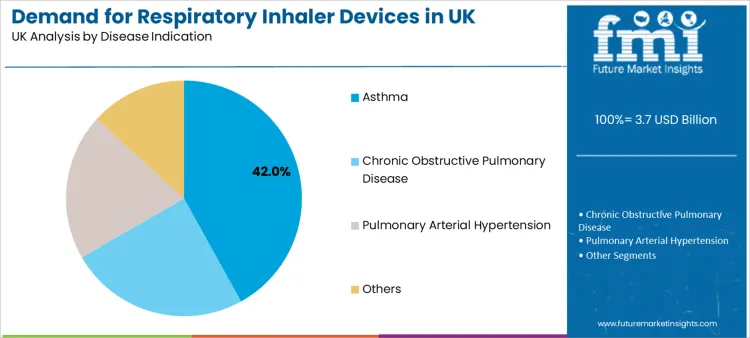
Asthma leads with a 42.0% share, reflecting broad patient prevalence and sustained controller plus reliever use. Device choice must fit symptom patterns, age profile, and adherence realities. GINA’s strategy report provides evidence-based direction for asthma management and prevention, reinforcing the importance of effective inhaled treatment delivery.
COPD demand is driven by long-term symptom management and exacerbation prevention. GOLD’s 2025 report provides global strategy direction for diagnosis, management, and prevention of COPD, supporting structured inhaled therapy escalation and device choice considerations. Pulmonary arterial hypertension contributes to specialist inhaled therapy contexts in narrower patient groups, with clinical frameworks covered within ESC/ERS guidelines for pulmonary hypertension diagnosis and treatment.
What are the Dynamics, Restraints, Opportunities, and Threats Shaping this Space?
What dynamics are strengthening UK demand fundamentals?
Chronic respiratory disease management drives repeat prescribing cycles and long-duration therapy persistence. NICE guidance supports structured diagnosis, monitoring, and long-term control approaches for asthma, reinforcing ongoing therapy engagement. Similar structured pathways exist for COPD management, supporting sustained device demand across adult populations.
Sustainability is also shaping device strategies. NHS England highlights that inhaler emissions account for around 3% of the NHS carbon footprint and identifies lower-carbon options such as dry powder inhalers. This drives procurement attention toward device substitution where clinically appropriate, without compromising patient outcomes.
What restraints can slow adoption shifts or limit technology substitution?
Technique burden remains a key restraint. A device that looks efficient on paper can underperform if patients struggle with actuation steps or inspiratory flow requirements. Switching devices also carries risk of temporary control loss if training is not managed well.
Digital device adoption faces practical barriers such as cost, reimbursement alignment, data governance requirements, and workflow complexity in busy primary care settings. UK-aligned asthma guidance also notes that digital inhalers are not recommended for routine use, limiting broad deployment for now.
Where do practical opportunities sit for suppliers and service partners?
Opportunity concentrates in three areas, adherence improvement, low-carbon innovation, and simplified combination therapy delivery. Smart inhaler ecosystems can support targeted patient groups where adherence tracking and technique feedback reduce exacerbations and emergency visits. Product planners often align innovation roadmaps with connected device categories such as smart inhalers, particularly where pharmacy-led programs and digital respiratory monitoring expand.
Low global warming potential propellants represent another opportunity. A reported example includes AstraZeneca’s progress toward eco-friendly inhaler propellant transition for a COPD inhaler, reflecting active industry movement in this direction. Quality and regulatory alignment also matter for scaling inhalation products.
The EMA’s updated guideline on pharmaceutical quality for inhalation and nasal medicinal products highlights expectations tied to product quality and recent advancements, influencing how device and formulation programs are developed.
What threats can disrupt demand expectations?
Supply disruptions and product recalls can create short-term prescribing instability, especially for high-volume inhaled medicines paired with proprietary devices. Policy-driven change pressure can also create risk if device switching targets are not matched with training capacity. Environmental pressure may accelerate transitions faster than some prescribing systems can safely deliver, creating a need for staged implementation planning supported by clinical governance.
How is Respiratory Inhaler Devices Demand Evolving across Key Regions in the UK?
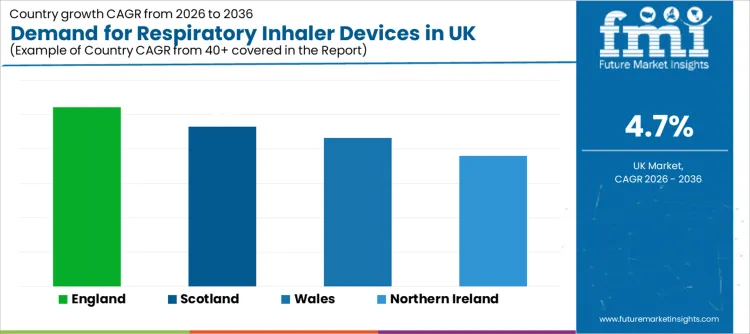
| Region | CAGR 2026 to 2036 |
|---|---|
| England | 5.2% |
| Scotland | 4.6% |
| Wales | 4.3% |
| Northern Ireland | 3.8% |
Why is England leading growth through Scale Prescribing Volumes and Care Pathway Standardisation?
England grows at 5.2%, supported by high prescribing volumes across primary care, dense specialist service networks, and large patient pools under long-term therapy management. Standardisation of device education pathways supports repeat demand, especially in asthma where disease control needs steady adherence. Sustainability programs targeting inhaler emissions also influence prescribing review activity, supporting gradual device mix refinement.
How is Scotland building steady gains through technique-led care models and prescribing review programs?
Scotland advances at 4.6%, shaped by structured chronic disease management and steady focus on inhaler technique improvement. Demand is supported by consistent asthma and COPD management needs.
Inhaler sustainability work and low-carbon prescribing initiatives also influence product mix decisions in certain local systems, supporting careful transitions where clinical suitability is confirmed.
What is driving Wales as respiratory prescribing stays anchored in long-term control needs?
Wales grows at 4.3%, supported by stable chronic respiratory care demand and the need for consistent therapy delivery across community health settings. Procurement and prescribing choices are influenced by workforce capacity for patient training and follow-up. Device families that reduce coordination burden and support repeatable education workflows can gain traction.
Why is Northern Ireland expanding at a measured pace through targeted service delivery and stable device preferences?
Northern Ireland increases at 3.8%, reflecting steady therapy demand with a smaller base and a careful approach to device switching. Stability in prescribing and patient familiarity remains important, particularly for older populations and patients with complex respiratory needs. Where device changes occur, they tend to be tied to training readiness and clinical review capacity.
What Defines the Competitive Landscape for Respiratory Inhaler Devices in the UK?
Competition is shaped by device performance reliability, patient usability, therapy compatibility, and the ability to scale manufacturing with consistent quality. Stakeholders also evaluate manufacturers through regulatory readiness, device training support programs, and supply resilience across national prescribing volumes.
GlaxoSmithKline plc (GSK) holds deep positioning through established respiratory portfolios and strong distribution reach across inhaler formats. AstraZeneca plc competes through combination therapy presence and ongoing device innovation priorities. Boehringer Ingelheim International GmbH competes through COPD-focused portfolios and long-term inhaled therapy positioning.
Teva Pharmaceutical Industries Ltd. competes through broad medicine access models and respiratory portfolio participation. Novartis AG competes through specialty respiratory and associated therapeutic development pathways where device design influences adherence and outcomes.
A growing differentiator is how companies approach sustainability and device transitions. NHS England has highlighted work to support clinically appropriate low-carbon inhaler use while engaging manufacturers on next generation propellant innovation. The firms that can deliver lower-carbon options without increasing patient handling complexity will be better positioned as procurement expectations mature.
Portfolio teams planning across multiple device families often align strategy with device categories such as metered dose inhalers and advanced therapy formats such as triple therapy inhalers when balancing long-term respiratory franchise direction.
Key Industry Participants
- GlaxoSmithKline plc (GSK)
- AstraZeneca plc
- Boehringer Ingelheim International GmbH
- Teva Pharmaceutical Industries Ltd.
- Novartis AG
Scope of Report
| Items | Values |
|---|---|
| Quantitative Units | USD Billion |
| Product | Dry Powder Inhalers; Metered Dose Inhalers; Nebulizers; Others |
| Technology | Manually Operated Inhaler Devices; Digitally Operated Inhaler Devices |
| Disease Indication | Asthma; Chronic Obstructive Pulmonary Disease; Pulmonary Arterial Hypertension |
| Regions Covered | England; Scotland; Wales; Northern Ireland |
| Key Companies Profiled | GlaxoSmithKline plc (GSK); AstraZeneca plc; Boehringer Ingelheim International GmbH; Teva Pharmaceutical Industries Ltd.; Novartis AG |
Respiratory Inhaler Devices Demand in the UK by Segments
By Product
- Dry Powder Inhalers
- Metered Dose Inhalers
- Nebulizers
- Others
By Technology
- Manually Operated Inhaler Devices
- Digitally Operated Inhaler Devices
By Disease Indication
- Asthma
- Chronic Obstructive Pulmonary Disease
- Pulmonary Arterial Hypertension
By Region
- England
- Scotland
- Wales
- Northern Ireland
Bibliography
- European Medicines Agency. (2025). Guideline on the pharmaceutical quality of inhalation and nasal medicinal products (Revision 1).
- Global Initiative for Asthma. (2024). Global Strategy for Asthma Management and Prevention (2024 Update).
- Global Initiative for Chronic Obstructive Lung Disease. (2024). Global Strategy for the Diagnosis, Management, and Prevention of COPD: 2025 Report.
- National Institute for Health and Care Excellence. (2024). Asthma: diagnosis, monitoring and chronic asthma management (NG245).
- National Institute for Health and Care Excellence. (2018). Chronic obstructive pulmonary disease in over 16s: diagnosis and management (NG115).
- NHS England. (2025). Five years of a greener NHS: progress and forward look.
- NHS England. (n.d.). Improving health outcomes for respiratory patients while reducing carbon emissions.
- European Society of Cardiology, & European Respiratory Society. (2022). 2022 ESC/ERS Guidelines for the diagnosis and treatment of pulmonary hypertension.
- British Thoracic Society, NICE, & SIGN. (2025). BTS/NICE/SIGN joint guideline on asthma: diagnosis, monitoring and management. Thorax.
Frequently Asked Questions
How big is the demand for respiratory inhaler devices in uk in 2026?
The demand for respiratory inhaler devices in uk is estimated to be valued at USD 3.7 billion in 2026.
What will be the size of respiratory inhaler devices in uk in 2036?
The market size for the respiratory inhaler devices in uk is projected to reach USD 5.9 billion by 2036.
How much will be the demand for respiratory inhaler devices in uk growth between 2026 and 2036?
The demand for respiratory inhaler devices in uk is expected to grow at a 4.7% CAGR between 2026 and 2036.
What are the key product types in the respiratory inhaler devices in uk?
The key product types in respiratory inhaler devices in uk are dry powder inhalers, metered dose inhalers and nebulizers.
Which technology segment is expected to contribute significant share in the respiratory inhaler devices in uk in 2026?
In terms of technology, manually operated inhaler devices segment is expected to command 72.5% share in the respiratory inhaler devices in uk in 2026.
Table of Content
- Executive Summary
- UK Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- UK Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- UK Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- UK Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product , 2026 to 2036
- Dry Powder Inhalers
- Metered Dose Inhalers
- Nebulizers
- Dry Powder Inhalers
- Y to o to Y Growth Trend Analysis By Product , 2021 to 2025
- Absolute $ Opportunity Analysis By Product , 2026 to 2036
- UK Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Technology
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Technology, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Technology, 2026 to 2036
- Manually Operated Inhaler Devices
- Digitally Operated Inhaler Devices
- Manually Operated Inhaler Devices
- Y to o to Y Growth Trend Analysis By Technology, 2021 to 2025
- Absolute $ Opportunity Analysis By Technology, 2026 to 2036
- UK Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Disease Indication
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Disease Indication, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Disease Indication, 2026 to 2036
- Asthma
- Chronic Obstructive Pulmonary Disease
- Pulmonary Arterial Hypertension
- Others
- Asthma
- Y to o to Y Growth Trend Analysis By Disease Indication, 2021 to 2025
- Absolute $ Opportunity Analysis By Disease Indication, 2026 to 2036
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product
- By Technology
- By Disease Indication
- Competition Analysis
- Competition Deep Dive
- GlaxoSmithKline plc (GSK)
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- AstraZeneca plc
- Boehringer Ingelheim International GmbH
- Teva Pharmaceutical Industries Ltd.
- Novartis AG
- GlaxoSmithKline plc (GSK)
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: UK Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: UK Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 3: UK Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 4: UK Market Value (USD Million) Forecast by Disease Indication, 2021 to 2036
- Table 5: UK Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 6: UK Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 7: UK Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 8: UK Market Value (USD Million) Forecast by Disease Indication, 2021 to 2036
List of Figures
- Figure 1: UK Market Pricing Analysis
- Figure 2: UK Market Value (USD Million) Forecast 2021-2036
- Figure 3: UK Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 4: UK Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 5: UK Market Attractiveness Analysis by Product
- Figure 6: UK Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 7: UK Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 8: UK Market Attractiveness Analysis by Technology
- Figure 9: UK Market Value Share and BPS Analysis by Disease Indication, 2026 and 2036
- Figure 10: UK Market Y-o-Y Growth Comparison by Disease Indication, 2026-2036
- Figure 11: UK Market Attractiveness Analysis by Disease Indication
- Figure 12: UK Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 13: UK Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 14: UK Market Attractiveness Analysis by Region
- Figure 15: UK Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: UK Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 17: UK Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 18: UK Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 19: UK Market Attractiveness Analysis by Product
- Figure 20: UK Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 21: UK Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 22: UK Market Attractiveness Analysis by Technology
- Figure 23: UK Market Value Share and BPS Analysis by Disease Indication, 2026 and 2036
- Figure 24: UK Market Y-o-Y Growth Comparison by Disease Indication, 2026-2036
- Figure 25: UK Market Attractiveness Analysis by Disease Indication
- Figure 26: UK Market - Tier Structure Analysis
- Figure 27: UK Market - Company Share Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

