Hot and cold therapy packs sit in a practical part of consumer healthcare. They are cheap to buy, easy to store, and familiar to both clinicians and households.
That mix matters in a market where buyers want relief options they can use quickly without adding much cost or clinical complexity. Demand is now being lifted by sports participation, home recovery routines, and a larger over-the-counter wellness aisle.
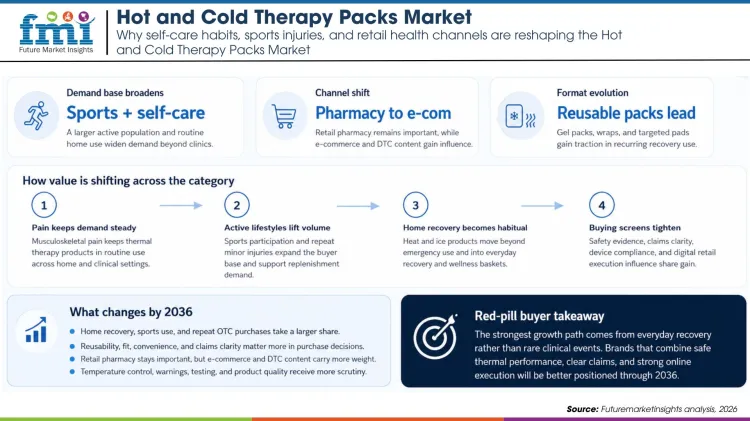
The World Health Organization continues to describe musculoskeletal conditions as a major source of disability worldwide, with low back pain alone affecting hundreds of millions of people. That keeps simple heat and cold products relevant because they are often one of the first interventions used before patients escalate to more expensive care.
The consumer side is strengthening at the same time. SFIA participation data show a very large active base in the United States, and fast-growing activities such as pickleball bring a steady stream of strains, soreness, and repeat purchases. This is one reason the market can grow through both retail and clinical channels without depending on a single use case.
CDC pain guidance keeps nonpharmacologic care in view, and heat or ice remains one of the most accessible tools for acute discomfort. That matters commercially because it supports sell-through in pharmacies, general merchandise, and e-commerce without requiring a specialist sales pitch.
The product mix is also improving. Reusable gel packs, wraps, and targeted pads fit better into recurring use, especially for households that manage minor injuries at home. As a result, the category is moving away from one-off emergency purchases and toward more deliberate basket-building across sports, wellness, and recovery.
| Market dimension | 2015-2025 | 2026-2036 |
|---|---|---|
| Primary demand base | Clinical use and basic home first-aid purchases dominated. | Home recovery, sports use, and repeat OTC purchases take a larger share. |
| Buyer priority | Low cost and broad availability mattered most. | Reusability, fit, convenience, and claims clarity matter more. |
| Channel mix | Retail pharmacy remained the obvious route to market. | Retail pharmacy stays important, but e-commerce and DTC content carry more weight. |
| Compliance focus | Basic safety and packaging requirements were usually enough. | Temperature control, warnings, and product testing receive more scrutiny. |
| Product format | Standard hot packs and cold packs led the market. | Reusable gel systems, wraps, and targeted pads gain share. |
FDA device categories for cold packs and disposable hot or cold packs make compliance a concrete buying issue, not just a labeling exercise. Buyers should check device listings, material integrity, leak performance, and any evidence around surface temperature or activation consistency before treating suppliers as interchangeable.
Channel economics have changed too. E-commerce continues to outpace overall retail growth, which means product pages, claims support, and fulfillment reliability directly influence sales. Brands that look strong on a shelf but weak online will give up share in a category where shoppers increasingly compare options before they buy.
Consumer product recalls in adjacent heating categories have already made merchants more cautious about thermal products. That should push suppliers to document heat retention curves, maximum surface temperatures, packaging warnings, and durability with more discipline than before.
The broad demand story remains favorable. High physical activity levels, simple pain-management guidance, and the convenience of at-home use give the category a stable base through 2036. For deeper segment sizing and regional outlooks, see the Future Market Insights report: Hot and Cold Therapy Packs Market (2026 - 2036) - https://www.futuremarketinsights.com/reports/hot-and-cold-therapy-packs-market
Growth in the Hot and Cold Therapy Packs Market is being supported by stronger end-user demand, operational efficiency needs, regulatory pressure, and wider adoption across relevant commercial and industrial applications.
High upfront costs, validation requirements, supply chain constraints, pricing pressure, and slower adoption among cost-sensitive buyers can restrict expansion in the Hot and Cold Therapy Packs Market.
Demand typically comes from manufacturers, service providers, healthcare or industrial operators, distributors, and specialized buyers that need reliable performance, compliance, and cost efficiency.
Regulations are pushing suppliers toward safer materials, better documentation, stronger quality controls, and products that help customers meet environmental, safety, or performance standards.
Companies should track raw material costs, technology upgrades, customer purchasing cycles, regional policy changes, and competitive moves that can alter pricing and adoption rates.
This report covers the coronary heart disease diagnostic imaging market through analysis of market size, revenue forecast, competitive landscape, demand outlook, growth drivers, restraints, imaging pathway shifts, supply-side developments, and strategic growth opportunities.
This report covers the mRNA vaccine formulation stability and stress test systems market through analysis of market size, market share, revenue forecast, system pricing benchmarks, company positioning, competitive landscape, demand outlook, growth drivers, restraints, technology trends, formulation testing requirements, assay integration, analytical method continuity, end-user adoption, supply chain developments, regulatory and quality expectations, investment opportunities, profitability outlook, and strategic growth opportunities during 2026 to 2036.
This report covers the cell therapy cryopreservation and thaw performance test platforms market through analysis of market size, market share, revenue forecast, platform segmentation, assay readout trends, cell type demand, preservation format usage, automation level adoption, end-user demand, workflow stage requirements, regional outlook, technology developments, handling standardization needs, method transfer practices, comparability testing, supply chain dynamics, regulatory and quality expectations, investment opportunities, profitability outlook, and strategic growth opportunities during 2026 to 2036.
The Gene Editing Off-Target Effect Detection Test Systems Market is segmented by Detection Approach (Cell-based Assays, Biochemical Assays, In Silico Tools, Orthogonal Validation), Product Format (Services, Assay Kits, Software, Instruments, Reagents), Workflow Stage (Site Nomination, Site Confirmation, Structural Follow-up, Release Support), Editing Modality (CRISPR-Cas9, Base Editing, Prime Editing, Cas12 Systems, Other Editors), Readout Technology (Amplicon NGS, Short-read WGS, Single-cell DNA, ChIP-seq Workflows, Long-read Sequencing), End User (Biopharma Companies, Academic Laboratories, CROs, CDMOs, Hospitals), Application (IND-enabling Studies, Discovery Screening, Cell Therapy Quality Assessment, In vivo Profiling, Comparability Studies), Deployment (Outsourced Testing, In-house Testing, Hybrid Models), and Region. Forecast for 2026 to 2036.
This report covers the at-home blood collection kits and devices market through analysis of market size, revenue forecast, competitive landscape, demand outlook, growth drivers, restraints, self-collection workflow trends, regulatory developments, and strategic growth opportunities.