Tray sealers have become more important because packaging is now treated as part of device performance, not just a finishing step. A weak seal can turn into a sterility failure, a recall event, or an audit problem.
That shift is raising the bar for equipment buyers. Medical device manufacturers and contract packagers now need sealing systems that support validation, traceability, and tighter process control while still handling commercial throughput.
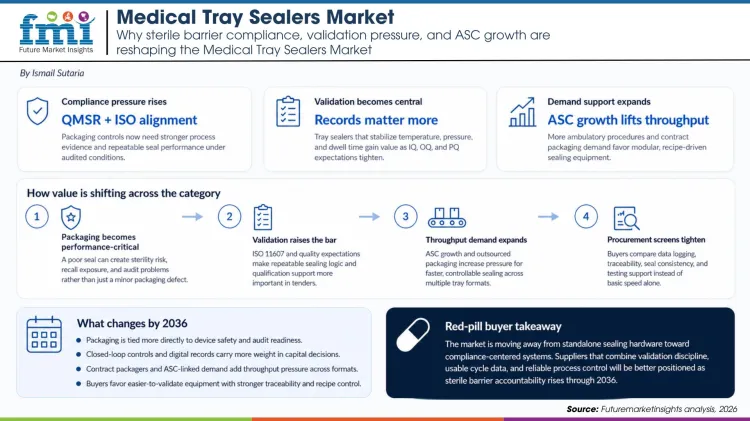
FDA's final Quality Management System Regulation and its incorporation of ISO 13485 principles push packaging teams toward more disciplined process evidence. That matters directly for tray sealing because buyers need equipment that can support traceable, repeatable sealing under audited conditions.
Europe is reinforcing the same message through EN ISO 11607 alignment and MDR expectations. In practice, this means notified bodies and quality teams will expect stronger evidence that sealing processes are validated and linked to sterile barrier performance rather than treated as routine production steps.
ISO 11607 has effectively made process validation a commercial requirement for credible suppliers. That favors tray sealers that can hold temperature, pressure, and dwell time within tight windows while also producing the records needed for IQ, OQ, and PQ work.
Clinical site-of-care change gives the market another tailwind. As ambulatory surgery centers handle more procedures, OEMs and contract packagers need reliable sealing at higher throughput and across more kit formats. This creates room for modular lines and recipe-driven equipment rather than basic standalone machines alone.
| Market dimension | 2015-2025 | 2026-2036 |
|---|---|---|
| Packaging role | Often treated as a downstream production function. | More clearly tied to device safety and audit readiness. |
| Validation burden | Manual documentation and fragmented controls were more common. | Closed-loop controls and digital records become more valuable. |
| End-market pull | Hospital-focused device packaging drove most decisions. | ASC growth and contract packaging expansion add throughput pressure. |
| Risk focus | Seal defects were important but not always front-of-mind in capital planning. | Recall exposure and traceability make seal integrity a board-level issue. |
| Preferred equipment profile | Basic sealing capability could still win some tenders. | Buyers favor controllable, logged, and easier-to-validate systems. |
Recall examples tied to seal strength and sterility show why the equipment decision cannot be separated from downstream risk. A lower-cost machine can become expensive very quickly if it introduces process drift, weak seals, or poor documentation.
ASTM methods such as F88, F1929, and F2096 make the validation burden tangible. Buyers should check whether the sealer stabilizes core parameters, exports usable cycle data, and supports the specific testing logic the quality team expects to run during qualification and routine monitoring.
As quality systems migrate toward tighter ISO-style expectations, equipment that supports recipe management, barcode-driven setup, and reliable e-records will look safer to buyers than machines that rely on operator habit. This is especially true where tray formats change often or compliance teams are already stretched.
European and US evidence expectations point in the same direction: fewer shortcuts, more process proof, and stronger packaging accountability. For broader market sizing and the 2026-2036 outlook, see the Future Market Insights report: Medical Tray Sealers Market (2026 - 2036) - https://www.futuremarketinsights.com/reports/medical-tray-sealers-market
Growth in the Medical Tray Sealers Market is being supported by stronger end-user demand, operational efficiency needs, regulatory pressure, and wider adoption across relevant commercial and industrial applications.
High upfront costs, validation requirements, supply chain constraints, pricing pressure, and slower adoption among cost-sensitive buyers can restrict expansion in the Medical Tray Sealers Market.
Demand typically comes from manufacturers, service providers, healthcare or industrial operators, distributors, and specialized buyers that need reliable performance, compliance, and cost efficiency.
Regulations are pushing suppliers toward safer materials, better documentation, stronger quality controls, and products that help customers meet environmental, safety, or performance standards.
Companies should track raw material costs, technology upgrades, customer purchasing cycles, regional policy changes, and competitive moves that can alter pricing and adoption rates.
The Packaging Extended Producer Responsibility Compliance Test Platforms Market is segmented by Deployment (Cloud, On-premise, Hybrid), Platform Type (Multi-jurisdiction, Single-country, PRO-linked), Functionality (Reporting Engines, Fee Modeling, Data Mapping, Audit Trails, Eco-modulation), End User (Brand Owners, Retailers, Importers, Converters, Marketplaces), Enterprise Size (Large Enterprises, Mid-market, SMEs), and Region. Forecast runs from 2026 to 2036.
The PPWR-Compliant Recyclable PE/PP Stand-Up Pouch Recycling Systems Market Is Segmented By Recycling Technology (Mechanical Systems, Chemical Systems, Hybrid Systems), Material Stream (PE Pouches, PP Pouches, Mixed PO), System Module (Sorting Lines, Washing Lines, Extrusion Lines, Digital Traceability, Testing Labs), End Use (Food Brands, Personal Care, Household Care, Pet Food, Industrial Goods), Plant Format (Retrofit Lines, Greenfield Plants, Tolling Units), And Region. Forecast For 2026 To 2036.
The Reusable Produce Mesh Bag Market Is Segmented By Material (Organic Cotton, Recycled Polyester, Cellulose Blends, Hemp), Product Type (Drawstring Bags, Flat Bags, Tare-Tag Bags, Moisture-Lock Bags), Distribution Channel (Online Retail, Specialty Stores, Supermarkets, Wholesale), End Use (Household, Retail Programs, Bulk Foods, Food Service), Pack Size (Multi-Pack Sets, Single Bags, Three-Pack, Six-Pack), And Region. Forecast for 2026 to 2036.
The Fiber Cushioning is segmented by Material type (Molded pulp, Paper pads, Corrugated cushions, Honeycomb cushions, Hybrid fiber), Form factor (Inserts, End caps, Wraps, Corner blocks, Trays), End use (Electronics, Industrial goods, Food bottles, Healthcare devices, Consumer durables), Sales channel (Custom converters, System suppliers, OEM direct, Distributors), Function (Shock absorption, Void fill, Bracing, Surface protection, Moisture buffering), and Region. Forecast for 2026 to 2036.
Molded Pulp Inserts is segmented by Product type (End caps, Trays, Clamshells, Cradles, Corner blocks), Pulp type (Transfer molded, Thermoformed, Thick wall, Processed pulp), Source (Recycled pulp, Virgin pulp, Blended pulp), End use (Electronics, Appliances, Industrial goods, Cosmetics, Medical devices), Sales channel (Custom OEM, Contract packaging, Distributor sales), and Region. Forecast for 2026 to 2036.