Checkpoint Inhibitor Refractory Cancer Market : Global Industry Analysis 2015 - 2024 and Opportunity Assessment 2025 - 2035
Checkpoint Inhibitor Refractory Cancer Market Size and Share Forecast Outlook 2025 to 2035
Historical Data Covered: 2015 - 2024 | Base Year: 2024 | Estimated Year: 2025 | Forecast Period: 2025 - 2035
Checkpoint Inhibitor Refractory Cancer Market Size and Share Forecast Outlook 2025 to 2035
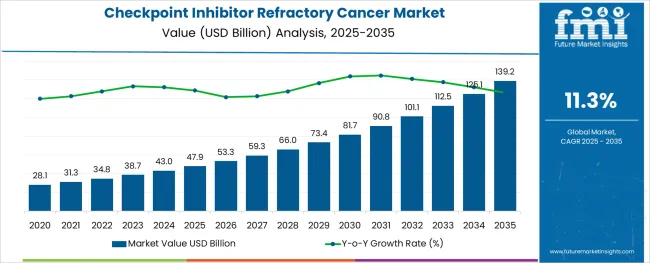
The Checkpoint Inhibitor Refractory Cancer Market is estimated to be valued at USD 47.9 billion in 2025 and is projected to reach USD 139.2 billion by 2035, registering a compound annual growth rate (CAGR) of 11.3% over the forecast period.
Quick Stats for Checkpoint Inhibitor Refractory Cancer Market
- Checkpoint Inhibitor Refractory Cancer Market Industry Value (2025): USD 47.9 billion
- Checkpoint Inhibitor Refractory Cancer Market Forecast Value (2035): USD 139.2 billion
- Checkpoint Inhibitor Refractory Cancer Market Forecast CAGR: 11.3%
- Leading Segment in Checkpoint Inhibitor Refractory Cancer Market in 2025: PD-1 Inhibitor (42.5%)
- Key Growth Region in Checkpoint Inhibitor Refractory Cancer Market: North America, Asia-Pacific, Europe
- Top Key Players in Checkpoint Inhibitor Refractory Cancer Market: Bristol‑Myers Squibb, 4D Pharma Plc, 4SC AG, Ascentage Pharma Group, Eisai Inc, AstraZeneca, Merck, Opdivo, Keytruda, Tecentriq
Rationale for Segmental Growth in the Checkpoint Inhibitor Refractory Cancer Market
Market Overview
The checkpoint inhibitor refractory cancer market is witnessing significant momentum as resistance to standard immunotherapies emerges as a critical challenge in oncology. Advances in understanding tumor immune evasion mechanisms have created avenues for specialized therapies that address refractory cases, offering renewed hope for patients who do not respond to first-line checkpoint inhibitors.
Continued investment in clinical trials and biomarker-driven drug development is shaping the competitive landscape, with pharmaceutical companies focusing on combination regimens and novel targets beyond traditional pathways. Regulatory agencies are prioritizing accelerated approvals in this space, reflecting the unmet clinical need and strong demand from oncologists for more effective options.
In the coming years, the market is expected to benefit from expanding research into tumor microenvironment modulation, improved patient stratification techniques, and strategic alliances between biotechnology firms and academic research centers that are paving the path for more durable and personalized treatment solutions.
Segmental Analysis
The market is segmented by Type and Application and region. By Type, the market is divided into PD-1 Inhibitor, CTLA-4 Inhibitor, PD-L1 Inhibitor, and Others. In terms of Application, the market is classified into Lung Cancer, Bladder Cancer, Melanoma, Hodgkin Lymphoma, and Others. Regionally, the market is classified into North America, Latin America, Western Europe, Eastern Europe, Balkan & Baltic Countries, Russia & Belarus, Central Asia, East Asia, South Asia & Pacific, and the Middle East & Africa.
Insights into the PD 1 Inhibitor Type Segment
When segmented by type, the PD 1 inhibitor segment is projected to command 42⋅5 share of the market in 2025, reinforcing its leadership position. This dominance has been supported by the well-established mechanism of action of PD 1 inhibitors, which has set the foundation for further development and refinement of therapies targeting refractory cases.
Widespread clinical familiarity and robust safety profiles have contributed to continued reliance on PD 1 based strategies, even in the face of resistance, often as a component of combination regimens. Advances in biomarker identification and dosage optimization have allowed for more precise application of PD 1 inhibitors in refractory populations, extending their utility beyond initial assumptions.
Pharmaceutical pipelines remain heavily concentrated on enhancing the efficacy of PD 1 inhibitors through adjunctive therapies, which has sustained investment and innovation in this segment, securing its leading share of the market.
Insights into the Lung Cancer Application Segment
Segmented by application, lung cancer is expected to hold 38⋅0 share of the checkpoint inhibitor refractory cancer market in 2025, establishing itself as the most significant application segment. This prominence has been reinforced by the high incidence and mortality associated with lung cancer, making it a critical focus area for therapeutic development.
The complex tumor biology and historically poor prognosis in refractory lung cancer cases have driven sustained clinical and commercial attention toward this indication. Enhanced screening programs and improved molecular characterization of lung tumors have enabled more targeted approaches, including refractory-specific interventions.
Additionally, strong engagement from both public and private sectors in funding lung cancer research has facilitated broader access to innovative treatments, fostering adoption of checkpoint inhibitor refractory therapies. The urgency to address unmet needs in this population has propelled lung cancer to the forefront of application areas within the market, consolidating its leadership position.
2018 to 2022 Global Checkpoint Inhibitor Refractory Cancer Demand Outlook Compared to Forecast from 2023 to 2033
The global demand for Checkpoint Inhibitor Refractory Cancer is projected to increase at a CAGR of 11.26% during the forecast period between 2025 and 2035, reaching a total of USD 139.2 Billion in 2035, according to a report from Future Market Insights (FMI). From 2020 to 2025, sales witnessed significant growth, registering a CAGR of 5.5%.
According to Future Market Insights, a market research and competitive intelligence provider, the Checkpoint Inhibitor Refractory Cancer market was valued at USD 47.9 Billion in 2025.
The key factors leading to the market growth include the geriatric population across the globe, as well as a rapid increase in the global prevalence of several cancers, mainly in emerging and developed economies, which is expected to boost market growth in the forthcoming years.
For instance, as the World Health Organization reports, cancer has become a primary cause of death globally. As per the updated release of Globocan report released by the International Agency for Research on Cancer (IARC), in December 2024, the global burden of cancer has risen to 43 million cases and deaths by cancer have increased to 10 million in 2024. Thus, an upsurge in the volume of the patient pool leads to a greater demand for the market, which is accelerating the growth of the global market during the analysis period.
Leading industry players are focusing on the development and launch of novel PD-1/PD-L1 inhibitors to treat various types of cancers. This escalated the availability of PD-1/PD-L1 immunotherapeutic in the market is expected to boost the checkpoint inhibitor refractory cancer market.
Key Drivers Supporting Expansion in the Checkpoint Inhibitor Refractory Cancer Market
Rise in Incidence of Cancer Globally to Fuel the Market Growth
The rise in the prevalence of cancer around the world is a major factor that will escalate the growth of the global checkpoint inhibitor refractory cancer market in the coming years. For instance, these checkpoint inhibitors are significantly used as front-line treatments for several types of cancer. As per the World Health Organization (WHO), cancer was one of the leading causes of death worldwide, accounting for around ten million deaths in 2024, or nearly one in six deaths. The most common cancers include breast, lung, colon and rectum, and prostate cancers. Thus, there is an increase in demand for checkpoint inhibitors globally.
There are various novel therapies, which have been approved in recent years. In addition, there is a gap between the demand and supply of these therapies, which has led to a huge unmet need. This, in turn, has given vendors an opportunity to conduct studies on drugs such as checkpoint inhibitors. In addition, the increasing approvals of these inhibitors are expected to increase their demand, which is anticipated to fuel the growth of the market.
Increase in Geriatric Population to Accelerate the Market Growth
The rise in cases of the geriatric population globally is projected to fuel the growth of the checkpoint inhibitor refractory cancer market over the analysis period. For instance, as per the Union for International Cancer Control (UICC), the elderly (people aged 65 years and more) are 11 times more likely to develop cancer, when compared to younger people. According to the United Nations, there are over 28.1 million people aged 65 or older, a number that is expected to reach 1.5 billion by 2050. According to the latest estimates and projections from UN DESA’s Population Division, one in six people in the world will be over the age of 65 by 2050, up from 1 in 11 in 2020.
The rise in research and development activities is expected to offer significant lucrative avenues for players in the checkpoint inhibitor refractory cancer market. For instance, market players have been increasingly investing in Research and Development activities, in order to meet the increasing demand. For instance, researchers at Johns Hopkins Kimmel Cancer Center are paving the way in developing novel immunotherapies called anti-PD-1 and anti-PD-L1 for people with advanced melanomas. The aim of the therapy is not to kill cancer cells directly, but to block a pathway, that protects tumor cells from components of the immune system that are capable and ready to fight cancer.
An increase in awareness among people about immune checkpoint inhibitors is projected to provide significant growth prospects for players in the global checkpoint inhibitor market. For instance, with the increase in awareness among people, the demand for these inhibitors is also increasing at a rapid pace globally.
Challenges Limiting Growth in the Checkpoint Inhibitor Refractory Cancer Industry
Stringent Government Regulations to Restrain the Market Growth
Globally, several laws have been introduced to monitor the quality as well as the effectiveness of inhibitors. The regulatory requirements are becoming increasingly stringent, as biologics progress through clinical trials and to commercialization. Due to the nature of biologics, which are produced by living cells, the process and the product must be validated as well as strictly monitored against critical quality attributes (CQAs), throughout the manufacturing process, not just at the end as for chemistry-based drugs. This creates hurdles to getting regulatory approval for the biologics, which in turn is expected to impede the growth of the market.
Further, cancer treatment can cause many side effects, such as depression, tiredness, and trouble eating. The National Cancer Institute and the American Cancer Society describe many of these problems and offer tips for preventing or coping with them. Several side effects can happen months or years after treatment. Hence, the risk of complications associated with the treatment is another factor that will hamper the market growth.
High Cost of Inhibitors to Limit the Market Growth
Cancer treatment costs create significant economic challenges for healthcare providers as well as patients, because of the impact they have on patient safety, quality of care, and overall healthcare. The cost of cancer treatment is high, mainly due to laboratory tests, hospitalization, and the administration of novel checkpoint inhibitors (PD-1 and PD-L1) inhibitor therapeutics. The treatment cost of these therapeutics depends on the stage of cancer. In addition, the high cost is leading to a rise in the preference for alternative treatment options such as surgeries.
For instance, on average, the cost of cancer treatment in India is around INR 503,118 (USD 6,300) with a minimum expense of INR 90,561 (USD 1,134) and a maximum cost of INR 27,67,149 (USD 34,650). Also, the cost of available checkpoint inhibitors is high. Thus, the high cost associated with cancer treatment is a major factor that will restrain the treatment adoption rate especially in developing regions.
Regional Trends Shaping Treatment Access and Innovation in Refractory Cancers
Will North America Continue Dominance in the Global Checkpoint Inhibitor Refractory Cancer Market?
Favorable Initiatives by Regulatory Authorities in the Region to Fuel the Market Growth
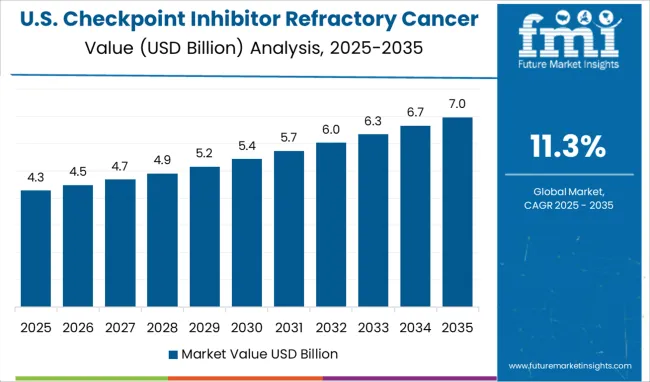
The Checkpoint Inhibitor Refractory Cancer Market in North America is expected to accumulate the highest market share of 39% in 2025.
Factors such as favorable initiatives by the regulatory authorities in the region, especially in the USA, coupled with the increasing prevalence of cancer are expected to boost the growth of the checkpoint inhibitor refractory cancer market in North America. For instance, these inhibitors are used to treat a variety of cancer. As per the American Cancer Society, in 2025, there will be an estimated 1.9 million new cancer cases diagnosed and 609,360 cancer deaths in the USA. This in turn is anticipated to augment the market’s revenue share in the region.
The USA is estimated to account for around 99.2% of the North America Checkpoint inhibitors market in 2025. The significant share in the US is mainly attributed to the rising incidence of several chronic diseases such as urothelial carcinomas, lung cancers, skin cancer, and diabetes in the region, which demand efficient treatment.
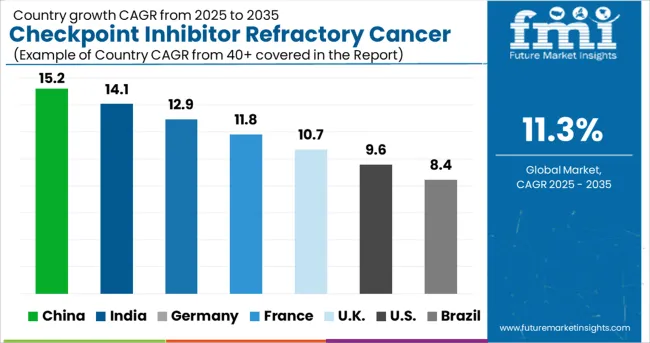
For instance, according to a report from the American Cancer Society (2024), lung cancer is the second leading cancer resulting in 135,720 deaths in men as well as women. The numbers also report that around 84% of all lung cancers are NSCLC (non-small cell lung cancer) and 13% (small cell lung cancer) in the USA, which is expected to continue in the forthcoming years, leading to greater demand for checkpoint Inhibitors thus, fueling the market in the region. The region is expected to hold the highest CAGR of 11.1% during the forecast period.
How Will the Growth of the Checkpoint Inhibitor Refractory Cancer Market unfold in Asia Pacific?
Increasing Government Engagement to Surge Cancer Diagnosis in the Region to Fuel the Market Growth
The Checkpoint Inhibitor Refractory Cancer Market in Asia Pacific is expected to accumulate the highest market share of 37% in 2025.
The regional growth is attributed to the factors such as the high incidence of cancer, growing government engagement to surge cancer diagnosis, and growing adoption of inorganic growth strategies like collaborations, acquisitions, and mergers by major players functioning in the market introduction of new immunotherapies with lesser side effects, and increasing geriatric population in this region, especially in India. For instance, as per the cancer report by the Indian Council of Medical Research (ICMR), India’s cancer cases could increase by 12% in the next five years, with 47.9 million people projected to suffer from the non-communicable disease by 2025, up from 43 million in 2024. This is a major factor that will stimulate market growth.
Besides, China is expected to hold a significant growth rate over the analysis period, attributed to factors including a large population base, and major investment from private and public sectors into healthcare infrastructure development. Also, the growing interest in immune checkpoint inhibitors, coupled with the increase in the number of cancer cases, is anticipated to aid the development of the regional market in the near future. The region is expected to hold the highest CAGR of 11.0% during the forecast period.
Checkpoint Inhibitor Refractory Cancer Industry Analysis by Cancer Type, Therapy, and End User
By Application, which Checkpoint Inhibitor Refractory Cancer Category will remain prominent?
Non-Small Cell Lung Segment to beat Competition in Untiring Market
On the basis of type, the global Checkpoint Inhibitor Refractory Cancer market is dominated by the Non-Small Cell Lung Segment, which accounts for a share of 37%. The segment is expected to hold a CAGR of 11.1% over the analysis period.
This significant share of non-small cell lung cancer has encouraged the majority of major players to concentrate on continuous innovation of advanced therapeutics and improving the efficacy of the present therapeutics.
Additionally, a growing number of diagnoses of NSCLC (non-small cell lung cancer), and the rising awareness in the nations of the developing and developed markets are supporting the growth of the non-small cell lung cancer segment.
By End-User, which Checkpoint Inhibitor Refractory Cancer Category dominates?
Hospital Pharmacies Segment to Drive the Checkpoint Inhibitor Refractory Cancer Market
Based on the End-User, the hospital pharmacies segment is expected to witness a significant growth of 48.3% in 2025, and the trend is expected to continue during the forecast period, expanding at a rapid rate of 11.0% CAGR over the analysis period.
The segment’s growth is attributed to the factors such as large-scale orders, robust supply chains as well as attempts by hospital facilities to sustain large-scale inventories for cancer therapy applications. Moreover, the presence of advanced equipment and the availability of well-developed infrastructure adds to the development of the segment.
Further, the growing patient admission to hospitals for cancer treatment is surging the use of checkpoint inhibitors in hospitals, which in turn is anticipated to accelerate the sales in hospital pharmacies.
Emerging Start-ups Unlocking Innovation in Refractory Cancer Therapeutics
Checkpoint Inhibitor Refractory Cancer Market start-up players are adopting various marketing strategies such as new product launches, geographical expansion, mergers and acquisitions, partnerships, and collaboration to identify the interest of potential patients and create a larger customer base. For instance,
- Founded in 2020, ALX Oncology is a developer of immuno-oncology therapies for cancer treatment. The lead product candidate ALX148 is a checkpoint inhibitor that comprises an affinity CD47 binding domain of SIRPα linked to an inactive Fc region of a human immunoglobulin. It binds CD47 and inhibits the CD47-SIRPα signaling pathway.
- Established in 2020, another start-up named Hummingbird Bioscience is a developer of antibody drugs for cancer treatment. It is involved in developing antibodies by using systems immunology and computational structural biology technology. The company is using platform technology for the design and development of therapeutic antibodies. The pipelined products are HMBD-001 a monoclonal antibody and HMBD-002 an anti-vista monoclonal antibody for cancer treatment.
- Founded in 2020, Shattuck Labs is a developer of an immunotherapy platform for oncology and other diseases. The company has developed ARC (Agonist Redirected Checkpoints), which hinges on the development of bispecific biologics that simultaneously block candidate checkpoint molecules in cancer while also stimulating TNF superfamily costimulatory receptors on T cells and innate cells. The company claims that ARC is adaptable to most of the checkpoint targets currently in clinical development.
Top Companies Leading the Checkpoint Inhibitor Refractory Cancer Market
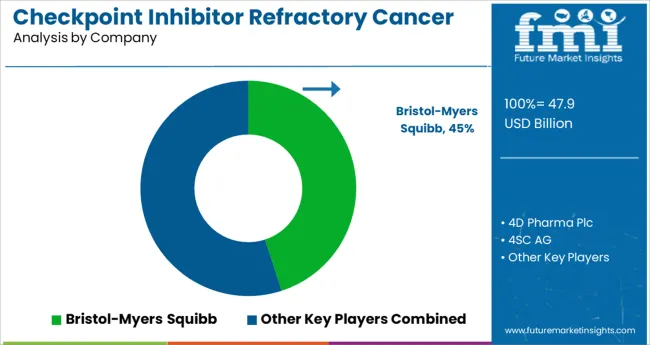
Prominent players in the Checkpoint Inhibitor Refractory Cancer market are Bristol-Myers Squibb, AstraZeneca, Merck, Genentech/Hoffmann-La Roche, Regeneron Pharmaceuticals, Merck KGaA, Pfizer, Bristol-Myers Squibb, Janssen Research and Development, LLC., 4D pharma plc., 4SC AG, OncoSec Medical, Mirati Therapeutics, Ascentage Pharma Group, and ENB Therapeutics, Inc., among others.
Recent Developments:
- In October 2024, Merck announced the USA Food and Drug Administration (FDA) approval for KEYTRUDA, Merck’s anti-PD-1 therapy, in combination with chemotherapy, with or without bevacizumab. The therapy is used for the treatment of patients with persistent, recurrent, or metastatic cervical cancer whose tumors express PD-L1 (Combined Positive Score [CPS] ≥1) as determined by an FDA-approved test.
- In January 2024, Novartis International AG acquired The Medicines Company. This acquisition helped Novartis in the treatment of the world’s leading cause of mortality and disability with a vaccine-like approach.
- In May 2024, the Food and Drug Administration (FDA) approved Roche’s Tecentriq, as a first-line monotherapy for certain people with metastatic non-small cell lung.
Scope of the Checkpoint Inhibitor Refractory Cancer Market Report
| Report Attribute | Details |
|---|---|
| Growth Rate | CAGR of 11.26% from 2025 to 2035 |
| Market Value in 2025 | USD 47.9 billion |
| Market Value in 2035 | USD 139.2 billion |
| Base Year for Estimation | 2025 |
| Historical Data | 2020 to 2025 |
| Forecast Period | 2025 to 2035 |
| Quantitative Units | Revenue in billion and CAGR from 2025 to 2035 |
| Report Coverage | Revenue Forecast, Company Ranking, Competitive Landscape, Growth Factors, Trends, and Pricing Analysis |
| Segments Covered | Type, Application, End-User, Region |
| Regions Covered | North America; Europe; Asia Pacific; Rest of the World |
| Key Countries Profiled | USA, Canada, Brazil, Mexico, Germany, UK, France, Spain, Italy, China, Japan, South Korea, Malaysia, Singapore, Australia, New Zealand, GCC Countries, South Africa, Israel |
| Key Companies Profiled | Bristol-Myers Squibb; AstraZeneca; Merck; Genentech/Hoffmann-La Roche; Regeneron Pharmaceuticals; Merck KGaA and Pfizer; Bristol-Myers Squibb; Janssen Research and Development, LLC; 4D pharma plc.; 4SC AG; OncoSec Medical; Mirati Therapeutics; Ascentage Pharma Group; ENB Therapeutics, Inc.; Exicure, Inc.; Evelo Biosciences, Inc./Merck Sharp & ; Dohme Corp.; Eisai; Kartos Therapeutics; Exelixis; ImmunityBio |
Top Investment Segments in the Checkpoint Inhibitor Refractory Cancer Industry Analysis
ByType:
- PD-1 Inhibitors
- PD-L1 Inhibitors
By Application:
- Hodgkin Lymphoma
- Kidney Cancer
- Melanoma
- Non-Small Cell Lung Cancer
- Others
By End-User:
- Hospital Pharmacies
- Retail Pharmacies
- Online Pharmacies
By Region:
- North America
- Europe
- Asia Pacific
- Rest of the World
Frequently Asked Questions
How big is the checkpoint inhibitor refractory cancer market in 2025?
The global checkpoint inhibitor refractory cancer market is estimated to be valued at USD 47.9 billion in 2025.
What will be the market size for the checkpoint inhibitor refractory cancer market in 2035?
It is projected to reach USD 139.2 billion by 2035.
How much will be the CAGR of the checkpoint inhibitor refractory cancer market between 2025 and 2035?
The market is expected to grow at a 11.3% CAGR between 2025 and 2035.
What are the key product types in the checkpoint inhibitor refractory cancer market?
The key product types are pd-1 inhibitor, ctla-4 inhibitor, pd-l1 inhibitor and others.
Which lung cancer segment is expected to dominate the checkpoint inhibitor refractory cancer market in 2025?
lung cancer segment is expected to dominate with a 38.0% industry share in 2025.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand-side Trends
- Supply-side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2020 to 2024 and Forecast, 2025 to 2035
- Historical Market Size Value (USD Millon) Analysis, 2020 to 2024
- Current and Future Market Size Value (USD Millon) Projections, 2025 to 2035
- Y-o-Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2020 to 2024 and Forecast 2025 to 2035
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Type
- Introduction / Key Findings
- Historical Market Size Value (USD Millon) Analysis By Type , 2020 to 2024
- Current and Future Market Size Value (USD Millon) Analysis and Forecast By Type , 2025 to 2035
- PD-1 Inhibitor
- CTLA-4 Inhibitor
- PD-L1 Inhibitor
- Others
- PD-1 Inhibitor
- Y-o-Y Growth Trend Analysis By Type , 2020 to 2024
- Absolute $ Opportunity Analysis By Type , 2025 to 2035
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Millon) Analysis By Application, 2020 to 2024
- Current and Future Market Size Value (USD Millon) Analysis and Forecast By Application, 2025 to 2035
- Lung Cancer
- Bladder Cancer
- Melanoma
- Hodgkin Lymphoma
- Others
- Lung Cancer
- Y-o-Y Growth Trend Analysis By Application, 2020 to 2024
- Absolute $ Opportunity Analysis By Application, 2025 to 2035
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Region
- Introduction
- Historical Market Size Value (USD Millon) Analysis By Region, 2020 to 2024
- Current Market Size Value (USD Millon) Analysis and Forecast By Region, 2025 to 2035
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Millon) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Millon) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- USA
- Canada
- Mexico
- By Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Application
- Key Takeaways
- Latin America Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Millon) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Millon) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Application
- Key Takeaways
- Western Europe Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Millon) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Millon) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Application
- Key Takeaways
- Eastern Europe Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Millon) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Millon) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Application
- Key Takeaways
- East Asia Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Millon) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Millon) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- China
- Japan
- South Korea
- By Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Application
- Key Takeaways
- South Asia and Pacific Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Millon) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Millon) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Application
- Key Takeaways
- Middle East & Africa Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Millon) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Millon) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Application
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Application
- Canada
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Application
- Mexico
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Application
- Brazil
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Application
- Chile
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Application
- Germany
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Application
- UK
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Application
- Italy
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Application
- Spain
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Application
- France
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Application
- India
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Application
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Application
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Application
- China
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Application
- Japan
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Application
- South Korea
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Application
- Russia
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Application
- Poland
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Application
- Hungary
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Application
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Application
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Application
- South Africa
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Application
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Type
- By Application
- Competition Analysis
- Competition Deep Dive
- Bristol‑Myers Squibb
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- 4D Pharma Plc
- 4SC AG
- Ascentage Pharma Group
- Eisai Inc
- AstraZeneca
- Merck
- Opdivo
- Keytruda
- Tecentriq
- Bristol‑Myers Squibb
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: Global Market Value (USD Millon) Forecast by Region, 2020-2035
- Table 2: North America Market Value (USD Millon) Forecast by Country, 2020-2035
- Table 3: Latin America Market Value (USD Millon) Forecast by Country, 2020-2035
- Table 4: Western Europe Market Value (USD Millon) Forecast by Country, 2020-2035
- Table 5: Eastern Europe Market Value (USD Millon) Forecast by Country, 2020-2035
- Table 6: East Asia Market Value (USD Millon) Forecast by Country, 2020-2035
- Table 7: South Asia and Pacific Market Value (USD Millon) Forecast by Country, 2020-2035
- Table 8: Middle East & Africa Market Value (USD Millon) Forecast by Country, 2020-2035
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Millon) Forecast 2020-2035
- Figure 3: Global Market Value (USD Millon) Share and BPS Analysis by Region, 2025 and 2035
- Figure 4: Global Market Y-o-Y Growth Comparison by Region, 2025-2035
- Figure 5: Global Market Attractiveness Analysis by Region
- Figure 6: North America Market Incremental $ Opportunity, 2025-2035
- Figure 7: Latin America Market Incremental $ Opportunity, 2025-2035
- Figure 8: Western Europe Market Incremental $ Opportunity, 2025-2035
- Figure 9: Eastern Europe Market Incremental $ Opportunity, 2025-2035
- Figure 10: East Asia Market Incremental $ Opportunity, 2025-2035
- Figure 11: South Asia and Pacific Market Incremental $ Opportunity, 2025-2035
- Figure 12: Middle East & Africa Market Incremental $ Opportunity, 2025-2035
- Figure 13: North America Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 14: Latin America Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 15: Western Europe Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 16: Eastern Europe Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 17: East Asia Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 18: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 19: Middle East & Africa Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 20: Global Market - Tier Structure Analysis
- Figure 21: Global Market - Company Share Analysis

